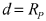Comparative Study: Li-S vs Sodium-Ion Battery Technologies
OCT 24, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Battery Technology Evolution and Objectives
The evolution of battery technology has witnessed significant advancements since the commercialization of lithium-ion batteries in the early 1990s. These developments have been primarily driven by increasing demands for higher energy density, longer cycle life, improved safety, and reduced costs across various applications ranging from portable electronics to electric vehicles and grid-scale energy storage systems.
Traditional lithium-ion batteries, while revolutionary, face inherent limitations in energy density, resource constraints, and safety concerns. This has prompted researchers and industry stakeholders to explore alternative battery chemistries with enhanced performance characteristics and sustainability profiles. Among these emerging technologies, lithium-sulfur (Li-S) and sodium-ion batteries represent promising candidates that could potentially address the limitations of conventional lithium-ion systems.
Li-S battery technology has garnered substantial attention due to its theoretical energy density of approximately 2,600 Wh/kg, which significantly exceeds the theoretical limits of lithium-ion batteries (around 350-400 Wh/kg). This remarkable energy density potential stems from sulfur's high theoretical capacity (1,675 mAh/g) and natural abundance, positioning Li-S batteries as potential game-changers for applications requiring high gravimetric energy density, such as electric aviation and advanced portable electronics.
Concurrently, sodium-ion battery technology has emerged as a compelling alternative, primarily motivated by sodium's abundance (approximately 1,000 times more abundant than lithium in the Earth's crust) and more favorable geographical distribution. While sodium-ion batteries typically offer lower energy densities compared to lithium-ion counterparts, recent advancements have narrowed this gap considerably, with some prototypes achieving energy densities of 150-160 Wh/kg.
The technical objectives for advancing both Li-S and sodium-ion technologies share common themes: overcoming cycle life limitations, improving rate capability, enhancing operational stability, and developing scalable manufacturing processes. For Li-S batteries, specific challenges include mitigating polysulfide shuttle effects and managing volume expansion during cycling. For sodium-ion batteries, key objectives include developing high-performance electrode materials compatible with sodium ions and optimizing electrolyte formulations.
Industry projections suggest that by 2030, Li-S batteries could achieve practical energy densities exceeding 500 Wh/kg with cycle lives approaching 1,000 cycles, while sodium-ion batteries might reach energy densities of 200 Wh/kg with comparable cycle performance to current lithium-ion technologies. These advancements would position both technologies as viable complements or alternatives to conventional lithium-ion batteries in specific application domains.
The comparative study of these technologies aims to evaluate their respective strengths, limitations, and potential application spaces, providing strategic insights for research prioritization, investment decisions, and technology roadmapping in the evolving landscape of electrochemical energy storage solutions.
Traditional lithium-ion batteries, while revolutionary, face inherent limitations in energy density, resource constraints, and safety concerns. This has prompted researchers and industry stakeholders to explore alternative battery chemistries with enhanced performance characteristics and sustainability profiles. Among these emerging technologies, lithium-sulfur (Li-S) and sodium-ion batteries represent promising candidates that could potentially address the limitations of conventional lithium-ion systems.
Li-S battery technology has garnered substantial attention due to its theoretical energy density of approximately 2,600 Wh/kg, which significantly exceeds the theoretical limits of lithium-ion batteries (around 350-400 Wh/kg). This remarkable energy density potential stems from sulfur's high theoretical capacity (1,675 mAh/g) and natural abundance, positioning Li-S batteries as potential game-changers for applications requiring high gravimetric energy density, such as electric aviation and advanced portable electronics.
Concurrently, sodium-ion battery technology has emerged as a compelling alternative, primarily motivated by sodium's abundance (approximately 1,000 times more abundant than lithium in the Earth's crust) and more favorable geographical distribution. While sodium-ion batteries typically offer lower energy densities compared to lithium-ion counterparts, recent advancements have narrowed this gap considerably, with some prototypes achieving energy densities of 150-160 Wh/kg.
The technical objectives for advancing both Li-S and sodium-ion technologies share common themes: overcoming cycle life limitations, improving rate capability, enhancing operational stability, and developing scalable manufacturing processes. For Li-S batteries, specific challenges include mitigating polysulfide shuttle effects and managing volume expansion during cycling. For sodium-ion batteries, key objectives include developing high-performance electrode materials compatible with sodium ions and optimizing electrolyte formulations.
