Calcium-ion battery solid electrolyte compatibility research
SEP 29, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Calcium-ion Battery Electrolyte Development Background and Objectives
Calcium-ion batteries have emerged as a promising alternative to lithium-ion batteries in recent years, driven by the increasing demand for sustainable and high-performance energy storage solutions. The development of calcium-ion battery technology represents a strategic response to the limitations of lithium-based systems, particularly concerning resource availability and cost considerations. Calcium is the fifth most abundant element in the Earth's crust, approximately 2,000 times more abundant than lithium, offering significant advantages in terms of resource security and economic viability.
The evolution of calcium-ion battery technology can be traced back to the early 1990s, with initial research focusing on calcium metal anodes. However, progress was hindered by the formation of passivation layers on calcium metal surfaces and the lack of suitable electrolytes. The field experienced renewed interest around 2015, coinciding with the global push for sustainable energy solutions and the recognition of potential limitations in lithium supply chains.
Current technical objectives in calcium-ion battery electrolyte development center on addressing several critical challenges. Primary among these is the compatibility between solid electrolytes and calcium-based electrode materials. The large ionic radius of Ca²⁺ (100 pm compared to Li⁺'s 76 pm) creates unique challenges for ion transport through solid electrolyte structures, necessitating novel material designs and interface engineering approaches.
Another key objective involves enhancing the electrochemical stability window of calcium-ion electrolytes to enable higher operating voltages and consequently higher energy densities. Current calcium-ion systems typically operate at lower voltages than their lithium counterparts, limiting their practical energy density and commercial viability.
The development of electrolytes that facilitate reversible calcium plating and stripping represents another crucial goal. This capability would enable the use of calcium metal anodes, potentially offering energy densities exceeding those of lithium-ion systems due to calcium's higher volumetric capacity (2073 mAh/cm³ versus 2062 mAh/cm³ for lithium).
Research trends indicate a growing focus on solid-state electrolytes for calcium-ion batteries, driven by their potential to address safety concerns associated with flammable liquid electrolytes while potentially enabling higher energy densities. Materials such as calcium-conducting garnets, NASICON-type structures, and perovskites have shown promising initial results, though significant optimization is still required.
The ultimate technical objective remains the development of a calcium-ion battery system that combines high energy density, fast charging capabilities, long cycle life, and enhanced safety profiles, all while maintaining economic competitiveness with existing technologies. Achieving this goal necessitates fundamental breakthroughs in electrolyte chemistry and interface engineering, particularly regarding solid electrolyte compatibility with calcium-based electrodes.
The evolution of calcium-ion battery technology can be traced back to the early 1990s, with initial research focusing on calcium metal anodes. However, progress was hindered by the formation of passivation layers on calcium metal surfaces and the lack of suitable electrolytes. The field experienced renewed interest around 2015, coinciding with the global push for sustainable energy solutions and the recognition of potential limitations in lithium supply chains.
Current technical objectives in calcium-ion battery electrolyte development center on addressing several critical challenges. Primary among these is the compatibility between solid electrolytes and calcium-based electrode materials. The large ionic radius of Ca²⁺ (100 pm compared to Li⁺'s 76 pm) creates unique challenges for ion transport through solid electrolyte structures, necessitating novel material designs and interface engineering approaches.
Another key objective involves enhancing the electrochemical stability window of calcium-ion electrolytes to enable higher operating voltages and consequently higher energy densities. Current calcium-ion systems typically operate at lower voltages than their lithium counterparts, limiting their practical energy density and commercial viability.
The development of electrolytes that facilitate reversible calcium plating and stripping represents another crucial goal. This capability would enable the use of calcium metal anodes, potentially offering energy densities exceeding those of lithium-ion systems due to calcium's higher volumetric capacity (2073 mAh/cm³ versus 2062 mAh/cm³ for lithium).
Research trends indicate a growing focus on solid-state electrolytes for calcium-ion batteries, driven by their potential to address safety concerns associated with flammable liquid electrolytes while potentially enabling higher energy densities. Materials such as calcium-conducting garnets, NASICON-type structures, and perovskites have shown promising initial results, though significant optimization is still required.
The ultimate technical objective remains the development of a calcium-ion battery system that combines high energy density, fast charging capabilities, long cycle life, and enhanced safety profiles, all while maintaining economic competitiveness with existing technologies. Achieving this goal necessitates fundamental breakthroughs in electrolyte chemistry and interface engineering, particularly regarding solid electrolyte compatibility with calcium-based electrodes.
