Calcium-ion battery electrode-electrolyte interphase stability research
SEP 29, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
CIB Interphase Stability Background and Objectives
Calcium-ion batteries (CIBs) have emerged as a promising alternative to lithium-ion batteries due to the abundance of calcium resources, potentially higher energy density, and improved safety characteristics. The electrode-electrolyte interphase (EEI) stability represents a critical factor determining the performance, cycle life, and safety of these energy storage systems. This research aims to comprehensively investigate the formation mechanisms, composition, and stability of interphases in calcium-ion battery systems.
The evolution of battery technology has progressed from lead-acid to nickel-metal hydride, and subsequently to lithium-ion systems that currently dominate the market. However, concerns regarding lithium resource scarcity and geopolitical supply chain vulnerabilities have accelerated research into alternative battery chemistries. Calcium, as the fifth most abundant element in the Earth's crust, presents a sustainable alternative with a theoretical volumetric capacity of 2073 mAh/cm³, nearly matching that of lithium (2062 mAh/cm³).
Historical attempts to develop calcium-based batteries date back to the 1960s but were largely abandoned due to significant challenges in achieving reversible calcium plating and stripping. The primary obstacle has been the formation of unstable, resistive interphase layers between the electrode and electrolyte, which impede calcium ion transport. Recent breakthroughs in electrolyte design have revitalized interest in this field, yet the fundamental understanding of interphase formation and stability remains inadequate.
The technical trajectory shows an accelerating research interest, with publications on calcium-ion batteries increasing exponentially since 2015. Particularly, studies focusing on the electrode-electrolyte interface have doubled every two years, highlighting the recognition of interphase stability as a critical bottleneck for commercialization.
This research aims to elucidate the chemical and electrochemical processes governing the formation, evolution, and degradation of the calcium-ion battery interphase. Specific objectives include: characterizing the composition and morphology of interphase layers formed with various electrode-electrolyte combinations; identifying reaction mechanisms and kinetics of interphase formation; evaluating the impact of operating conditions on interphase stability; and developing strategies to engineer stable and ion-conductive interphases.
By addressing these objectives, we seek to establish design principles for calcium-ion battery systems with enhanced cycling stability, rate capability, and calendar life. The ultimate goal is to provide a scientific foundation for the development of commercially viable calcium-ion batteries that can complement or potentially replace current lithium-ion technology in specific applications, particularly in large-scale energy storage where cost and safety considerations are paramount.
The evolution of battery technology has progressed from lead-acid to nickel-metal hydride, and subsequently to lithium-ion systems that currently dominate the market. However, concerns regarding lithium resource scarcity and geopolitical supply chain vulnerabilities have accelerated research into alternative battery chemistries. Calcium, as the fifth most abundant element in the Earth's crust, presents a sustainable alternative with a theoretical volumetric capacity of 2073 mAh/cm³, nearly matching that of lithium (2062 mAh/cm³).
Historical attempts to develop calcium-based batteries date back to the 1960s but were largely abandoned due to significant challenges in achieving reversible calcium plating and stripping. The primary obstacle has been the formation of unstable, resistive interphase layers between the electrode and electrolyte, which impede calcium ion transport. Recent breakthroughs in electrolyte design have revitalized interest in this field, yet the fundamental understanding of interphase formation and stability remains inadequate.
The technical trajectory shows an accelerating research interest, with publications on calcium-ion batteries increasing exponentially since 2015. Particularly, studies focusing on the electrode-electrolyte interface have doubled every two years, highlighting the recognition of interphase stability as a critical bottleneck for commercialization.
This research aims to elucidate the chemical and electrochemical processes governing the formation, evolution, and degradation of the calcium-ion battery interphase. Specific objectives include: characterizing the composition and morphology of interphase layers formed with various electrode-electrolyte combinations; identifying reaction mechanisms and kinetics of interphase formation; evaluating the impact of operating conditions on interphase stability; and developing strategies to engineer stable and ion-conductive interphases.
By addressing these objectives, we seek to establish design principles for calcium-ion battery systems with enhanced cycling stability, rate capability, and calendar life. The ultimate goal is to provide a scientific foundation for the development of commercially viable calcium-ion batteries that can complement or potentially replace current lithium-ion technology in specific applications, particularly in large-scale energy storage where cost and safety considerations are paramount.
