Emerging trends in PEC water splitting material sciences.
SEP 4, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PEC Water Splitting Background and Objectives
Photoelectrochemical (PEC) water splitting has emerged as a promising technology for sustainable hydrogen production, offering a pathway to convert solar energy directly into chemical energy. The evolution of this field dates back to the 1970s when Fujishima and Honda first demonstrated the photocatalytic splitting of water using titanium dioxide electrodes. Since then, significant advancements have been made in understanding the fundamental principles and developing more efficient materials for PEC water splitting.
The technological trajectory of PEC water splitting has been characterized by continuous improvements in photoelectrode materials, device architectures, and system integration. Early research focused primarily on metal oxide semiconductors such as TiO2, Fe2O3, and WO3. However, these materials faced limitations in terms of solar light absorption, charge carrier mobility, and surface catalytic activity, resulting in low solar-to-hydrogen conversion efficiencies.
Recent years have witnessed a paradigm shift towards more sophisticated material designs, including nanostructured materials, heterojunctions, plasmonic structures, and quantum-confined systems. These advanced materials aim to address the fundamental challenges of PEC water splitting: efficient light absorption across the solar spectrum, effective charge separation and transport, and catalytically active surfaces for water oxidation and reduction reactions.
The primary objective of current research in PEC water splitting materials science is to develop photoelectrode materials that can achieve solar-to-hydrogen conversion efficiencies exceeding 10% with stability over thousands of hours under real-world conditions. This requires a multidisciplinary approach combining insights from solid-state physics, electrochemistry, materials science, and nanotechnology.
Emerging trends in this field include the exploration of earth-abundant materials to replace precious metal catalysts, the development of tandem photoelectrochemical cells to maximize solar spectrum utilization, and the integration of artificial intelligence for accelerated materials discovery and optimization. Additionally, there is growing interest in bifunctional materials that can simultaneously perform water oxidation and reduction reactions, simplifying device architectures and potentially reducing costs.
The ultimate goal of PEC water splitting research is to establish a viable technology for large-scale, sustainable hydrogen production that can compete economically with conventional hydrogen production methods. This would contribute significantly to the global transition towards a hydrogen-based economy and help address climate change challenges by reducing dependence on fossil fuels.
The technological trajectory of PEC water splitting has been characterized by continuous improvements in photoelectrode materials, device architectures, and system integration. Early research focused primarily on metal oxide semiconductors such as TiO2, Fe2O3, and WO3. However, these materials faced limitations in terms of solar light absorption, charge carrier mobility, and surface catalytic activity, resulting in low solar-to-hydrogen conversion efficiencies.
Recent years have witnessed a paradigm shift towards more sophisticated material designs, including nanostructured materials, heterojunctions, plasmonic structures, and quantum-confined systems. These advanced materials aim to address the fundamental challenges of PEC water splitting: efficient light absorption across the solar spectrum, effective charge separation and transport, and catalytically active surfaces for water oxidation and reduction reactions.
The primary objective of current research in PEC water splitting materials science is to develop photoelectrode materials that can achieve solar-to-hydrogen conversion efficiencies exceeding 10% with stability over thousands of hours under real-world conditions. This requires a multidisciplinary approach combining insights from solid-state physics, electrochemistry, materials science, and nanotechnology.
Emerging trends in this field include the exploration of earth-abundant materials to replace precious metal catalysts, the development of tandem photoelectrochemical cells to maximize solar spectrum utilization, and the integration of artificial intelligence for accelerated materials discovery and optimization. Additionally, there is growing interest in bifunctional materials that can simultaneously perform water oxidation and reduction reactions, simplifying device architectures and potentially reducing costs.
The ultimate goal of PEC water splitting research is to establish a viable technology for large-scale, sustainable hydrogen production that can compete economically with conventional hydrogen production methods. This would contribute significantly to the global transition towards a hydrogen-based economy and help address climate change challenges by reducing dependence on fossil fuels.
Market Analysis for Hydrogen Production Technologies
The global hydrogen production market is experiencing significant growth, driven by increasing demand for clean energy solutions and decarbonization efforts across industries. Currently valued at approximately $130 billion, the market is projected to reach $220 billion by 2030, with a compound annual growth rate of 6.8%. Green hydrogen production, particularly through photoelectrochemical (PEC) water splitting technologies, represents one of the fastest-growing segments within this market.
Traditional hydrogen production methods, dominated by steam methane reforming (SMR), account for over 75% of current production capacity. However, these methods generate substantial carbon emissions, creating a strategic opportunity for PEC water splitting technologies to gain market share as environmental regulations tighten globally. The cost differential remains significant, with conventional hydrogen production at $1-2/kg compared to $4-6/kg for PEC-based methods, though this gap is narrowing rapidly due to technological advancements in material sciences.