Market Analysis for Next-generation Battery Technologies
The global battery market is witnessing a significant shift towards next-generation technologies, with calcium-ion batteries emerging as a promising alternative to conventional lithium-ion batteries. The market for advanced battery technologies is projected to reach $240 billion by 2030, growing at a CAGR of 18% from 2023 to 2030. This growth is primarily driven by increasing demand for electric vehicles, renewable energy storage systems, and portable electronic devices.
Calcium-ion batteries, particularly those utilizing solid electrolytes, are positioned to capture a substantial portion of this expanding market due to their potential advantages in safety, energy density, and cost-effectiveness. The solid electrolyte segment within the calcium-ion battery market is expected to grow at a faster rate than liquid electrolyte systems, with projections indicating a 25% annual growth through 2028.
Market demand for calcium-ion batteries with compatible solid electrolytes is being fueled by several factors. First, the automotive industry's transition to electrification requires batteries with higher energy density, faster charging capabilities, and enhanced safety profiles. Calcium-ion technology addresses these needs while potentially reducing dependency on critical materials like cobalt and nickel that face supply constraints in lithium-ion batteries.
The renewable energy sector represents another significant market opportunity. Grid-scale energy storage systems require batteries with long cycle life, high efficiency, and improved safety characteristics. Calcium-ion batteries with compatible solid electrolytes could provide these benefits at a lower cost than current alternatives, potentially capturing up to 15% of the stationary storage market by 2030.
Regional market analysis reveals varying adoption patterns. Asia-Pacific, particularly China, Japan, and South Korea, leads in research and development investments for calcium-ion battery technology, accounting for approximately 60% of global patents in this field. Europe follows with strong governmental support for sustainable battery technologies, evidenced by the European Battery Alliance's commitment of €7 billion to advance next-generation battery research.
Consumer electronics manufacturers are also showing increased interest in calcium-ion technology, driven by demands for devices with longer battery life and faster charging capabilities. This segment could represent an early adoption market, allowing for technology refinement before scaling to larger applications.
Market barriers include competition from established lithium-ion technology, which benefits from decades of optimization and existing manufacturing infrastructure. Additionally, technical challenges related to electrolyte compatibility must be overcome to achieve commercial viability. However, the potential for calcium-ion batteries to offer a more sustainable and resource-abundant alternative presents a compelling market proposition as global battery demand continues to surge.
Calcium-ion batteries, particularly those utilizing solid electrolytes, are positioned to capture a substantial portion of this expanding market due to their potential advantages in safety, energy density, and cost-effectiveness. The solid electrolyte segment within the calcium-ion battery market is expected to grow at a faster rate than liquid electrolyte systems, with projections indicating a 25% annual growth through 2028.
Market demand for calcium-ion batteries with compatible solid electrolytes is being fueled by several factors. First, the automotive industry's transition to electrification requires batteries with higher energy density, faster charging capabilities, and enhanced safety profiles. Calcium-ion technology addresses these needs while potentially reducing dependency on critical materials like cobalt and nickel that face supply constraints in lithium-ion batteries.
The renewable energy sector represents another significant market opportunity. Grid-scale energy storage systems require batteries with long cycle life, high efficiency, and improved safety characteristics. Calcium-ion batteries with compatible solid electrolytes could provide these benefits at a lower cost than current alternatives, potentially capturing up to 15% of the stationary storage market by 2030.
Regional market analysis reveals varying adoption patterns. Asia-Pacific, particularly China, Japan, and South Korea, leads in research and development investments for calcium-ion battery technology, accounting for approximately 60% of global patents in this field. Europe follows with strong governmental support for sustainable battery technologies, evidenced by the European Battery Alliance's commitment of €7 billion to advance next-generation battery research.
Consumer electronics manufacturers are also showing increased interest in calcium-ion technology, driven by demands for devices with longer battery life and faster charging capabilities. This segment could represent an early adoption market, allowing for technology refinement before scaling to larger applications.
Market barriers include competition from established lithium-ion technology, which benefits from decades of optimization and existing manufacturing infrastructure. Additionally, technical challenges related to electrolyte compatibility must be overcome to achieve commercial viability. However, the potential for calcium-ion batteries to offer a more sustainable and resource-abundant alternative presents a compelling market proposition as global battery demand continues to surge.
