Comparing Chrome Plating and Electroless Nickel for Conductivity
APR 8, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Chrome Plating vs Electroless Nickel Background and Objectives
Surface finishing technologies have evolved significantly over the past century, with chrome plating and electroless nickel emerging as two dominant coating solutions for enhancing electrical conductivity in industrial applications. Chrome plating, developed in the early 1900s, revolutionized the automotive and aerospace industries by providing superior hardness and corrosion resistance. Meanwhile, electroless nickel coating, introduced in the 1940s, offered unique advantages through its chemical deposition process, eliminating the need for external electrical current.
The evolution of these technologies has been driven by increasing demands for reliable electrical connections in electronic devices, telecommunications equipment, and precision instruments. As miniaturization trends accelerate and performance requirements become more stringent, the selection between chrome plating and electroless nickel has become increasingly critical for manufacturers seeking optimal conductivity solutions.
Chrome plating technology has progressed from decorative applications to functional coatings, with hexavalent and trivalent chromium processes offering different performance characteristics. The technology has adapted to environmental regulations while maintaining its reputation for exceptional durability and low contact resistance. Recent developments focus on improving throwing power and reducing environmental impact through advanced electrolyte formulations.
Electroless nickel technology has similarly advanced, with phosphorus and boron-based alloys providing varying degrees of conductivity and corrosion protection. The self-catalytic nature of electroless nickel deposition enables uniform coating thickness on complex geometries, making it particularly valuable for intricate electronic components where consistent electrical performance is paramount.
The primary objective of comparing these technologies centers on establishing clear performance benchmarks for electrical conductivity applications. This evaluation aims to determine optimal coating selection criteria based on substrate materials, operating environments, and specific conductivity requirements. Understanding the fundamental differences in deposition mechanisms, microstructure formation, and resulting electrical properties enables informed decision-making for critical applications.
Secondary objectives include assessing long-term stability, cost-effectiveness, and environmental compliance factors that influence technology adoption. The comparison seeks to identify application-specific advantages and limitations, providing guidance for engineers selecting appropriate coating technologies for next-generation electronic systems and high-performance electrical connections.
The evolution of these technologies has been driven by increasing demands for reliable electrical connections in electronic devices, telecommunications equipment, and precision instruments. As miniaturization trends accelerate and performance requirements become more stringent, the selection between chrome plating and electroless nickel has become increasingly critical for manufacturers seeking optimal conductivity solutions.
Chrome plating technology has progressed from decorative applications to functional coatings, with hexavalent and trivalent chromium processes offering different performance characteristics. The technology has adapted to environmental regulations while maintaining its reputation for exceptional durability and low contact resistance. Recent developments focus on improving throwing power and reducing environmental impact through advanced electrolyte formulations.
Electroless nickel technology has similarly advanced, with phosphorus and boron-based alloys providing varying degrees of conductivity and corrosion protection. The self-catalytic nature of electroless nickel deposition enables uniform coating thickness on complex geometries, making it particularly valuable for intricate electronic components where consistent electrical performance is paramount.
The primary objective of comparing these technologies centers on establishing clear performance benchmarks for electrical conductivity applications. This evaluation aims to determine optimal coating selection criteria based on substrate materials, operating environments, and specific conductivity requirements. Understanding the fundamental differences in deposition mechanisms, microstructure formation, and resulting electrical properties enables informed decision-making for critical applications.
Secondary objectives include assessing long-term stability, cost-effectiveness, and environmental compliance factors that influence technology adoption. The comparison seeks to identify application-specific advantages and limitations, providing guidance for engineers selecting appropriate coating technologies for next-generation electronic systems and high-performance electrical connections.
Market Demand for High-Conductivity Surface Coatings
The global market for high-conductivity surface coatings has experienced substantial growth driven by the expanding electronics industry, automotive sector electrification, and increasing demand for reliable electrical connections. Traditional chrome plating and electroless nickel coatings represent two dominant technologies serving this market, each addressing specific conductivity requirements across diverse applications.
Electronics manufacturing constitutes the largest market segment for conductive surface coatings, where miniaturization trends and higher current densities demand superior electrical performance. Printed circuit boards, connectors, and semiconductor packaging applications require coatings that maintain low contact resistance while providing corrosion protection. The automotive industry's transition toward electric vehicles has created unprecedented demand for high-conductivity coatings in battery systems, charging infrastructure, and power electronics components.
Aerospace and defense applications represent a premium market segment where conductivity requirements are coupled with stringent reliability standards. These sectors prioritize coatings that maintain electrical performance under extreme environmental conditions, including temperature cycling, humidity exposure, and mechanical stress. The growing satellite communication and radar systems markets further amplify demand for specialized conductive coatings.
Industrial equipment manufacturers increasingly require surface coatings that combine electrical conductivity with wear resistance and durability. Power generation equipment, industrial automation systems, and renewable energy infrastructure drive consistent demand for reliable conductive surface treatments. The telecommunications sector's expansion, particularly with 5G network deployment, has created new opportunities for high-frequency applications requiring specific conductivity characteristics.
Market dynamics reveal a clear preference shift toward environmentally compliant coating technologies. Regulatory pressures surrounding hexavalent chromium usage in traditional chrome plating have accelerated adoption of alternative solutions, including electroless nickel systems. This regulatory landscape creates both challenges for established chrome plating operations and opportunities for innovative coating technologies.
Regional market patterns show concentrated demand in Asia-Pacific manufacturing hubs, North American automotive centers, and European industrial regions. Supply chain considerations increasingly influence coating selection decisions, as manufacturers seek reliable, cost-effective solutions that meet both performance and regulatory requirements while supporting sustainable manufacturing practices.
Electronics manufacturing constitutes the largest market segment for conductive surface coatings, where miniaturization trends and higher current densities demand superior electrical performance. Printed circuit boards, connectors, and semiconductor packaging applications require coatings that maintain low contact resistance while providing corrosion protection. The automotive industry's transition toward electric vehicles has created unprecedented demand for high-conductivity coatings in battery systems, charging infrastructure, and power electronics components.