Regional market analysis reveals Asia-Pacific as the dominant player, controlling approximately 40% of the hydrogen production market, followed by Europe (30%) and North America (20%). Europe leads in green hydrogen initiatives, with substantial investments in PEC research and development programs. Countries like Germany, Japan, and South Korea have established national hydrogen strategies that specifically target advancements in photoelectrochemical technologies.
Industry segmentation shows transportation and industrial applications as the primary demand drivers, collectively representing over 60% of the market. The transportation sector, particularly fuel cell vehicles, is expected to be a major growth catalyst for PEC water splitting technologies, with projected annual growth rates exceeding 15% through 2030.
Investment trends indicate a surge in funding for PEC material science research, with venture capital investments increasing by 35% annually since 2018. Major energy corporations are strategically repositioning their portfolios to include PEC water splitting technologies, with several announcing partnerships with research institutions and material science startups.
Market barriers include high initial capital requirements, technological scalability challenges, and competition from other emerging hydrogen production methods such as electrolysis. However, the unique advantages of PEC systems—including potential for higher efficiency and lower operational costs—position them favorably in the long-term market landscape.
Consumer and industrial adoption patterns suggest accelerating interest in green hydrogen solutions, with over 65% of surveyed industrial users expressing willingness to pay premium prices for carbon-neutral hydrogen. This market sentiment creates a favorable environment for continued investment in PEC water splitting material sciences and related technologies.
Traditional hydrogen production methods, dominated by steam methane reforming (SMR), account for over 75% of current production capacity. However, these methods generate substantial carbon emissions, creating a strategic opportunity for PEC water splitting technologies to gain market share as environmental regulations tighten globally. The cost differential remains significant, with conventional hydrogen production at $1-2/kg compared to $4-6/kg for PEC-based methods, though this gap is narrowing rapidly due to technological advancements in material sciences.
Regional market analysis reveals Asia-Pacific as the dominant player, controlling approximately 40% of the hydrogen production market, followed by Europe (30%) and North America (20%). Europe leads in green hydrogen initiatives, with substantial investments in PEC research and development programs. Countries like Germany, Japan, and South Korea have established national hydrogen strategies that specifically target advancements in photoelectrochemical technologies.
Industry segmentation shows transportation and industrial applications as the primary demand drivers, collectively representing over 60% of the market. The transportation sector, particularly fuel cell vehicles, is expected to be a major growth catalyst for PEC water splitting technologies, with projected annual growth rates exceeding 15% through 2030.
Investment trends indicate a surge in funding for PEC material science research, with venture capital investments increasing by 35% annually since 2018. Major energy corporations are strategically repositioning their portfolios to include PEC water splitting technologies, with several announcing partnerships with research institutions and material science startups.
Market barriers include high initial capital requirements, technological scalability challenges, and competition from other emerging hydrogen production methods such as electrolysis. However, the unique advantages of PEC systems—including potential for higher efficiency and lower operational costs—position them favorably in the long-term market landscape.
Consumer and industrial adoption patterns suggest accelerating interest in green hydrogen solutions, with over 65% of surveyed industrial users expressing willingness to pay premium prices for carbon-neutral hydrogen. This market sentiment creates a favorable environment for continued investment in PEC water splitting material sciences and related technologies.
Current Challenges in PEC Material Development
Despite significant advancements in photoelectrochemical (PEC) water splitting materials over the past decade, several critical challenges continue to impede widespread commercial implementation. The primary obstacle remains the insufficient solar-to-hydrogen (STH) conversion efficiency of current materials. While theoretical maximum efficiency approaches 30%, most practical PEC systems struggle to exceed 10%, with commercially viable applications requiring at least 15-20% efficiency under standard conditions.
Material stability presents another formidable challenge, as many high-performance semiconductors suffer from photocorrosion when exposed to aqueous electrolytes under illumination. This degradation significantly reduces device lifetime, with most current materials demonstrating stability measured in hours rather than the years required for commercial viability. The harsh oxidative and reductive environments at the semiconductor-electrolyte interface accelerate this deterioration process.
Cost-effectiveness remains a substantial barrier, with many high-performance materials incorporating expensive noble metals as co-catalysts or rare earth elements in their composition. The complex fabrication processes often involve high-temperature treatments, vacuum deposition techniques, and precise control of nanostructuring, all contributing to prohibitive manufacturing costs that limit scalability.
Charge carrier dynamics within PEC materials continue to present significant challenges. Recombination losses at grain boundaries, surface states, and bulk defects dramatically reduce quantum efficiency. Additionally, the band alignment between semiconductors and electrolytes often requires complex engineering to facilitate efficient charge transfer while maintaining photovoltage.
The development of tandem or Z-scheme systems to overcome the thermodynamic limitations of single-absorber systems introduces additional integration challenges. Interface engineering between multiple absorber layers and ensuring compatible processing conditions for different materials significantly complicate fabrication processes.