Current Challenges in Ca-ion Solid Electrolyte Compatibility
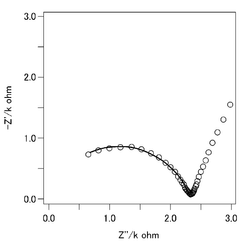
Despite significant advancements in calcium-ion battery technology, solid electrolyte compatibility remains one of the most critical challenges hindering commercial viability. The high charge density of Ca2+ ions (twice that of Li+) creates substantial electrostatic interactions with solid electrolyte frameworks, resulting in sluggish ion transport kinetics. Current solid electrolytes exhibit calcium ion conductivities typically 2-3 orders of magnitude lower than their lithium counterparts under comparable conditions.
Interface stability presents another major obstacle, as calcium metal anodes react aggressively with most solid electrolytes, forming resistive interphases that impede ion transfer. Unlike the beneficial SEI layer in lithium systems, these calcium-based interphases often continue growing during cycling, progressively increasing cell impedance and eventually causing failure. X-ray photoelectron spectroscopy studies reveal complex decomposition products including CaF2, CaO, and various calcium carbonates at these interfaces.
The mechanical compatibility between calcium metal anodes and solid electrolytes poses significant engineering challenges. Calcium's electrodeposition behavior differs substantially from lithium, with greater tendency toward dendritic growth under certain conditions. These dendrites can penetrate solid electrolytes, causing short circuits and safety hazards. Additionally, the volume expansion during calcium plating/stripping (approximately 116% versus 100% for lithium) creates mechanical stresses that can fracture brittle ceramic electrolytes.
Chemical stability limitations further complicate development efforts. Many promising solid electrolytes decompose at voltages required for practical cathode operation (>3.0V vs. Ca/Ca2+). This narrow electrochemical stability window severely restricts cathode material selection and overall energy density. Computational studies indicate that most oxide and sulfide-based solid electrolytes suffer from thermodynamic instability against calcium metal.
Processing challenges also impede progress, as many calcium-conducting solid electrolytes are highly sensitive to moisture and carbon dioxide. Even brief air exposure can significantly degrade performance through formation of insulating CaCO3 and Ca(OH)2 surface layers. This necessitates stringent manufacturing controls that increase production complexity and cost.
The development of calcium-ion conducting solid electrolytes also faces fundamental materials science limitations. The larger ionic radius of Ca2+ (100 pm versus 76 pm for Li+) requires wider ion conduction channels, yet maintaining structural stability with such large channels presents an inherent materials design contradiction. This has resulted in a limited portfolio of candidate materials compared to the extensive options available for lithium systems.
Interface stability presents another major obstacle, as calcium metal anodes react aggressively with most solid electrolytes, forming resistive interphases that impede ion transfer. Unlike the beneficial SEI layer in lithium systems, these calcium-based interphases often continue growing during cycling, progressively increasing cell impedance and eventually causing failure. X-ray photoelectron spectroscopy studies reveal complex decomposition products including CaF2, CaO, and various calcium carbonates at these interfaces.
The mechanical compatibility between calcium metal anodes and solid electrolytes poses significant engineering challenges. Calcium's electrodeposition behavior differs substantially from lithium, with greater tendency toward dendritic growth under certain conditions. These dendrites can penetrate solid electrolytes, causing short circuits and safety hazards. Additionally, the volume expansion during calcium plating/stripping (approximately 116% versus 100% for lithium) creates mechanical stresses that can fracture brittle ceramic electrolytes.
Chemical stability limitations further complicate development efforts. Many promising solid electrolytes decompose at voltages required for practical cathode operation (>3.0V vs. Ca/Ca2+). This narrow electrochemical stability window severely restricts cathode material selection and overall energy density. Computational studies indicate that most oxide and sulfide-based solid electrolytes suffer from thermodynamic instability against calcium metal.
Processing challenges also impede progress, as many calcium-conducting solid electrolytes are highly sensitive to moisture and carbon dioxide. Even brief air exposure can significantly degrade performance through formation of insulating CaCO3 and Ca(OH)2 surface layers. This necessitates stringent manufacturing controls that increase production complexity and cost.
