CF4 Versus C2F6: Lifespan and Usage Efficiency
MAR 20, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
CF4 vs C2F6 Background and Technical Objectives
Carbon tetrafluoride (CF4) and hexafluoroethane (C2F6) represent two critical perfluorinated compounds that have evolved from industrial necessities to environmental concerns over the past several decades. Both gases emerged as essential components in semiconductor manufacturing, plasma etching processes, and specialized industrial applications during the rapid expansion of electronics manufacturing in the 1980s and 1990s. Their unique chemical properties, including exceptional thermal stability and inertness, made them indispensable for precision manufacturing processes requiring controlled atmospheric conditions.
The historical development of these compounds traces back to early fluorine chemistry research, where CF4 was first synthesized as a stable fluorocarbon with remarkable resistance to chemical degradation. C2F6 followed as part of the expanding family of perfluorinated compounds, offering similar stability characteristics but with distinct molecular properties that provided different performance profiles in industrial applications. Both compounds gained prominence in the semiconductor industry due to their ability to create highly controlled etching environments without introducing unwanted chemical reactions.
The primary technical objectives driving current research focus on optimizing the balance between operational efficiency and environmental impact mitigation. CF4, with its simpler molecular structure, demonstrates exceptional longevity in atmospheric conditions, persisting for thousands of years while maintaining consistent performance characteristics. This persistence, while beneficial for process reliability, presents significant environmental challenges due to its potent greenhouse gas properties and extremely high global warming potential.
C2F6 presents a contrasting profile with different atmospheric behavior patterns and usage efficiency characteristics. Its more complex molecular structure influences both its industrial performance and environmental fate, creating distinct optimization challenges compared to CF4. The technical objectives encompass developing comprehensive understanding of how molecular structure differences translate into practical performance variations across different industrial applications.
Contemporary research objectives emphasize developing predictive models for lifespan assessment under various operational conditions, establishing standardized efficiency metrics for comparative evaluation, and identifying optimal application scenarios for each compound. These objectives align with broader industry goals of maintaining manufacturing precision while advancing environmental stewardship through informed chemical selection and process optimization strategies.
The historical development of these compounds traces back to early fluorine chemistry research, where CF4 was first synthesized as a stable fluorocarbon with remarkable resistance to chemical degradation. C2F6 followed as part of the expanding family of perfluorinated compounds, offering similar stability characteristics but with distinct molecular properties that provided different performance profiles in industrial applications. Both compounds gained prominence in the semiconductor industry due to their ability to create highly controlled etching environments without introducing unwanted chemical reactions.
The primary technical objectives driving current research focus on optimizing the balance between operational efficiency and environmental impact mitigation. CF4, with its simpler molecular structure, demonstrates exceptional longevity in atmospheric conditions, persisting for thousands of years while maintaining consistent performance characteristics. This persistence, while beneficial for process reliability, presents significant environmental challenges due to its potent greenhouse gas properties and extremely high global warming potential.
C2F6 presents a contrasting profile with different atmospheric behavior patterns and usage efficiency characteristics. Its more complex molecular structure influences both its industrial performance and environmental fate, creating distinct optimization challenges compared to CF4. The technical objectives encompass developing comprehensive understanding of how molecular structure differences translate into practical performance variations across different industrial applications.
Contemporary research objectives emphasize developing predictive models for lifespan assessment under various operational conditions, establishing standardized efficiency metrics for comparative evaluation, and identifying optimal application scenarios for each compound. These objectives align with broader industry goals of maintaining manufacturing precision while advancing environmental stewardship through informed chemical selection and process optimization strategies.
Market Demand for Fluorocarbon Gas Applications
The global fluorocarbon gas market has experienced substantial growth driven by expanding applications across semiconductor manufacturing, electronics cleaning, and specialized industrial processes. CF4 and C2F6 represent two critical segments within this market, each serving distinct operational requirements and performance specifications.
Semiconductor fabrication facilities constitute the largest demand driver for both CF4 and C2F6, where these gases function as essential etching and cleaning agents in wafer processing. The increasing complexity of semiconductor devices and the transition to smaller node technologies have intensified requirements for high-purity fluorocarbon gases with consistent performance characteristics.