Industry projections suggest that by 2030, Li-S batteries could achieve practical energy densities exceeding 500 Wh/kg with cycle lives approaching 1,000 cycles, while sodium-ion batteries might reach energy densities of 200 Wh/kg with comparable cycle performance to current lithium-ion technologies. These advancements would position both technologies as viable complements or alternatives to conventional lithium-ion batteries in specific application domains.
The comparative study of these technologies aims to evaluate their respective strengths, limitations, and potential application spaces, providing strategic insights for research prioritization, investment decisions, and technology roadmapping in the evolving landscape of electrochemical energy storage solutions.
Market Demand Analysis for Next-Gen Batteries
The global battery market is experiencing unprecedented growth driven by the rapid expansion of electric vehicles (EVs), renewable energy storage systems, and portable electronics. Current projections indicate the advanced battery market will reach $168 billion by 2030, with a compound annual growth rate exceeding 18% between 2023-2030. This remarkable growth trajectory creates an urgent demand for next-generation battery technologies that overcome the limitations of traditional lithium-ion batteries.
Market analysis reveals several critical pain points in current battery technologies that next-generation solutions must address. Energy density limitations restrict EV range capabilities, with consumers consistently ranking "range anxiety" as a primary barrier to adoption. Cost factors remain significant, with battery packs still representing 30-40% of total EV manufacturing costs despite recent price reductions. Additionally, safety concerns persist following high-profile thermal runaway incidents, while sustainability issues regarding raw material sourcing and end-of-life recycling face increasing regulatory scrutiny.
Both lithium-sulfur (Li-S) and sodium-ion technologies are positioned to address these market demands, albeit with different strengths. Li-S batteries attract significant interest from aerospace, defense, and premium EV segments due to their theoretical energy density of 2,600 Wh/kg—nearly five times that of conventional lithium-ion batteries. Market research indicates the Li-S battery segment could capture 5-8% of the specialty battery market by 2028, particularly in applications where weight reduction justifies premium pricing.
Sodium-ion batteries, conversely, target cost-sensitive market segments including grid storage, entry-level EVs, and developing markets. With sodium resources being 1,000 times more abundant than lithium and more geographically distributed, these batteries address growing concerns about lithium supply chain vulnerabilities. Industry forecasts suggest sodium-ion technology could capture 10-15% of the stationary storage market by 2030, with particular strength in regions facing lithium access challenges.
Regional market analysis shows differentiated demand patterns. European markets prioritize sustainability and circular economy principles, creating favorable conditions for sodium-ion adoption. Asian markets, particularly China, are investing heavily in both technologies, with CATL's recent sodium-ion battery announcements signaling serious commercial intent. North American markets show strong interest in Li-S technology for defense applications and premium EVs, while expressing growing concern about critical mineral dependencies.
Consumer and industrial surveys indicate willingness to adopt these emerging technologies provided they deliver meaningful improvements in specific applications. For Li-S, the primary market pull comes from applications where weight reduction creates substantial value. For sodium-ion, price sensitivity and supply chain resilience drive market interest, particularly in grid-scale implementations where energy density is less critical than lifetime cost considerations.
Market analysis reveals several critical pain points in current battery technologies that next-generation solutions must address. Energy density limitations restrict EV range capabilities, with consumers consistently ranking "range anxiety" as a primary barrier to adoption. Cost factors remain significant, with battery packs still representing 30-40% of total EV manufacturing costs despite recent price reductions. Additionally, safety concerns persist following high-profile thermal runaway incidents, while sustainability issues regarding raw material sourcing and end-of-life recycling face increasing regulatory scrutiny.
Both lithium-sulfur (Li-S) and sodium-ion technologies are positioned to address these market demands, albeit with different strengths. Li-S batteries attract significant interest from aerospace, defense, and premium EV segments due to their theoretical energy density of 2,600 Wh/kg—nearly five times that of conventional lithium-ion batteries. Market research indicates the Li-S battery segment could capture 5-8% of the specialty battery market by 2028, particularly in applications where weight reduction justifies premium pricing.
Sodium-ion batteries, conversely, target cost-sensitive market segments including grid storage, entry-level EVs, and developing markets. With sodium resources being 1,000 times more abundant than lithium and more geographically distributed, these batteries address growing concerns about lithium supply chain vulnerabilities. Industry forecasts suggest sodium-ion technology could capture 10-15% of the stationary storage market by 2030, with particular strength in regions facing lithium access challenges.