Standardization issues further hinder progress, as inconsistent testing protocols and reporting methods make direct comparisons between different research efforts difficult. The lack of universally accepted benchmarking standards complicates the assessment of genuine technological advances in the field.
Environmental concerns have also emerged regarding the toxicity of certain high-performance materials, particularly those containing lead, cadmium, or other heavy metals. Developing environmentally benign alternatives without sacrificing performance represents a critical research direction for sustainable implementation of PEC technology.
Material stability presents another formidable challenge, as many high-performance semiconductors suffer from photocorrosion when exposed to aqueous electrolytes under illumination. This degradation significantly reduces device lifetime, with most current materials demonstrating stability measured in hours rather than the years required for commercial viability. The harsh oxidative and reductive environments at the semiconductor-electrolyte interface accelerate this deterioration process.
Cost-effectiveness remains a substantial barrier, with many high-performance materials incorporating expensive noble metals as co-catalysts or rare earth elements in their composition. The complex fabrication processes often involve high-temperature treatments, vacuum deposition techniques, and precise control of nanostructuring, all contributing to prohibitive manufacturing costs that limit scalability.
Charge carrier dynamics within PEC materials continue to present significant challenges. Recombination losses at grain boundaries, surface states, and bulk defects dramatically reduce quantum efficiency. Additionally, the band alignment between semiconductors and electrolytes often requires complex engineering to facilitate efficient charge transfer while maintaining photovoltage.
The development of tandem or Z-scheme systems to overcome the thermodynamic limitations of single-absorber systems introduces additional integration challenges. Interface engineering between multiple absorber layers and ensuring compatible processing conditions for different materials significantly complicate fabrication processes.
Standardization issues further hinder progress, as inconsistent testing protocols and reporting methods make direct comparisons between different research efforts difficult. The lack of universally accepted benchmarking standards complicates the assessment of genuine technological advances in the field.
Environmental concerns have also emerged regarding the toxicity of certain high-performance materials, particularly those containing lead, cadmium, or other heavy metals. Developing environmentally benign alternatives without sacrificing performance represents a critical research direction for sustainable implementation of PEC technology.
State-of-the-Art PEC Material Solutions
01 Metal oxide-based photoanodes for PEC water splitting
Metal oxide semiconductors are widely used as photoanode materials in photoelectrochemical (PEC) water splitting due to their stability, abundance, and suitable band structures. These materials, including titanium dioxide, hematite, and tungsten trioxide, can be modified through doping, nanostructuring, or surface treatments to enhance their light absorption, charge separation, and catalytic properties. The optimization of metal oxide photoanodes is crucial for improving the overall efficiency of PEC water splitting systems for hydrogen production.- Metal oxide-based photocatalysts for PEC water splitting: Metal oxide semiconductors are widely used as photocatalysts in photoelectrochemical (PEC) water splitting due to their stability, abundance, and suitable band structures. These materials, including titanium dioxide, hematite, and tungsten oxide, can absorb light to generate electron-hole pairs that drive water splitting reactions. Various modification strategies such as doping, surface treatment, and nanostructuring are employed to enhance their light absorption, charge separation, and catalytic activity for improved hydrogen production efficiency.
- Nanostructured materials for enhanced PEC performance: Nanostructured materials offer advantages for PEC water splitting due to their high surface area, shortened charge carrier transport distances, and enhanced light absorption properties. These include nanowires, nanotubes, nanosheets, and hierarchical nanostructures that can be engineered to optimize the interface between the semiconductor and electrolyte. The controlled morphology at the nanoscale facilitates efficient charge separation and transfer, reducing recombination losses and improving overall water splitting efficiency.
- Composite and heterojunction materials for improved charge separation: Composite and heterojunction materials combine different semiconductors or incorporate conductive components to enhance charge separation and reduce recombination in PEC water splitting systems. These materials create energetically favorable interfaces that promote directional electron flow, extending charge carrier lifetimes. Examples include semiconductor-semiconductor heterojunctions, metal-semiconductor composites, and carbon-based hybrid materials that synergistically improve light harvesting, charge transport, and catalytic activity for water splitting.
- Novel electrode materials and fabrication methods: Advanced electrode materials and innovative fabrication techniques are being developed to enhance PEC water splitting performance. These include solution-based processing methods, electrodeposition, atomic layer deposition, and other scalable techniques to create photoelectrodes with optimized properties. The focus is on developing materials with appropriate band alignment, high conductivity, and stability in aqueous environments under illumination, while also considering cost-effectiveness and environmental compatibility for practical applications.
- PEC system design and integration technologies: System-level design and integration approaches for PEC water splitting focus on optimizing the overall device architecture to maximize solar-to-hydrogen conversion efficiency. This includes the development of tandem configurations, integrated photovoltaic-electrolyzer systems, and modular designs that can be scaled for practical applications. Considerations include light management strategies, electrolyte optimization, membrane integration, and product separation techniques to create efficient and durable water splitting systems for renewable hydrogen production.
