Analyze Safety Protocols for Chlorine Trifluoride Usage
FEB 13, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Chlorine Trifluoride Safety Background and Objectives
Chlorine trifluoride (ClF3) represents one of the most reactive and hazardous oxidizing agents utilized in industrial applications, particularly within semiconductor manufacturing, nuclear fuel processing, and specialized chemical synthesis operations. This compound exhibits extreme reactivity with virtually all organic and inorganic materials, including substances traditionally considered non-flammable such as water, sand, and asbestos. Historical incidents involving ClF3 have demonstrated catastrophic consequences, including the 1950s industrial accidents that resulted in facility destruction and severe personnel injuries, underscoring the critical necessity for comprehensive safety protocols.
The evolution of ClF3 safety management has progressed through distinct phases since its initial industrial adoption in the 1940s. Early applications in the Manhattan Project revealed fundamental handling challenges, prompting the development of specialized containment systems and emergency response procedures. Subsequent decades witnessed refinement of safety standards driven by accumulated operational experience and advancing materials science, particularly in the development of passivated metal alloys and fluoropolymer sealing technologies capable of withstanding ClF3 exposure.
Current safety objectives center on establishing multi-layered protection systems that address the compound's unique hazard profile. Primary goals include preventing uncontrolled reactions through rigorous containment engineering, implementing real-time monitoring systems for early leak detection, and developing rapid response protocols for incident mitigation. The technical challenge extends beyond simple containment to encompass personnel protection, environmental safeguarding, and maintaining operational continuity while managing this extraordinarily reactive substance.
The strategic importance of advancing ClF3 safety protocols stems from its irreplaceable role in certain high-value manufacturing processes, particularly in semiconductor etching and uranium hexafluoride production. As industrial demand intensifies and regulatory requirements become more stringent, organizations face mounting pressure to demonstrate robust safety management capabilities. This necessitates continuous improvement in engineering controls, procedural safeguards, and emergency preparedness frameworks to minimize risk exposure while maintaining operational efficiency in facilities handling this exceptionally hazardous material.
The evolution of ClF3 safety management has progressed through distinct phases since its initial industrial adoption in the 1940s. Early applications in the Manhattan Project revealed fundamental handling challenges, prompting the development of specialized containment systems and emergency response procedures. Subsequent decades witnessed refinement of safety standards driven by accumulated operational experience and advancing materials science, particularly in the development of passivated metal alloys and fluoropolymer sealing technologies capable of withstanding ClF3 exposure.
Current safety objectives center on establishing multi-layered protection systems that address the compound's unique hazard profile. Primary goals include preventing uncontrolled reactions through rigorous containment engineering, implementing real-time monitoring systems for early leak detection, and developing rapid response protocols for incident mitigation. The technical challenge extends beyond simple containment to encompass personnel protection, environmental safeguarding, and maintaining operational continuity while managing this extraordinarily reactive substance.
The strategic importance of advancing ClF3 safety protocols stems from its irreplaceable role in certain high-value manufacturing processes, particularly in semiconductor etching and uranium hexafluoride production. As industrial demand intensifies and regulatory requirements become more stringent, organizations face mounting pressure to demonstrate robust safety management capabilities. This necessitates continuous improvement in engineering controls, procedural safeguards, and emergency preparedness frameworks to minimize risk exposure while maintaining operational efficiency in facilities handling this exceptionally hazardous material.
Industrial Demand for ClF3 Applications
Chlorine trifluoride has established itself as a critical chemical reagent across multiple industrial sectors, driven by its exceptional reactivity and unique chemical properties. The semiconductor manufacturing industry represents one of the most significant demand centers, where ClF3 serves as an essential chamber cleaning agent for chemical vapor deposition equipment. Its ability to effectively remove silicon and metal deposits from reactor walls makes it indispensable in maintaining production efficiency and product quality in microelectronics fabrication facilities.
The nuclear fuel processing sector constitutes another major application domain for chlorine trifluoride. Its powerful fluorinating capabilities enable the conversion of uranium compounds into uranium hexafluoride, a crucial intermediate in nuclear fuel enrichment processes. This application has sustained consistent demand despite stringent regulatory oversight and specialized handling requirements inherent to the nuclear industry.
Aerospace and rocket propulsion research continues to explore ClF3 as a potential oxidizer due to its hypergolic properties and high specific impulse characteristics. While practical implementation remains limited by safety considerations, ongoing research programs maintain specialized demand for experimental and testing purposes. The compound's ability to ignite spontaneously with most organic materials presents both opportunities and challenges in propulsion system development.
The specialty chemicals sector utilizes chlorine trifluoride in synthesizing various fluorinated compounds and intermediates. Pharmaceutical manufacturers employ it in producing fluorinated active pharmaceutical ingredients, where selective fluorination can significantly enhance drug efficacy and metabolic stability. This niche but high-value application segment demonstrates steady growth aligned with expanding fluorochemistry applications in drug discovery.