Regional market analysis shows differentiated demand patterns. European markets prioritize sustainability and circular economy principles, creating favorable conditions for sodium-ion adoption. Asian markets, particularly China, are investing heavily in both technologies, with CATL's recent sodium-ion battery announcements signaling serious commercial intent. North American markets show strong interest in Li-S technology for defense applications and premium EVs, while expressing growing concern about critical mineral dependencies.
Consumer and industrial surveys indicate willingness to adopt these emerging technologies provided they deliver meaningful improvements in specific applications. For Li-S, the primary market pull comes from applications where weight reduction creates substantial value. For sodium-ion, price sensitivity and supply chain resilience drive market interest, particularly in grid-scale implementations where energy density is less critical than lifetime cost considerations.
Current Status and Technical Barriers
Lithium-sulfur (Li-S) and sodium-ion battery technologies represent two promising alternatives to conventional lithium-ion batteries, each at different stages of development and facing unique technical challenges. Li-S batteries have progressed significantly in laboratory settings, with theoretical energy densities reaching up to 2,600 Wh/kg, substantially higher than current lithium-ion technologies. However, commercial deployment remains limited due to persistent technical barriers.
The primary challenge for Li-S batteries involves the "shuttle effect," where soluble polysulfide intermediates migrate between electrodes during cycling, causing rapid capacity fading and shortened battery life. Current Li-S prototypes typically achieve only 300-500 cycles before significant degradation occurs, compared to 1,000+ cycles for commercial lithium-ion batteries. Additionally, Li-S batteries suffer from low coulombic efficiency (70-80%) and poor rate capability, limiting their practical applications.
Sodium-ion battery technology has advanced considerably in recent years, with several companies announcing pilot production lines. These batteries offer theoretical energy densities of 300-400 Wh/kg, lower than Li-S but competitive with some lithium-ion chemistries. The technology benefits from abundant sodium resources, potentially reducing material costs by 30-40% compared to lithium-based systems.
Technical barriers for sodium-ion batteries include electrode material stability issues, with current cathode materials experiencing structural degradation during repeated sodium insertion/extraction. The larger ionic radius of sodium (1.02Å vs. 0.76Å for lithium) creates challenges for intercalation chemistry, resulting in lower energy density and power capability. Current sodium-ion prototypes demonstrate cycle life of 2,000-3,000 cycles but at lower depth of discharge than commercial requirements.
Geographically, Li-S research is concentrated in North America and Europe, with significant advancements from research institutions in the United States, Germany, and the UK. Companies like OXIS Energy (UK) and Sion Power (US) have made notable progress toward commercialization. Sodium-ion battery development shows stronger presence in Asia, particularly China, where companies like CATL and HiNa Battery have established pilot production facilities.
Both technologies face manufacturing scalability challenges. Li-S batteries require specialized production environments to handle elemental sulfur and prevent moisture contamination. Sodium-ion batteries face fewer manufacturing hurdles but require redesigned production equipment compared to lithium-ion manufacturing lines. Current production costs remain 2-3 times higher than established lithium-ion technologies, presenting a significant barrier to market entry despite promising performance characteristics.
The primary challenge for Li-S batteries involves the "shuttle effect," where soluble polysulfide intermediates migrate between electrodes during cycling, causing rapid capacity fading and shortened battery life. Current Li-S prototypes typically achieve only 300-500 cycles before significant degradation occurs, compared to 1,000+ cycles for commercial lithium-ion batteries. Additionally, Li-S batteries suffer from low coulombic efficiency (70-80%) and poor rate capability, limiting their practical applications.
Sodium-ion battery technology has advanced considerably in recent years, with several companies announcing pilot production lines. These batteries offer theoretical energy densities of 300-400 Wh/kg, lower than Li-S but competitive with some lithium-ion chemistries. The technology benefits from abundant sodium resources, potentially reducing material costs by 30-40% compared to lithium-based systems.
Technical barriers for sodium-ion batteries include electrode material stability issues, with current cathode materials experiencing structural degradation during repeated sodium insertion/extraction. The larger ionic radius of sodium (1.02Å vs. 0.76Å for lithium) creates challenges for intercalation chemistry, resulting in lower energy density and power capability. Current sodium-ion prototypes demonstrate cycle life of 2,000-3,000 cycles but at lower depth of discharge than commercial requirements.