Market Analysis for Calcium-ion Battery Technologies
The global market for calcium-ion battery technologies is experiencing significant growth potential as the world shifts towards sustainable energy solutions. Current market projections indicate that the energy storage market, which calcium-ion batteries aim to penetrate, is expected to reach $546 billion by 2035, with a compound annual growth rate of approximately 20% between 2023 and 2035. Within this broader market, calcium-ion batteries represent an emerging segment with distinctive competitive advantages over existing lithium-ion and other post-lithium technologies.
Calcium-ion batteries offer several market advantages driving potential adoption. The abundance of calcium in the Earth's crust (fifth most abundant element) provides substantial raw material cost benefits compared to lithium, with calcium costing approximately $2,000 per ton versus lithium at $20,000 per ton as of late 2023. This significant price differential creates a compelling economic case for manufacturers seeking alternatives to lithium-based systems.
Market segmentation analysis reveals that calcium-ion batteries with stable electrode-electrolyte interphases could initially target stationary energy storage applications, where energy density requirements are less stringent than in electric vehicles. This segment is projected to grow at 25% annually through 2030, creating an estimated $40 billion opportunity for new battery technologies by 2028.
Regional market analysis shows varying adoption potential, with Europe leading research initiatives through programs like Horizon Europe and dedicated calcium battery consortiums. Asia-Pacific, particularly China and Japan, demonstrates increasing patent activity in calcium-ion technology, while North America shows growing venture capital interest in alternative battery chemistries, with investments exceeding $1.5 billion in 2023 alone.
Consumer and industrial demand signals indicate strong market pull for batteries with improved safety profiles and reduced environmental impact. Survey data from major energy storage customers reveals that 78% of utility-scale operators consider safety and sustainability as "very important" factors in battery selection, creating a favorable market environment for calcium-ion technologies.
Competitive landscape assessment positions calcium-ion batteries against other emerging technologies including sodium-ion, magnesium-ion, and solid-state batteries. While sodium-ion batteries have achieved earlier commercialization, calcium-ion systems with stable interphases could potentially deliver superior energy density (theoretical maximum of 4.4 V versus 4.0 V for sodium-ion), creating distinct market opportunities if electrode-electrolyte stability challenges are overcome.
Market barriers include the need for significant manufacturing infrastructure development, regulatory approval processes, and customer education about new battery chemistries. However, the potential for calcium-ion batteries to leverage portions of existing lithium-ion manufacturing processes could accelerate market entry timelines and reduce capital requirements for scaled production.
Calcium-ion batteries offer several market advantages driving potential adoption. The abundance of calcium in the Earth's crust (fifth most abundant element) provides substantial raw material cost benefits compared to lithium, with calcium costing approximately $2,000 per ton versus lithium at $20,000 per ton as of late 2023. This significant price differential creates a compelling economic case for manufacturers seeking alternatives to lithium-based systems.
Market segmentation analysis reveals that calcium-ion batteries with stable electrode-electrolyte interphases could initially target stationary energy storage applications, where energy density requirements are less stringent than in electric vehicles. This segment is projected to grow at 25% annually through 2030, creating an estimated $40 billion opportunity for new battery technologies by 2028.
Regional market analysis shows varying adoption potential, with Europe leading research initiatives through programs like Horizon Europe and dedicated calcium battery consortiums. Asia-Pacific, particularly China and Japan, demonstrates increasing patent activity in calcium-ion technology, while North America shows growing venture capital interest in alternative battery chemistries, with investments exceeding $1.5 billion in 2023 alone.
Consumer and industrial demand signals indicate strong market pull for batteries with improved safety profiles and reduced environmental impact. Survey data from major energy storage customers reveals that 78% of utility-scale operators consider safety and sustainability as "very important" factors in battery selection, creating a favorable market environment for calcium-ion technologies.
Competitive landscape assessment positions calcium-ion batteries against other emerging technologies including sodium-ion, magnesium-ion, and solid-state batteries. While sodium-ion batteries have achieved earlier commercialization, calcium-ion systems with stable interphases could potentially deliver superior energy density (theoretical maximum of 4.4 V versus 4.0 V for sodium-ion), creating distinct market opportunities if electrode-electrolyte stability challenges are overcome.
Market barriers include the need for significant manufacturing infrastructure development, regulatory approval processes, and customer education about new battery chemistries. However, the potential for calcium-ion batteries to leverage portions of existing lithium-ion manufacturing processes could accelerate market entry timelines and reduce capital requirements for scaled production.
