Cryopreservation vs Controlled-Rate Freezing: Which is Better?
FEB 25, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Cryopreservation Technology Background and Objectives
Cryopreservation represents a critical biotechnology that enables the long-term preservation of biological materials at ultra-low temperatures, typically below -130°C, with the primary objective of maintaining cellular viability and functional integrity. The fundamental principle involves reducing metabolic activities to negligible levels by transitioning biological specimens into a state of suspended animation, thereby preventing biochemical degradation and structural damage over extended storage periods.
The historical development of cryopreservation traces back to the mid-20th century when researchers first discovered that certain cells could survive freezing processes when protected by cryoprotective agents. This breakthrough laid the foundation for modern preservation techniques that have become indispensable across multiple sectors including reproductive medicine, regenerative therapies, biobanking, and pharmaceutical research. The evolution from simple freezing methods to sophisticated cryopreservation protocols reflects decades of scientific advancement in understanding ice crystal formation, osmotic stress, and cellular response mechanisms.
Contemporary cryopreservation technology aims to achieve several interconnected objectives. The primary goal is to maximize post-thaw cell viability and functionality, ensuring that preserved biological materials retain their original characteristics and therapeutic potential. This requires minimizing ice crystal formation within cells, which can cause mechanical damage to cellular structures and compromise membrane integrity. Additionally, the technology seeks to establish standardized protocols that ensure reproducibility and reliability across different specimen types and application contexts.
Another critical objective involves developing cost-effective and scalable preservation solutions that can accommodate the growing demands of biomedical research and clinical applications. As personalized medicine and cell-based therapies advance, the need for efficient long-term storage systems becomes increasingly urgent. Furthermore, cryopreservation technology strives to expand its applicability to complex biological systems, including tissue constructs, organoids, and potentially whole organs, which present unique challenges due to their structural complexity and varied cellular compositions.
The comparative analysis between traditional cryopreservation approaches and controlled-rate freezing methods emerges from the need to optimize preservation outcomes while addressing specific limitations inherent in each technique. Understanding these methodological differences is essential for advancing preservation science and selecting appropriate protocols for diverse biological materials.
The historical development of cryopreservation traces back to the mid-20th century when researchers first discovered that certain cells could survive freezing processes when protected by cryoprotective agents. This breakthrough laid the foundation for modern preservation techniques that have become indispensable across multiple sectors including reproductive medicine, regenerative therapies, biobanking, and pharmaceutical research. The evolution from simple freezing methods to sophisticated cryopreservation protocols reflects decades of scientific advancement in understanding ice crystal formation, osmotic stress, and cellular response mechanisms.
Contemporary cryopreservation technology aims to achieve several interconnected objectives. The primary goal is to maximize post-thaw cell viability and functionality, ensuring that preserved biological materials retain their original characteristics and therapeutic potential. This requires minimizing ice crystal formation within cells, which can cause mechanical damage to cellular structures and compromise membrane integrity. Additionally, the technology seeks to establish standardized protocols that ensure reproducibility and reliability across different specimen types and application contexts.
Another critical objective involves developing cost-effective and scalable preservation solutions that can accommodate the growing demands of biomedical research and clinical applications. As personalized medicine and cell-based therapies advance, the need for efficient long-term storage systems becomes increasingly urgent. Furthermore, cryopreservation technology strives to expand its applicability to complex biological systems, including tissue constructs, organoids, and potentially whole organs, which present unique challenges due to their structural complexity and varied cellular compositions.
The comparative analysis between traditional cryopreservation approaches and controlled-rate freezing methods emerges from the need to optimize preservation outcomes while addressing specific limitations inherent in each technique. Understanding these methodological differences is essential for advancing preservation science and selecting appropriate protocols for diverse biological materials.
Market Demand for Cell and Tissue Preservation Solutions
The global market for cell and tissue preservation solutions has experienced substantial growth driven by expanding applications across regenerative medicine, biobanking, assisted reproductive technologies, and pharmaceutical research. Healthcare institutions, research laboratories, and biotechnology companies increasingly require reliable preservation methods to maintain cellular viability and functionality during long-term storage. The demand stems from critical needs in organ transplantation programs, stem cell therapies, vaccine production, and personalized medicine initiatives where preserved biological materials serve as foundational resources.
Biobanking facilities worldwide face mounting pressure to store diverse biological specimens including blood samples, tissue biopsies, and genetic materials for future clinical and research purposes. This sector particularly values preservation technologies that minimize ice crystal formation and cellular damage, as sample integrity directly impacts downstream analytical accuracy and therapeutic efficacy. The controlled-rate freezing method has gained traction in facilities requiring standardized protocols and reproducible outcomes, while traditional cryopreservation remains prevalent in settings prioritizing rapid processing and cost efficiency.