Geographically, Li-S research is concentrated in North America and Europe, with significant advancements from research institutions in the United States, Germany, and the UK. Companies like OXIS Energy (UK) and Sion Power (US) have made notable progress toward commercialization. Sodium-ion battery development shows stronger presence in Asia, particularly China, where companies like CATL and HiNa Battery have established pilot production facilities.
Both technologies face manufacturing scalability challenges. Li-S batteries require specialized production environments to handle elemental sulfur and prevent moisture contamination. Sodium-ion batteries face fewer manufacturing hurdles but require redesigned production equipment compared to lithium-ion manufacturing lines. Current production costs remain 2-3 times higher than established lithium-ion technologies, presenting a significant barrier to market entry despite promising performance characteristics.
Current Technical Solutions Comparison
01 Electrode materials for Li-S batteries
Various materials can be used as electrodes in lithium-sulfur batteries to improve performance. These include carbon-based materials, metal oxides, and composite structures that can enhance sulfur utilization and prevent polysulfide shuttling. The electrode design focuses on increasing conductivity, providing sufficient space for sulfur expansion, and maintaining structural integrity during cycling, which ultimately leads to higher capacity and longer cycle life.- Electrode materials for Li-S batteries: Various electrode materials can be used in lithium-sulfur batteries to improve performance. These materials include carbon-based structures, metal oxides, and composite materials that can enhance sulfur utilization, reduce polysulfide shuttling, and improve cycle life. The electrode design often focuses on containing sulfur within conductive frameworks to maintain electrical contact and prevent capacity loss during cycling.
- Electrolyte compositions for sodium-ion batteries: Specialized electrolyte formulations are crucial for sodium-ion batteries to ensure efficient sodium ion transport and electrode stability. These electrolytes typically contain sodium salts dissolved in organic solvents, with additives to form stable solid electrolyte interphase layers. The composition can significantly affect battery performance metrics such as capacity retention, rate capability, and operating temperature range.
- Cathode materials for dual-ion battery systems: Advanced cathode materials are being developed for both Li-S and sodium-ion battery systems. These materials often incorporate layered structures, polyanionic compounds, or transition metal oxides that can accommodate the insertion and extraction of either lithium or sodium ions. The design focuses on maintaining structural stability during repeated cycling while providing high energy density and power output.
- Separator technologies for preventing polysulfide shuttling: Specialized separator technologies are essential in Li-S batteries to prevent the polysulfide shuttling effect, which causes capacity fading. These separators often incorporate functional coatings, nanomaterials, or polymer modifications that can physically or chemically interact with polysulfides to prevent their migration between electrodes. This technology is critical for extending the cycle life of Li-S batteries.
- Anode protection strategies for alkali metal batteries: Various strategies are employed to protect anodes in both Li-S and sodium-ion batteries from degradation mechanisms. These include artificial solid electrolyte interphase formation, protective coatings, and structural modifications to accommodate volume changes during cycling. For sodium-ion batteries, carbon-based materials are often used instead of metallic sodium to improve safety and cycling stability.
02 Electrolyte compositions for sodium-ion batteries
Specialized electrolyte formulations for sodium-ion batteries can significantly impact battery performance. These electrolytes typically contain sodium salts dissolved in organic solvents, sometimes with additives to form stable solid electrolyte interphases. The composition of the electrolyte affects ionic conductivity, electrode-electrolyte interface stability, and overall battery safety, which are crucial factors for developing high-performance sodium-ion energy storage systems.Expand Specific Solutions03 Cathode protection strategies for Li-S batteries
Various approaches are employed to protect sulfur cathodes in Li-S batteries from degradation. These include encapsulation techniques, protective coatings, and the use of interlayers or functional separators that can trap polysulfides. These protection strategies aim to prevent the dissolution of active material, reduce the shuttle effect, and maintain the structural integrity of the cathode during cycling, thereby extending battery life and improving energy density.Expand Specific Solutions04 Anode materials for sodium-ion batteries
Innovative anode materials for sodium-ion batteries focus on accommodating the larger sodium ions compared to lithium. These materials include hard carbons, alloys, phosphorus-based compounds, and titanium-based oxides. The design of these anodes aims to provide sufficient sodium storage capacity, good rate capability, and structural stability during repeated sodium insertion and extraction, which are essential for developing cost-effective and sustainable energy storage alternatives to lithium-ion batteries.Expand Specific Solutions05 Hybrid and dual-ion battery systems
Hybrid battery systems that combine features of both Li-S and sodium-ion technologies offer unique advantages. These systems may utilize dual-ion mechanisms, shared components, or complementary electrode materials to achieve improved energy density, power capability, or cycling stability. Such hybrid approaches can potentially overcome limitations of individual battery chemistries while leveraging their respective strengths, leading to more versatile energy storage solutions for various applications.Expand Specific Solutions
Key Industry Players and Competitive Landscape
The lithium-sulfur (Li-S) and sodium-ion battery technology landscape is currently in a transitional phase, moving from research to early commercialization. The market is projected to grow significantly as industries seek alternatives to traditional lithium-ion batteries, with sodium-ion technology gaining momentum due to abundant raw materials and lower costs. Companies like Faradion Ltd. and Broadbit Batteries Oy are pioneering sodium-ion solutions, while academic institutions including MIT, Cornell, and Monash University are advancing Li-S technology. Research organizations such as CNRS and industrial players including Hyundai Motor Co. and Phillips 66 are investing in both technologies, with Chinese manufacturers like Guoxuan High-Tech and Jiangsu Zenergy positioning themselves as key players in scaling production capabilities for future market adoption.