Aerospace and defense applications represent a premium market segment where conductivity requirements are coupled with stringent reliability standards. These sectors prioritize coatings that maintain electrical performance under extreme environmental conditions, including temperature cycling, humidity exposure, and mechanical stress. The growing satellite communication and radar systems markets further amplify demand for specialized conductive coatings.
Industrial equipment manufacturers increasingly require surface coatings that combine electrical conductivity with wear resistance and durability. Power generation equipment, industrial automation systems, and renewable energy infrastructure drive consistent demand for reliable conductive surface treatments. The telecommunications sector's expansion, particularly with 5G network deployment, has created new opportunities for high-frequency applications requiring specific conductivity characteristics.
Market dynamics reveal a clear preference shift toward environmentally compliant coating technologies. Regulatory pressures surrounding hexavalent chromium usage in traditional chrome plating have accelerated adoption of alternative solutions, including electroless nickel systems. This regulatory landscape creates both challenges for established chrome plating operations and opportunities for innovative coating technologies.
Regional market patterns show concentrated demand in Asia-Pacific manufacturing hubs, North American automotive centers, and European industrial regions. Supply chain considerations increasingly influence coating selection decisions, as manufacturers seek reliable, cost-effective solutions that meet both performance and regulatory requirements while supporting sustainable manufacturing practices.
Current State of Chrome and Electroless Nickel Technologies
Chrome plating technology has reached a mature state with well-established industrial processes that have been refined over decades. The current chrome plating process typically involves hexavalent chromium solutions, which can achieve excellent conductivity levels ranging from 7.6 to 12.8 × 10^6 S/m depending on the plating conditions and substrate preparation. Modern chrome plating facilities utilize advanced rectifier systems and automated process controls to maintain consistent thickness and quality across large production volumes.
However, the chrome plating industry faces significant regulatory pressures due to environmental and health concerns associated with hexavalent chromium compounds. This has led to the development of trivalent chromium alternatives, though these newer processes generally exhibit slightly lower conductivity performance and require more complex bath chemistry management. The transition to environmentally compliant chrome plating solutions remains an ongoing challenge for manufacturers seeking to maintain optimal electrical performance.
Electroless nickel technology has evolved considerably, with multiple formulation types now available to address specific application requirements. Standard electroless nickel-phosphorus coatings typically contain 7-12% phosphorus and achieve conductivity values between 1.4 to 2.8 × 10^6 S/m. The conductivity performance is heavily influenced by the phosphorus content, with lower phosphorus formulations generally providing better electrical properties but reduced corrosion resistance.
Recent advances in electroless nickel include the development of nickel-boron formulations that offer superior conductivity compared to traditional nickel-phosphorus systems. These newer formulations can achieve conductivity levels approaching 4.5 × 10^6 S/m while maintaining the inherent advantages of uniform thickness distribution and excellent throwing power that characterize electroless processes.
Both technologies currently face technical limitations that impact their conductivity performance. Chrome plating processes are constrained by the need for precise current density control and substrate preparation requirements, while electroless nickel systems must balance conductivity optimization with bath stability and coating adhesion properties. The industry continues to invest in research aimed at overcoming these fundamental trade-offs through advanced chemistry development and process optimization techniques.
However, the chrome plating industry faces significant regulatory pressures due to environmental and health concerns associated with hexavalent chromium compounds. This has led to the development of trivalent chromium alternatives, though these newer processes generally exhibit slightly lower conductivity performance and require more complex bath chemistry management. The transition to environmentally compliant chrome plating solutions remains an ongoing challenge for manufacturers seeking to maintain optimal electrical performance.
Electroless nickel technology has evolved considerably, with multiple formulation types now available to address specific application requirements. Standard electroless nickel-phosphorus coatings typically contain 7-12% phosphorus and achieve conductivity values between 1.4 to 2.8 × 10^6 S/m. The conductivity performance is heavily influenced by the phosphorus content, with lower phosphorus formulations generally providing better electrical properties but reduced corrosion resistance.
Recent advances in electroless nickel include the development of nickel-boron formulations that offer superior conductivity compared to traditional nickel-phosphorus systems. These newer formulations can achieve conductivity levels approaching 4.5 × 10^6 S/m while maintaining the inherent advantages of uniform thickness distribution and excellent throwing power that characterize electroless processes.
Both technologies currently face technical limitations that impact their conductivity performance. Chrome plating processes are constrained by the need for precise current density control and substrate preparation requirements, while electroless nickel systems must balance conductivity optimization with bath stability and coating adhesion properties. The industry continues to invest in research aimed at overcoming these fundamental trade-offs through advanced chemistry development and process optimization techniques.
Existing Conductivity Enhancement Solutions
01 Electroless nickel plating processes and compositions
Various electroless nickel plating processes have been developed to deposit nickel coatings on substrates without the use of external electrical current. These processes typically involve chemical reduction of nickel ions in solution using reducing agents such as hypophosphite or borohydride. The resulting nickel-phosphorus or nickel-boron alloy coatings provide excellent corrosion resistance and uniform thickness distribution. The plating bath compositions, operating conditions, and substrate preparation methods are critical factors affecting the quality and conductivity of the deposited nickel layers.- Electroless nickel plating processes and compositions: Various electroless nickel plating processes have been developed to deposit nickel coatings on substrates without the use of external electrical current. These processes typically involve chemical reduction of nickel ions in solution using reducing agents such as hypophosphite or borohydride. The resulting nickel-phosphorus or nickel-boron alloy coatings provide excellent corrosion resistance and uniform thickness distribution. The plating bath compositions, operating conditions, and substrate preparation methods are optimized to achieve desired coating properties including conductivity, hardness, and adhesion.
- Chrome plating methods and applications: Chrome plating techniques involve the electrodeposition of chromium onto various substrates to provide decorative finishes, corrosion protection, and wear resistance. The plating processes utilize chromic acid-based electrolytes under controlled current density and temperature conditions. Different types of chrome plating include decorative chrome, hard chrome, and functional chrome, each serving specific industrial applications. The plating parameters are adjusted to control the deposit thickness, brightness, and surface characteristics of the chromium layer.