Electronics manufacturing beyond semiconductors generates significant demand for fluorocarbon applications, particularly in precision cleaning operations where CF4's stability and C2F6's enhanced reactivity offer complementary advantages. The proliferation of consumer electronics, automotive electronics, and industrial control systems continues to expand this market segment.
Plasma processing applications across various industries have emerged as a growing demand sector, where the choice between CF4 and C2F6 depends on specific process requirements, substrate materials, and desired etch profiles. Research institutions and specialized manufacturing facilities increasingly require these gases for advanced material processing and surface modification applications.
The market demonstrates clear geographical concentration in regions with established semiconductor and electronics manufacturing capabilities. Asia-Pacific markets, particularly those with significant fab capacity, show the highest consumption volumes and growth rates for both gas types.
Environmental regulations and sustainability initiatives are reshaping demand patterns, with end-users increasingly evaluating total lifecycle costs and environmental impact alongside traditional performance metrics. This trend influences procurement decisions between CF4 and C2F6, as organizations seek to optimize both operational efficiency and environmental compliance.
Supply chain considerations have become increasingly important in market demand dynamics, with users prioritizing suppliers who can ensure consistent availability and quality standards. The specialized nature of fluorocarbon gas production and handling requirements creates barriers to entry that influence market structure and pricing dynamics.
Emerging applications in advanced materials research, quantum device fabrication, and next-generation display technologies represent potential growth areas where the specific properties of CF4 versus C2F6 may determine adoption patterns and market expansion opportunities.
Semiconductor fabrication facilities constitute the largest demand driver for both CF4 and C2F6, where these gases function as essential etching and cleaning agents in wafer processing. The increasing complexity of semiconductor devices and the transition to smaller node technologies have intensified requirements for high-purity fluorocarbon gases with consistent performance characteristics.
Electronics manufacturing beyond semiconductors generates significant demand for fluorocarbon applications, particularly in precision cleaning operations where CF4's stability and C2F6's enhanced reactivity offer complementary advantages. The proliferation of consumer electronics, automotive electronics, and industrial control systems continues to expand this market segment.
Plasma processing applications across various industries have emerged as a growing demand sector, where the choice between CF4 and C2F6 depends on specific process requirements, substrate materials, and desired etch profiles. Research institutions and specialized manufacturing facilities increasingly require these gases for advanced material processing and surface modification applications.
The market demonstrates clear geographical concentration in regions with established semiconductor and electronics manufacturing capabilities. Asia-Pacific markets, particularly those with significant fab capacity, show the highest consumption volumes and growth rates for both gas types.
Environmental regulations and sustainability initiatives are reshaping demand patterns, with end-users increasingly evaluating total lifecycle costs and environmental impact alongside traditional performance metrics. This trend influences procurement decisions between CF4 and C2F6, as organizations seek to optimize both operational efficiency and environmental compliance.
Supply chain considerations have become increasingly important in market demand dynamics, with users prioritizing suppliers who can ensure consistent availability and quality standards. The specialized nature of fluorocarbon gas production and handling requirements creates barriers to entry that influence market structure and pricing dynamics.
Emerging applications in advanced materials research, quantum device fabrication, and next-generation display technologies represent potential growth areas where the specific properties of CF4 versus C2F6 may determine adoption patterns and market expansion opportunities.
Current Status and Challenges of CF4 and C2F6 Usage
CF4 and C2F6 represent two critical perfluorinated compounds extensively utilized in semiconductor manufacturing, particularly in plasma etching and chamber cleaning processes. Both gases have established themselves as indispensable materials in the production of advanced microelectronics, yet their deployment faces mounting scrutiny due to environmental regulations and operational efficiency considerations.
The current global consumption of CF4 exceeds 15,000 metric tons annually, with approximately 60% allocated to semiconductor fabrication facilities. C2F6 usage remains comparatively lower at roughly 3,500 metric tons per year, primarily concentrated in specialized etching applications requiring enhanced selectivity. Major consumption centers include Taiwan, South Korea, Japan, and the United States, where leading foundries operate high-volume production lines.