Faradion Ltd.
Technical Solution: Faradion has pioneered sodium-ion battery technology as a sustainable alternative to lithium-ion batteries. Their proprietary technology utilizes sodium salts that are abundant and widely available, making them significantly more cost-effective than lithium-based alternatives. Faradion's approach involves using hard carbon anodes and layered oxide cathodes (typically Na₂/₃[Fe₁/₂Mn₁/₂]O₂), optimized for high energy density applications. Their cells deliver energy densities of approximately 140-160 Wh/kg, which while lower than top-tier Li-ion cells, represents industry-leading performance for sodium-ion technology. The company has developed a drop-in manufacturing process compatible with existing lithium-ion production lines, allowing for rapid scalability without significant capital investment. Faradion's batteries demonstrate excellent thermal stability and safety characteristics, operating effectively across a wider temperature range (-30°C to +60°C) than many lithium-ion alternatives.
Strengths: Cost-effective (30-40% lower cost than Li-ion), uses abundant materials, compatible with existing manufacturing infrastructure, superior safety profile with no thermal runaway, and excellent low-temperature performance. Weaknesses: Lower energy density compared to advanced Li-ion and Li-S systems, still facing challenges in cycle life optimization for certain applications, and relatively early in commercial-scale deployment.
Massachusetts Institute of Technology
Technical Solution: MIT has developed groundbreaking research in both Li-S and sodium-ion battery technologies. For Li-S batteries, MIT researchers have created a novel electrode structure using a hierarchical arrangement of carbon nanotubes coated with lithium sulfide particles, addressing the polysulfide shuttle effect that typically degrades Li-S performance. This architecture effectively traps polysulfides while maintaining high sulfur utilization, achieving energy densities approaching 500 Wh/kg with improved cycle stability. In the sodium-ion domain, MIT has pioneered the development of Prussian Blue analogs as cathode materials, offering a framework that can accommodate sodium ions with minimal structural strain. Their research has demonstrated sodium-ion cells with energy densities of approximately 150 Wh/kg and cycle life exceeding 2,000 cycles. MIT's comparative studies have highlighted the complementary nature of these technologies, suggesting Li-S for high-energy applications and sodium-ion for cost-sensitive, high-power applications.
Strengths: World-class fundamental research capabilities, innovative approaches to solving key technical challenges in both technologies, strong industry partnerships for technology transfer, and comprehensive understanding of materials science aspects. Weaknesses: Technologies still primarily at laboratory scale rather than commercial production, requiring further development for manufacturing scalability, and facing competition from established battery manufacturers with existing production infrastructure.
Core Patents and Research Breakthroughs
Lithium/dissolved polysulfide rechargeable lithium- sulfur batteries and methods of making the same
PatentWO2014164595A1
Innovation
- A rechargeable lithium-sulfur battery design featuring a binder-free, self-weaving fibrous carbon cathode with a polysulfide catholyte containing high-order polysulfides, where amorphous microparticles of elemental sulfur or lithium sulfide form on the cathode, facilitating charge transport and trapping polysulfides within the carbon structure, eliminating the need for additional conductivity enhancers.