- Composite plating with enhanced electrical conductivity: Composite plating technologies combine metallic coatings with conductive particles or additives to enhance electrical conductivity properties. These methods involve co-deposition of metal matrices with dispersed conductive materials such as carbon nanotubes, graphene, or metal particles. The composite coatings exhibit improved electrical performance compared to pure metal deposits while maintaining other desirable properties such as corrosion resistance and mechanical strength. Process parameters including particle concentration, agitation, and current density are controlled to achieve uniform particle distribution.
- Multi-layer plating structures for conductivity optimization: Multi-layer plating structures are designed with alternating or sequential layers of different metals to optimize electrical conductivity and other functional properties. These structures typically combine layers such as copper, nickel, and chromium in specific sequences to achieve desired conductivity levels while providing corrosion protection and aesthetic appearance. The layer thickness, interface quality, and deposition sequence are carefully controlled to minimize contact resistance and maximize overall conductivity. Pre-treatment and intermediate treatment steps between layers ensure proper adhesion and interface quality.
- Surface treatment and post-plating processes for conductivity improvement: Various surface treatment and post-plating processes are employed to enhance the electrical conductivity of plated surfaces. These treatments include heat treatment, mechanical polishing, chemical passivation, and surface activation methods. The processes modify the surface morphology, reduce oxide formation, and improve contact resistance of the plated layers. Additional treatments such as sealing, coating with conductive polymers, or application of contact-enhancing agents further optimize the electrical performance of the finished products.
02 Chrome plating methods and applications
Chrome plating techniques involve the electrodeposition of chromium onto various substrates to provide decorative appearance, corrosion resistance, and wear resistance. Different types of chrome plating include decorative chrome, hard chrome, and functional chrome coatings. The plating process parameters such as current density, bath temperature, and electrolyte composition significantly influence the properties of the chromium layer including its electrical conductivity, hardness, and adhesion to the substrate.Expand Specific Solutions03 Composite plating combining chrome and nickel layers
Multi-layer plating systems combining chrome and nickel layers have been developed to optimize both conductivity and protective properties. These composite structures typically involve depositing an electroless nickel underlayer followed by a chrome top layer, or vice versa. The combination leverages the advantages of both metals, with nickel providing good conductivity and corrosion resistance as a base layer, while chrome offers enhanced surface hardness and aesthetic appeal. The interface between layers and overall coating architecture are important for achieving desired electrical and mechanical properties.Expand Specific Solutions04 Conductive additives and alloy modifications
Enhancement of electrical conductivity in nickel and chrome plating can be achieved through the incorporation of conductive additives or alloying elements. Various dopants and co-deposited materials have been investigated to improve the electrical properties of plated layers. Modifications to the plating bath chemistry, including the addition of specific metal salts or organic compounds, can influence the microstructure and grain size of the deposited metal, thereby affecting its conductivity. Post-plating heat treatments may also be employed to optimize the electrical characteristics.Expand Specific Solutions05 Surface treatment and conductivity measurement techniques
Various surface treatment methods have been developed to prepare substrates prior to plating and to enhance the conductivity of plated surfaces. These include cleaning, etching, and activation processes that ensure proper adhesion and electrical contact. Testing and measurement techniques for evaluating the electrical conductivity of chrome and electroless nickel coatings involve methods such as four-point probe measurements, contact resistance testing, and surface resistivity analysis. Quality control procedures ensure that the plated layers meet specified conductivity requirements for their intended applications.Expand Specific Solutions
Key Players in Surface Coating and Plating Industry
The chrome plating versus electroless nickel conductivity comparison represents a mature surface treatment industry experiencing steady growth, with the global electroplating market valued at approximately $15 billion and projected to reach $20 billion by 2028. The industry is in a consolidation phase, driven by environmental regulations favoring REACH-compliant solutions. Technology maturity varies significantly across players: established giants like Toyota Motor Corp., Samsung Electro-Mechanics, and Texas Instruments have integrated these technologies into mass production, while specialized companies like Savroc Oy are pioneering next-generation TripleHard chromium coatings and Modumetal develops advanced nanolaminated alternatives. Japanese companies including Murata Manufacturing, Canon, and IBIDEN demonstrate high technical sophistication in precision applications, whereas emerging players like BGT Materials are exploring graphene-enhanced conductive coatings, indicating ongoing innovation despite the industry's maturity.
International Business Machines Corp.
Technical Solution: IBM has extensively researched both chrome plating and electroless nickel for semiconductor and electronic packaging applications. Their studies focus on conductivity optimization for high-frequency applications, where chrome plating demonstrates superior performance with lower electrical resistance. IBM's research shows that chrome-plated surfaces maintain better signal integrity in high-speed circuits due to lower skin effect losses. They have also developed modified electroless nickel formulations with reduced phosphorus content to improve conductivity while maintaining the self-limiting thickness advantage of the electroless process.
Strengths: Deep technical expertise, advanced research capabilities, focus on high-performance applications. Weaknesses: Solutions primarily targeted at high-end applications, may not be cost-effective for standard commercial uses.
Texas Instruments Incorporated
Technical Solution: Texas Instruments utilizes both chrome plating and electroless nickel in their semiconductor packaging and lead frame manufacturing processes. Their comparative studies indicate that chrome plating offers superior conductivity with approximately 3-5 times lower electrical resistance compared to electroless nickel coatings. TI's process optimization focuses on achieving uniform chrome deposits for consistent electrical performance across large-scale production. They employ electroless nickel as a barrier layer in multi-layer plating systems, where its uniform thickness distribution provides excellent base preparation for subsequent conductive layers while offering adequate conductivity for power management applications.
Strengths: Large-scale manufacturing expertise, proven reliability in semiconductor applications, cost-effective production processes. Weaknesses: Limited to specific application requirements, process complexity increases with multi-layer systems.