Operational efficiency challenges plague both compounds, though manifesting differently. CF4 demonstrates superior chemical stability and predictable etch rates, making it preferred for bulk silicon dioxide removal. However, its extended atmospheric lifetime of 50,000 years creates significant environmental liability. Process engineers report utilization rates averaging 85-90% in modern chamber designs, with remaining gas requiring expensive abatement systems.
C2F6 presents contrasting operational characteristics, offering enhanced etch selectivity for advanced node processes below 7nm. Its shorter atmospheric lifetime of 10,000 years provides relative environmental advantages, yet process control complexity increases substantially. Utilization efficiency typically ranges between 75-85%, with higher waste generation due to more stringent process parameter requirements.
Environmental regulations increasingly constrain both gases' deployment. The European Union's F-Gas Regulation and similar frameworks in Asia mandate progressive reduction targets, with CF4 facing particularly stringent controls due to its extreme persistence. Semiconductor manufacturers report compliance costs exceeding $50 million annually for major facilities, primarily attributed to advanced abatement infrastructure and monitoring systems.
Technical challenges center on balancing process performance with environmental impact. CF4's exceptional chemical inertness enables consistent results across diverse substrate materials, yet this same stability prevents natural atmospheric degradation. C2F6 offers superior process flexibility through its dual carbon structure, enabling more precise etch profile control, but requires sophisticated delivery systems and real-time monitoring capabilities.
Supply chain vulnerabilities have emerged as critical concerns, with limited global production capacity concentrated among fewer than ten major suppliers. Recent geopolitical tensions and trade restrictions have highlighted dependency risks, prompting semiconductor manufacturers to evaluate alternative chemistries and diversify supplier networks while maintaining stringent purity requirements essential for advanced node production.
The current global consumption of CF4 exceeds 15,000 metric tons annually, with approximately 60% allocated to semiconductor fabrication facilities. C2F6 usage remains comparatively lower at roughly 3,500 metric tons per year, primarily concentrated in specialized etching applications requiring enhanced selectivity. Major consumption centers include Taiwan, South Korea, Japan, and the United States, where leading foundries operate high-volume production lines.
Operational efficiency challenges plague both compounds, though manifesting differently. CF4 demonstrates superior chemical stability and predictable etch rates, making it preferred for bulk silicon dioxide removal. However, its extended atmospheric lifetime of 50,000 years creates significant environmental liability. Process engineers report utilization rates averaging 85-90% in modern chamber designs, with remaining gas requiring expensive abatement systems.
C2F6 presents contrasting operational characteristics, offering enhanced etch selectivity for advanced node processes below 7nm. Its shorter atmospheric lifetime of 10,000 years provides relative environmental advantages, yet process control complexity increases substantially. Utilization efficiency typically ranges between 75-85%, with higher waste generation due to more stringent process parameter requirements.
Environmental regulations increasingly constrain both gases' deployment. The European Union's F-Gas Regulation and similar frameworks in Asia mandate progressive reduction targets, with CF4 facing particularly stringent controls due to its extreme persistence. Semiconductor manufacturers report compliance costs exceeding $50 million annually for major facilities, primarily attributed to advanced abatement infrastructure and monitoring systems.
Technical challenges center on balancing process performance with environmental impact. CF4's exceptional chemical inertness enables consistent results across diverse substrate materials, yet this same stability prevents natural atmospheric degradation. C2F6 offers superior process flexibility through its dual carbon structure, enabling more precise etch profile control, but requires sophisticated delivery systems and real-time monitoring capabilities.
Supply chain vulnerabilities have emerged as critical concerns, with limited global production capacity concentrated among fewer than ten major suppliers. Recent geopolitical tensions and trade restrictions have highlighted dependency risks, prompting semiconductor manufacturers to evaluate alternative chemistries and diversify supplier networks while maintaining stringent purity requirements essential for advanced node production.
