Optimize Chlorine Trifluoride for Precision Etching Applications
FEB 13, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
ClF3 Etching Background and Technical Objectives
Chlorine trifluoride (ClF3) has emerged as a critical etchant in semiconductor manufacturing due to its exceptional reactivity and ability to etch silicon-based materials at room temperature. Since its introduction to the microelectronics industry in the 1980s, ClF3 has been recognized for its unique chemical properties that enable highly efficient material removal. Unlike traditional plasma-based etching processes, ClF3 operates through direct chemical reaction mechanisms, offering distinct advantages in specific manufacturing scenarios where conventional methods face limitations.
The evolution of semiconductor device architectures toward increasingly complex three-dimensional structures and sub-nanometer feature sizes has intensified the demand for precision etching technologies. Modern integrated circuits require etching processes that can achieve atomic-level control while maintaining high selectivity between different material layers. ClF3's strong oxidizing nature and spontaneous reactivity with silicon, silicon dioxide, and various metal films position it as a promising candidate for next-generation fabrication processes, particularly in applications involving high-aspect-ratio structures and temperature-sensitive substrates.
However, the inherent challenges associated with ClF3 utilization have historically limited its widespread adoption. The compound's extreme reactivity poses significant safety concerns and material compatibility issues, while its tendency to produce non-volatile reaction byproducts can compromise etch uniformity and surface quality. Additionally, achieving precise control over etch rates and profiles remains technically demanding, as ClF3 reactions are highly exothermic and can lead to localized thermal effects that degrade dimensional accuracy.
The primary technical objective of this research initiative is to develop optimized ClF3 delivery and process control methodologies that enable precision etching with sub-nanometer dimensional control and enhanced selectivity ratios exceeding 100:1 for critical material interfaces. Specific goals include establishing stable process windows that minimize surface roughness to below 0.5 nanometers RMS, developing real-time monitoring systems for etch depth control, and implementing advanced containment strategies that ensure safe handling while maintaining process reproducibility. Furthermore, the research aims to characterize reaction kinetics under various temperature and pressure conditions to establish predictive models for industrial-scale implementation, ultimately bridging the gap between ClF3's theoretical potential and practical manufacturing requirements.
The evolution of semiconductor device architectures toward increasingly complex three-dimensional structures and sub-nanometer feature sizes has intensified the demand for precision etching technologies. Modern integrated circuits require etching processes that can achieve atomic-level control while maintaining high selectivity between different material layers. ClF3's strong oxidizing nature and spontaneous reactivity with silicon, silicon dioxide, and various metal films position it as a promising candidate for next-generation fabrication processes, particularly in applications involving high-aspect-ratio structures and temperature-sensitive substrates.
However, the inherent challenges associated with ClF3 utilization have historically limited its widespread adoption. The compound's extreme reactivity poses significant safety concerns and material compatibility issues, while its tendency to produce non-volatile reaction byproducts can compromise etch uniformity and surface quality. Additionally, achieving precise control over etch rates and profiles remains technically demanding, as ClF3 reactions are highly exothermic and can lead to localized thermal effects that degrade dimensional accuracy.
The primary technical objective of this research initiative is to develop optimized ClF3 delivery and process control methodologies that enable precision etching with sub-nanometer dimensional control and enhanced selectivity ratios exceeding 100:1 for critical material interfaces. Specific goals include establishing stable process windows that minimize surface roughness to below 0.5 nanometers RMS, developing real-time monitoring systems for etch depth control, and implementing advanced containment strategies that ensure safe handling while maintaining process reproducibility. Furthermore, the research aims to characterize reaction kinetics under various temperature and pressure conditions to establish predictive models for industrial-scale implementation, ultimately bridging the gap between ClF3's theoretical potential and practical manufacturing requirements.
Market Demand for Precision Etching Solutions
The semiconductor manufacturing industry continues to drive substantial demand for advanced precision etching solutions, with chlorine trifluoride emerging as a critical enabler for next-generation device fabrication. As integrated circuit geometries shrink below seven nanometers and three-dimensional architectures become standard, conventional etching chemistries face increasing limitations in achieving the required selectivity, anisotropy, and surface quality. This technological transition creates urgent market requirements for optimized etching agents capable of handling complex material stacks including high-k dielectrics, metal gates, and advanced interconnect structures.