The assisted reproductive technology sector represents a significant demand driver, with fertility clinics requiring optimal preservation of oocytes, embryos, and sperm. Patient expectations for high post-thaw survival rates have intensified scrutiny of freezing methodologies, prompting clinics to evaluate comparative advantages between vitrification-based cryopreservation and programmable controlled-rate systems. Market preferences vary regionally based on regulatory frameworks, reimbursement policies, and clinical practice standards.
Pharmaceutical and biotechnology industries contribute substantial demand through cell line banking, therapeutic cell manufacturing, and biologics production. These sectors prioritize scalability, automation compatibility, and regulatory compliance in preservation solutions. The shift toward cell-based therapies and advanced biologics has created specialized requirements for preserving complex cellular products while maintaining potency and safety profiles throughout distribution chains.
Emerging markets in Asia-Pacific and Latin America demonstrate accelerating adoption rates as healthcare infrastructure modernizes and research capabilities expand. However, cost sensitivity in these regions influences technology selection, with institutions balancing performance requirements against capital investment and operational expenses associated with different preservation approaches.
Biobanking facilities worldwide face mounting pressure to store diverse biological specimens including blood samples, tissue biopsies, and genetic materials for future clinical and research purposes. This sector particularly values preservation technologies that minimize ice crystal formation and cellular damage, as sample integrity directly impacts downstream analytical accuracy and therapeutic efficacy. The controlled-rate freezing method has gained traction in facilities requiring standardized protocols and reproducible outcomes, while traditional cryopreservation remains prevalent in settings prioritizing rapid processing and cost efficiency.
The assisted reproductive technology sector represents a significant demand driver, with fertility clinics requiring optimal preservation of oocytes, embryos, and sperm. Patient expectations for high post-thaw survival rates have intensified scrutiny of freezing methodologies, prompting clinics to evaluate comparative advantages between vitrification-based cryopreservation and programmable controlled-rate systems. Market preferences vary regionally based on regulatory frameworks, reimbursement policies, and clinical practice standards.
Pharmaceutical and biotechnology industries contribute substantial demand through cell line banking, therapeutic cell manufacturing, and biologics production. These sectors prioritize scalability, automation compatibility, and regulatory compliance in preservation solutions. The shift toward cell-based therapies and advanced biologics has created specialized requirements for preserving complex cellular products while maintaining potency and safety profiles throughout distribution chains.
Emerging markets in Asia-Pacific and Latin America demonstrate accelerating adoption rates as healthcare infrastructure modernizes and research capabilities expand. However, cost sensitivity in these regions influences technology selection, with institutions balancing performance requirements against capital investment and operational expenses associated with different preservation approaches.
Current Status and Challenges in Freezing Methods
Cryopreservation and controlled-rate freezing represent two fundamental approaches in the preservation of biological materials, yet both face significant technical challenges that limit their widespread application. The current landscape reveals a complex interplay between technological capabilities and biological constraints that researchers and practitioners must navigate.
Traditional controlled-rate freezing has been the cornerstone of cell and tissue preservation for decades, utilizing programmable freezers to reduce temperature at predetermined rates, typically between 1°C to 10°C per minute. This method aims to balance the competing risks of intracellular ice formation and osmotic injury. However, the technology faces persistent challenges including high equipment costs, lengthy processing times, and variable success rates across different cell types. The requirement for precise temperature control and the need for cryoprotective agents at specific concentrations add layers of complexity to the process.
Vitrification-based cryopreservation has emerged as an alternative approach, achieving an ice-free solidified state through ultra-rapid cooling rates exceeding 15,000°C per minute. While this method eliminates ice crystal damage, it introduces its own set of challenges. The high concentrations of cryoprotective agents required for successful vitrification can induce cellular toxicity and osmotic stress. Additionally, the technical difficulty of achieving uniform cooling rates in larger sample volumes remains a significant barrier to scaling up this technology.
Both methods struggle with common fundamental issues that transcend their technical differences. Cryoprotective agent toxicity remains a critical concern, as these chemicals necessary for preventing ice damage can simultaneously harm cellular structures and functions. The phenomenon of chilling injury, occurring even before ice formation begins, affects membrane integrity and cellular metabolism. Post-thaw recovery rates vary dramatically across cell types, with some cells demonstrating robust survival while others show minimal viability.
Geographic distribution of expertise and infrastructure reveals disparities in technological advancement. North America and Europe maintain leadership in research and clinical applications, with established biobanking facilities and standardized protocols. Asian countries, particularly Japan and China, have made substantial investments in cryopreservation research, focusing on reproductive medicine and regenerative therapies. However, developing regions face significant barriers including limited access to specialized equipment, inadequate cold chain infrastructure, and insufficient technical training programs.
The standardization gap presents another major challenge across both methodologies. Lack of universal protocols for different biological materials leads to inconsistent outcomes and difficulties in comparing research results across institutions. Quality control measures remain inadequately defined, and the long-term stability of cryopreserved materials under various storage conditions requires further investigation to ensure reliability and reproducibility.