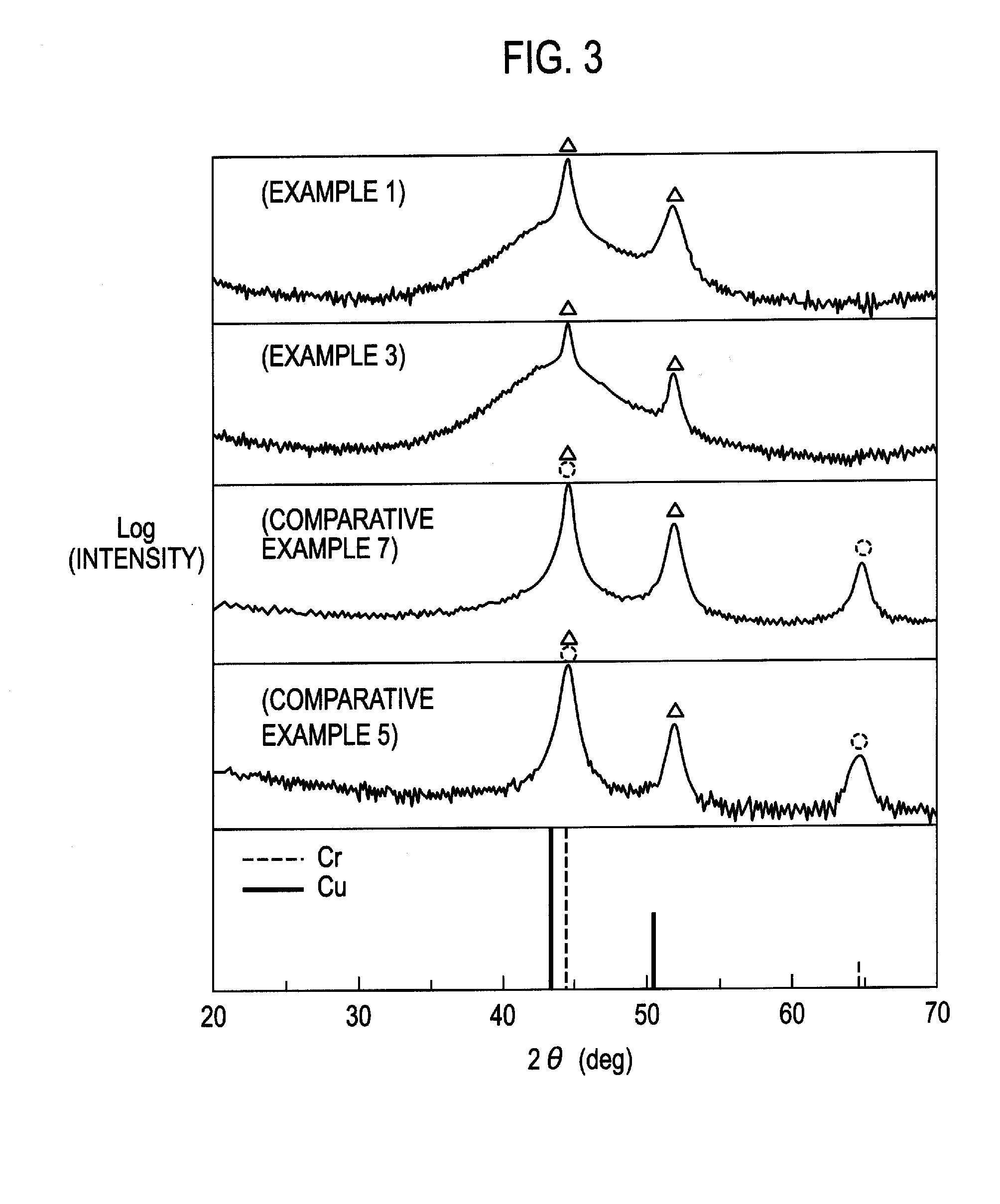
Core Patents in Chrome and Electroless Nickel Processes
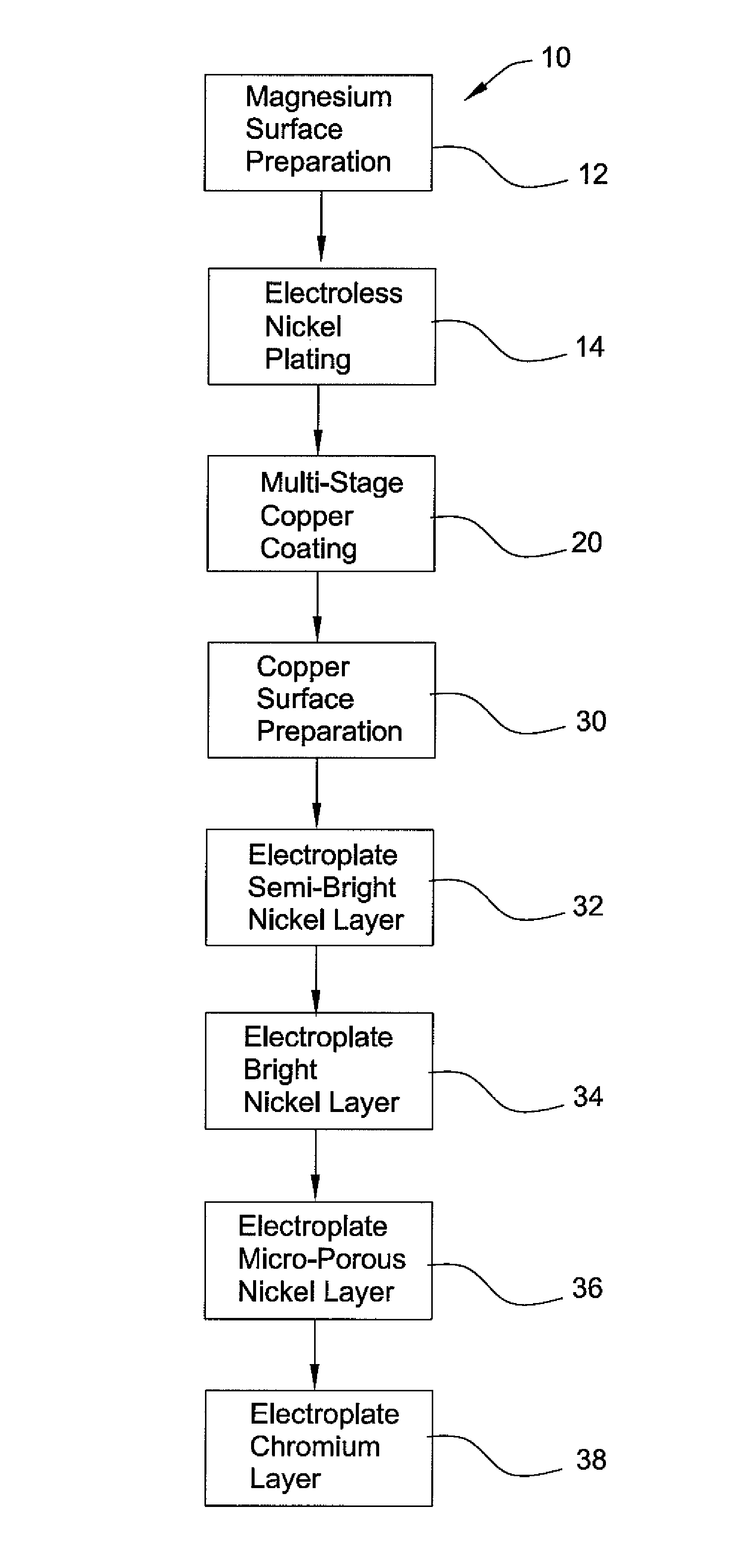
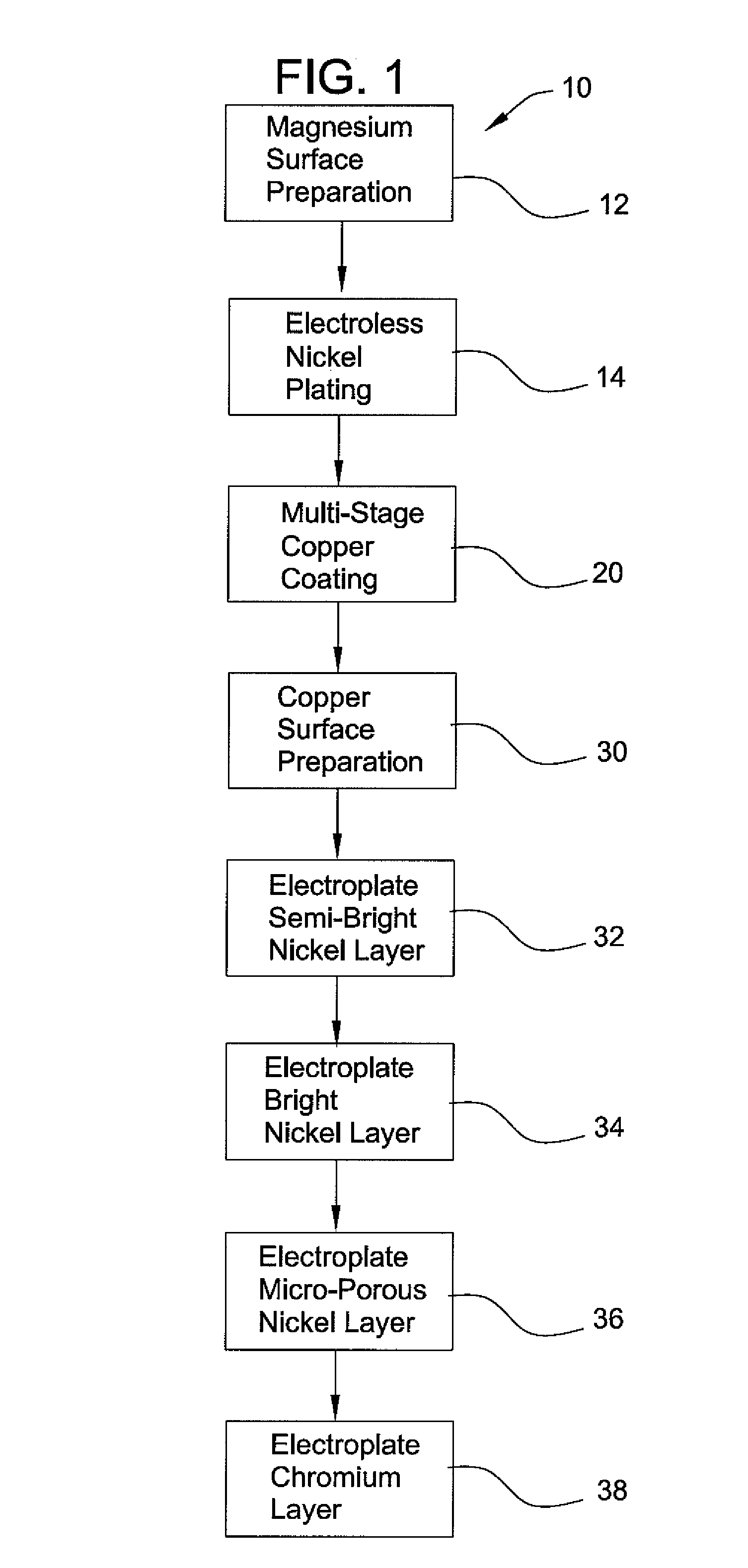
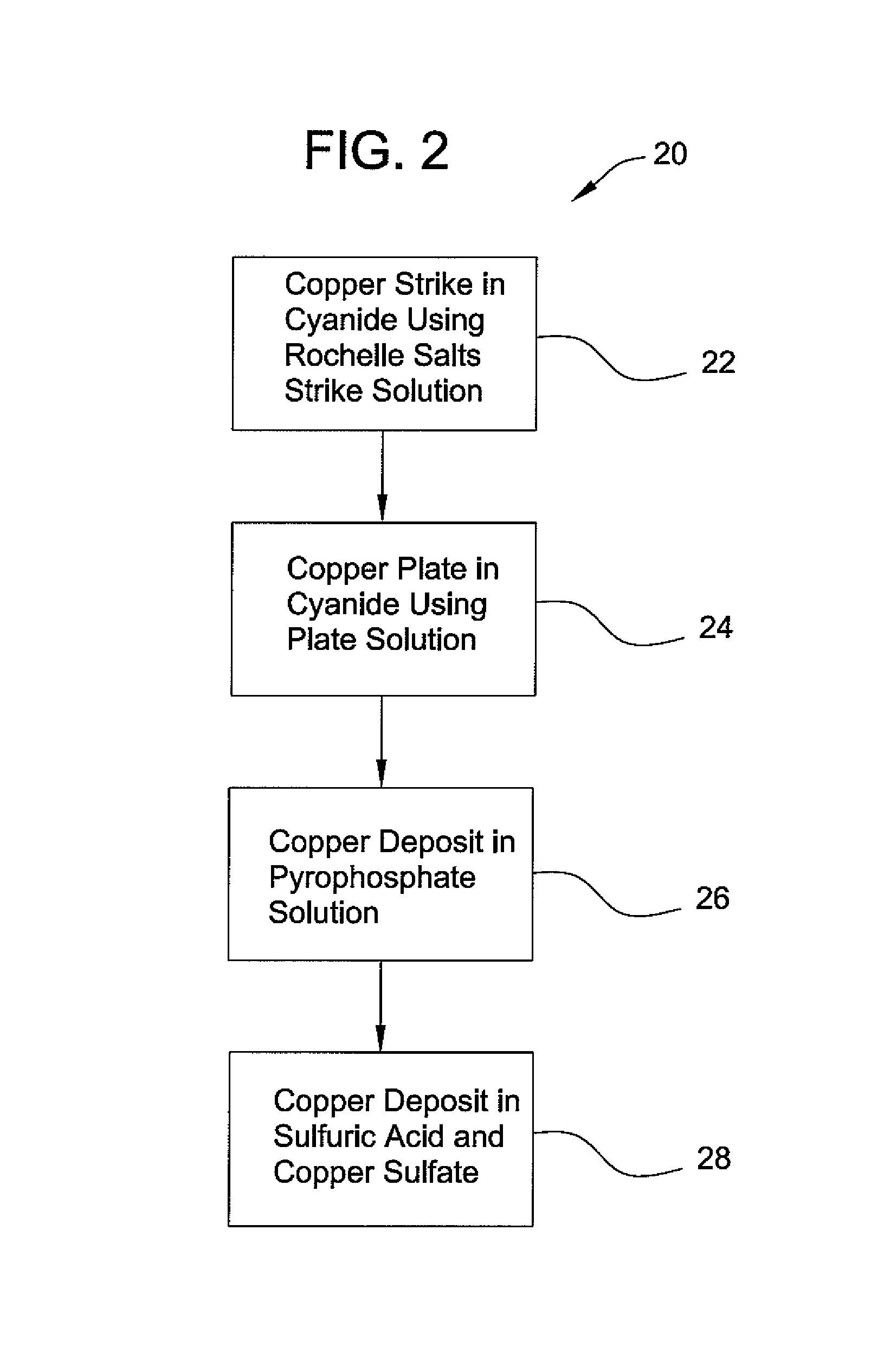