The proliferation of emerging applications beyond traditional computing amplifies market demand significantly. Power electronics manufacturers require precise etching for wide-bandgap semiconductors such as silicon carbide and gallium nitride, where chlorine trifluoride demonstrates superior performance compared to alternative chemistries. The photonics sector demands ultra-smooth etched surfaces for waveguide fabrication and optical component integration, while MEMS and sensor manufacturers seek controllable etching processes for intricate three-dimensional structures with high aspect ratios.
Market pressure intensifies from the expanding adoption of advanced packaging technologies including through-silicon vias, fan-out wafer-level packaging, and chiplet integration. These approaches necessitate precision etching across diverse substrate materials with stringent dimensional control and minimal damage to underlying layers. The automotive electronics sector particularly drives demand growth, as electrification and autonomous driving systems require robust semiconductor components manufactured with reliable precision etching processes.
Environmental regulations and sustainability initiatives reshape market requirements, pushing manufacturers toward etching solutions that balance performance with reduced environmental impact. Optimized chlorine trifluoride formulations addressing safety concerns, waste reduction, and process efficiency gain competitive advantage. Additionally, the geopolitical emphasis on semiconductor supply chain resilience stimulates regional capacity expansion, creating distributed demand for advanced etching technologies across multiple geographic markets. This diversification pattern generates opportunities for tailored solutions addressing specific regional manufacturing capabilities and application focuses.
The proliferation of emerging applications beyond traditional computing amplifies market demand significantly. Power electronics manufacturers require precise etching for wide-bandgap semiconductors such as silicon carbide and gallium nitride, where chlorine trifluoride demonstrates superior performance compared to alternative chemistries. The photonics sector demands ultra-smooth etched surfaces for waveguide fabrication and optical component integration, while MEMS and sensor manufacturers seek controllable etching processes for intricate three-dimensional structures with high aspect ratios.
Market pressure intensifies from the expanding adoption of advanced packaging technologies including through-silicon vias, fan-out wafer-level packaging, and chiplet integration. These approaches necessitate precision etching across diverse substrate materials with stringent dimensional control and minimal damage to underlying layers. The automotive electronics sector particularly drives demand growth, as electrification and autonomous driving systems require robust semiconductor components manufactured with reliable precision etching processes.
Environmental regulations and sustainability initiatives reshape market requirements, pushing manufacturers toward etching solutions that balance performance with reduced environmental impact. Optimized chlorine trifluoride formulations addressing safety concerns, waste reduction, and process efficiency gain competitive advantage. Additionally, the geopolitical emphasis on semiconductor supply chain resilience stimulates regional capacity expansion, creating distributed demand for advanced etching technologies across multiple geographic markets. This diversification pattern generates opportunities for tailored solutions addressing specific regional manufacturing capabilities and application focuses.
Current ClF3 Etching Challenges and Constraints
Chlorine trifluoride (ClF3) has emerged as a promising etchant for advanced semiconductor manufacturing due to its exceptional reactivity and ability to etch materials that resist conventional fluorine-based chemistries. However, its implementation in precision etching applications faces significant technical barriers that currently limit widespread industrial adoption. The primary challenge stems from ClF3's extreme reactivity, which makes controlled etching exceptionally difficult to achieve consistently across nanoscale features.
Process control represents the most critical constraint in ClF3 etching operations. The compound's hypergolic nature and violent reactions with most materials create substantial difficulties in maintaining stable process conditions. Temperature management becomes particularly problematic, as even minor fluctuations can trigger runaway reactions or cause dramatic variations in etch rates. This sensitivity necessitates sophisticated thermal control systems that add complexity and cost to manufacturing equipment.
Material compatibility issues pose another fundamental constraint. ClF3 aggressively attacks most conventional chamber materials, including many metals and ceramics typically used in semiconductor processing equipment. This reactivity severely limits the selection of suitable reactor components and necessitates expensive specialized materials such as nickel alloys or passivated surfaces. The degradation of chamber components over time leads to particle generation and contamination risks that compromise yield in high-volume manufacturing environments.
Selectivity control remains inadequately developed for ClF3-based processes. While the etchant demonstrates capability across diverse materials, achieving the precise selectivity ratios required for modern multi-layer device structures proves challenging. The lack of established inhibitor chemistries or surface passivation techniques comparable to those available for plasma-based etching limits pattern fidelity and dimensional control. This deficiency becomes increasingly problematic as feature sizes shrink below ten nanometers.