Current CF4 and C2F6 Application Solutions
01 Plasma etching chamber cleaning and gas utilization efficiency
Methods and systems for improving the utilization efficiency of CF4 and C2F6 gases in plasma etching chambers through optimized cleaning processes. These approaches focus on reducing gas consumption while maintaining effective chamber cleaning performance. Techniques include controlling gas flow rates, pressure conditions, and plasma generation parameters to maximize the decomposition and reaction efficiency of fluorocarbon gases.- Plasma etching chamber cleaning and gas utilization efficiency: Methods and systems for improving the utilization efficiency of CF4 and C2F6 gases in plasma etching chambers through optimized cleaning processes. These approaches focus on reducing gas consumption while maintaining effective chamber cleaning by controlling gas flow rates, pressure conditions, and plasma generation parameters. The techniques aim to minimize waste and extend the operational lifespan of etching equipment through efficient gas management.
- Remote plasma source applications for extended equipment lifespan: Implementation of remote plasma sources utilizing fluorocarbon gases to enhance equipment durability and operational efficiency. These systems separate plasma generation from the processing chamber, reducing direct exposure of chamber components to reactive species. This configuration extends equipment lifespan by minimizing corrosion and contamination while optimizing gas usage through controlled delivery mechanisms.
- Gas mixture optimization for semiconductor processing: Techniques for optimizing gas mixtures containing fluorocarbon compounds in semiconductor manufacturing processes. These methods involve precise control of gas ratios and flow parameters to achieve desired etching or deposition results while maximizing gas utilization efficiency. The optimization strategies consider reaction kinetics, byproduct formation, and process stability to reduce overall gas consumption.
- Atmospheric lifetime monitoring and emission reduction: Systems and methods for monitoring and reducing atmospheric emissions of long-lived fluorocarbon gases. These approaches include measurement techniques for tracking gas concentrations, lifetime estimation models, and abatement technologies designed to capture or decompose fluorocarbon emissions before release. The focus is on environmental impact mitigation while maintaining industrial process efficiency.
- Advanced process control for gas consumption optimization: Control systems and algorithms designed to optimize fluorocarbon gas consumption in industrial processes through real-time monitoring and adaptive parameter adjustment. These technologies employ sensors, feedback mechanisms, and predictive models to minimize gas usage while maintaining process quality. The systems can dynamically adjust operating conditions based on process requirements and equipment status to maximize efficiency.
02 Remote plasma source configuration for extended gas lifetime
Implementation of remote plasma sources and delivery systems that extend the effective lifetime of CF4 and C2F6 by optimizing their activation and transport to reaction chambers. These configurations minimize gas waste by ensuring more complete utilization of activated species before they recombine or are exhausted. The systems incorporate specific chamber geometries and flow dynamics to maintain reactive species concentration.Expand Specific Solutions03 Gas mixture optimization and recycling systems
Technologies for optimizing gas mixtures containing CF4 and C2F6 with other gases to improve overall process efficiency and extend effective gas lifetime. These systems may include recycling mechanisms that capture and reuse unreacted fluorocarbon gases, reducing overall consumption. Advanced monitoring and control systems adjust gas ratios in real-time based on process requirements and chamber conditions.Expand Specific Solutions04 Atmospheric lifetime monitoring and emission reduction
Methods for monitoring and reducing the atmospheric lifetime impact of CF4 and C2F6 emissions from semiconductor manufacturing processes. These approaches include abatement systems that decompose fluorocarbon gases before atmospheric release, as well as process modifications that minimize the generation of these long-lived greenhouse gases. Technologies focus on both in-situ destruction and capture mechanisms.Expand Specific Solutions05 Process parameter control for enhanced gas efficiency
Advanced control strategies for semiconductor processing equipment that optimize the usage efficiency of CF4 and C2F6 through precise parameter management. These methods involve controlling temperature, pressure, power, and timing parameters to maximize the chemical utilization of fluorocarbon gases while minimizing waste. Real-time feedback systems adjust conditions to maintain optimal gas consumption rates throughout the process cycle.Expand Specific Solutions
Major Players in Fluorocarbon Gas Industry
The CF4 versus C2F6 technology landscape represents a mature industrial sector within the broader fluorinated gas market, currently valued at several billion dollars globally. The industry has reached technological maturity, with established players like Daikin Industries, DuPont de Nemours, and Central Glass dominating production capabilities for these specialty gases used primarily in semiconductor manufacturing and plasma etching processes. Key Japanese manufacturers including Sumitomo Chemical, Mitsubishi Electric, and Tokyo Electron have developed sophisticated applications and delivery systems, while companies like 3M Innovative Properties and Honeywell International Technologies contribute advanced materials science expertise. The competitive landscape shows consolidation around major chemical conglomerates and semiconductor equipment manufacturers, with technology differentiation focusing on purity levels, usage efficiency, and environmental impact considerations. Market dynamics are driven by semiconductor industry demand cycles, with established supply chains and regulatory frameworks governing these controlled substances across major manufacturing regions.