02 Novel composite materials for enhanced PEC performance
Composite materials combining different semiconductors, catalysts, or conductive substrates can significantly improve PEC water splitting efficiency. These heterojunction structures facilitate better charge separation, extended light absorption range, and improved stability. Examples include semiconductor/metal composites, core-shell structures, and layered materials that create synergistic effects. The strategic design of these composite materials addresses multiple limitations of single-component systems and leads to higher solar-to-hydrogen conversion efficiencies.Expand Specific Solutions03 Nanostructured materials for improved PEC water splitting
Nanostructuring of materials for PEC water splitting offers advantages such as increased surface area, shortened charge carrier transport distances, and enhanced light absorption. Various nanostructures including nanowires, nanotubes, nanosheets, and hierarchical structures can be synthesized to optimize the photoelectrochemical properties. These nanostructured materials provide more active sites for water splitting reactions and improve charge separation efficiency, resulting in higher photocurrent densities and better overall performance.Expand Specific Solutions04 Earth-abundant catalysts for PEC water splitting
Earth-abundant materials are being developed as alternatives to precious metal catalysts for PEC water splitting. These include transition metal phosphides, sulfides, nitrides, and carbides that demonstrate high catalytic activity for hydrogen or oxygen evolution reactions. The development of these cost-effective catalysts focuses on optimizing their composition, structure, and integration with photoactive materials to achieve efficient and sustainable solar hydrogen production systems without relying on scarce and expensive noble metals.Expand Specific Solutions05 PEC device architectures and system integration
Advanced PEC device architectures and system integration approaches are essential for practical water splitting applications. These include tandem configurations, wireless designs, and integrated systems that combine light harvesting, catalysis, and separation components. Innovations in electrode design, membrane technology, and reactor engineering aim to address challenges related to efficiency, stability, and scalability. These integrated systems often incorporate strategies for product separation, electrolyte management, and optimal light utilization to maximize solar-to-hydrogen conversion efficiency.Expand Specific Solutions
Leading Research Groups and Industry Players
The PEC water splitting materials field is currently in a growth phase, characterized by increasing research intensity and early commercialization efforts. The market is expanding rapidly, driven by global clean hydrogen initiatives, with projections suggesting significant growth as renewable hydrogen production scales. Technologically, the field shows varied maturity levels across players. Academic institutions like King Abdullah University of Science & Technology, University of Michigan, and Technical Institute of Physics & Chemistry CAS are advancing fundamental research, while companies including SABIC, Toyota, and EvolOH are developing commercial applications. Emerging players like AquaHydrex and EvolOH are introducing innovative electrolyzer technologies, while established corporations such as Toray Industries and Xerox are leveraging their materials expertise to enter this space, creating a dynamic competitive landscape.
King Abdullah University of Science & Technology
Technical Solution: KAUST has developed advanced bismuth vanadate (BiVO4) photoanodes with strategic doping of molybdenum and gradient tungsten for PEC water splitting. Their approach incorporates a dual-layer structure with optimized band alignment that significantly enhances charge separation efficiency. The university's research team has pioneered a novel hydrothermal synthesis method that creates highly crystalline BiVO4 with controlled facet exposure, maximizing photocatalytic activity. Additionally, KAUST has developed proprietary cobalt phosphate (CoPi) co-catalysts that are deposited on the photoanode surface using photo-assisted electrodeposition, reducing the onset potential by approximately 0.3V and increasing photocurrent density by up to 3 mA/cm² under standard AM 1.5G illumination.
Strengths: Exceptional expertise in nanomaterial engineering and surface modification techniques that enhance charge carrier separation and transport. Their integrated systems approach combines materials science with device engineering for practical applications. Weaknesses: Some of their advanced materials require complex synthesis procedures that may limit large-scale manufacturing feasibility and increase production costs.
William Marsh Rice University
Technical Solution: Rice University has pioneered innovative 2D material-based photoelectrodes for PEC water splitting, focusing on MXenes and transition metal dichalcogenides (TMDs). Their research team has developed a proprietary exfoliation technique that produces large-area, defect-controlled 2D nanosheets with tunable bandgaps. A significant breakthrough includes their heterostructure engineering approach, where they stack different 2D materials to create artificial Z-scheme systems that mimic natural photosynthesis. These systems demonstrate photocurrent densities exceeding 10 mA/cm² under simulated sunlight. Rice has also developed a novel atomic layer deposition method for creating ultrathin protective layers on photoelectrodes, extending operational stability from hours to weeks while maintaining over 90% of initial performance in harsh electrolyte conditions.
Strengths: World-class expertise in 2D materials synthesis and characterization, with exceptional control over nanoscale architecture and interfaces. Their materials show remarkable stability in corrosive electrolytes. Weaknesses: The complex fabrication processes for their advanced heterostructures may present challenges for scaling up production, and some rare elements used in their systems could limit commercial viability.