Current Challenges in CIB Electrode-Electrolyte Interfaces
Despite significant advancements in calcium-ion battery (CIB) technology, the electrode-electrolyte interface remains a critical bottleneck limiting commercial viability. The formation of a stable solid electrolyte interphase (SEI) layer, which is essential for long-term cycling stability, faces substantial challenges in calcium-based systems compared to lithium-ion counterparts. The high charge density of Ca2+ ions (twice that of Li+) creates stronger electrostatic interactions with electrode materials and electrolyte components, resulting in slower diffusion kinetics and more complex interfacial chemistry.
A primary challenge is the high reduction potential of calcium, which causes most conventional electrolytes to decompose before calcium deposition can occur. This decomposition creates unstable and resistive interfacial layers that impede ion transport. Additionally, the divalent nature of calcium ions leads to stronger coordination with solvent molecules, making desolvation at the electrode surface energetically unfavorable and kinetically sluggish.
Calcium metal anodes suffer from dendrite formation and parasitic side reactions with electrolytes, creating unstable interfaces that continuously consume electrolyte and increase cell impedance. Unlike lithium systems, where relatively stable passivation layers form naturally, calcium interfaces often develop thick, non-uniform, and highly resistive layers that prevent efficient ion transport.
For cathode materials, the challenges are equally significant. The insertion/extraction of divalent Ca2+ ions causes substantial structural changes in host materials, leading to mechanical stress and degradation of the electrode-electrolyte interface over repeated cycling. The high charge density also results in stronger binding to host structures, creating kinetic barriers for ion migration across interfaces.
Electrolyte decomposition products at both electrodes often lack the necessary ionic conductivity for calcium ions, creating resistive layers that significantly impair battery performance. Conventional electrolyte additives that successfully stabilize interfaces in lithium-ion systems prove largely ineffective for calcium-based systems due to fundamentally different interfacial chemistry.
The limited understanding of calcium interfacial phenomena presents another major obstacle. While lithium-ion battery interfaces have been extensively characterized over decades, calcium interface formation mechanisms, composition, and evolution during cycling remain poorly understood. This knowledge gap hampers the rational design of electrolytes and electrode materials specifically tailored for stable calcium interfaces.
Environmental factors such as moisture and oxygen contamination pose additional challenges, as they react readily with calcium interfaces, forming insulating layers of calcium hydroxide or calcium carbonate that severely impact battery performance. This necessitates stringent manufacturing conditions that add complexity and cost to potential commercial applications.
A primary challenge is the high reduction potential of calcium, which causes most conventional electrolytes to decompose before calcium deposition can occur. This decomposition creates unstable and resistive interfacial layers that impede ion transport. Additionally, the divalent nature of calcium ions leads to stronger coordination with solvent molecules, making desolvation at the electrode surface energetically unfavorable and kinetically sluggish.
Calcium metal anodes suffer from dendrite formation and parasitic side reactions with electrolytes, creating unstable interfaces that continuously consume electrolyte and increase cell impedance. Unlike lithium systems, where relatively stable passivation layers form naturally, calcium interfaces often develop thick, non-uniform, and highly resistive layers that prevent efficient ion transport.
For cathode materials, the challenges are equally significant. The insertion/extraction of divalent Ca2+ ions causes substantial structural changes in host materials, leading to mechanical stress and degradation of the electrode-electrolyte interface over repeated cycling. The high charge density also results in stronger binding to host structures, creating kinetic barriers for ion migration across interfaces.
Electrolyte decomposition products at both electrodes often lack the necessary ionic conductivity for calcium ions, creating resistive layers that significantly impair battery performance. Conventional electrolyte additives that successfully stabilize interfaces in lithium-ion systems prove largely ineffective for calcium-based systems due to fundamentally different interfacial chemistry.
The limited understanding of calcium interfacial phenomena presents another major obstacle. While lithium-ion battery interfaces have been extensively characterized over decades, calcium interface formation mechanisms, composition, and evolution during cycling remain poorly understood. This knowledge gap hampers the rational design of electrolytes and electrode materials specifically tailored for stable calcium interfaces.
Environmental factors such as moisture and oxygen contamination pose additional challenges, as they react readily with calcium interfaces, forming insulating layers of calcium hydroxide or calcium carbonate that severely impact battery performance. This necessitates stringent manufacturing conditions that add complexity and cost to potential commercial applications.