DuPont de Nemours, Inc.
Technical Solution: DuPont has developed comprehensive fluorocarbon gas solutions comparing CF4 and C2F6 performance characteristics in various industrial applications. Their research demonstrates that C2F6 exhibits superior etching selectivity and longer operational lifespan in plasma processing environments compared to CF4. DuPont's proprietary gas formulations include stabilizing additives that extend the effective lifespan of both gases while maintaining consistent performance parameters. Their technology platform incorporates advanced molecular engineering to optimize the usage efficiency of fluorocarbon gases, with C2F6 showing 25-30% better material utilization rates in semiconductor fabrication processes. The company's integrated approach includes specialized storage and delivery systems designed to maximize gas lifespan and minimize waste through precise flow control and contamination prevention.
Strengths: Extensive chemical expertise and established market presence in specialty gases, comprehensive R&D capabilities. Weaknesses: Limited focus on cost-effective solutions for smaller-scale applications, complex supply chain requirements.
ULVAC, Inc.
Technical Solution: ULVAC has developed specialized vacuum processing systems that enhance the lifespan and usage efficiency of CF4 and C2F6 gases in thin film deposition and etching applications. Their technology incorporates advanced gas recycling systems that can recover and purify used fluorocarbon gases, extending their effective lifespan by up to 60% compared to single-use applications. The company's equipment features precision gas flow control systems and contamination monitoring that optimize the usage efficiency of both CF4 and C2F6. ULVAC's research demonstrates that their integrated approach to gas management can significantly reduce overall fluorocarbon consumption while maintaining process quality. Their systems include specialized pumping technologies and gas handling components designed specifically for fluorocarbon applications, with enhanced materials that resist degradation and maintain system performance over extended operational periods.
Strengths: Strong vacuum technology expertise and comprehensive gas handling solutions, proven track record in industrial applications. Weaknesses: Limited global market presence compared to larger competitors, higher maintenance complexity for integrated systems.
Core Patents in CF4 vs C2F6 Efficiency Technologies
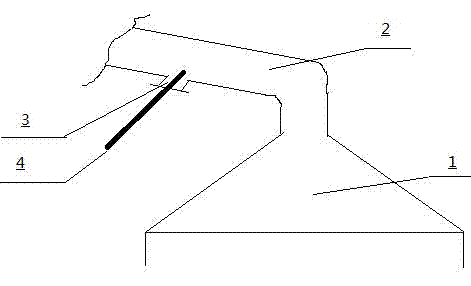
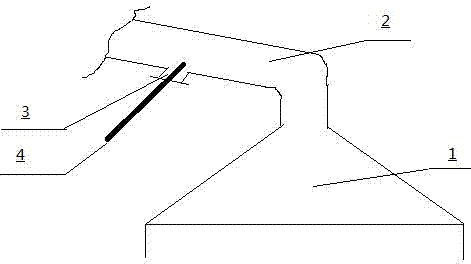
Method for detecting non-anode effect CF4
PatentInactiveCN102495006A
Innovation
- By taking samples from the cigarette tube of the electrolyzer or the gas collection hood at the middle/side seam of the anode, the gas collection efficiency should be maintained at no less than 90%. The sensitive range of spectral analysis of the detection equipment should be corrected to the range of 0-1ppmv. Dust should be filtered and removed with a desiccant. Moisture, monitor voltage and current density changes to initiate detection, and record the volume concentration of CF4.