Method of chrome plating magnesium and magnesium alloys
PatentInactiveUS8152985B2
Innovation
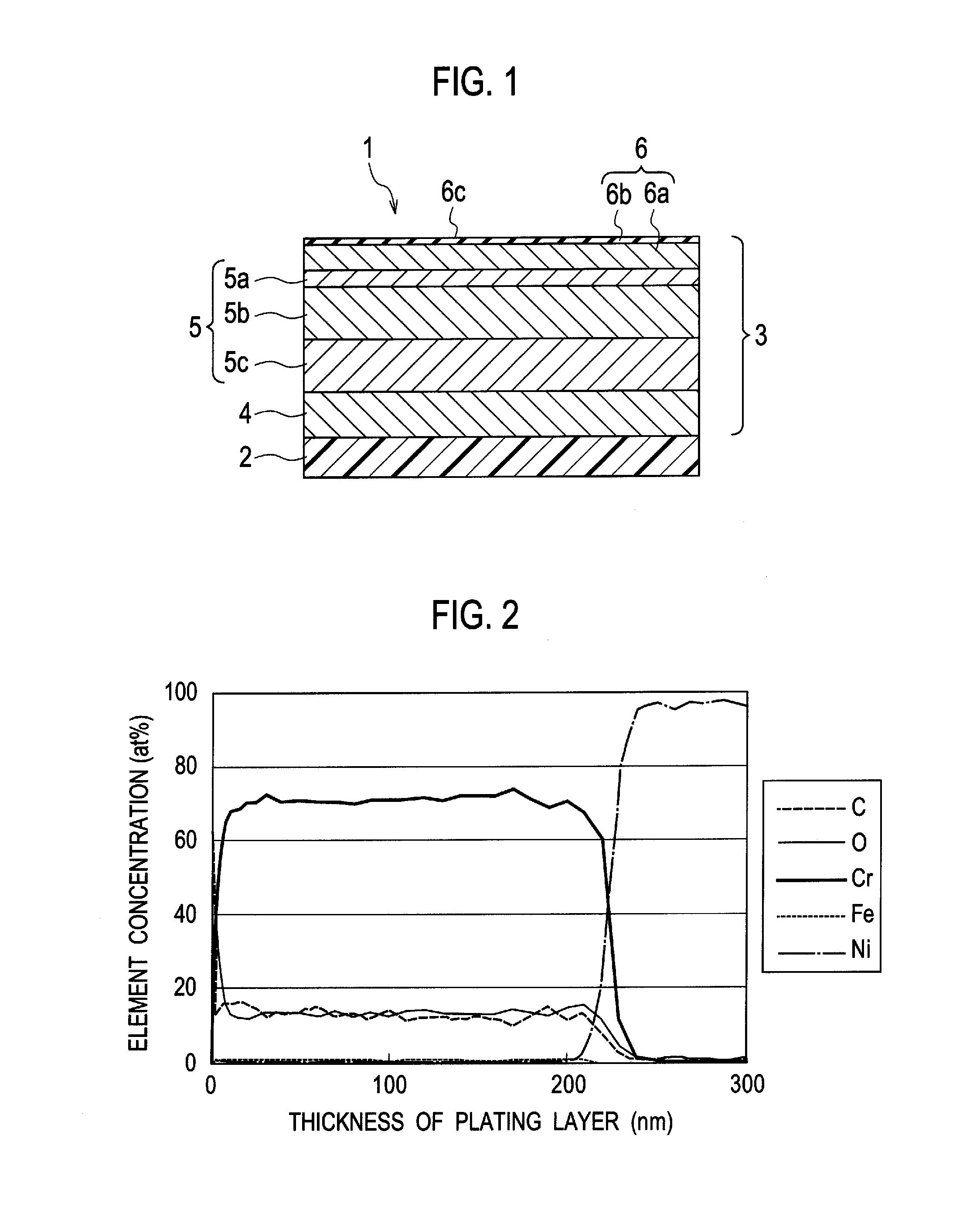
- A process involving electroless nickel plating, a multi-stage copper coating transition zone, and multiple layers of electrodeposited nickel to create a corrosion-resistant system suitable for direct chromium over plating, ensuring interlayer adherence and impermeability.