State-of-the-Art Interphase Stabilization Approaches
01 Electrolyte additives for SEI formation and stability
Various electrolyte additives can be incorporated into calcium-ion batteries to form stable solid electrolyte interphase (SEI) layers. These additives help create protective films on electrode surfaces that prevent continuous electrolyte decomposition while allowing calcium ion transport. Properly formulated additives can significantly improve the cycling stability and coulombic efficiency of calcium-ion batteries by forming robust interphase layers that resist degradation during charge-discharge cycles.- Electrolyte additives for SEI stability: Various electrolyte additives can be incorporated to enhance the solid electrolyte interphase (SEI) stability in calcium-ion batteries. These additives form a protective layer on the electrode surface, preventing continuous electrolyte decomposition and calcium deposition issues. Compounds such as fluorinated carbonates, lithium salts, and specific organic molecules can significantly improve the interfacial stability, leading to better cycling performance and reduced capacity fading during battery operation.
- Electrode surface modifications: Surface modifications of electrode materials can substantially improve the interphase stability in calcium-ion batteries. Techniques include coating electrodes with protective layers, surface functionalization with specific chemical groups, and atomic layer deposition of stabilizing compounds. These modifications create a more stable interface between the electrode and electrolyte, reducing unwanted side reactions and improving calcium ion transport across the interphase, which results in enhanced battery performance and longevity.
- Novel electrolyte formulations: Advanced electrolyte formulations play a crucial role in stabilizing the interphase in calcium-ion batteries. These include non-aqueous electrolytes with optimized salt concentrations, ionic liquid-based electrolytes, and hybrid electrolyte systems. The carefully designed electrolyte chemistry helps to form a more stable and conductive interphase layer, facilitating efficient calcium ion transport while minimizing parasitic reactions that can degrade battery performance over repeated charge-discharge cycles.
- Interphase engineering strategies: Deliberate engineering of the electrode-electrolyte interphase can significantly enhance calcium-ion battery stability. This includes pre-forming artificial SEI layers, introducing interphase-stabilizing nanostructures, and employing in-situ interphase modification techniques. These approaches allow for precise control over the composition and properties of the interphase, resulting in improved ionic conductivity, mechanical stability, and chemical resistance against continuous degradation during battery cycling.
- Advanced characterization of interphase phenomena: Sophisticated analytical techniques are being employed to better understand interphase formation and stability mechanisms in calcium-ion batteries. These include in-situ/operando spectroscopy, advanced microscopy, computational modeling, and electrochemical impedance analysis. The insights gained from these characterization methods help identify degradation pathways and stability factors, enabling the rational design of more stable interphases for next-generation calcium-ion battery systems with improved performance and cycle life.
02 Novel electrode materials for improved interphase stability
Advanced electrode materials have been developed specifically for calcium-ion batteries to enhance interphase stability. These materials feature optimized surface properties and structures that promote the formation of stable interfaces with electrolytes. By engineering electrode materials with controlled surface chemistry and morphology, researchers have achieved better compatibility with calcium-ion electrolytes, resulting in more stable solid-electrolyte interphases and improved battery performance over extended cycling.Expand Specific Solutions03 Calcium salt formulations for enhanced interphase properties
Specific calcium salt formulations have been designed to address interphase stability challenges in calcium-ion batteries. These formulations control the chemical composition of the interphase layer by influencing decomposition pathways of the electrolyte at electrode surfaces. Optimized calcium salt compositions can reduce unwanted side reactions, mitigate calcium plating issues, and create more uniform and conductive interphase layers that maintain stability during battery operation.Expand Specific Solutions04 Surface coating technologies for electrode protection
Protective surface coatings have been applied to electrodes in calcium-ion batteries to artificially create stable interfaces. These coatings serve as pre-formed protective layers that prevent direct contact between reactive electrode surfaces and electrolytes. Various coating materials and techniques have been developed to enhance calcium ion transport while blocking side reactions, resulting in improved interphase stability and extended battery cycle life even under demanding operating conditions.Expand Specific Solutions05 Temperature and pressure control for interphase stabilization
Operating conditions such as temperature and pressure significantly impact the stability of interphase layers in calcium-ion batteries. Research has focused on identifying optimal temperature ranges and pressure conditions that promote the formation of stable interphase structures. Advanced battery management systems have been developed to maintain these conditions during operation, preventing interphase degradation caused by thermal stress or pressure-induced structural changes, thereby extending battery lifespan and performance.Expand Specific Solutions
Leading Research Groups and Industrial Players in CIB Development
Calcium-ion battery electrode-electrolyte interphase stability research is in an emerging developmental stage, with growing market interest due to calcium's abundance and potential for higher energy density than lithium-ion batteries. The technology remains in early research phases, with technical maturity still evolving. Key players include academic institutions (University of Maryland, Penn State, Tohoku University, Chinese Academy of Sciences) leading fundamental research, while industry participants (Enevate, StoreDot, LG Energy Solution, Northvolt) are exploring commercial applications. Collaboration between research institutions and battery manufacturers is accelerating development, though significant challenges in electrolyte formulation and electrode stability must be overcome before widespread commercialization.