Emerging applications in advanced materials processing and surface treatment technologies are gradually expanding the industrial demand landscape. Research into novel fluoropolymer synthesis routes and specialized coating technologies continues to identify new potential uses. However, the extreme hazards associated with ClF3 handling impose significant barriers to widespread adoption, constraining market expansion primarily to facilities with sophisticated safety infrastructure and specialized technical expertise. The overall demand trajectory reflects a balance between compelling technical advantages and formidable safety management requirements.
The nuclear fuel processing sector constitutes another major application domain for chlorine trifluoride. Its powerful fluorinating capabilities enable the conversion of uranium compounds into uranium hexafluoride, a crucial intermediate in nuclear fuel enrichment processes. This application has sustained consistent demand despite stringent regulatory oversight and specialized handling requirements inherent to the nuclear industry.
Aerospace and rocket propulsion research continues to explore ClF3 as a potential oxidizer due to its hypergolic properties and high specific impulse characteristics. While practical implementation remains limited by safety considerations, ongoing research programs maintain specialized demand for experimental and testing purposes. The compound's ability to ignite spontaneously with most organic materials presents both opportunities and challenges in propulsion system development.
The specialty chemicals sector utilizes chlorine trifluoride in synthesizing various fluorinated compounds and intermediates. Pharmaceutical manufacturers employ it in producing fluorinated active pharmaceutical ingredients, where selective fluorination can significantly enhance drug efficacy and metabolic stability. This niche but high-value application segment demonstrates steady growth aligned with expanding fluorochemistry applications in drug discovery.
Emerging applications in advanced materials processing and surface treatment technologies are gradually expanding the industrial demand landscape. Research into novel fluoropolymer synthesis routes and specialized coating technologies continues to identify new potential uses. However, the extreme hazards associated with ClF3 handling impose significant barriers to widespread adoption, constraining market expansion primarily to facilities with sophisticated safety infrastructure and specialized technical expertise. The overall demand trajectory reflects a balance between compelling technical advantages and formidable safety management requirements.
Current Safety Challenges in ClF3 Handling
Chlorine trifluoride handling presents formidable safety challenges that stem from its extreme reactivity and hazardous properties. The compound reacts violently with virtually all organic and inorganic materials, including substances traditionally considered non-flammable such as concrete, sand, and asbestos. This universal reactivity creates unprecedented containment difficulties, as even trace amounts of incompatible materials can trigger catastrophic reactions. The substance ignites spontaneously upon contact with most materials at room temperature, generating toxic byproducts including hydrogen fluoride and chlorine gas, which pose severe respiratory and corrosive hazards to personnel.
Material compatibility remains one of the most critical challenges in ClF3 operations. While nickel, monel, and certain fluorinated polymers demonstrate relative resistance after passivation, maintaining the integrity of passivation layers requires meticulous control. Any breach in these protective layers, whether from mechanical damage, thermal cycling, or chemical degradation, can expose reactive substrate materials and initiate violent reactions. The selection and maintenance of appropriate containment materials demand continuous monitoring and specialized expertise that many facilities lack.
Personnel exposure risks are amplified by ClF3's ability to penetrate conventional protective equipment. Standard chemical-resistant materials offer inadequate protection, necessitating specialized gear that is often cumbersome and limits operational efficiency. The compound's high vapor pressure and tendency to generate corrosive fumes create atmospheric contamination risks even in well-ventilated spaces. Emergency response protocols face unique complications, as traditional fire suppression methods prove ineffective or counterproductive when dealing with ClF3 incidents.
Storage and transportation present additional layers of complexity. The substance requires specialized pressure vessels capable of withstanding both chemical attack and potential pressure buildup from decomposition reactions. Temperature control systems must maintain narrow operational windows while preventing condensation or overheating. Leak detection systems face technical limitations, as many conventional sensors cannot survive exposure to ClF3 vapors. The absence of standardized transportation protocols and limited carrier availability further constrain industrial applications.
Human error and procedural deviations constitute persistent vulnerability factors in ClF3 operations. The unforgiving nature of the compound leaves minimal margin for mistakes, yet the complexity of handling procedures increases the likelihood of human error. Training programs struggle to adequately prepare personnel for the full spectrum of potential scenarios without exposing them to unacceptable risks during practical exercises.
Material compatibility remains one of the most critical challenges in ClF3 operations. While nickel, monel, and certain fluorinated polymers demonstrate relative resistance after passivation, maintaining the integrity of passivation layers requires meticulous control. Any breach in these protective layers, whether from mechanical damage, thermal cycling, or chemical degradation, can expose reactive substrate materials and initiate violent reactions. The selection and maintenance of appropriate containment materials demand continuous monitoring and specialized expertise that many facilities lack.