Safety and handling constraints significantly impact operational feasibility. ClF3's toxicity, corrosiveness, and spontaneous ignition characteristics with organic materials demand extraordinary safety protocols and specialized infrastructure. The requirements for emergency response systems, specialized storage facilities, and extensive operator training create substantial barriers to adoption, particularly for facilities not previously equipped for handling such hazardous materials. Environmental regulations regarding ClF3 emissions and waste disposal further complicate implementation strategies.
Process control represents the most critical constraint in ClF3 etching operations. The compound's hypergolic nature and violent reactions with most materials create substantial difficulties in maintaining stable process conditions. Temperature management becomes particularly problematic, as even minor fluctuations can trigger runaway reactions or cause dramatic variations in etch rates. This sensitivity necessitates sophisticated thermal control systems that add complexity and cost to manufacturing equipment.
Material compatibility issues pose another fundamental constraint. ClF3 aggressively attacks most conventional chamber materials, including many metals and ceramics typically used in semiconductor processing equipment. This reactivity severely limits the selection of suitable reactor components and necessitates expensive specialized materials such as nickel alloys or passivated surfaces. The degradation of chamber components over time leads to particle generation and contamination risks that compromise yield in high-volume manufacturing environments.
Selectivity control remains inadequately developed for ClF3-based processes. While the etchant demonstrates capability across diverse materials, achieving the precise selectivity ratios required for modern multi-layer device structures proves challenging. The lack of established inhibitor chemistries or surface passivation techniques comparable to those available for plasma-based etching limits pattern fidelity and dimensional control. This deficiency becomes increasingly problematic as feature sizes shrink below ten nanometers.
Safety and handling constraints significantly impact operational feasibility. ClF3's toxicity, corrosiveness, and spontaneous ignition characteristics with organic materials demand extraordinary safety protocols and specialized infrastructure. The requirements for emergency response systems, specialized storage facilities, and extensive operator training create substantial barriers to adoption, particularly for facilities not previously equipped for handling such hazardous materials. Environmental regulations regarding ClF3 emissions and waste disposal further complicate implementation strategies.
Mainstream ClF3 Etching Process Solutions
01 Precision manufacturing equipment for chlorine trifluoride production
Specialized manufacturing equipment and apparatus designed for the precise production and handling of chlorine trifluoride. These systems incorporate advanced control mechanisms, precision valves, and monitoring devices to ensure accurate production parameters and safe handling of this highly reactive chemical compound.- Precision manufacturing equipment for chlorine trifluoride production: Specialized manufacturing equipment and apparatus designed for the precise production and handling of chlorine trifluoride. These systems incorporate advanced control mechanisms, precision valves, and monitoring devices to ensure accurate production parameters and safe handling of this highly reactive compound. The equipment includes precision flow control systems, temperature regulation devices, and automated production lines.
- Precision purification and separation methods: Advanced purification and separation techniques for obtaining high-purity chlorine trifluoride. These methods involve precision distillation processes, selective absorption techniques, and multi-stage purification systems that can achieve extremely high purity levels. The processes utilize precise temperature and pressure control to separate chlorine trifluoride from impurities and byproducts.
- Precision measurement and detection systems: Sophisticated measurement and detection systems for monitoring chlorine trifluoride concentration, purity, and process parameters. These systems employ advanced sensors, analytical instruments, and real-time monitoring technologies to provide precise measurements of chemical composition, flow rates, and environmental conditions. The detection methods ensure accurate quality control throughout production and application processes.
- Precision storage and transportation containers: Specialized containers and storage systems designed for the safe and precise handling of chlorine trifluoride. These containers feature advanced sealing mechanisms, corrosion-resistant materials, and precision pressure control systems. The designs incorporate safety features, leak detection systems, and materials specifically selected to withstand the highly reactive nature of the compound while maintaining precise environmental conditions.
- Precision application and etching processes: Precision application methods for using chlorine trifluoride in semiconductor manufacturing, surface treatment, and etching processes. These techniques involve controlled delivery systems, precise dosing mechanisms, and automated application processes that ensure uniform treatment and accurate material removal. The methods incorporate real-time monitoring and feedback control to achieve precise etching depths and surface characteristics.