Breakthrough Innovations in Photocatalysts
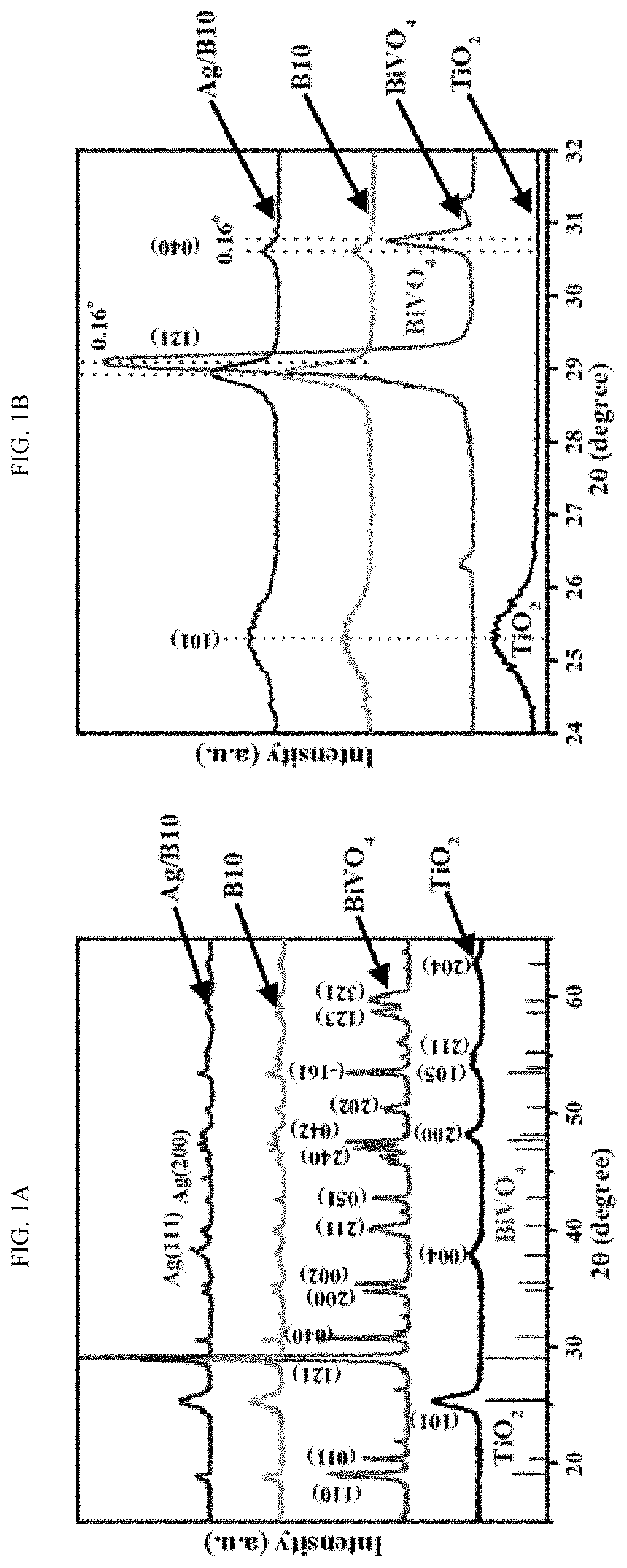
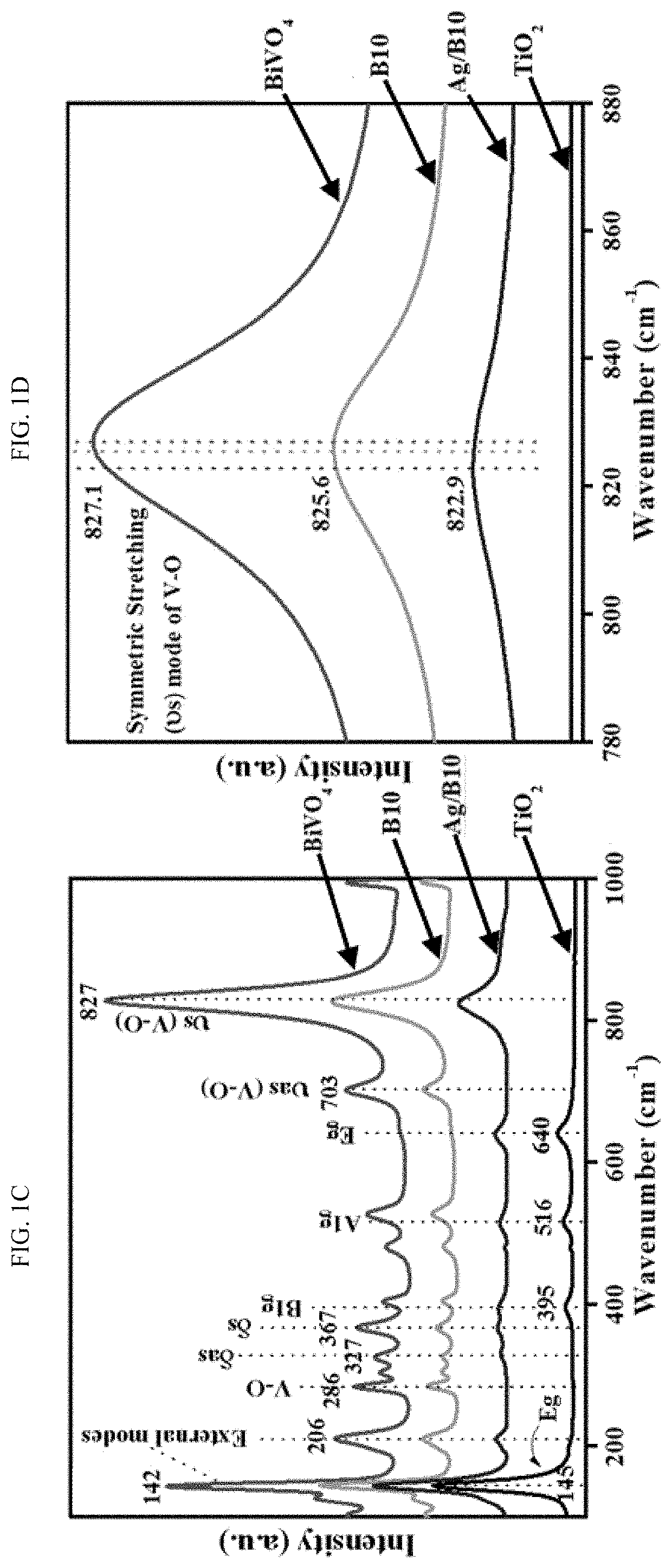
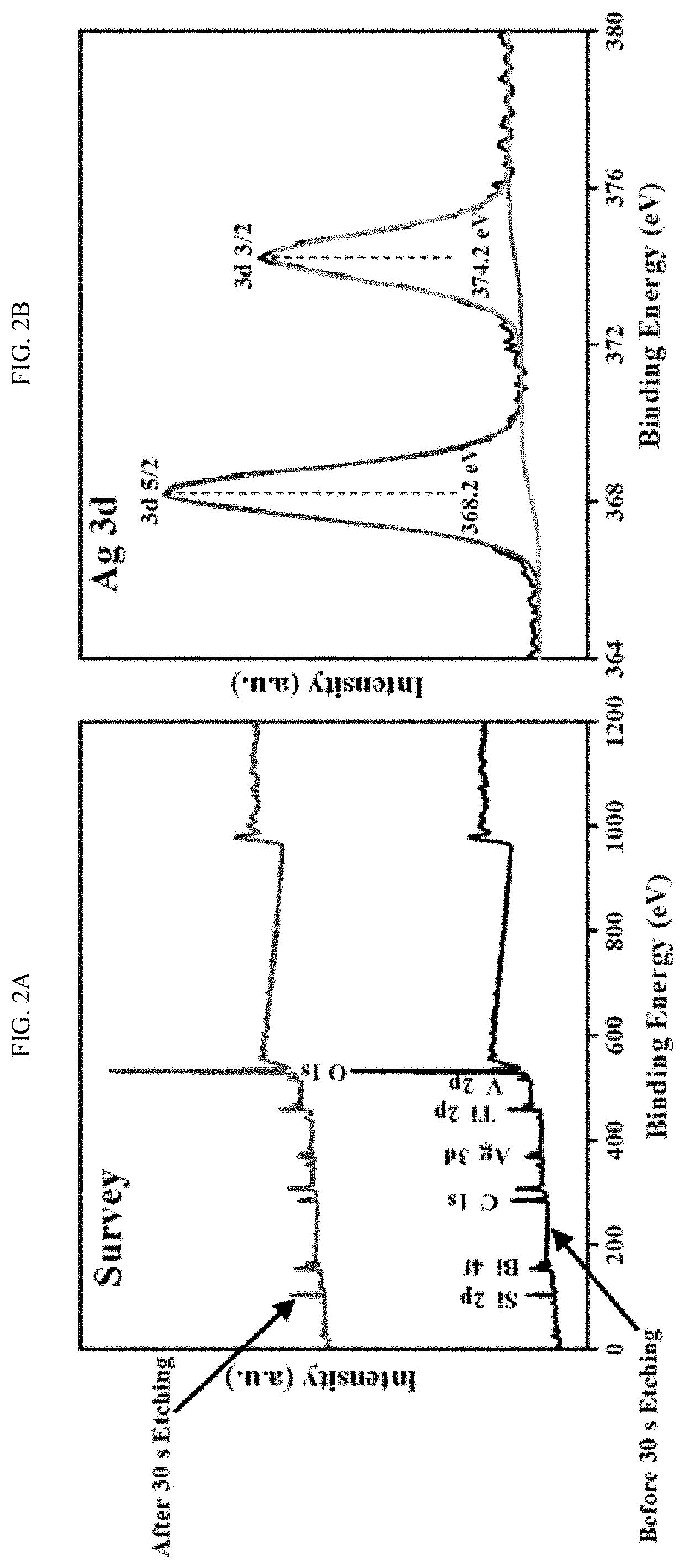
EFFICIENT METHOD FOR PLASMON-AIDED SOLAR WATER-SPLITTING USING (BiVO4)X–(TiO2)1-X TERNARY NANOCOMPOSITES
PatentActiveUS20200024754A1
Innovation
- A nanocomposite series (BiVO4)x-(TiO2)1-x is synthesized using a solvothermal method, with Ag nanoparticles impregnated on the surface to enhance plasmon resonance, resulting in a regular morphology, increased surface area, and improved polydispersity, which enhances solar-driven water splitting.