Traditional controlled-rate freezing has been the cornerstone of cell and tissue preservation for decades, utilizing programmable freezers to reduce temperature at predetermined rates, typically between 1°C to 10°C per minute. This method aims to balance the competing risks of intracellular ice formation and osmotic injury. However, the technology faces persistent challenges including high equipment costs, lengthy processing times, and variable success rates across different cell types. The requirement for precise temperature control and the need for cryoprotective agents at specific concentrations add layers of complexity to the process.
Vitrification-based cryopreservation has emerged as an alternative approach, achieving an ice-free solidified state through ultra-rapid cooling rates exceeding 15,000°C per minute. While this method eliminates ice crystal damage, it introduces its own set of challenges. The high concentrations of cryoprotective agents required for successful vitrification can induce cellular toxicity and osmotic stress. Additionally, the technical difficulty of achieving uniform cooling rates in larger sample volumes remains a significant barrier to scaling up this technology.
Both methods struggle with common fundamental issues that transcend their technical differences. Cryoprotective agent toxicity remains a critical concern, as these chemicals necessary for preventing ice damage can simultaneously harm cellular structures and functions. The phenomenon of chilling injury, occurring even before ice formation begins, affects membrane integrity and cellular metabolism. Post-thaw recovery rates vary dramatically across cell types, with some cells demonstrating robust survival while others show minimal viability.
Geographic distribution of expertise and infrastructure reveals disparities in technological advancement. North America and Europe maintain leadership in research and clinical applications, with established biobanking facilities and standardized protocols. Asian countries, particularly Japan and China, have made substantial investments in cryopreservation research, focusing on reproductive medicine and regenerative therapies. However, developing regions face significant barriers including limited access to specialized equipment, inadequate cold chain infrastructure, and insufficient technical training programs.
The standardization gap presents another major challenge across both methodologies. Lack of universal protocols for different biological materials leads to inconsistent outcomes and difficulties in comparing research results across institutions. Quality control measures remain inadequately defined, and the long-term stability of cryopreserved materials under various storage conditions requires further investigation to ensure reliability and reproducibility.
Mainstream Freezing Protocols and Technical Solutions
01 Controlled-rate freezing apparatus and systems
Specialized apparatus and systems designed for controlled-rate freezing in cryopreservation processes. These systems typically include programmable cooling units, temperature sensors, and control mechanisms that allow precise regulation of cooling rates. The equipment enables automated freezing protocols with specific temperature gradients to minimize cellular damage during the freezing process. Advanced systems may incorporate real-time monitoring and feedback control to maintain optimal freezing conditions throughout the cryopreservation procedure.- Controlled-rate freezing apparatus and systems: Specialized apparatus and systems designed for controlled-rate freezing in cryopreservation processes. These systems typically include programmable cooling chambers, temperature sensors, and automated control mechanisms to precisely regulate the cooling rate during the freezing process. The equipment allows for customizable freezing protocols and can accommodate various sample types and volumes, ensuring optimal preservation conditions.
- Cryoprotective agent formulations and compositions: Development of cryoprotective solutions and media compositions that protect biological materials during controlled-rate freezing. These formulations include various cryoprotectants, stabilizers, and buffering agents that minimize ice crystal formation and cellular damage. The compositions are optimized for different cell types and tissues to maintain viability and functionality after thawing.
- Methods for freezing biological cells and tissues: Specific protocols and methodologies for implementing controlled-rate freezing of various biological materials including cells, tissues, and organs. These methods define optimal cooling rates, temperature transition points, and holding periods at specific temperatures. The techniques address challenges such as intracellular ice formation and osmotic stress during the freezing process.
- Monitoring and control systems for freezing parameters: Advanced monitoring and control technologies for real-time tracking and adjustment of freezing parameters during cryopreservation. These systems incorporate sensors, feedback mechanisms, and automated control algorithms to maintain precise temperature profiles and cooling rates. The technology ensures reproducibility and consistency across different freezing cycles.
- Storage and thawing protocols for cryopreserved materials: Comprehensive protocols for post-freezing storage conditions and controlled thawing procedures of cryopreserved biological materials. These methods specify optimal storage temperatures, container types, and thawing rates to maximize cell recovery and viability. The protocols also address long-term storage stability and quality control measures for preserved samples.