02 Precision etching and cleaning processes using chlorine trifluoride
Methods and systems for utilizing chlorine trifluoride in precision etching and cleaning applications, particularly in semiconductor manufacturing. These processes leverage the highly reactive nature of the compound to achieve precise material removal and surface cleaning with controlled reaction rates and uniform results.Expand Specific Solutions03 Precision flow control and delivery systems
Advanced flow control systems and delivery mechanisms designed to handle chlorine trifluoride with high precision. These systems include precision flow meters, pressure regulators, and automated delivery apparatus that ensure accurate dosing and controlled distribution of the chemical in various industrial applications.Expand Specific Solutions04 Precision measurement and detection devices
Specialized measurement instruments and detection devices for monitoring chlorine trifluoride concentration, purity, and process parameters with high precision. These devices employ advanced sensing technologies and calibration methods to provide accurate real-time data for process control and quality assurance.Expand Specific Solutions05 Precision storage and containment systems
Engineered storage vessels and containment systems specifically designed for the safe and precise storage of chlorine trifluoride. These systems incorporate specialized materials, pressure control mechanisms, and safety features to maintain product integrity and ensure precise inventory management while preventing contamination and degradation.Expand Specific Solutions
Core Patents in ClF3 Precision Etching
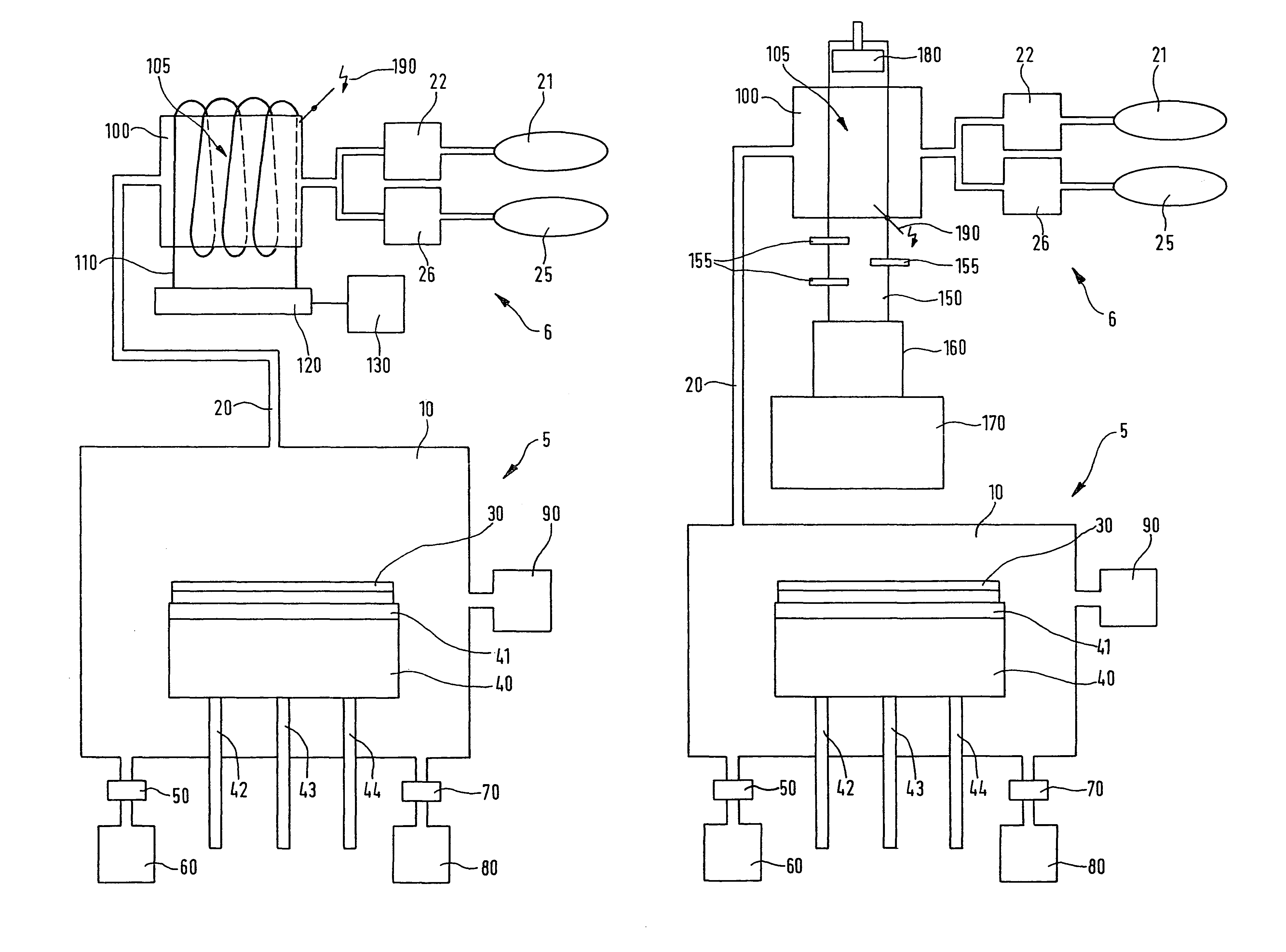
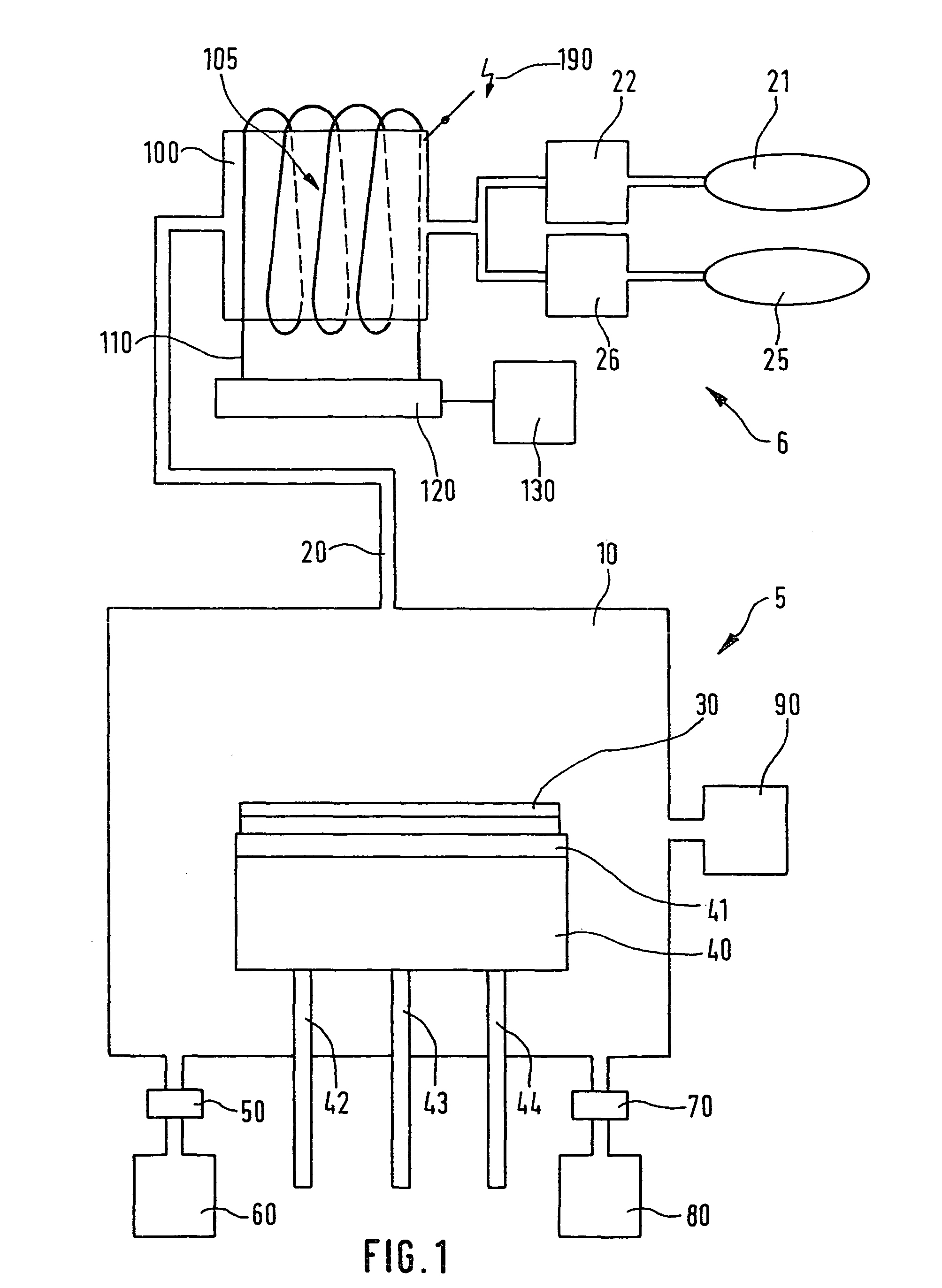
Method for etching a layer of a silicon semiconductor substrate
PatentWO2009010391A1
Innovation
- The method involves using an etching gas mixture that includes chlorine trifluoride (ClF3) with additional chlorine (Cl2) or hydrogen chloride (HCl), which enhances the selectivity of SiGe etching over silicon, allowing for reduced etching of silicon while maintaining or increasing the etching rate of SiGe, thereby enabling the production of thinner and more critically dimensioned silicon structures without damaging functional silicon layers.