Personnel exposure risks are amplified by ClF3's ability to penetrate conventional protective equipment. Standard chemical-resistant materials offer inadequate protection, necessitating specialized gear that is often cumbersome and limits operational efficiency. The compound's high vapor pressure and tendency to generate corrosive fumes create atmospheric contamination risks even in well-ventilated spaces. Emergency response protocols face unique complications, as traditional fire suppression methods prove ineffective or counterproductive when dealing with ClF3 incidents.
Storage and transportation present additional layers of complexity. The substance requires specialized pressure vessels capable of withstanding both chemical attack and potential pressure buildup from decomposition reactions. Temperature control systems must maintain narrow operational windows while preventing condensation or overheating. Leak detection systems face technical limitations, as many conventional sensors cannot survive exposure to ClF3 vapors. The absence of standardized transportation protocols and limited carrier availability further constrain industrial applications.
Human error and procedural deviations constitute persistent vulnerability factors in ClF3 operations. The unforgiving nature of the compound leaves minimal margin for mistakes, yet the complexity of handling procedures increases the likelihood of human error. Training programs struggle to adequately prepare personnel for the full spectrum of potential scenarios without exposing them to unacceptable risks during practical exercises.
Existing ClF3 Safety Protocol Solutions
01 Containment and storage systems for chlorine trifluoride
Specialized containment vessels and storage systems are designed to safely handle chlorine trifluoride due to its highly reactive and corrosive nature. These systems typically incorporate corrosion-resistant materials such as nickel alloys, monel, or specialized fluoropolymers that can withstand the aggressive chemical properties. The containment systems include pressure relief mechanisms, temperature control features, and leak detection systems to prevent accidental releases and ensure safe storage conditions.- Storage and containment systems for chlorine trifluoride: Specialized storage vessels and containment systems are designed to safely handle chlorine trifluoride due to its highly reactive and corrosive nature. These systems incorporate materials resistant to fluorine compounds and feature pressure relief mechanisms, temperature control systems, and leak detection capabilities. The containment structures are engineered to prevent reactions with moisture and other incompatible substances while maintaining structural integrity under various operating conditions.
- Material compatibility and corrosion resistance: Selection of appropriate materials that can withstand exposure to chlorine trifluoride is critical for safety. Certain metals, alloys, and coatings demonstrate superior resistance to the highly oxidizing and corrosive properties of this compound. Surface treatments and passivation techniques are employed to create protective layers that prevent degradation. Material testing protocols ensure long-term compatibility and structural stability in chlorine trifluoride service environments.
- Handling and transfer equipment safety features: Specialized equipment for transferring and handling chlorine trifluoride incorporates multiple safety features including automated shut-off valves, pressure monitoring systems, and emergency containment protocols. The equipment design minimizes human exposure and includes fail-safe mechanisms to prevent accidental releases. Transfer systems utilize inert atmospheres and are constructed with materials that maintain integrity during normal and emergency operations.
- Detection and monitoring systems: Advanced detection systems continuously monitor for chlorine trifluoride leaks and concentration levels in work environments. These systems employ various sensing technologies to provide early warning of potential hazards. Real-time monitoring capabilities enable rapid response to abnormal conditions, while data logging functions support safety compliance and incident analysis. Integration with alarm systems ensures immediate notification of personnel in case of detected releases.
- Emergency response and neutralization methods: Comprehensive emergency response protocols address potential chlorine trifluoride incidents through specialized neutralization agents and containment procedures. Emergency equipment includes appropriate personal protective gear, neutralizing materials, and spill containment systems designed specifically for reactive fluorine compounds. Response procedures incorporate isolation techniques, ventilation strategies, and decontamination processes to minimize exposure risks and environmental impact during emergency situations.