Scalability and Commercialization Pathways
The transition from laboratory-scale PEC water splitting systems to commercially viable technologies represents a significant challenge in the field. Current laboratory demonstrations typically operate at scales of a few square centimeters, while commercial applications would require systems spanning hundreds or thousands of square meters. This scaling disparity necessitates innovative approaches to manufacturing, system integration, and cost reduction.
Manufacturing scalability presents the first major hurdle. Traditional fabrication methods for high-performance photoelectrodes often involve complex processes such as atomic layer deposition or molecular beam epitaxy, which are prohibitively expensive at large scales. Recent developments in solution-based processing, roll-to-roll manufacturing, and electrodeposition show promise for cost-effective mass production of PEC materials.
Cost considerations remain paramount for commercial viability. Current PEC systems achieve solar-to-hydrogen efficiencies of 5-15%, with production costs estimated at $10-15/kg H₂. To compete with conventional hydrogen production methods ($1-3/kg H₂), significant improvements in both efficiency and manufacturing costs are required. Material innovations using earth-abundant elements rather than precious metals represent a crucial pathway toward cost reduction.
System durability presents another commercialization challenge. Laboratory demonstrations often achieve stability over hundreds of hours, while commercial applications require operational lifetimes of 5-10 years (40,000+ hours). Recent advances in protective coatings and self-healing materials show promise for extending device lifetimes under real-world conditions.
Several commercialization pathways are emerging in the industry. The modular approach involves developing standardized PEC panels that can be deployed at various scales, similar to photovoltaic installations. This allows for gradual market entry and distributed hydrogen production. Alternatively, the integrated approach focuses on large-scale centralized facilities that combine PEC water splitting with existing industrial infrastructure.
Strategic partnerships between material science innovators, manufacturing experts, and energy companies are accelerating commercialization efforts. Notable examples include collaborations between academic institutions developing novel materials and industrial partners with manufacturing expertise. Government initiatives supporting renewable hydrogen production are further catalyzing these partnerships through targeted funding programs.
Market entry strategies increasingly focus on niche applications where hydrogen's value exceeds bulk commodity prices. These include specialized industrial processes, remote power applications, and green chemistry initiatives where the premium for carbon-neutral hydrogen justifies higher production costs during early commercialization phases.
Manufacturing scalability presents the first major hurdle. Traditional fabrication methods for high-performance photoelectrodes often involve complex processes such as atomic layer deposition or molecular beam epitaxy, which are prohibitively expensive at large scales. Recent developments in solution-based processing, roll-to-roll manufacturing, and electrodeposition show promise for cost-effective mass production of PEC materials.
Cost considerations remain paramount for commercial viability. Current PEC systems achieve solar-to-hydrogen efficiencies of 5-15%, with production costs estimated at $10-15/kg H₂. To compete with conventional hydrogen production methods ($1-3/kg H₂), significant improvements in both efficiency and manufacturing costs are required. Material innovations using earth-abundant elements rather than precious metals represent a crucial pathway toward cost reduction.
System durability presents another commercialization challenge. Laboratory demonstrations often achieve stability over hundreds of hours, while commercial applications require operational lifetimes of 5-10 years (40,000+ hours). Recent advances in protective coatings and self-healing materials show promise for extending device lifetimes under real-world conditions.
Several commercialization pathways are emerging in the industry. The modular approach involves developing standardized PEC panels that can be deployed at various scales, similar to photovoltaic installations. This allows for gradual market entry and distributed hydrogen production. Alternatively, the integrated approach focuses on large-scale centralized facilities that combine PEC water splitting with existing industrial infrastructure.
Strategic partnerships between material science innovators, manufacturing experts, and energy companies are accelerating commercialization efforts. Notable examples include collaborations between academic institutions developing novel materials and industrial partners with manufacturing expertise. Government initiatives supporting renewable hydrogen production are further catalyzing these partnerships through targeted funding programs.