The development of calcium-ion conducting solid electrolytes also faces fundamental materials science limitations. The larger ionic radius of Ca2+ (100 pm versus 76 pm for Li+) requires wider ion conduction channels, yet maintaining structural stability with such large channels presents an inherent materials design contradiction. This has resulted in a limited portfolio of candidate materials compared to the extensive options available for lithium systems.
Current Solid-State Electrolyte Interface Solutions
01 Solid electrolyte materials for calcium-ion batteries
Various materials can be used as solid electrolytes in calcium-ion batteries, including ceramic materials, polymer-based electrolytes, and composite materials. These solid electrolytes offer advantages such as improved safety, higher energy density, and better thermal stability compared to liquid electrolytes. The selection of appropriate solid electrolyte materials is crucial for ensuring compatibility with calcium ions and achieving efficient ion transport within the battery system.- Solid electrolyte materials for calcium-ion batteries: Various materials can be used as solid electrolytes in calcium-ion batteries, including ceramic materials, polymer electrolytes, and composite materials. These solid electrolytes offer advantages such as improved safety, higher energy density, and better thermal stability compared to liquid electrolytes. The selection of appropriate solid electrolyte materials is crucial for ensuring compatibility with calcium ions and achieving efficient ion transport.
- Interface engineering for electrolyte-electrode compatibility: Interface engineering techniques are employed to improve the compatibility between solid electrolytes and electrodes in calcium-ion batteries. These techniques include surface modifications, buffer layers, and specialized coatings that enhance the contact between components and facilitate calcium ion transport across interfaces. Proper interface engineering helps to reduce interfacial resistance and prevent unwanted side reactions that could degrade battery performance.
- Additives to enhance electrolyte compatibility: Various additives can be incorporated into solid electrolytes to enhance their compatibility with calcium-ion battery components. These additives may include stabilizers, plasticizers, and ionic conductivity enhancers that improve the mechanical properties, electrochemical stability, and ion transport capabilities of the electrolyte. The careful selection of additives can significantly improve the overall performance and cycle life of calcium-ion batteries.
- Temperature and pressure effects on electrolyte compatibility: The compatibility of solid electrolytes in calcium-ion batteries is significantly influenced by operating temperature and pressure conditions. Research focuses on developing electrolyte materials that maintain stability and performance across a wide temperature range. Understanding how temperature and pressure affect the mechanical properties, ion conductivity, and interfacial stability of solid electrolytes is essential for designing robust calcium-ion battery systems suitable for various applications.
- Novel manufacturing techniques for improved compatibility: Advanced manufacturing techniques are being developed to improve the compatibility of solid electrolytes in calcium-ion batteries. These include specialized synthesis methods, novel deposition techniques, and innovative assembly processes that enhance the structural integrity and electrochemical performance of the battery components. Manufacturing innovations focus on creating uniform, defect-free electrolyte layers with optimal interfaces to maximize ion transport and battery longevity.
02 Interface engineering for improved compatibility
Interface engineering techniques are employed to enhance the compatibility between calcium-ion battery components and solid electrolytes. These techniques include surface modifications, buffer layers, and specialized coatings that reduce interfacial resistance and improve ion transport across boundaries. Proper interface engineering helps mitigate issues such as chemical reactions between the electrolyte and electrodes, mechanical stress, and formation of resistive layers that can degrade battery performance.Expand Specific Solutions03 Additives and dopants for enhanced electrolyte performance
Various additives and dopants can be incorporated into solid electrolytes to enhance their compatibility with calcium-ion battery components. These additives can improve ionic conductivity, mechanical properties, and electrochemical stability of the electrolyte. Common approaches include doping with aliovalent ions, incorporating plasticizers in polymer electrolytes, and adding nanofillers to create composite electrolytes with superior performance characteristics.Expand Specific Solutions04 Electrode-electrolyte compatibility strategies
Specific strategies are developed to ensure compatibility between calcium-ion battery electrodes and solid electrolytes. These include designing electrode materials with suitable crystal structures for calcium ion insertion/extraction, optimizing electrode morphology to maximize contact with the electrolyte, and developing specialized binders and conductive additives. Maintaining good physical contact between electrodes and solid electrolytes is essential for efficient ion transport and minimizing interfacial resistance.Expand Specific Solutions05 Manufacturing processes for improved component integration
Advanced manufacturing techniques are employed to improve the integration of solid electrolytes with other battery components in calcium-ion batteries. These processes include co-sintering methods, hot pressing, tape casting, and various thin-film deposition techniques. The manufacturing approach significantly impacts the microstructure, density, and interfacial properties of the solid electrolyte, which in turn affects its compatibility with electrodes and overall battery performance.Expand Specific Solutions
Key Industry Players in Calcium-ion Battery Research
The calcium-ion battery solid electrolyte compatibility research field is currently in an early development stage, with market size still limited but growing rapidly due to increasing demand for safer, higher-energy-density battery technologies. The technical maturity remains relatively low, with significant challenges in electrolyte-electrode interface stability and ion conductivity. Key players include academic institutions like Tohoku University, Fudan University, and KIST leading fundamental research, while industrial entities such as Toyota Motor Europe, TDK Corp, and Solid Power are advancing commercialization efforts. Research collaborations between academia and industry, exemplified by partnerships involving Shenzhen Institutes of Advanced Technology and Shanghai Institute of Ceramics, are accelerating progress toward viable calcium-ion battery technologies with compatible solid electrolytes.