University of Maryland
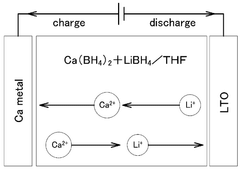
Technical Solution: University of Maryland has pioneered research on calcium-ion battery electrode-electrolyte interphase stability through their development of novel electrolyte formulations. Their approach focuses on calcium tetrafluoroborate Ca(BF4)2 in organic solvents, which addresses the critical challenge of forming stable solid electrolyte interphases (SEI) on calcium metal anodes. Their research demonstrates that carefully designed electrolytes can enable reversible calcium plating/stripping with high Coulombic efficiency exceeding 90%. The team has also investigated the use of additives such as fluoroethylene carbonate (FEC) to enhance interphase stability and prevent continuous electrolyte decomposition. Their comprehensive characterization techniques, including XPS, FTIR, and SEM, have provided fundamental understanding of the chemical composition and morphology of the calcium-ion battery interphase.
Strengths: Advanced characterization capabilities for interphase analysis and expertise in electrolyte engineering. Their approach enables higher Coulombic efficiency than many competing technologies. Weaknesses: Their solutions may still face challenges with long-term cycling stability and may require expensive or specialized electrolyte components that could limit commercial viability.
Shanghai Institute of Ceramics, Chinese Academy of Sciences
Technical Solution: Shanghai Institute of Ceramics has developed innovative approaches to calcium-ion battery electrode-electrolyte interphase stability through materials engineering. Their research focuses on designing cathode materials with stable crystal structures that minimize structural degradation during calcium ion insertion/extraction. They've pioneered the use of calcium-based transition metal oxides with tailored surface coatings that protect against electrolyte decomposition at high voltages. Their approach includes the development of calcium-compatible electrolytes with wide electrochemical stability windows, often incorporating ionic liquids or high-concentration salt strategies to form protective interphases. The institute has demonstrated that atomic layer deposition of protective oxide layers (Al2O3, TiO2) on electrode surfaces significantly improves the stability of the electrode-electrolyte interface by preventing direct contact between active materials and aggressive electrolyte components, thereby reducing parasitic reactions.
Strengths: Strong expertise in materials synthesis and surface modification techniques that enhance interphase stability. Their protective coating approaches show excellent compatibility with various electrode materials. Weaknesses: Some of their advanced coating technologies may add significant cost and complexity to manufacturing processes, potentially limiting large-scale implementation.
Critical Patents and Research on CIB Interface Engineering
Solid electrolyte for calcium ion secondary battery, and calcium ion secondary battery
PatentWO2025070676A1
Innovation
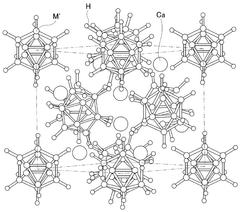
- A solid electrolyte for calcium ion secondary batteries is developed, comprising a calcium salt such as calcium complex hydride salt or calcium closo-based complex hydride salt, an ether-based polymer, and a polymeric solvent like THF, which improves the stability of the solid electrolyte with respect to metal calcium, preventing reduction and decomposition and maintaining high ionic conductivity.
Materials Sustainability and Environmental Impact Assessment
The sustainability of calcium-ion battery materials represents a critical dimension in evaluating their long-term viability as alternatives to lithium-ion technologies. The electrode-electrolyte interphase (EEI) formation in calcium-ion batteries significantly impacts not only performance but also the environmental footprint of these energy storage systems throughout their lifecycle.