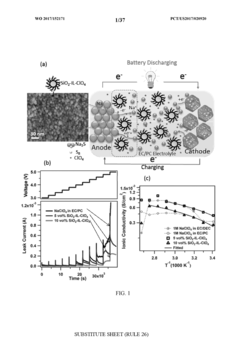
Stable room-temperature sodium-sulfur battery
PatentWO2017152171A1
Innovation
- A sodium-ion conducting battery design featuring a microporous and mesoporous carbon-sulfur composite cathode and a liquid carbonate electrolyte with an ionic liquid tethered to silica nanoparticles, which stabilizes the sodium anode and confines sulfur within the carbon pores, enabling a solid-state electrochemical reaction and preventing the formation of soluble polysulfides.
Environmental Impact Assessment
The environmental impact of battery technologies has become a critical consideration in the transition to sustainable energy systems. When comparing Li-S (Lithium-Sulfur) and Sodium-ion battery technologies, several environmental factors must be evaluated across their entire lifecycle.
Li-S batteries offer significant environmental advantages in terms of raw material sourcing. Sulfur is an abundant by-product of petroleum refining, effectively repurposing what would otherwise be industrial waste. This contrasts favorably with traditional lithium-ion batteries that require cobalt and nickel, materials associated with concerning mining practices and limited availability.
However, Li-S technology faces environmental challenges related to the polysulfide shuttle effect, which can lead to reduced cycle life and potentially more frequent battery replacement. This shorter lifespan could increase the overall environmental footprint through additional manufacturing and disposal requirements.
Sodium-ion batteries present a compelling environmental case through their use of abundant, widely distributed raw materials. Sodium is approximately 1,000 times more abundant than lithium in the Earth's crust, and its extraction typically has lower environmental impact. The technology also commonly utilizes aluminum rather than copper for current collectors, reducing both cost and environmental burden.
Manufacturing processes for both technologies demonstrate different environmental profiles. Current Li-S production methods often require energy-intensive processes and specialized environments. Sodium-ion manufacturing can leverage existing lithium-ion production infrastructure with modifications, potentially reducing the environmental impact of transitioning to this technology.
End-of-life considerations reveal that Li-S batteries may offer advantages in recycling potential, as sulfur is relatively easy to recover and repurpose. Sodium-ion batteries, while theoretically recyclable, are still developing efficient recycling protocols as the technology matures.
Carbon footprint analysis indicates that both technologies could significantly reduce greenhouse gas emissions compared to conventional lithium-ion batteries, particularly when considering full lifecycle assessments. Sodium-ion batteries may have a slight edge due to less energy-intensive material extraction and processing requirements.
Water usage and pollution risks differ between the technologies, with sodium extraction typically requiring less water than lithium brine operations. However, both technologies must address potential environmental risks from electrolyte leakage and improper disposal.
The environmental impact assessment must also consider scalability factors, as the ability to deploy these technologies at grid scale will determine their ultimate contribution to reducing global carbon emissions from energy storage applications.
Li-S batteries offer significant environmental advantages in terms of raw material sourcing. Sulfur is an abundant by-product of petroleum refining, effectively repurposing what would otherwise be industrial waste. This contrasts favorably with traditional lithium-ion batteries that require cobalt and nickel, materials associated with concerning mining practices and limited availability.
However, Li-S technology faces environmental challenges related to the polysulfide shuttle effect, which can lead to reduced cycle life and potentially more frequent battery replacement. This shorter lifespan could increase the overall environmental footprint through additional manufacturing and disposal requirements.
Sodium-ion batteries present a compelling environmental case through their use of abundant, widely distributed raw materials. Sodium is approximately 1,000 times more abundant than lithium in the Earth's crust, and its extraction typically has lower environmental impact. The technology also commonly utilizes aluminum rather than copper for current collectors, reducing both cost and environmental burden.
Manufacturing processes for both technologies demonstrate different environmental profiles. Current Li-S production methods often require energy-intensive processes and specialized environments. Sodium-ion manufacturing can leverage existing lithium-ion production infrastructure with modifications, potentially reducing the environmental impact of transitioning to this technology.
End-of-life considerations reveal that Li-S batteries may offer advantages in recycling potential, as sulfur is relatively easy to recover and repurpose. Sodium-ion batteries, while theoretically recyclable, are still developing efficient recycling protocols as the technology matures.
Carbon footprint analysis indicates that both technologies could significantly reduce greenhouse gas emissions compared to conventional lithium-ion batteries, particularly when considering full lifecycle assessments. Sodium-ion batteries may have a slight edge due to less energy-intensive material extraction and processing requirements.