02 Cryoprotective agent formulations and compositions
Development of cryoprotective solutions and media compositions that protect biological materials during controlled-rate freezing. These formulations typically contain various cryoprotectants that prevent ice crystal formation and reduce osmotic stress during freezing and thawing. The compositions may include penetrating and non-penetrating cryoprotectants in specific concentrations optimized for different cell types and tissues. Advanced formulations incorporate multiple protective agents to enhance cell viability and post-thaw recovery rates.Expand Specific Solutions03 Controlled-rate freezing protocols and methods
Specific protocols and methodologies for implementing controlled-rate freezing in cryopreservation applications. These methods define optimal cooling rates, holding temperatures, and phase transition management during the freezing process. The protocols typically involve multi-step cooling procedures with different rates at various temperature ranges to minimize intracellular ice formation. Advanced methods may include seeding techniques and specific temperature profiles tailored for different biological materials such as cells, tissues, or reproductive materials.Expand Specific Solutions04 Cryopreservation of reproductive cells and tissues
Specialized applications of controlled-rate freezing for preserving reproductive materials including sperm, oocytes, embryos, and reproductive tissues. These applications require precise control of freezing parameters to maintain cellular integrity and functional viability. The techniques address specific challenges related to reproductive cell cryopreservation such as zona pellucida hardening, cytoskeletal damage, and chromosomal abnormalities. Methods may include specific equilibration times, cooling rates, and warming protocols optimized for different reproductive cell types.Expand Specific Solutions05 Monitoring and quality control in controlled-rate freezing
Systems and methods for monitoring, controlling, and ensuring quality during controlled-rate freezing processes. These approaches include real-time temperature monitoring, ice nucleation detection, and post-thaw viability assessment. Quality control measures may involve validation of freezing protocols, equipment calibration, and standardization of procedures. Advanced monitoring systems incorporate sensors and data logging capabilities to track critical parameters throughout the cryopreservation process and ensure reproducibility and compliance with established protocols.Expand Specific Solutions
Major Players in Cryopreservation Equipment and Services
The cryopreservation and controlled-rate freezing sector represents a maturing technology landscape within the broader biopreservation market, currently transitioning from early adoption to mainstream clinical and research applications. The market demonstrates robust growth driven by expanding cell therapy, regenerative medicine, and organ transplantation demands. Technology maturity varies significantly across players: established equipment manufacturers like BioLife Solutions, Asymptote Ltd., and Praxair Technology deliver commercialized controlled-rate freezing systems, while innovators such as Cradle Healthcare (Until Labs) pioneer advanced vitrification approaches for extended organ preservation. Academic institutions including University of California, University of Minnesota, and University of North Carolina contribute foundational research advancing cryoprotectant formulations and freezing protocols. Specialized firms like CryoCrate and Cool Lab focus on niche transport and storage solutions, whereas Lifeline Scientific emphasizes organ perfusion technologies. Chinese entities including Shanghai Origincell and University of Shanghai for Science & Technology strengthen regional capabilities, reflecting global market expansion and increasing competition across automated systems, biopreservation media, and integrated cryochain solutions.
Cook Medical Technologies LLC
Technical Solution: Cook Medical has developed controlled-rate freezing protocols specifically for reproductive tissue cryopreservation, including oocytes, embryos, and ovarian tissue. Their methodology employs programmable freezers that execute multi-step cooling profiles, typically starting at -2°C/min to -40°C, followed by rapid transfer to liquid nitrogen storage at -196°C. The company integrates permeating cryoprotectants like dimethyl sulfoxide (DMSO) and ethylene glycol with non-permeating agents to achieve optimal osmotic balance during freezing. Their systems feature real-time temperature monitoring and automated seeding initiation to control ice nucleation, resulting in post-thaw survival rates exceeding 80% for embryos and 65-75% for oocytes in clinical applications.
Strengths: Specialized expertise in reproductive medicine with clinically validated protocols; precise control over ice crystal formation through automated seeding. Weaknesses: Limited application scope beyond reproductive tissues; protocol optimization required for different cell types and tissue structures.
Shanghai Origincell Biological Cryo Equipment Co. Ltd.
Technical Solution: Shanghai Origincell focuses on manufacturing controlled-rate freezing equipment for biological sample preservation in research and clinical settings. Their programmable freezers feature multi-stage cooling protocols with temperature control precision of ±0.3°C and cooling rate adjustability from 0.1°C/min to 50°C/min. The systems incorporate liquid nitrogen delivery mechanisms with automated flow regulation to maintain consistent cooling profiles throughout the freezing chamber. Their technology addresses the comparison between rapid cryopreservation and controlled-rate freezing by offering flexible programming options that can simulate both approaches, allowing researchers to optimize protocols for specific cell types. The equipment includes data logging capabilities for protocol validation and regulatory compliance documentation.
Strengths: Cost-effective solutions for Asian markets; flexible programming allowing protocol customization and comparative studies between freezing methods. Weaknesses: Limited global market presence compared to established Western manufacturers; less extensive clinical validation data for diverse cell types.
Core Patents in Controlled-Rate Freezing Technologies
Freezing of Biological Material
PatentActiveUS20200154699A1
Innovation
- A method and apparatus that control the phase transition time of biological samples during freezing to minimize ice crystal formation by determining a cooling profile that allows the phase transition to occur in less than 12 minutes, potentially eliminating the need for cryoprotectants and improving viability.