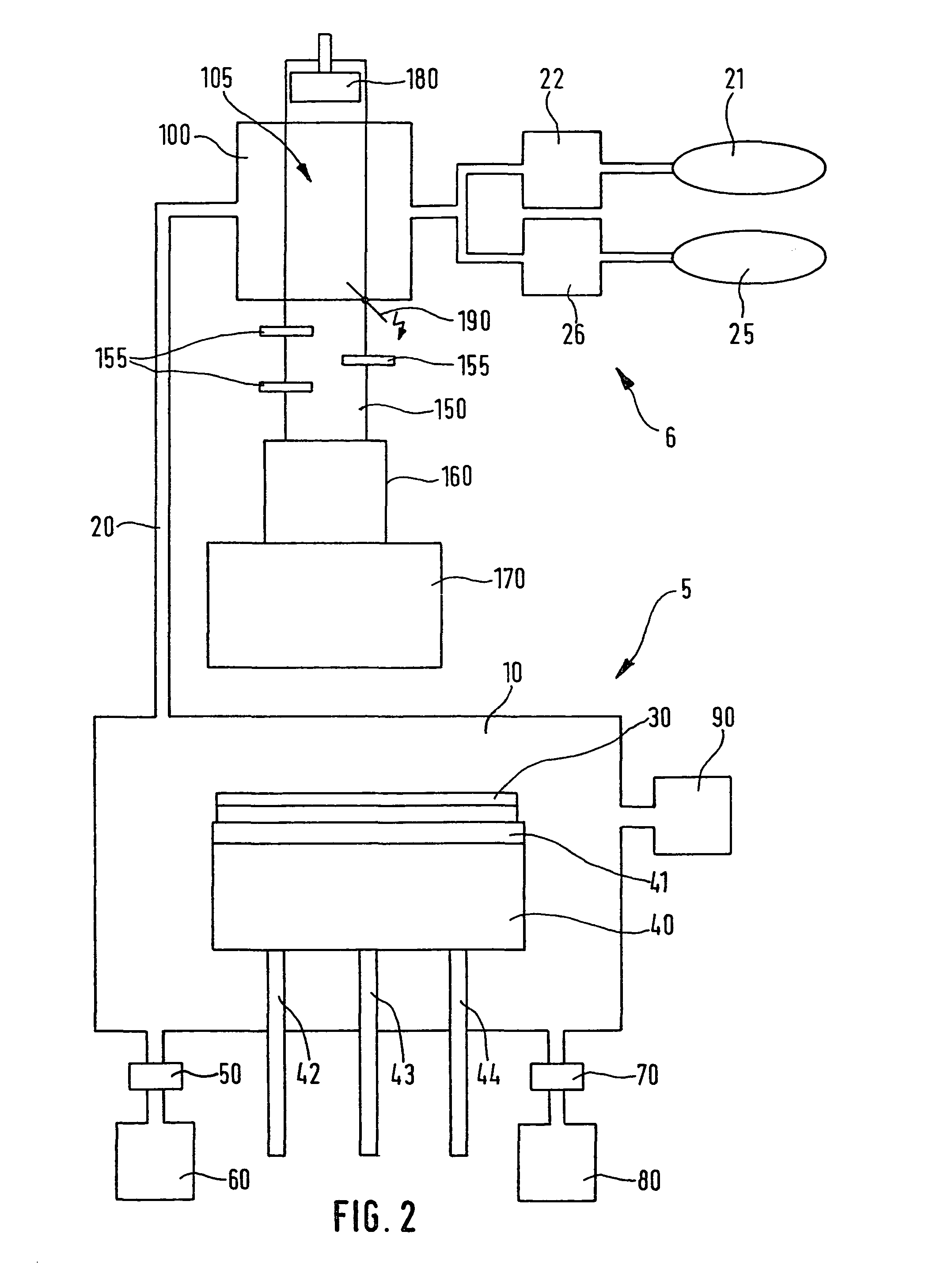
Device and method for producing chlorine trifluoride and system for etching semiconductor substrates using this device
PatentInactiveUS8382940B2
Innovation
- A device using a plasma reactor to generate chlorine trifluoride from harmless precursor gases, such as F2, Cl2, SF6, NF3, or HCl, through high-density plasma reactions, allowing for on-site production and direct use in etching processes, thereby avoiding the need for hazardous stockpiling and improving efficiency.