Market entry strategies increasingly focus on niche applications where hydrogen's value exceeds bulk commodity prices. These include specialized industrial processes, remote power applications, and green chemistry initiatives where the premium for carbon-neutral hydrogen justifies higher production costs during early commercialization phases.
Environmental Impact and Sustainability Assessment
Photoelectrochemical (PEC) water splitting represents a promising pathway toward sustainable hydrogen production, yet its environmental implications warrant thorough examination. The materials used in PEC systems, while enabling clean hydrogen generation, may themselves carry significant environmental footprints throughout their lifecycle.
Primary materials in advanced PEC systems often include rare earth elements and precious metals that require energy-intensive mining and processing operations. These extraction activities can lead to habitat destruction, soil degradation, and water pollution in mining regions. For instance, the production of semiconductor materials like gallium arsenide or cadmium telluride involves toxic compounds that pose environmental risks if improperly managed during manufacturing or disposal phases.
Water consumption represents another critical environmental consideration. While PEC systems split water to produce hydrogen, the manufacturing processes for PEC materials themselves can be water-intensive. This creates a potential sustainability paradox where water-saving technology requires significant water inputs during production. Comprehensive water footprint analyses indicate that advanced material fabrication techniques may require between 2,000-4,000 liters of ultrapure water per square meter of photoelectrode material.
Carbon emissions associated with PEC material production must also be evaluated against the emissions avoided through hydrogen utilization. Life cycle assessments reveal that current manufacturing processes for high-efficiency PEC materials generate substantial carbon footprints, potentially requiring 3-5 years of system operation to achieve carbon payback. However, emerging low-temperature synthesis methods and energy-efficient manufacturing techniques are progressively reducing this environmental burden.
Material durability and end-of-life management present additional sustainability challenges. Current PEC materials often demonstrate degradation under operating conditions, necessitating replacement and creating waste streams. Developing circular economy approaches for PEC materials, including recovery and recycling protocols for valuable components, remains an underdeveloped research area despite its importance for long-term sustainability.
Recent innovations in bio-inspired and earth-abundant materials offer promising pathways to reduce environmental impacts. These include iron oxide-based photoelectrodes, carbon nitride photocatalysts, and biomass-derived sensitizers that minimize reliance on rare or toxic elements while maintaining reasonable conversion efficiencies. Such materials typically demonstrate 30-50% lower environmental impact scores across multiple indicators compared to conventional alternatives.
Standardization of environmental impact assessment methodologies specifically tailored to PEC technologies remains crucial for meaningful comparison between different material systems and manufacturing approaches. This would enable researchers and industry to prioritize truly sustainable innovation pathways rather than focusing exclusively on efficiency metrics.
Primary materials in advanced PEC systems often include rare earth elements and precious metals that require energy-intensive mining and processing operations. These extraction activities can lead to habitat destruction, soil degradation, and water pollution in mining regions. For instance, the production of semiconductor materials like gallium arsenide or cadmium telluride involves toxic compounds that pose environmental risks if improperly managed during manufacturing or disposal phases.
Water consumption represents another critical environmental consideration. While PEC systems split water to produce hydrogen, the manufacturing processes for PEC materials themselves can be water-intensive. This creates a potential sustainability paradox where water-saving technology requires significant water inputs during production. Comprehensive water footprint analyses indicate that advanced material fabrication techniques may require between 2,000-4,000 liters of ultrapure water per square meter of photoelectrode material.
Carbon emissions associated with PEC material production must also be evaluated against the emissions avoided through hydrogen utilization. Life cycle assessments reveal that current manufacturing processes for high-efficiency PEC materials generate substantial carbon footprints, potentially requiring 3-5 years of system operation to achieve carbon payback. However, emerging low-temperature synthesis methods and energy-efficient manufacturing techniques are progressively reducing this environmental burden.
Material durability and end-of-life management present additional sustainability challenges. Current PEC materials often demonstrate degradation under operating conditions, necessitating replacement and creating waste streams. Developing circular economy approaches for PEC materials, including recovery and recycling protocols for valuable components, remains an underdeveloped research area despite its importance for long-term sustainability.
Recent innovations in bio-inspired and earth-abundant materials offer promising pathways to reduce environmental impacts. These include iron oxide-based photoelectrodes, carbon nitride photocatalysts, and biomass-derived sensitizers that minimize reliance on rare or toxic elements while maintaining reasonable conversion efficiencies. Such materials typically demonstrate 30-50% lower environmental impact scores across multiple indicators compared to conventional alternatives.
Standardization of environmental impact assessment methodologies specifically tailored to PEC technologies remains crucial for meaningful comparison between different material systems and manufacturing approaches. This would enable researchers and industry to prioritize truly sustainable innovation pathways rather than focusing exclusively on efficiency metrics.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!