02 Material compatibility and corrosion resistance
Selection of appropriate materials that can resist the highly corrosive nature of chlorine trifluoride is critical for safety. Research focuses on identifying and testing materials that maintain structural integrity when exposed to this compound. This includes evaluation of various metal alloys, ceramics, and polymer coatings that demonstrate resistance to fluorination and oxidation reactions. Surface treatment methods and protective coatings are developed to enhance material durability and prevent degradation during handling and processing operations.Expand Specific Solutions03 Safety monitoring and detection systems
Advanced monitoring systems are implemented to detect leaks, measure concentration levels, and provide early warning of potential hazards associated with chlorine trifluoride. These systems incorporate gas sensors, pressure monitors, and automated alarm mechanisms that can quickly identify abnormal conditions. Real-time monitoring technologies enable immediate response to safety threats, including automatic shutdown procedures and emergency ventilation activation to protect personnel and facilities.Expand Specific Solutions04 Handling and transfer equipment design
Specialized equipment and procedures are developed for the safe transfer and handling of chlorine trifluoride in industrial applications. This includes design of transfer lines, valves, pumps, and dispensing systems that minimize exposure risks and prevent contamination. Equipment features include double-wall construction, inert gas purging systems, and fail-safe mechanisms that automatically isolate the chemical in case of equipment malfunction. Proper grounding and static discharge prevention measures are integrated into handling systems.Expand Specific Solutions05 Emergency response and neutralization methods
Protocols and systems for emergency response to chlorine trifluoride incidents include neutralization techniques, spill containment procedures, and personnel protection measures. Development of effective neutralizing agents and absorbent materials that can safely react with or contain spills is essential. Emergency equipment such as specialized fire suppression systems, protective barriers, and decontamination facilities are designed specifically for chlorine trifluoride incidents. Training programs and safety procedures ensure personnel are prepared to respond appropriately to various emergency scenarios.Expand Specific Solutions
Major Players in ClF3 Production and Usage
The chlorine trifluoride safety protocols landscape reflects an emerging yet highly specialized sector within fluorine chemistry, characterized by limited but strategically positioned players. The market remains in early development stages, driven primarily by semiconductor manufacturing and specialized chemical applications, with modest current scale but significant growth potential tied to electronics industry expansion. Technology maturity varies considerably across participants: established chemical giants like Honeywell International Technologies, DAIKIN INDUSTRIES, and The Chemours Co. demonstrate advanced handling capabilities and comprehensive safety infrastructure, while Chinese manufacturers including Fujian Deer Technology, Peric Special Gases, and Zhejiang Juhua Technology Center are rapidly developing competencies. Regional research institutes such as Xi'an Modern Chemistry Research Institute and Zhejiang Chemical Industry Research Institute contribute foundational safety research. The competitive landscape indicates consolidation around companies with robust fluorine chemistry expertise, specialized containment systems, and established regulatory compliance frameworks, positioning this as a high-barrier, expertise-intensive market segment requiring substantial investment in safety engineering and materials science capabilities.
Honeywell International Technologies Ltd.
Technical Solution: Honeywell has developed comprehensive safety protocols for handling chlorine trifluoride (ClF3) based on their extensive experience in fluorine chemistry and specialty gas production. Their approach includes multi-layered containment systems utilizing passivated nickel or Monel alloy vessels that resist ClF3's extreme reactivity. The company implements strict moisture control protocols maintaining less than 10 ppm water content in storage environments, as ClF3 reacts violently with water. Their safety framework incorporates automated monitoring systems with real-time leak detection using infrared spectroscopy, emergency shutdown procedures with inert gas purging capabilities, and specialized personal protective equipment including full-face supplied air respirators and fluoropolymer-coated protective suits. Temperature control systems maintain storage below 150°C to prevent thermal decomposition, while pressure relief systems are designed with rupture discs venting to caustic scrubbers for safe neutralization.
Strengths: Extensive industrial gas handling experience, proven track record in fluorine chemistry, integrated monitoring systems. Weaknesses: High implementation costs, requires specialized infrastructure and trained personnel.
Peric Special Gases Co., Ltd.
Technical Solution: Peric Special Gases has developed practical safety protocols for ClF3 handling tailored to specialty gas distribution and semiconductor industry applications. Their approach focuses on small-scale handling safety, utilizing cabinet-based containment systems with integrated ventilation providing minimum 150 air changes per hour. The protocol emphasizes pre-delivery site assessment including verification of compatible materials, adequate ventilation infrastructure, and emergency response capabilities. Peric's safety procedures include cylinder passivation verification before filling, pressure cycling tests to detect micro-leaks, and valve protection systems using nickel diaphragm valves with metal-to-metal seating. Their handling guidelines specify minimum approach distances, mandatory buddy system for all ClF3 operations, and staged emergency response plans coordinated with local hazmat teams. The company provides customer training programs covering ClF3 properties, proper connection procedures using torque specifications to prevent over-tightening damage, and leak response protocols emphasizing evacuation over intervention. Storage protocols require segregation from incompatible materials with minimum 6-meter separation distances and dedicated gas cabinets with seismic restraints rated for local seismic zones.
Strengths: Practical small-scale handling expertise, strong customer training programs, semiconductor industry application knowledge. Weaknesses: Limited large-scale industrial experience, protocols primarily focused on cylinder-based systems rather than bulk handling.