Safety and Environmental Regulations for ClF3
Chlorine trifluoride (ClF3) is classified as a highly hazardous substance under multiple international regulatory frameworks due to its extreme reactivity, corrosiveness, and toxicity. In the United States, the Occupational Safety and Health Administration (OSHA) mandates strict permissible exposure limits (PEL) of 0.1 ppm as a ceiling value for workplace environments. The Environmental Protection Agency (EPA) categorizes ClF3 under the Emergency Planning and Community Right-to-Know Act (EPCRA), requiring facilities that store or use quantities exceeding threshold planning quantities to submit detailed emergency response plans and maintain comprehensive safety data sheets accessible to local authorities and emergency responders.
European Union regulations under REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) impose stringent registration requirements for ClF3 manufacturers and importers, demanding extensive toxicological and ecotoxicological data. The substance is listed under Annex XVII restrictions, limiting its use to industrial applications with demonstrated containment capabilities. Transportation of ClF3 falls under UN Class 2.3 (toxic gases) and Class 8 (corrosive substances) classifications, requiring specialized containers meeting DOT and IMDG Code specifications, along with mandatory hazard communication protocols during transit.
Workplace safety protocols necessitate engineering controls including closed-loop handling systems, automated dispensing equipment, and continuous atmospheric monitoring with immediate alarm systems. Personal protective equipment requirements extend beyond standard chemical handling gear to include supplied-air respirators, chemical-resistant suits rated for hydrofluoric acid exposure, and emergency shower stations with specialized neutralizing agents. Facilities must implement rigorous training programs covering emergency response procedures, leak detection protocols, and proper disposal methods for contaminated materials.
Environmental discharge regulations prohibit direct release of ClF3 or its reaction products into air, water, or soil. Waste streams containing ClF3 residues must undergo neutralization through controlled hydrolysis or reaction with appropriate scrubbing systems before disposal. The semiconductor industry has developed industry-specific guidelines through SEMI Standards, establishing best practices for point-of-use abatement systems and real-time emission monitoring to ensure compliance with local air quality regulations and minimize environmental impact from precision etching operations.
European Union regulations under REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) impose stringent registration requirements for ClF3 manufacturers and importers, demanding extensive toxicological and ecotoxicological data. The substance is listed under Annex XVII restrictions, limiting its use to industrial applications with demonstrated containment capabilities. Transportation of ClF3 falls under UN Class 2.3 (toxic gases) and Class 8 (corrosive substances) classifications, requiring specialized containers meeting DOT and IMDG Code specifications, along with mandatory hazard communication protocols during transit.
Workplace safety protocols necessitate engineering controls including closed-loop handling systems, automated dispensing equipment, and continuous atmospheric monitoring with immediate alarm systems. Personal protective equipment requirements extend beyond standard chemical handling gear to include supplied-air respirators, chemical-resistant suits rated for hydrofluoric acid exposure, and emergency shower stations with specialized neutralizing agents. Facilities must implement rigorous training programs covering emergency response procedures, leak detection protocols, and proper disposal methods for contaminated materials.