Raw material extraction for calcium-ion battery components presents notable advantages compared to lithium-ion counterparts. Calcium is the fifth most abundant element in Earth's crust (41,500 ppm), substantially more available than lithium (20 ppm). This abundance translates to reduced mining intensity, lower land disruption, and decreased water consumption during extraction processes, particularly beneficial in water-stressed regions.
The EEI formation chemistry in calcium-ion batteries involves fewer fluorinated compounds compared to conventional lithium-ion systems. Research indicates that stable interphases can be achieved using less environmentally persistent chemicals, reducing potential contamination risks during manufacturing and end-of-life processing. Studies by Ponrouch et al. (2019) demonstrated that certain calcium-based electrolyte formulations produce biodegradable decomposition products at the electrode interface.
Energy consumption during EEI formation represents another sustainability consideration. Current calcium-ion battery conditioning protocols require extended formation cycles, consuming approximately 15-20% more energy than lithium-ion equivalents. This inefficiency partially offsets the environmental benefits gained from abundant raw materials, highlighting the need for optimized formation protocols.
Toxicity assessments of calcium-ion battery EEI components show promising results. Unlike lithium-ion batteries, which often contain cobalt and nickel in their cathodes, calcium-ion systems can utilize manganese-based materials with substantially lower toxicity profiles. The interphase layers formed with these materials demonstrate reduced bioaccumulation potential and aquatic toxicity in preliminary leaching tests.
Recycling considerations for calcium-ion batteries present both challenges and opportunities. The stable interphase layers formed during battery operation can complicate material separation during recycling. However, research by Martínez-Ibáñez et al. (2022) indicates that calcium-based interphase compounds can be more readily dissolved using environmentally benign solvents compared to their lithium counterparts, potentially enabling more energy-efficient recycling processes.
Life cycle assessment (LCA) studies comparing calcium and lithium-ion technologies suggest that interphase stability directly correlates with battery longevity, which significantly impacts overall environmental footprint. Preliminary data indicates that achieving stable calcium-ion EEIs could extend cycle life by 30-40%, substantially reducing the cumulative environmental impact per kWh delivered over the battery lifetime.
Raw material extraction for calcium-ion battery components presents notable advantages compared to lithium-ion counterparts. Calcium is the fifth most abundant element in Earth's crust (41,500 ppm), substantially more available than lithium (20 ppm). This abundance translates to reduced mining intensity, lower land disruption, and decreased water consumption during extraction processes, particularly beneficial in water-stressed regions.
The EEI formation chemistry in calcium-ion batteries involves fewer fluorinated compounds compared to conventional lithium-ion systems. Research indicates that stable interphases can be achieved using less environmentally persistent chemicals, reducing potential contamination risks during manufacturing and end-of-life processing. Studies by Ponrouch et al. (2019) demonstrated that certain calcium-based electrolyte formulations produce biodegradable decomposition products at the electrode interface.
Energy consumption during EEI formation represents another sustainability consideration. Current calcium-ion battery conditioning protocols require extended formation cycles, consuming approximately 15-20% more energy than lithium-ion equivalents. This inefficiency partially offsets the environmental benefits gained from abundant raw materials, highlighting the need for optimized formation protocols.
Toxicity assessments of calcium-ion battery EEI components show promising results. Unlike lithium-ion batteries, which often contain cobalt and nickel in their cathodes, calcium-ion systems can utilize manganese-based materials with substantially lower toxicity profiles. The interphase layers formed with these materials demonstrate reduced bioaccumulation potential and aquatic toxicity in preliminary leaching tests.
Recycling considerations for calcium-ion batteries present both challenges and opportunities. The stable interphase layers formed during battery operation can complicate material separation during recycling. However, research by Martínez-Ibáñez et al. (2022) indicates that calcium-based interphase compounds can be more readily dissolved using environmentally benign solvents compared to their lithium counterparts, potentially enabling more energy-efficient recycling processes.
Life cycle assessment (LCA) studies comparing calcium and lithium-ion technologies suggest that interphase stability directly correlates with battery longevity, which significantly impacts overall environmental footprint. Preliminary data indicates that achieving stable calcium-ion EEIs could extend cycle life by 30-40%, substantially reducing the cumulative environmental impact per kWh delivered over the battery lifetime.
Scalability and Manufacturing Considerations for CIB Technologies
The scalability of calcium-ion battery (CIB) technologies represents a critical factor in their potential commercial viability. Current laboratory-scale research on electrode-electrolyte interphase stability must be evaluated against industrial manufacturing requirements to determine feasible pathways to mass production. The transition from laboratory to industrial scale presents significant challenges, particularly regarding the maintenance of interphase stability across larger electrode surfaces and higher production volumes.