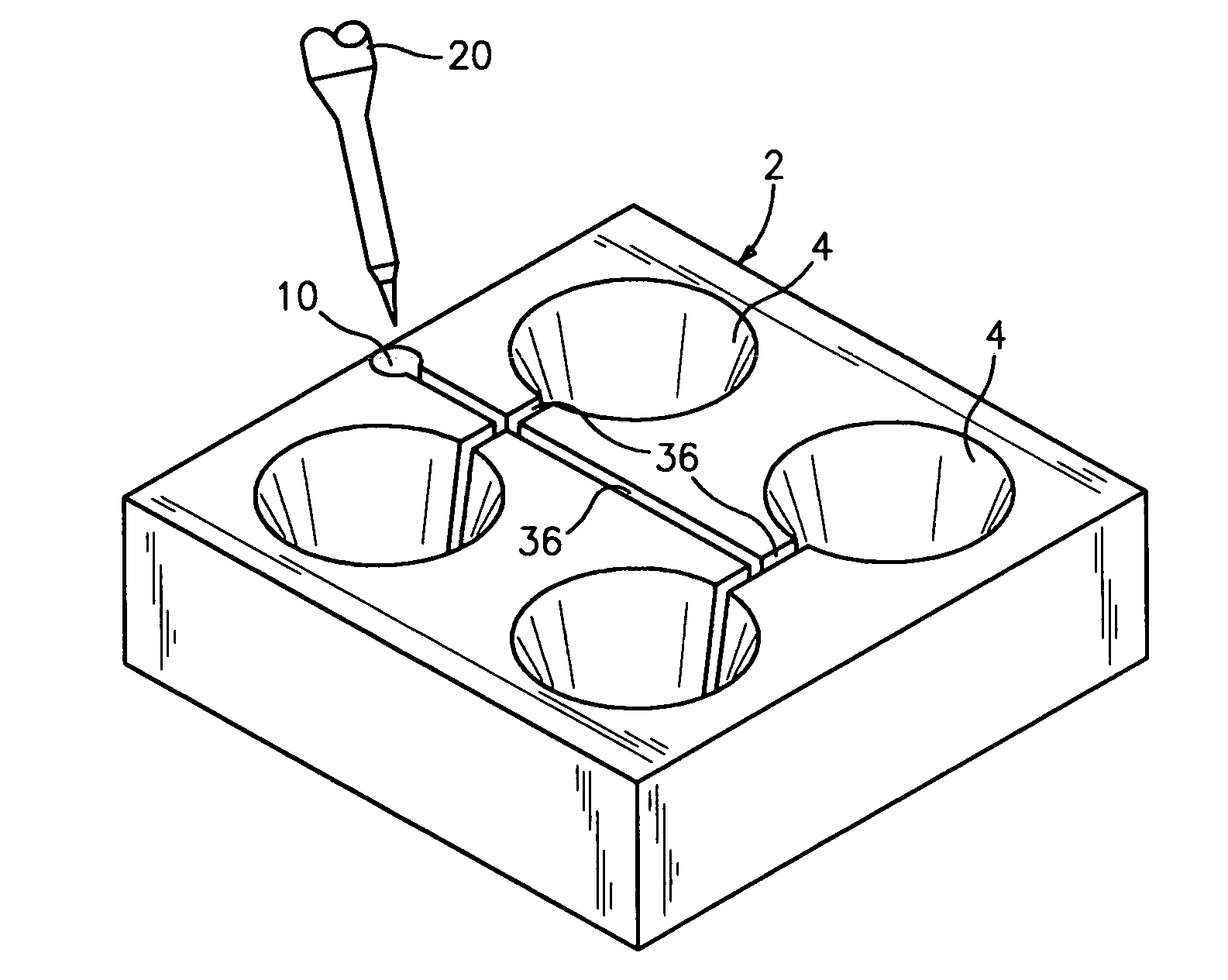
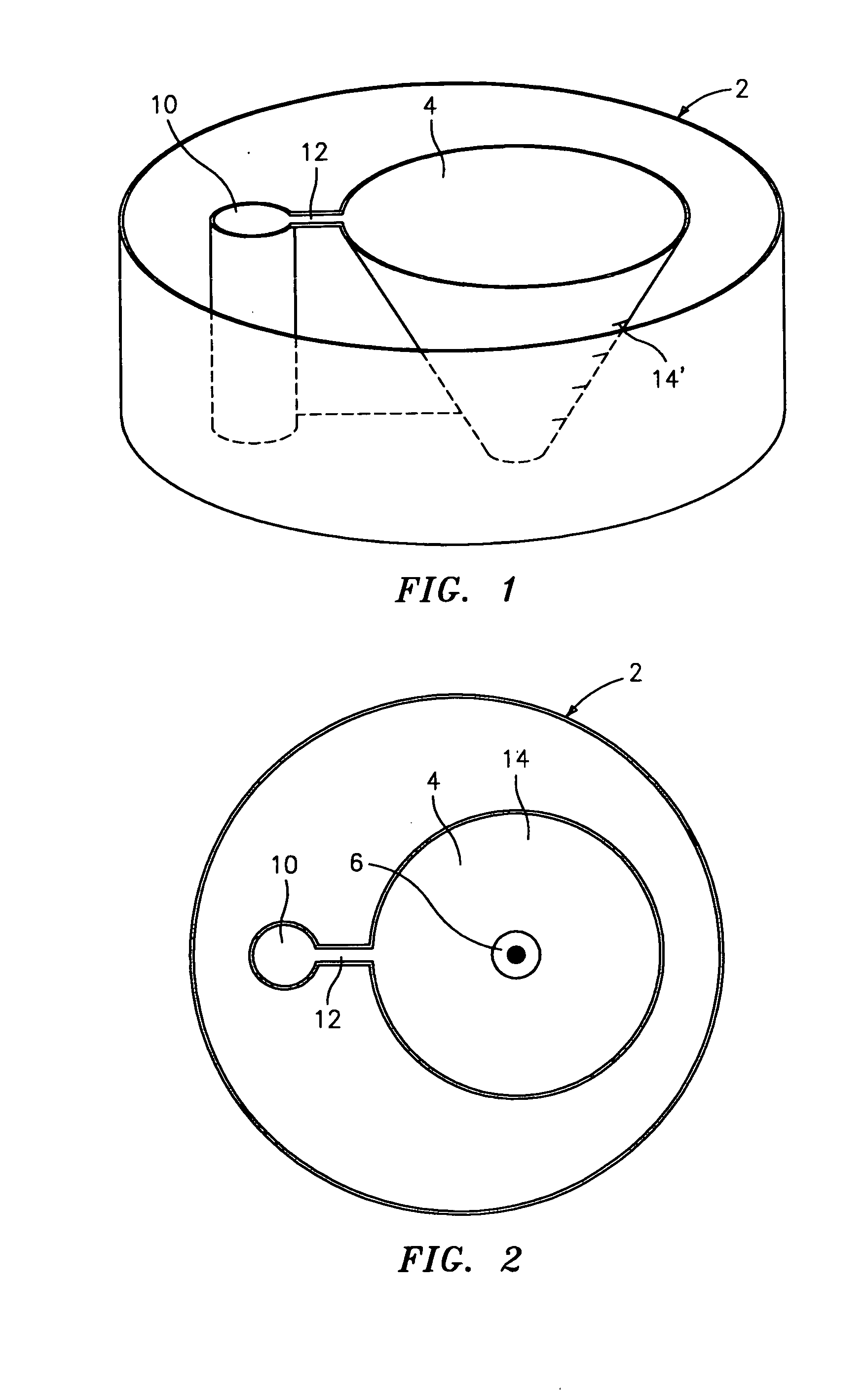
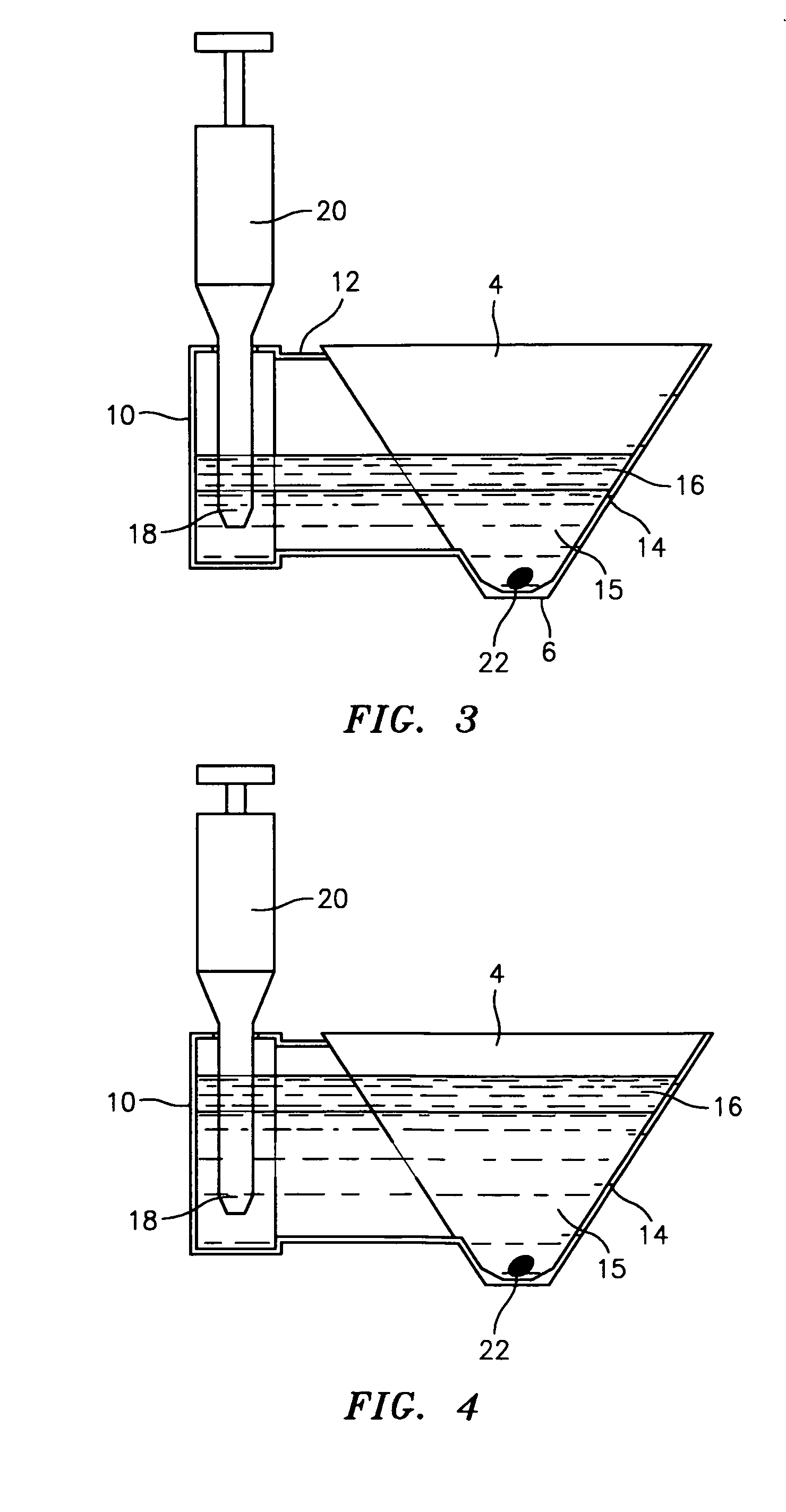
Flooding dish and method for changing media in the dish in the preparation of mammalian specimen culture and for cryo-preservation, freezing, vitrification and the thawing and warming of such specimens
PatentActiveUS20100297600A1
Innovation
- A dish with conical or pyramidal wells and integral adjunct passages allows for sequential media infusion and withdrawal without disturbing specimens, reducing handling and enabling consistent treatment protocols for cryo-preservation and thawing.
Quality Standards and Regulatory Requirements
The regulatory landscape governing cryopreservation and controlled-rate freezing methods is complex and varies significantly across jurisdictions, reflecting the critical importance of ensuring biological material integrity and patient safety. International standards organizations, including the International Organization for Standardization (ISO) and the American Association of Tissue Banks (AATB), have established comprehensive guidelines that address both methodological approaches. These standards encompass critical parameters such as cooling rates, cryoprotectant concentrations, storage temperatures, and documentation requirements that must be maintained throughout the preservation process.