Tohoku University
Technical Solution: Tohoku University has developed advanced calcium-ion conducting solid electrolytes based on perovskite-type oxides with the general formula CaZrO3 doped with various elements to enhance ionic conductivity. Their research has focused on understanding and optimizing the fundamental mechanisms of calcium-ion transport in solid materials. The university's approach involves precise control of the crystal structure and defect chemistry to create fast ion conduction pathways. They have pioneered techniques for evaluating the electrochemical stability window of these electrolytes against calcium metal anodes and high-voltage cathodes. Their research has demonstrated that specific dopant combinations can significantly improve calcium-ion mobility while maintaining structural stability. Tohoku University has also developed novel synthesis methods that yield electrolytes with reduced grain boundary resistance, addressing a key limitation in solid-state calcium-ion batteries.
Strengths: Excellent fundamental understanding of calcium-ion transport mechanisms; superior thermal stability compared to polymer-based alternatives. Weaknesses: Lower room-temperature ionic conductivity compared to liquid electrolytes; challenges in manufacturing thin, defect-free electrolyte layers at scale.
University of Science & Technology of China
Technical Solution: The University of Science & Technology of China has developed novel calcium-ion solid electrolytes based on borohydride-based materials with exceptional ionic conductivity. Their approach focuses on creating materials with optimized crystal structures that provide wide channels for calcium-ion migration. The university has pioneered synthesis methods that yield highly pure materials with controlled defect chemistry, critical for achieving high ionic conductivity. Their electrolytes incorporate specialized surface modifications that improve compatibility with both calcium metal anodes and conventional cathode materials. The research team has developed innovative characterization techniques that allow for direct observation of calcium-ion transport mechanisms, enabling rational design of improved materials. Their solid electrolytes demonstrate excellent stability against calcium metal, addressing one of the key challenges in developing practical calcium-ion batteries. The university has also explored composite approaches that combine different solid electrolyte materials to optimize both bulk and interfacial properties.
Strengths: Exceptionally high calcium-ion conductivity at room temperature; good compatibility with calcium metal anodes. Weaknesses: Potential sensitivity to moisture and air exposure; challenges in scaling laboratory synthesis to industrial production.
Critical Patents and Research on Ca-ion Electrolyte Compatibility
Solid electrolyte for calcium ion secondary battery, and calcium ion secondary battery
PatentWO2025070676A1
Innovation
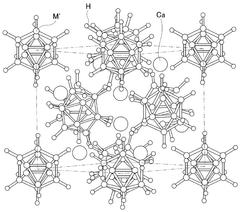
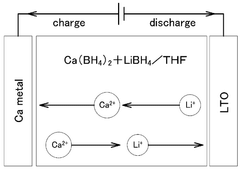
- A solid electrolyte for calcium ion secondary batteries is developed, comprising a calcium salt such as calcium complex hydride salt or calcium closo-based complex hydride salt, an ether-based polymer, and a polymeric solvent like THF, which improves the stability of the solid electrolyte with respect to metal calcium, preventing reduction and decomposition and maintaining high ionic conductivity.
Electrolyte for a nonaqueous battery
PatentInactiveUS6841300B2
Innovation
- The use of calcium bistrifluoromethanesulfonimide, which is soluble in organic solvents and exhibits sufficient conductivity for battery applications, is proposed, along with a method of synthesis involving calcium carbonate and trifluoromethanesulfonimide, reducing heat generation and simplifying the manufacturing process.