Critical Safety Technologies for ClF3 Management
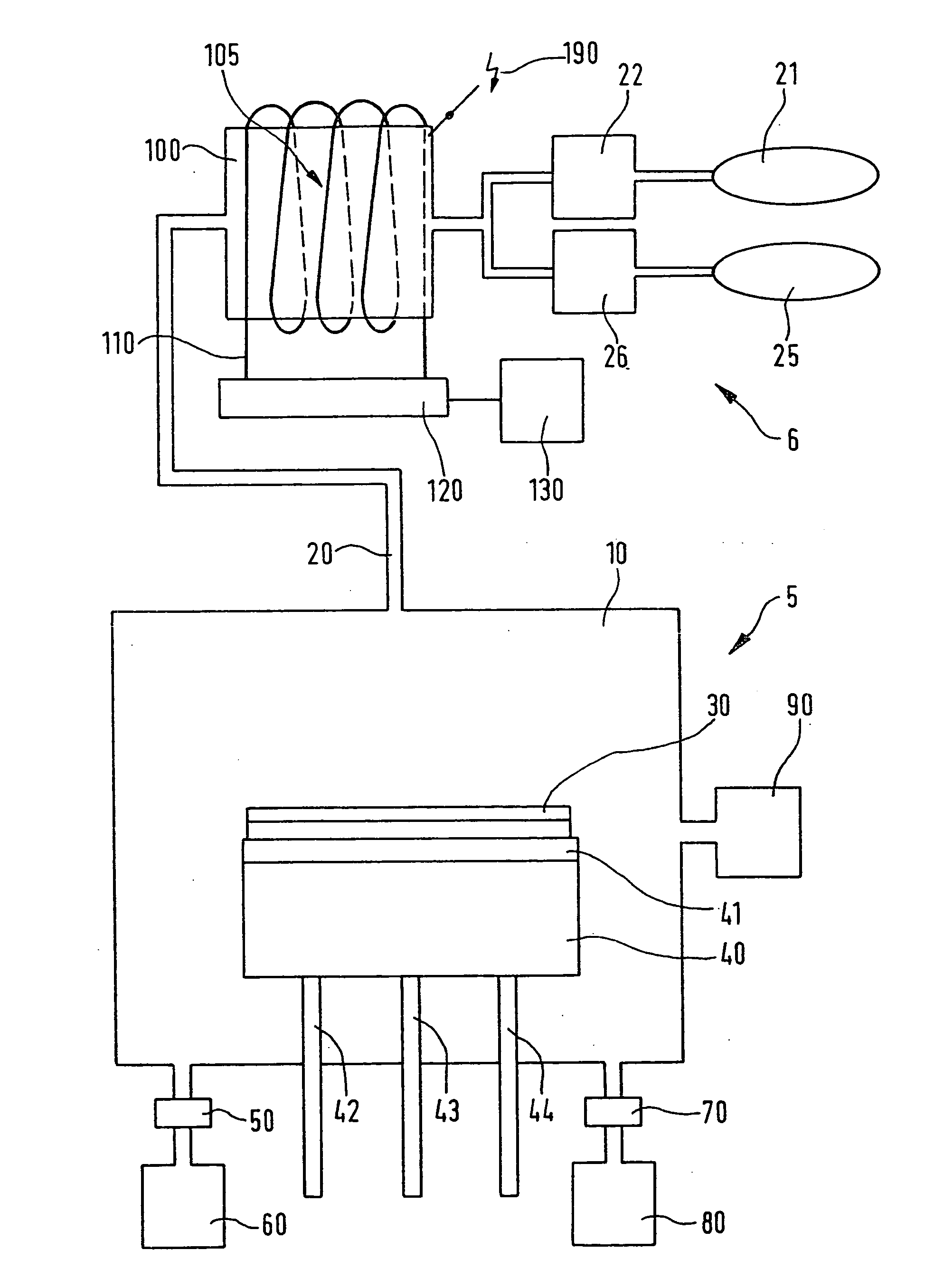
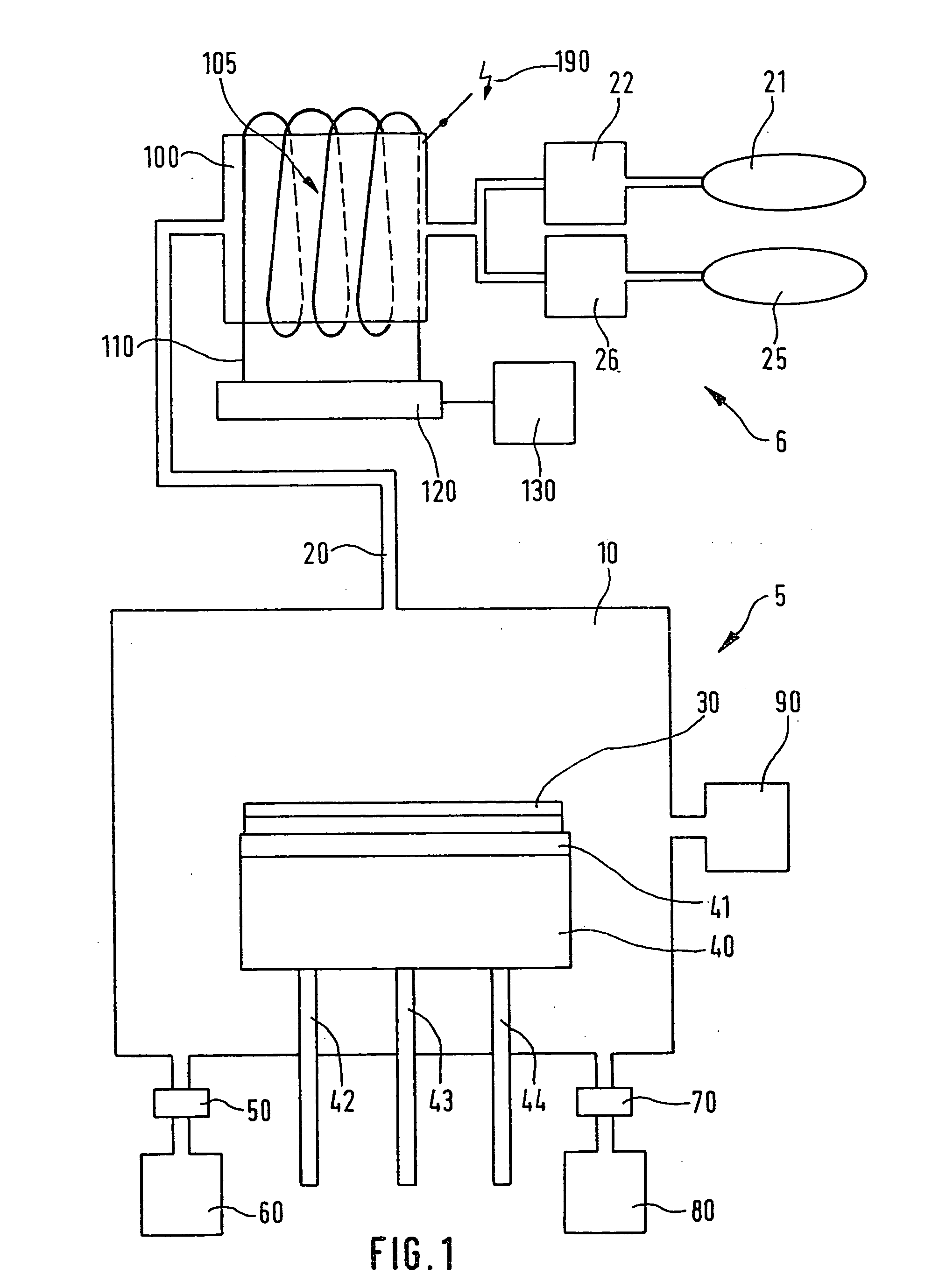
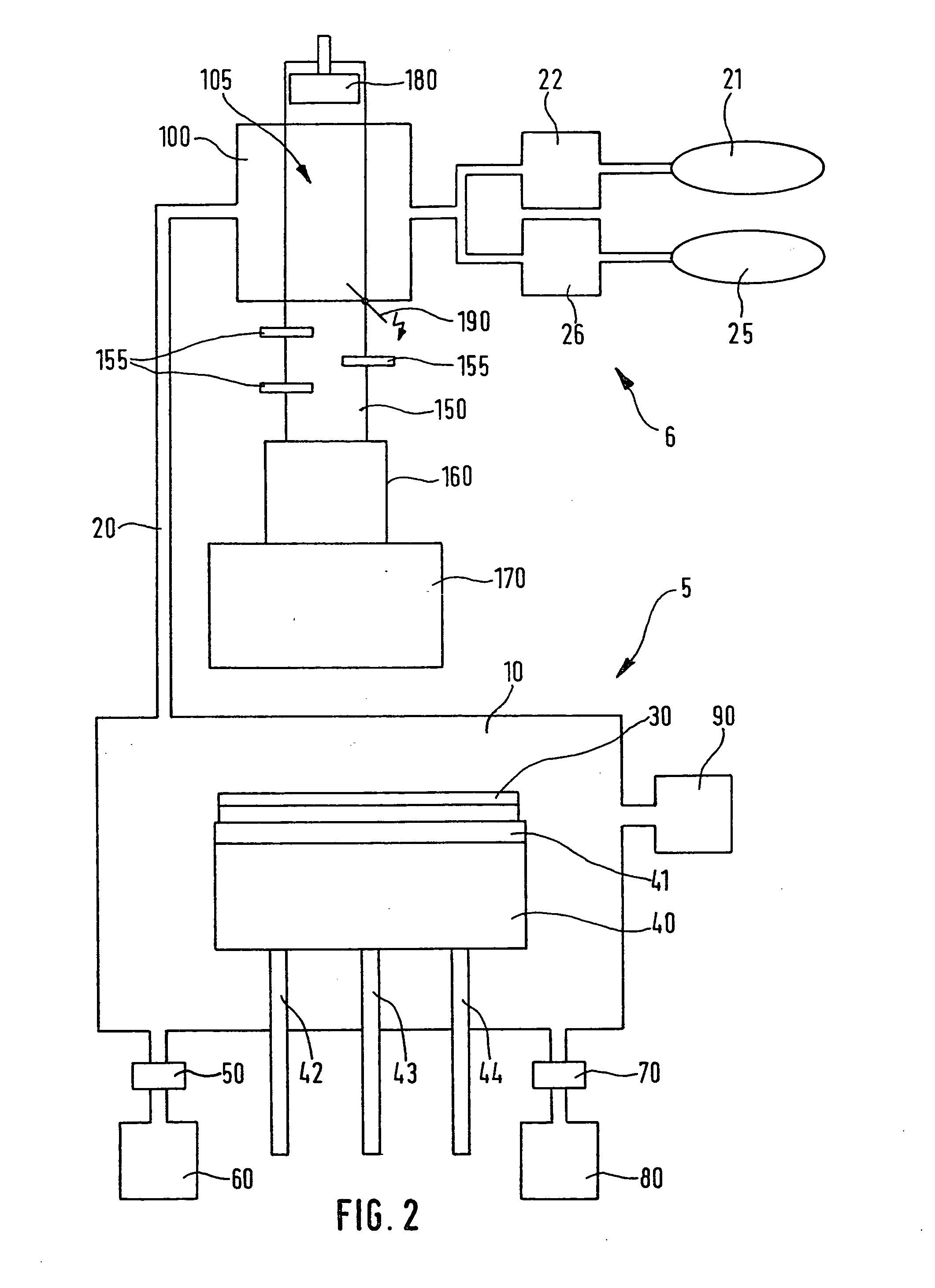
Process for chlorine trifluoride chamber cleaning
PatentInactiveUS5926743A
Innovation
- A method using diluted chlorine trifluoride (ClF3) with a differential etch process, where areas with heavy deposits are heated to a higher temperature than areas with lighter deposits, and a nonreactive gas is used to protect delicate areas from overetching, allowing for efficient cleaning without PFCs.
Device and method for the production of chlorotriflouride and system for etching semiconductor substrates using said device
PatentInactiveUS20060006057A1
Innovation
- A device using a plasma reactor to generate chlorine trifluoride from harmless precursor gases, such as SF6, NF3, or F2, under high-density plasma conditions, allowing for direct on-site production and efficient etching of semiconductor substrates without the need for hazardous material storage.
Regulatory Framework for Hazardous Fluorinating Agents
The regulatory landscape governing chlorine trifluoride (ClF3) and similar hazardous fluorinating agents is characterized by multi-layered frameworks spanning international conventions, national legislation, and industry-specific standards. At the international level, the handling and transportation of ClF3 fall under the United Nations Recommendations