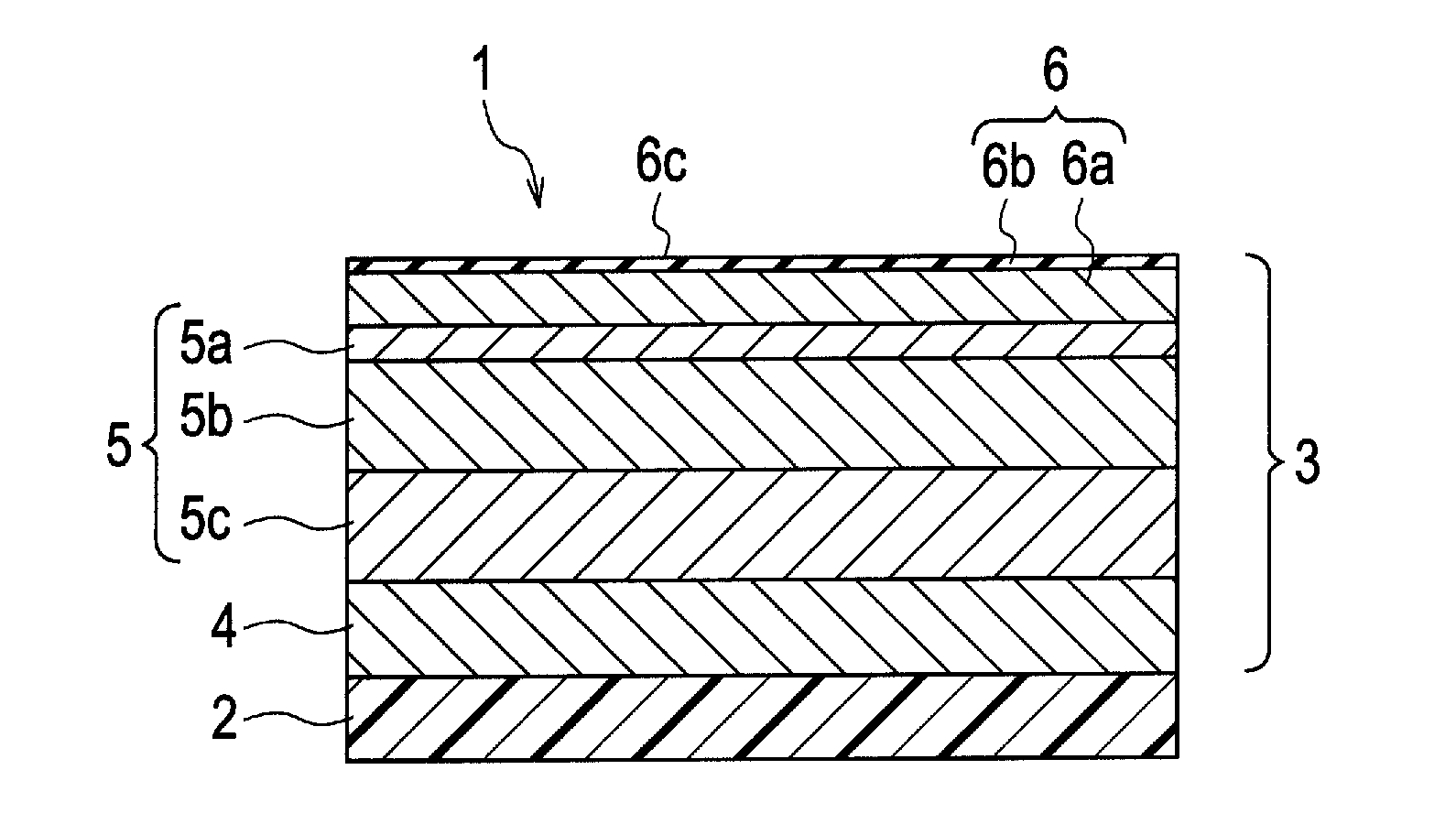
Chrome-plated part and manufacturing method of the same
PatentActiveUS20120052319A1
Innovation
- A chrome-plated part with a substrate, a bright nickel plating layer, a noble potential nickel plating layer with an electric potential difference between 40 mV to 150 mV, and a trivalent chrome plating layer with a microporous or microcrack structure, where the trivalent chrome plating layer is formed using a wet plating method and includes additives like carbon, oxygen, and iron to enhance corrosion resistance.
Environmental Regulations for Chrome Plating Processes
Chrome plating processes face increasingly stringent environmental regulations worldwide due to the use of hexavalent chromium compounds, which are classified as carcinogenic and highly toxic substances. The European Union's REACH regulation has placed severe restrictions on chromium VI usage, requiring extensive authorization procedures and risk management measures. Similarly, the United States Environmental Protection Agency has implemented strict air emission standards under the National Emission Standards for Hazardous Air Pollutants, mandating advanced ventilation systems and emission control technologies.
The regulatory landscape encompasses multiple aspects of chrome plating operations, including wastewater discharge limits, air quality standards, and worker safety requirements. Facilities must comply with maximum allowable concentrations of chromium compounds in effluent streams, typically ranging from 0.1 to 2.0 mg/L depending on local jurisdictions. Air emission standards require implementation of high-efficiency mist suppressants and scrubbing systems to minimize hexavalent chromium release into the atmosphere.