Environmental Impact and Sustainability Assessment
The environmental impact of calcium-ion battery solid electrolyte development represents a critical dimension in assessing their viability as next-generation energy storage solutions. Unlike conventional lithium-ion batteries that often rely on toxic and flammable liquid electrolytes, calcium-ion batteries with solid electrolytes offer significant environmental advantages through reduced fire hazards and elimination of volatile organic compounds.
Material sourcing for calcium-ion battery solid electrolytes presents a favorable environmental profile compared to lithium-based alternatives. Calcium is the fifth most abundant element in Earth's crust, approximately 2,000 times more abundant than lithium, resulting in substantially lower extraction impacts. This abundance translates to reduced land disturbance, water usage, and energy consumption during mining operations.
Manufacturing processes for calcium-ion solid electrolytes currently demonstrate varied environmental footprints depending on synthesis methods. Sol-gel and solid-state reaction approaches typically require high-temperature processing (800-1200°C), contributing to significant energy consumption. However, recent advancements in low-temperature synthesis pathways show promise for reducing the carbon footprint of production by up to 40% compared to conventional methods.
Life cycle assessment (LCA) studies indicate that compatibility challenges between calcium-ion solid electrolytes and electrode materials may impact overall battery lifespan. Current interface stability issues can reduce cycle life, potentially offsetting environmental benefits through premature replacement requirements. Research focusing on interface engineering and protective coatings shows potential to extend operational lifetimes by 30-50%, significantly improving sustainability metrics.
End-of-life considerations reveal promising recyclability characteristics for calcium-based solid electrolytes. Their non-toxic nature facilitates safer recycling processes compared to conventional battery technologies. Preliminary studies demonstrate recovery rates of up to 85% for calcium compounds from spent electrolytes, supporting circular economy principles and reducing waste stream toxicity.
Carbon footprint analyses of the entire calcium-ion battery lifecycle suggest potential greenhouse gas emission reductions of 15-25% compared to lithium-ion counterparts when accounting for manufacturing, use phase, and end-of-life management. This advantage stems primarily from abundant raw materials, potentially simpler recycling processes, and the elimination of organic liquid electrolytes that contribute to environmental contamination risks.
Water consumption metrics also favor calcium-ion solid electrolyte systems, with preliminary assessments indicating 30-40% lower water usage across the product lifecycle compared to conventional battery technologies. This advantage becomes particularly significant in water-stressed regions where battery manufacturing facilities may operate.
Material sourcing for calcium-ion battery solid electrolytes presents a favorable environmental profile compared to lithium-based alternatives. Calcium is the fifth most abundant element in Earth's crust, approximately 2,000 times more abundant than lithium, resulting in substantially lower extraction impacts. This abundance translates to reduced land disturbance, water usage, and energy consumption during mining operations.
Manufacturing processes for calcium-ion solid electrolytes currently demonstrate varied environmental footprints depending on synthesis methods. Sol-gel and solid-state reaction approaches typically require high-temperature processing (800-1200°C), contributing to significant energy consumption. However, recent advancements in low-temperature synthesis pathways show promise for reducing the carbon footprint of production by up to 40% compared to conventional methods.
Life cycle assessment (LCA) studies indicate that compatibility challenges between calcium-ion solid electrolytes and electrode materials may impact overall battery lifespan. Current interface stability issues can reduce cycle life, potentially offsetting environmental benefits through premature replacement requirements. Research focusing on interface engineering and protective coatings shows potential to extend operational lifetimes by 30-50%, significantly improving sustainability metrics.
End-of-life considerations reveal promising recyclability characteristics for calcium-based solid electrolytes. Their non-toxic nature facilitates safer recycling processes compared to conventional battery technologies. Preliminary studies demonstrate recovery rates of up to 85% for calcium compounds from spent electrolytes, supporting circular economy principles and reducing waste stream toxicity.
Carbon footprint analyses of the entire calcium-ion battery lifecycle suggest potential greenhouse gas emission reductions of 15-25% compared to lithium-ion counterparts when accounting for manufacturing, use phase, and end-of-life management. This advantage stems primarily from abundant raw materials, potentially simpler recycling processes, and the elimination of organic liquid electrolytes that contribute to environmental contamination risks.