Water usage and pollution risks differ between the technologies, with sodium extraction typically requiring less water than lithium brine operations. However, both technologies must address potential environmental risks from electrolyte leakage and improper disposal.
The environmental impact assessment must also consider scalability factors, as the ability to deploy these technologies at grid scale will determine their ultimate contribution to reducing global carbon emissions from energy storage applications.
Supply Chain Resilience
The supply chain resilience for battery technologies represents a critical factor in their commercial viability and widespread adoption. When comparing Li-S and sodium-ion battery technologies, significant differences emerge in their respective supply chain structures and vulnerabilities.
Lithium-sulfur batteries rely heavily on lithium metal, which faces considerable geopolitical constraints. Currently, lithium extraction is concentrated in the "Lithium Triangle" of South America (Chile, Argentina, and Bolivia) and Australia, creating potential bottlenecks. However, the sulfur component offers a distinct advantage as it is abundantly available as a byproduct of petroleum refining, providing cost benefits and supply stability.
Sodium-ion batteries present a compelling alternative from a supply chain perspective. Sodium is the sixth most abundant element in the Earth's crust, with reserves widely distributed globally. This geographical diversity significantly reduces geopolitical risks associated with resource concentration. The extraction and processing of sodium compounds generally require less specialized equipment and fewer environmental controls than lithium extraction, potentially enabling more localized production.
Both technologies face challenges in scaling manufacturing processes. Li-S batteries require specialized handling of lithium metal and management of polysulfide shuttling issues, necessitating advanced manufacturing capabilities. Sodium-ion production, while theoretically simpler, still requires development of standardized manufacturing protocols as the technology matures.
The cathode materials represent another point of divergence. Li-S batteries utilize sulfur, which is inexpensive and abundant. In contrast, sodium-ion batteries often employ materials like sodium iron phosphate or sodium manganese oxide, which require established supply chains similar to those of lithium-ion batteries but with potentially fewer critical material constraints.
From a resilience perspective, sodium-ion technology offers greater insulation from supply disruptions due to its reliance on more widely available materials. However, Li-S technology benefits from leveraging portions of existing lithium-ion manufacturing infrastructure, potentially accelerating deployment despite supply constraints for lithium itself.
Recent global events, including pandemic-related disruptions and increasing trade tensions, have highlighted the importance of supply chain diversification in battery technologies. Both Li-S and sodium-ion technologies could contribute to reducing dependency on single-source materials and geographically concentrated supply chains that currently characterize conventional lithium-ion batteries.
Lithium-sulfur batteries rely heavily on lithium metal, which faces considerable geopolitical constraints. Currently, lithium extraction is concentrated in the "Lithium Triangle" of South America (Chile, Argentina, and Bolivia) and Australia, creating potential bottlenecks. However, the sulfur component offers a distinct advantage as it is abundantly available as a byproduct of petroleum refining, providing cost benefits and supply stability.
Sodium-ion batteries present a compelling alternative from a supply chain perspective. Sodium is the sixth most abundant element in the Earth's crust, with reserves widely distributed globally. This geographical diversity significantly reduces geopolitical risks associated with resource concentration. The extraction and processing of sodium compounds generally require less specialized equipment and fewer environmental controls than lithium extraction, potentially enabling more localized production.
Both technologies face challenges in scaling manufacturing processes. Li-S batteries require specialized handling of lithium metal and management of polysulfide shuttling issues, necessitating advanced manufacturing capabilities. Sodium-ion production, while theoretically simpler, still requires development of standardized manufacturing protocols as the technology matures.
The cathode materials represent another point of divergence. Li-S batteries utilize sulfur, which is inexpensive and abundant. In contrast, sodium-ion batteries often employ materials like sodium iron phosphate or sodium manganese oxide, which require established supply chains similar to those of lithium-ion batteries but with potentially fewer critical material constraints.
From a resilience perspective, sodium-ion technology offers greater insulation from supply disruptions due to its reliance on more widely available materials. However, Li-S technology benefits from leveraging portions of existing lithium-ion manufacturing infrastructure, potentially accelerating deployment despite supply constraints for lithium itself.
Recent global events, including pandemic-related disruptions and increasing trade tensions, have highlighted the importance of supply chain diversification in battery technologies. Both Li-S and sodium-ion technologies could contribute to reducing dependency on single-source materials and geographically concentrated supply chains that currently characterize conventional lithium-ion batteries.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!