Environmental discharge regulations prohibit direct release of ClF3 or its reaction products into air, water, or soil. Waste streams containing ClF3 residues must undergo neutralization through controlled hydrolysis or reaction with appropriate scrubbing systems before disposal. The semiconductor industry has developed industry-specific guidelines through SEMI Standards, establishing best practices for point-of-use abatement systems and real-time emission monitoring to ensure compliance with local air quality regulations and minimize environmental impact from precision etching operations.
Process Control and Selectivity Enhancement Methods
Effective process control in chlorine trifluoride etching requires sophisticated real-time monitoring systems that track critical parameters including chamber pressure, gas flow rates, substrate temperature, and reaction byproduct concentrations. Advanced optical emission spectroscopy and mass spectrometry enable precise detection of etching endpoints and process drift, allowing immediate corrective adjustments. Temperature management proves particularly crucial, as ClF3 reactivity exhibits strong thermal dependence; maintaining substrate temperatures within narrow windows of 150-300°C through closed-loop heating systems ensures consistent etch rates while preventing thermal damage to underlying layers.
Selectivity enhancement represents a fundamental challenge given ClF3's aggressive nature toward most materials. Strategic implementation of plasma-assisted etching techniques allows modulation of radical species concentrations, enabling preferential attack on target materials while reducing damage to masking layers and adjacent structures. Pulsed delivery methods, alternating between etching and passivation cycles, demonstrate significant improvements in selectivity ratios by allowing controlled surface chemistry modification between active etching phases.
Chemical additives serve as effective selectivity modulators, with nitrogen trifluoride and sulfur hexafluoride showing promise as diluents that reduce overall reactivity while maintaining directional etching characteristics. Surface passivation strategies employing fluorocarbon precursors create protective polymer layers on non-target materials, achieving selectivity ratios exceeding 50:1 for silicon dioxide over silicon nitride in optimized conditions.
Pressure regime optimization significantly impacts selectivity outcomes. Operating at reduced pressures below 100 mTorr enhances anisotropic etching profiles through increased mean free path, while moderate pressures of 500-1000 mTorr facilitate chemical selectivity through collision-mediated reaction pathways. Dynamic pressure ramping protocols, adjusting pressure profiles throughout the etch process, enable multi-objective optimization balancing throughput, selectivity, and profile control.
Mask material selection and surface preparation critically influence selectivity performance. Nickel and chromium-based hard masks demonstrate superior resistance to ClF3 attack compared to conventional photoresist materials, enabling deeper etching with maintained pattern fidelity. Pre-treatment protocols involving fluorine-based surface conditioning establish stable interface chemistry that enhances subsequent selectivity during main etching sequences.
Selectivity enhancement represents a fundamental challenge given ClF3's aggressive nature toward most materials. Strategic implementation of plasma-assisted etching techniques allows modulation of radical species concentrations, enabling preferential attack on target materials while reducing damage to masking layers and adjacent structures. Pulsed delivery methods, alternating between etching and passivation cycles, demonstrate significant improvements in selectivity ratios by allowing controlled surface chemistry modification between active etching phases.
Chemical additives serve as effective selectivity modulators, with nitrogen trifluoride and sulfur hexafluoride showing promise as diluents that reduce overall reactivity while maintaining directional etching characteristics. Surface passivation strategies employing fluorocarbon precursors create protective polymer layers on non-target materials, achieving selectivity ratios exceeding 50:1 for silicon dioxide over silicon nitride in optimized conditions.
Pressure regime optimization significantly impacts selectivity outcomes. Operating at reduced pressures below 100 mTorr enhances anisotropic etching profiles through increased mean free path, while moderate pressures of 500-1000 mTorr facilitate chemical selectivity through collision-mediated reaction pathways. Dynamic pressure ramping protocols, adjusting pressure profiles throughout the etch process, enable multi-objective optimization balancing throughput, selectivity, and profile control.
Mask material selection and surface preparation critically influence selectivity performance. Nickel and chromium-based hard masks demonstrate superior resistance to ClF3 attack compared to conventional photoresist materials, enabling deeper etching with maintained pattern fidelity. Pre-treatment protocols involving fluorine-based surface conditioning establish stable interface chemistry that enhances subsequent selectivity during main etching sequences.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!