on the Transport of Dangerous Goods, which classifies it as a Class 2.3 toxic gas and Class 8 corrosive substance. The International Maritime Dangerous Goods Code and the International Air Transport Association regulations further impose stringent packaging, labeling, and documentation requirements for cross-border movement of such materials.
Within the United States, the Occupational Safety and Health Administration establishes permissible exposure limits and mandates comprehensive hazard communication programs under the Hazard Communication Standard. The Environmental Protection Agency regulates ClF3 under the Clean Air Act amendments, particularly concerning accidental release prevention through Risk Management Plans for facilities storing threshold quantities. The Department of Transportation enforces specific packaging standards under Title 49 of the Code of Federal Regulations, requiring specialized containers capable of withstanding the extreme reactivity of fluorinating agents.
European Union member states adhere to the Registration, Evaluation, Authorization and Restriction of Chemicals regulation, which demands extensive safety data and exposure assessments for ClF3 usage. The Classification, Labeling and Packaging regulation harmonizes hazard communication across member states, ensuring consistent warning systems. Additionally, the Seveso III Directive mandates safety management systems for establishments handling dangerous substances above specified thresholds.
Industry-specific guidelines complement governmental regulations, with organizations such as the Compressed Gas Association publishing technical bulletins addressing material compatibility, valve selection, and emergency response procedures. The American National Standards Institute coordinates voluntary consensus standards for fluorine handling systems, while the National Fire Protection Association provides codes addressing storage facility design and fire suppression systems tailored to reactive fluorinating agents. These regulatory instruments collectively establish a comprehensive compliance framework that facilities must navigate to ensure lawful and safe operations involving chlorine trifluoride.
Within the United States, the Occupational Safety and Health Administration establishes permissible exposure limits and mandates comprehensive hazard communication programs under the Hazard Communication Standard. The Environmental Protection Agency regulates ClF3 under the Clean Air Act amendments, particularly concerning accidental release prevention through Risk Management Plans for facilities storing threshold quantities. The Department of Transportation enforces specific packaging standards under Title 49 of the Code of Federal Regulations, requiring specialized containers capable of withstanding the extreme reactivity of fluorinating agents.