Quality control protocols for both methods require rigorous validation procedures to demonstrate consistent performance and reproducibility. Controlled-rate freezing systems must undergo regular calibration and maintenance verification, with documented evidence of temperature uniformity and cooling rate accuracy. Cryopreservation facilities are mandated to implement quality management systems compliant with Good Manufacturing Practice (GMP) or Good Tissue Practice (GTP) standards, depending on the application domain. These systems necessitate comprehensive standard operating procedures, personnel training records, and environmental monitoring data.
Regulatory agencies such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and national health authorities impose specific requirements based on the intended use of preserved materials. For clinical applications, both methods must demonstrate compliance with stringent sterility assurance levels, endotoxin limits, and viability thresholds. The FDA's 21 CFR Part 1271 specifically governs human cells and tissues, requiring establishments to register and list their products while maintaining detailed records of processing methods.
Emerging regulatory trends emphasize risk-based approaches and post-market surveillance, requiring manufacturers and facilities to implement continuous monitoring systems. Traceability requirements mandate complete documentation from collection through storage and eventual use, ensuring accountability throughout the cold chain. As regenerative medicine and cell therapy applications expand, regulatory frameworks continue evolving to address novel challenges while maintaining the fundamental principle of ensuring preserved material quality and safety across both technological approaches.
Quality control protocols for both methods require rigorous validation procedures to demonstrate consistent performance and reproducibility. Controlled-rate freezing systems must undergo regular calibration and maintenance verification, with documented evidence of temperature uniformity and cooling rate accuracy. Cryopreservation facilities are mandated to implement quality management systems compliant with Good Manufacturing Practice (GMP) or Good Tissue Practice (GTP) standards, depending on the application domain. These systems necessitate comprehensive standard operating procedures, personnel training records, and environmental monitoring data.