Occupational safety regulations mandate comprehensive worker protection protocols, including specialized ventilation systems, personal protective equipment requirements, and regular health monitoring programs. The Occupational Safety and Health Administration has established permissible exposure limits as low as 5 micrograms per cubic meter for hexavalent chromium in workplace air, necessitating continuous monitoring and control measures.
Waste management regulations require proper handling, treatment, and disposal of chromium-containing sludges and rinse waters. Many jurisdictions classify these materials as hazardous waste, requiring specialized treatment facilities and documentation procedures. The regulatory burden includes mandatory environmental impact assessments, periodic compliance audits, and substantial financial penalties for violations.
These environmental constraints significantly impact the operational costs and feasibility of chrome plating processes, driving increased interest in alternative technologies such as electroless nickel plating. The regulatory trend toward further restrictions on chromium usage creates additional pressure for industries to evaluate environmentally compliant alternatives while maintaining required conductivity performance standards.
The regulatory landscape encompasses multiple aspects of chrome plating operations, including wastewater discharge limits, air quality standards, and worker safety requirements. Facilities must comply with maximum allowable concentrations of chromium compounds in effluent streams, typically ranging from 0.1 to 2.0 mg/L depending on local jurisdictions. Air emission standards require implementation of high-efficiency mist suppressants and scrubbing systems to minimize hexavalent chromium release into the atmosphere.
Occupational safety regulations mandate comprehensive worker protection protocols, including specialized ventilation systems, personal protective equipment requirements, and regular health monitoring programs. The Occupational Safety and Health Administration has established permissible exposure limits as low as 5 micrograms per cubic meter for hexavalent chromium in workplace air, necessitating continuous monitoring and control measures.
Waste management regulations require proper handling, treatment, and disposal of chromium-containing sludges and rinse waters. Many jurisdictions classify these materials as hazardous waste, requiring specialized treatment facilities and documentation procedures. The regulatory burden includes mandatory environmental impact assessments, periodic compliance audits, and substantial financial penalties for violations.
These environmental constraints significantly impact the operational costs and feasibility of chrome plating processes, driving increased interest in alternative technologies such as electroless nickel plating. The regulatory trend toward further restrictions on chromium usage creates additional pressure for industries to evaluate environmentally compliant alternatives while maintaining required conductivity performance standards.
Cost-Performance Analysis of Coating Technologies
The cost-performance analysis of chrome plating versus electroless nickel coating reveals significant differences in both initial investment and long-term operational economics. Chrome plating typically requires lower upfront material costs, with chromium chemicals being relatively inexpensive compared to nickel-based solutions. However, the total cost of ownership must account for equipment requirements, processing complexity, and regulatory compliance expenses.
Chrome plating operations demand substantial infrastructure investments, including specialized rectifiers, ventilation systems, and waste treatment facilities. The hexavalent chromium process requires extensive safety measures and environmental controls, significantly increasing operational costs. Labor costs are also elevated due to the need for skilled technicians and stringent safety protocols. Additionally, the multi-step process involving copper and nickel underlayers before chrome deposition extends processing time and increases energy consumption.
Electroless nickel coating presents a different cost structure with higher material costs but potentially lower operational expenses. The autocatalytic process eliminates the need for complex electrical systems and reduces energy consumption. Processing equipment is generally simpler and requires less maintenance, though the chemical bath management demands precise control and regular replenishment. The single-step application process reduces labor requirements and processing time compared to traditional chrome plating sequences.
Performance evaluation demonstrates that electroless nickel provides superior conductivity consistency across complex geometries, eliminating the edge effects common in electroplated chrome. The uniform thickness distribution of electroless nickel reduces material waste and improves yield rates. Chrome plating, while offering excellent surface hardness and wear resistance, may exhibit conductivity variations due to current density differences during electrodeposition.
Long-term performance considerations favor electroless nickel in many applications due to its corrosion resistance and thermal stability. The reduced maintenance requirements and extended service life often offset the higher initial material costs. However, chrome plating remains cost-effective for applications requiring maximum surface hardness and where conductivity uniformity is less critical.
Environmental compliance costs increasingly favor electroless nickel processes, as chrome plating faces stricter regulations and potential phase-out scenarios. The transition costs from hexavalent to trivalent chrome systems add complexity to the economic analysis, making electroless nickel more attractive for future-oriented manufacturing strategies.
Chrome plating operations demand substantial infrastructure investments, including specialized rectifiers, ventilation systems, and waste treatment facilities. The hexavalent chromium process requires extensive safety measures and environmental controls, significantly increasing operational costs. Labor costs are also elevated due to the need for skilled technicians and stringent safety protocols. Additionally, the multi-step process involving copper and nickel underlayers before chrome deposition extends processing time and increases energy consumption.
Electroless nickel coating presents a different cost structure with higher material costs but potentially lower operational expenses. The autocatalytic process eliminates the need for complex electrical systems and reduces energy consumption. Processing equipment is generally simpler and requires less maintenance, though the chemical bath management demands precise control and regular replenishment. The single-step application process reduces labor requirements and processing time compared to traditional chrome plating sequences.
Performance evaluation demonstrates that electroless nickel provides superior conductivity consistency across complex geometries, eliminating the edge effects common in electroplated chrome. The uniform thickness distribution of electroless nickel reduces material waste and improves yield rates. Chrome plating, while offering excellent surface hardness and wear resistance, may exhibit conductivity variations due to current density differences during electrodeposition.
Long-term performance considerations favor electroless nickel in many applications due to its corrosion resistance and thermal stability. The reduced maintenance requirements and extended service life often offset the higher initial material costs. However, chrome plating remains cost-effective for applications requiring maximum surface hardness and where conductivity uniformity is less critical.
Environmental compliance costs increasingly favor electroless nickel processes, as chrome plating faces stricter regulations and potential phase-out scenarios. The transition costs from hexavalent to trivalent chrome systems add complexity to the economic analysis, making electroless nickel more attractive for future-oriented manufacturing strategies.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!