European Union member states adhere to the Registration, Evaluation, Authorization and Restriction of Chemicals regulation, which demands extensive safety data and exposure assessments for ClF3 usage. The Classification, Labeling and Packaging regulation harmonizes hazard communication across member states, ensuring consistent warning systems. Additionally, the Seveso III Directive mandates safety management systems for establishments handling dangerous substances above specified thresholds.
Industry-specific guidelines complement governmental regulations, with organizations such as the Compressed Gas Association publishing technical bulletins addressing material compatibility, valve selection, and emergency response procedures. The American National Standards Institute coordinates voluntary consensus standards for fluorine handling systems, while the National Fire Protection Association provides codes addressing storage facility design and fire suppression systems tailored to reactive fluorinating agents. These regulatory instruments collectively establish a comprehensive compliance framework that facilities must navigate to ensure lawful and safe operations involving chlorine trifluoride.
Emergency Response Systems for ClF3 Incidents
Emergency response systems for chlorine trifluoride incidents require specialized infrastructure and protocols due to the compound's extreme reactivity and hazardous nature. Effective response frameworks must integrate detection capabilities, containment strategies, and coordinated action plans that account for ClF3's unique chemical properties, including its ability to ignite concrete, sand, and water. The establishment of comprehensive emergency systems represents a critical component of safe ClF3 handling operations.
Detection and alarm systems form the first line of defense in ClF3 emergency response. Advanced gas detection sensors capable of identifying ClF3 leaks at trace concentrations must be strategically positioned throughout storage and handling areas. These systems typically employ infrared spectroscopy or electrochemical sensors with response times under three seconds. Integration with automated shutdown mechanisms ensures immediate isolation of affected areas, while multi-stage alarm protocols alert personnel at escalating threat levels. Redundant detection arrays prevent single-point failures that could delay critical response actions.
Containment infrastructure specifically designed for ClF3 incidents includes specialized suppression systems that avoid conventional water-based approaches. Dry chemical systems utilizing materials such as sodium chloride or specialized fluoride-absorbing compounds provide effective initial response options. Secondary containment vessels constructed from nickel alloys or passivated steel prevent environmental release during transfer operations. Remote-operated isolation valves enable rapid system segmentation without requiring personnel exposure to hazardous atmospheres.
Personnel protection and evacuation protocols constitute essential elements of emergency response planning. Self-contained breathing apparatus rated for highly corrosive atmospheres must be readily accessible, with personnel trained in donning procedures achievable within sixty seconds. Designated evacuation routes account for potential contamination zones and prevailing wind patterns. Decontamination stations equipped with appropriate neutralizing agents and medical supplies support safe personnel recovery following exposure incidents.
Coordination with external emergency services requires specialized training programs that familiarize first responders with ClF3 hazards. Pre-incident planning documents provide critical information regarding facility layout, chemical inventories, and recommended response tactics. Mutual aid agreements with specialized hazardous materials teams ensure access to expertise and equipment beyond on-site capabilities. Regular joint exercises validate response procedures and identify improvement opportunities before actual incidents occur.
Detection and alarm systems form the first line of defense in ClF3 emergency response. Advanced gas detection sensors capable of identifying ClF3 leaks at trace concentrations must be strategically positioned throughout storage and handling areas. These systems typically employ infrared spectroscopy or electrochemical sensors with response times under three seconds. Integration with automated shutdown mechanisms ensures immediate isolation of affected areas, while multi-stage alarm protocols alert personnel at escalating threat levels. Redundant detection arrays prevent single-point failures that could delay critical response actions.
Containment infrastructure specifically designed for ClF3 incidents includes specialized suppression systems that avoid conventional water-based approaches. Dry chemical systems utilizing materials such as sodium chloride or specialized fluoride-absorbing compounds provide effective initial response options. Secondary containment vessels constructed from nickel alloys or passivated steel prevent environmental release during transfer operations. Remote-operated isolation valves enable rapid system segmentation without requiring personnel exposure to hazardous atmospheres.
Personnel protection and evacuation protocols constitute essential elements of emergency response planning. Self-contained breathing apparatus rated for highly corrosive atmospheres must be readily accessible, with personnel trained in donning procedures achievable within sixty seconds. Designated evacuation routes account for potential contamination zones and prevailing wind patterns. Decontamination stations equipped with appropriate neutralizing agents and medical supplies support safe personnel recovery following exposure incidents.
Coordination with external emergency services requires specialized training programs that familiarize first responders with ClF3 hazards. Pre-incident planning documents provide critical information regarding facility layout, chemical inventories, and recommended response tactics. Mutual aid agreements with specialized hazardous materials teams ensure access to expertise and equipment beyond on-site capabilities. Regular joint exercises validate response procedures and identify improvement opportunities before actual incidents occur.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!