Regulatory agencies such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and national health authorities impose specific requirements based on the intended use of preserved materials. For clinical applications, both methods must demonstrate compliance with stringent sterility assurance levels, endotoxin limits, and viability thresholds. The FDA's 21 CFR Part 1271 specifically governs human cells and tissues, requiring establishments to register and list their products while maintaining detailed records of processing methods.
Emerging regulatory trends emphasize risk-based approaches and post-market surveillance, requiring manufacturers and facilities to implement continuous monitoring systems. Traceability requirements mandate complete documentation from collection through storage and eventual use, ensuring accountability throughout the cold chain. As regenerative medicine and cell therapy applications expand, regulatory frameworks continue evolving to address novel challenges while maintaining the fundamental principle of ensuring preserved material quality and safety across both technological approaches.
Cost-Benefit Analysis of Freezing Methods
When evaluating cryopreservation versus controlled-rate freezing methods, the economic dimension represents a critical decision factor for laboratories, biobanks, and clinical facilities. Initial capital investment differs substantially between these approaches. Controlled-rate freezers require sophisticated programmable equipment with precise temperature control mechanisms, typically ranging from $15,000 to $50,000 per unit depending on capacity and features. In contrast, basic cryopreservation can be initiated with standard cryogenic storage containers and liquid nitrogen dewars, with entry costs starting around $3,000 to $10,000, though automated systems increase this investment considerably.
Operational expenditures present a contrasting economic profile. Controlled-rate freezing incurs higher electricity consumption due to mechanical refrigeration systems, with annual energy costs potentially reaching $2,000 to $5,000 per unit. Maintenance contracts and periodic calibration add another $1,500 to $3,000 annually. Conversely, cryopreservation's primary recurring cost centers on liquid nitrogen supply, which varies significantly by geographic location and consumption volume, typically ranging from $0.50 to $2.00 per liter. High-throughput facilities may consume hundreds of liters monthly, generating substantial ongoing expenses.
Personnel training and labor costs constitute another differentiating factor. Controlled-rate freezing demands specialized operator training to program freezing protocols and troubleshoot equipment malfunctions, requiring 20-40 hours of initial training investment. Cryopreservation procedures, while requiring careful handling protocols for safety, generally involve shorter training periods of 10-20 hours. However, the manual nature of some cryopreservation workflows may increase labor time per sample processed.
Sample recovery rates directly impact cost-effectiveness through their influence on experimental success and material waste. Controlled-rate freezing typically achieves post-thaw viability rates of 70-90% for most cell types when properly optimized, while direct immersion cryopreservation may yield 60-85% viability depending on cell characteristics and cryoprotectant formulations. The economic value of improved recovery rates must be weighed against the differential equipment and operational costs, with high-value samples justifying premium preservation methods.
Scalability considerations further influence long-term cost structures. Controlled-rate freezers face capacity constraints requiring additional equipment purchases as sample volumes grow, whereas cryogenic storage can be expanded more incrementally through additional dewars. For institutions processing thousands of samples annually, the per-sample cost differential becomes a pivotal strategic consideration in method selection.
Operational expenditures present a contrasting economic profile. Controlled-rate freezing incurs higher electricity consumption due to mechanical refrigeration systems, with annual energy costs potentially reaching $2,000 to $5,000 per unit. Maintenance contracts and periodic calibration add another $1,500 to $3,000 annually. Conversely, cryopreservation's primary recurring cost centers on liquid nitrogen supply, which varies significantly by geographic location and consumption volume, typically ranging from $0.50 to $2.00 per liter. High-throughput facilities may consume hundreds of liters monthly, generating substantial ongoing expenses.
Personnel training and labor costs constitute another differentiating factor. Controlled-rate freezing demands specialized operator training to program freezing protocols and troubleshoot equipment malfunctions, requiring 20-40 hours of initial training investment. Cryopreservation procedures, while requiring careful handling protocols for safety, generally involve shorter training periods of 10-20 hours. However, the manual nature of some cryopreservation workflows may increase labor time per sample processed.
Sample recovery rates directly impact cost-effectiveness through their influence on experimental success and material waste. Controlled-rate freezing typically achieves post-thaw viability rates of 70-90% for most cell types when properly optimized, while direct immersion cryopreservation may yield 60-85% viability depending on cell characteristics and cryoprotectant formulations. The economic value of improved recovery rates must be weighed against the differential equipment and operational costs, with high-value samples justifying premium preservation methods.
Scalability considerations further influence long-term cost structures. Controlled-rate freezers face capacity constraints requiring additional equipment purchases as sample volumes grow, whereas cryogenic storage can be expanded more incrementally through additional dewars. For institutions processing thousands of samples annually, the per-sample cost differential becomes a pivotal strategic consideration in method selection.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!