Water consumption metrics also favor calcium-ion solid electrolyte systems, with preliminary assessments indicating 30-40% lower water usage across the product lifecycle compared to conventional battery technologies. This advantage becomes particularly significant in water-stressed regions where battery manufacturing facilities may operate.
Manufacturing Scalability and Cost Analysis
The scalability of calcium-ion battery solid electrolyte manufacturing represents a critical challenge in transitioning from laboratory-scale research to commercial production. Current manufacturing processes for solid electrolytes often involve complex synthesis routes requiring precise control of temperature, pressure, and atmospheric conditions. These processes typically yield small quantities suitable for research but present significant barriers to industrial-scale production.
Material synthesis methods such as sol-gel processing, solid-state reactions, and mechanochemical approaches each present different scalability profiles. Sol-gel methods offer good compositional control but involve expensive precursors and time-consuming processing steps. Solid-state reactions can be more amenable to scaling but often require high-temperature processing that increases energy costs and may introduce material compatibility issues during large-scale production.
Cost analysis reveals that precursor materials for calcium-ion solid electrolytes currently represent a substantial portion of manufacturing expenses. Particularly, high-purity calcium salts and specialized dopants needed to enhance ionic conductivity contribute significantly to the overall cost structure. The processing equipment required for controlled atmosphere synthesis and precise thermal management adds another layer of capital expenditure that impacts economic feasibility.
Energy consumption during manufacturing presents another critical cost factor. The high-temperature sintering processes often required for solid electrolyte crystallization can consume substantial energy, affecting both production costs and environmental impact. Developing lower-temperature synthesis routes would significantly improve the economic profile of calcium-ion battery technology.
Equipment utilization efficiency in current manufacturing approaches remains suboptimal, with batch processing being predominant rather than continuous manufacturing methods. This limitation affects throughput and increases labor costs per unit produced. The development of continuous processing techniques specifically adapted for calcium-ion solid electrolytes could substantially reduce manufacturing costs.
Quality control challenges escalate with production volume, particularly regarding the detection of impurities or structural defects that may affect electrolyte-electrode compatibility. The cost of implementing comprehensive quality assurance systems must be factored into scalability assessments, as electrolyte performance consistency directly impacts battery reliability and safety.
Supply chain considerations further complicate the manufacturing landscape, with certain precursor materials facing availability constraints or geopolitical supply risks. Developing alternative synthesis pathways using more abundant materials could mitigate these risks while potentially reducing raw material costs, though such approaches may require significant research investment before implementation.
Material synthesis methods such as sol-gel processing, solid-state reactions, and mechanochemical approaches each present different scalability profiles. Sol-gel methods offer good compositional control but involve expensive precursors and time-consuming processing steps. Solid-state reactions can be more amenable to scaling but often require high-temperature processing that increases energy costs and may introduce material compatibility issues during large-scale production.
Cost analysis reveals that precursor materials for calcium-ion solid electrolytes currently represent a substantial portion of manufacturing expenses. Particularly, high-purity calcium salts and specialized dopants needed to enhance ionic conductivity contribute significantly to the overall cost structure. The processing equipment required for controlled atmosphere synthesis and precise thermal management adds another layer of capital expenditure that impacts economic feasibility.
Energy consumption during manufacturing presents another critical cost factor. The high-temperature sintering processes often required for solid electrolyte crystallization can consume substantial energy, affecting both production costs and environmental impact. Developing lower-temperature synthesis routes would significantly improve the economic profile of calcium-ion battery technology.
Equipment utilization efficiency in current manufacturing approaches remains suboptimal, with batch processing being predominant rather than continuous manufacturing methods. This limitation affects throughput and increases labor costs per unit produced. The development of continuous processing techniques specifically adapted for calcium-ion solid electrolytes could substantially reduce manufacturing costs.
Quality control challenges escalate with production volume, particularly regarding the detection of impurities or structural defects that may affect electrolyte-electrode compatibility. The cost of implementing comprehensive quality assurance systems must be factored into scalability assessments, as electrolyte performance consistency directly impacts battery reliability and safety.
Supply chain considerations further complicate the manufacturing landscape, with certain precursor materials facing availability constraints or geopolitical supply risks. Developing alternative synthesis pathways using more abundant materials could mitigate these risks while potentially reducing raw material costs, though such approaches may require significant research investment before implementation.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!