Manufacturing considerations for CIB electrodes require specialized equipment and processes that can accommodate calcium's distinct chemical properties. Unlike lithium-ion battery production lines, which are now well-established, calcium-ion battery manufacturing may require modifications to existing infrastructure or entirely new production methodologies. The electrode-electrolyte interphase formation, which is crucial for battery performance and longevity, must be consistently reproducible in high-volume manufacturing environments.
Material supply chains represent another critical consideration for CIB scalability. While calcium is approximately 2,000 times more abundant in the Earth's crust than lithium, the processing of high-purity calcium compounds suitable for battery applications is not yet industrialized at the scale required for mass production. Additionally, electrolyte components that promote stable interphase formation may include specialized additives that currently lack established supply chains.
Cost modeling analyses indicate that CIB technologies could potentially offer significant cost advantages over lithium-ion batteries, primarily due to calcium's greater abundance and lower raw material costs. However, these advantages may be offset by higher initial manufacturing investments and potentially more complex processing requirements to ensure interphase stability. Current estimates suggest a 15-30% potential cost reduction compared to lithium-ion batteries, contingent upon achieving comparable production scales and yields.
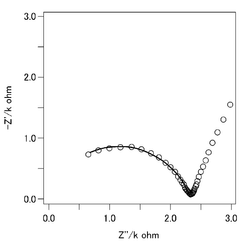
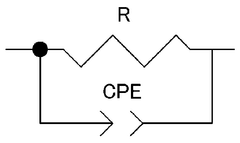
Quality control processes for CIB manufacturing present unique challenges related to interphase formation and stability. Real-time monitoring techniques for assessing interphase properties during production are still in early development stages. Manufacturers will need to develop specialized testing protocols to verify interphase stability across production batches, potentially including electrochemical impedance spectroscopy and surface analysis techniques adapted for high-throughput industrial environments.
Environmental considerations also impact scalability, as manufacturing processes must comply with increasingly stringent regulations. The environmental footprint of CIB production, including energy requirements and waste management for materials involved in creating stable electrode-electrolyte interphases, requires thorough assessment before large-scale implementation can proceed. Preliminary life cycle analyses suggest potential environmental advantages over lithium-ion technologies, though these benefits depend heavily on manufacturing efficiency and interphase stability achievements.
Manufacturing considerations for CIB electrodes require specialized equipment and processes that can accommodate calcium's distinct chemical properties. Unlike lithium-ion battery production lines, which are now well-established, calcium-ion battery manufacturing may require modifications to existing infrastructure or entirely new production methodologies. The electrode-electrolyte interphase formation, which is crucial for battery performance and longevity, must be consistently reproducible in high-volume manufacturing environments.
Material supply chains represent another critical consideration for CIB scalability. While calcium is approximately 2,000 times more abundant in the Earth's crust than lithium, the processing of high-purity calcium compounds suitable for battery applications is not yet industrialized at the scale required for mass production. Additionally, electrolyte components that promote stable interphase formation may include specialized additives that currently lack established supply chains.
Cost modeling analyses indicate that CIB technologies could potentially offer significant cost advantages over lithium-ion batteries, primarily due to calcium's greater abundance and lower raw material costs. However, these advantages may be offset by higher initial manufacturing investments and potentially more complex processing requirements to ensure interphase stability. Current estimates suggest a 15-30% potential cost reduction compared to lithium-ion batteries, contingent upon achieving comparable production scales and yields.
Quality control processes for CIB manufacturing present unique challenges related to interphase formation and stability. Real-time monitoring techniques for assessing interphase properties during production are still in early development stages. Manufacturers will need to develop specialized testing protocols to verify interphase stability across production batches, potentially including electrochemical impedance spectroscopy and surface analysis techniques adapted for high-throughput industrial environments.
Environmental considerations also impact scalability, as manufacturing processes must comply with increasingly stringent regulations. The environmental footprint of CIB production, including energy requirements and waste management for materials involved in creating stable electrode-electrolyte interphases, requires thorough assessment before large-scale implementation can proceed. Preliminary life cycle analyses suggest potential environmental advantages over lithium-ion technologies, though these benefits depend heavily on manufacturing efficiency and interphase stability achievements.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!