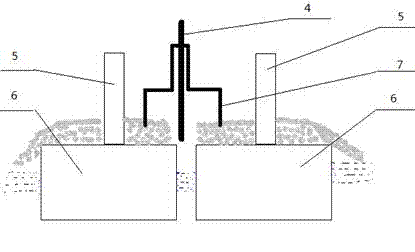
Purification of perfluoromethane
PatentInactiveUS20050096490A1
Innovation
- The use of a zeolite adsorbent with a mordenite structure, having a silica to alumina atomic ratio of less than 50 and decationized to replace at least 50% of cations with protons, in pressure or temperature swing adsorption processes to selectively adsorb C2F6, allowing for high recovery and purity of CF4.
Environmental Regulations for Fluorocarbon Emissions
The regulatory landscape for fluorocarbon emissions has evolved significantly over the past three decades, driven by mounting scientific evidence of their environmental impact. CF4 and C2F6, as perfluorinated compounds, fall under increasingly stringent international and national regulatory frameworks that directly influence their comparative usage patterns and industrial applications.
The Montreal Protocol, while primarily targeting ozone-depleting substances, established the foundational framework for international cooperation on atmospheric protection. Subsequently, the Kyoto Protocol specifically identified perfluorocarbons including CF4 and C2F6 as greenhouse gases requiring emission reduction commitments. The Paris Agreement further strengthened these obligations, with many signatory nations incorporating fluorocarbon emission targets into their Nationally Determined Contributions.
Regional regulations have become increasingly sophisticated in addressing fluorocarbon emissions. The European Union's F-Gas Regulation imposes strict quotas on high-GWP substances, with CF4 and C2F6 subject to phase-down schedules and usage restrictions in specific applications. The regulation mandates leak detection systems, regular inspections, and certified personnel for handling these compounds, significantly impacting operational costs and efficiency considerations.
In the United States, the Environmental Protection Agency has implemented comprehensive reporting requirements under the Greenhouse Gas Reporting Program, requiring facilities using CF4 and C2F6 to monitor and report emissions with high precision. The American Innovation and Manufacturing Act of 2020 further restricts the production and consumption of high-GWP substances, creating market pressures that favor compounds with lower environmental impact profiles.
Asian markets have developed distinct regulatory approaches, with Japan implementing voluntary reduction targets through industry partnerships, while China has integrated fluorocarbon controls into its national carbon neutrality strategy. These varying regulatory intensities create regional disparities in CF4 versus C2F6 adoption patterns, influencing global supply chains and technology transfer decisions.
Emerging regulations focus on lifecycle assessment requirements, mandating comprehensive evaluation of environmental impacts from production through disposal. This regulatory trend particularly affects the comparative assessment of CF4 and C2F6, as their different atmospheric lifespans and usage efficiencies translate into varying compliance costs and strategic implications for industrial users seeking long-term regulatory certainty.
The Montreal Protocol, while primarily targeting ozone-depleting substances, established the foundational framework for international cooperation on atmospheric protection. Subsequently, the Kyoto Protocol specifically identified perfluorocarbons including CF4 and C2F6 as greenhouse gases requiring emission reduction commitments. The Paris Agreement further strengthened these obligations, with many signatory nations incorporating fluorocarbon emission targets into their Nationally Determined Contributions.
Regional regulations have become increasingly sophisticated in addressing fluorocarbon emissions. The European Union's F-Gas Regulation imposes strict quotas on high-GWP substances, with CF4 and C2F6 subject to phase-down schedules and usage restrictions in specific applications. The regulation mandates leak detection systems, regular inspections, and certified personnel for handling these compounds, significantly impacting operational costs and efficiency considerations.
In the United States, the Environmental Protection Agency has implemented comprehensive reporting requirements under the Greenhouse Gas Reporting Program, requiring facilities using CF4 and C2F6 to monitor and report emissions with high precision. The American Innovation and Manufacturing Act of 2020 further restricts the production and consumption of high-GWP substances, creating market pressures that favor compounds with lower environmental impact profiles.
Asian markets have developed distinct regulatory approaches, with Japan implementing voluntary reduction targets through industry partnerships, while China has integrated fluorocarbon controls into its national carbon neutrality strategy. These varying regulatory intensities create regional disparities in CF4 versus C2F6 adoption patterns, influencing global supply chains and technology transfer decisions.
Emerging regulations focus on lifecycle assessment requirements, mandating comprehensive evaluation of environmental impacts from production through disposal. This regulatory trend particularly affects the comparative assessment of CF4 and C2F6, as their different atmospheric lifespans and usage efficiencies translate into varying compliance costs and strategic implications for industrial users seeking long-term regulatory certainty.
Lifecycle Assessment and Sustainability Analysis
The lifecycle assessment of CF4 and C2F6 reveals significant differences in their environmental impact profiles throughout their operational lifespans. CF4 demonstrates exceptional chemical stability with an atmospheric lifetime of approximately 50,000 years, while C2F6 exhibits a shorter but still substantial lifetime of around 10,000 years. This fundamental difference creates cascading effects across multiple sustainability metrics, influencing both immediate operational efficiency and long-term environmental stewardship considerations.
From a carbon footprint perspective, both compounds present challenges due to their high global warming potential values. CF4 carries a GWP of 7,390 over a 100-year timeframe, whereas C2F6 exhibits an even higher GWP of 12,200. However, the extended atmospheric persistence of CF4 means that emissions today will continue contributing to climate change for millennia, creating intergenerational environmental debt that must be factored into comprehensive sustainability assessments.
Resource utilization efficiency analysis indicates that C2F6 generally requires lower operational volumes to achieve equivalent processing results in semiconductor manufacturing applications. This translates to reduced raw material consumption per unit of production output, potentially offsetting some of its higher GWP impact through decreased absolute emission quantities. The manufacturing energy requirements for both compounds are comparable, though C2F6 production involves slightly more complex synthesis pathways.
End-of-life management presents distinct challenges for each compound. CF4's extreme stability makes it virtually impossible to decompose through conventional waste treatment methods, necessitating specialized abatement technologies such as plasma destruction or catalytic reduction systems. C2F6, while still requiring advanced treatment, can be processed through thermal destruction at lower energy inputs, improving overall waste management sustainability.
The circular economy potential differs markedly between these compounds. CF4's longevity in industrial systems allows for potential recovery and reuse applications, though current recycling infrastructure remains limited. C2F6's shorter operational lifespan but higher processing efficiency creates opportunities for optimized usage protocols that minimize waste generation while maintaining production quality standards.
Regulatory compliance costs increasingly favor compounds with lower environmental persistence. Carbon pricing mechanisms and emerging fluorinated gas regulations create economic incentives for transitioning toward shorter-lived alternatives, making C2F6 potentially more sustainable from a regulatory risk perspective despite its higher immediate GWP impact.
From a carbon footprint perspective, both compounds present challenges due to their high global warming potential values. CF4 carries a GWP of 7,390 over a 100-year timeframe, whereas C2F6 exhibits an even higher GWP of 12,200. However, the extended atmospheric persistence of CF4 means that emissions today will continue contributing to climate change for millennia, creating intergenerational environmental debt that must be factored into comprehensive sustainability assessments.
Resource utilization efficiency analysis indicates that C2F6 generally requires lower operational volumes to achieve equivalent processing results in semiconductor manufacturing applications. This translates to reduced raw material consumption per unit of production output, potentially offsetting some of its higher GWP impact through decreased absolute emission quantities. The manufacturing energy requirements for both compounds are comparable, though C2F6 production involves slightly more complex synthesis pathways.
End-of-life management presents distinct challenges for each compound. CF4's extreme stability makes it virtually impossible to decompose through conventional waste treatment methods, necessitating specialized abatement technologies such as plasma destruction or catalytic reduction systems. C2F6, while still requiring advanced treatment, can be processed through thermal destruction at lower energy inputs, improving overall waste management sustainability.
The circular economy potential differs markedly between these compounds. CF4's longevity in industrial systems allows for potential recovery and reuse applications, though current recycling infrastructure remains limited. C2F6's shorter operational lifespan but higher processing efficiency creates opportunities for optimized usage protocols that minimize waste generation while maintaining production quality standards.
Regulatory compliance costs increasingly favor compounds with lower environmental persistence. Carbon pricing mechanisms and emerging fluorinated gas regulations create economic incentives for transitioning toward shorter-lived alternatives, making C2F6 potentially more sustainable from a regulatory risk perspective despite its higher immediate GWP impact.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!