Evaluating biocompatibility in laryngoscope materials.
JUL 14, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Laryngoscope Biocompatibility Background and Objectives
Laryngoscopes have been an essential tool in medical practice for over a century, enabling healthcare professionals to visualize and access the larynx and vocal cords. As these devices come into direct contact with sensitive tissues in the upper airway, the biocompatibility of materials used in their construction is of paramount importance. The evolution of laryngoscope design has been closely tied to advancements in material science and bioengineering, with a constant focus on improving safety, efficacy, and patient comfort.
The primary objective of evaluating biocompatibility in laryngoscope materials is to ensure that these devices can be used safely without causing adverse reactions or tissue damage. This involves assessing the materials' potential for cytotoxicity, irritation, sensitization, and systemic toxicity. Additionally, the evaluation aims to identify materials that resist microbial colonization and can withstand repeated sterilization processes without degradation.
Historically, laryngoscopes were primarily made of metal alloys, which provided durability but posed challenges in terms of weight and potential for tissue trauma. The introduction of plastics and composite materials in the mid-20th century marked a significant shift in laryngoscope design, offering lighter weight options with improved ergonomics. However, these new materials brought their own set of biocompatibility concerns, necessitating rigorous testing and evaluation protocols.
Recent technological advancements have led to the exploration of novel materials such as biocompatible polymers, antimicrobial coatings, and even biodegradable components for single-use devices. These innovations aim to address longstanding issues such as cross-contamination risks and environmental sustainability while maintaining or improving the device's performance and safety profile.
The current landscape of laryngoscope biocompatibility research is characterized by a multidisciplinary approach, combining expertise from materials science, biomedical engineering, and clinical medicine. Researchers are focusing on developing materials that not only meet basic safety requirements but also enhance functionality, such as improving grip in wet conditions or reducing glare during procedures.
As healthcare practices evolve, there is an increasing emphasis on personalized medicine and patient-specific care. This trend is reflected in the objectives of biocompatibility research for laryngoscopes, with efforts directed towards developing materials that can be tailored to individual patient needs or specific clinical scenarios. Furthermore, the global push towards more sustainable healthcare practices is driving research into eco-friendly materials that maintain high biocompatibility standards.
In conclusion, the background and objectives of evaluating biocompatibility in laryngoscope materials encompass a rich history of medical device innovation, coupled with an ongoing commitment to patient safety and clinical efficacy. The field continues to evolve, driven by technological advancements and changing healthcare paradigms, with the ultimate goal of creating laryngoscopes that are safer, more effective, and better suited to the diverse needs of modern medical practice.
The primary objective of evaluating biocompatibility in laryngoscope materials is to ensure that these devices can be used safely without causing adverse reactions or tissue damage. This involves assessing the materials' potential for cytotoxicity, irritation, sensitization, and systemic toxicity. Additionally, the evaluation aims to identify materials that resist microbial colonization and can withstand repeated sterilization processes without degradation.
Historically, laryngoscopes were primarily made of metal alloys, which provided durability but posed challenges in terms of weight and potential for tissue trauma. The introduction of plastics and composite materials in the mid-20th century marked a significant shift in laryngoscope design, offering lighter weight options with improved ergonomics. However, these new materials brought their own set of biocompatibility concerns, necessitating rigorous testing and evaluation protocols.
Recent technological advancements have led to the exploration of novel materials such as biocompatible polymers, antimicrobial coatings, and even biodegradable components for single-use devices. These innovations aim to address longstanding issues such as cross-contamination risks and environmental sustainability while maintaining or improving the device's performance and safety profile.
The current landscape of laryngoscope biocompatibility research is characterized by a multidisciplinary approach, combining expertise from materials science, biomedical engineering, and clinical medicine. Researchers are focusing on developing materials that not only meet basic safety requirements but also enhance functionality, such as improving grip in wet conditions or reducing glare during procedures.
As healthcare practices evolve, there is an increasing emphasis on personalized medicine and patient-specific care. This trend is reflected in the objectives of biocompatibility research for laryngoscopes, with efforts directed towards developing materials that can be tailored to individual patient needs or specific clinical scenarios. Furthermore, the global push towards more sustainable healthcare practices is driving research into eco-friendly materials that maintain high biocompatibility standards.
In conclusion, the background and objectives of evaluating biocompatibility in laryngoscope materials encompass a rich history of medical device innovation, coupled with an ongoing commitment to patient safety and clinical efficacy. The field continues to evolve, driven by technological advancements and changing healthcare paradigms, with the ultimate goal of creating laryngoscopes that are safer, more effective, and better suited to the diverse needs of modern medical practice.
Market Analysis for Biocompatible Laryngoscopes
The market for biocompatible laryngoscopes has experienced significant growth in recent years, driven by increasing awareness of patient safety and the need for improved medical devices. The global laryngoscope market, which includes biocompatible devices, is projected to reach a substantial value by 2025, with a compound annual growth rate (CAGR) exceeding 6% during the forecast period.
The demand for biocompatible laryngoscopes is primarily fueled by the rising number of surgical procedures, particularly in the fields of anesthesiology, emergency medicine, and otolaryngology. As healthcare systems worldwide continue to expand and modernize, the adoption of advanced medical devices, including biocompatible laryngoscopes, is expected to increase.
One of the key factors driving market growth is the growing emphasis on infection control and prevention of cross-contamination in healthcare settings. Biocompatible materials used in laryngoscopes reduce the risk of adverse reactions and infections, making them increasingly preferred by healthcare professionals and patients alike.
The market is also influenced by technological advancements in laryngoscope design and materials. Manufacturers are investing in research and development to create innovative biocompatible materials that offer improved durability, flexibility, and optical properties. This trend is expected to continue, further stimulating market growth and product differentiation.
Geographically, North America and Europe currently dominate the biocompatible laryngoscope market, owing to well-established healthcare infrastructure and higher healthcare expenditure. However, the Asia-Pacific region is anticipated to witness the fastest growth in the coming years, driven by improving healthcare facilities, increasing medical tourism, and rising disposable incomes in emerging economies such as China and India.
The market landscape is characterized by the presence of both established medical device manufacturers and innovative start-ups. Key players are focusing on strategic collaborations, mergers, and acquisitions to expand their product portfolios and geographical presence. Additionally, there is a growing trend towards the development of disposable laryngoscopes made from biocompatible materials, addressing concerns related to sterilization and cross-contamination.
Despite the positive outlook, the market faces challenges such as stringent regulatory requirements for medical devices and the high cost associated with advanced biocompatible materials. However, ongoing research in material science and increasing healthcare investments are expected to mitigate these challenges and drive sustained market growth in the foreseeable future.
The demand for biocompatible laryngoscopes is primarily fueled by the rising number of surgical procedures, particularly in the fields of anesthesiology, emergency medicine, and otolaryngology. As healthcare systems worldwide continue to expand and modernize, the adoption of advanced medical devices, including biocompatible laryngoscopes, is expected to increase.
One of the key factors driving market growth is the growing emphasis on infection control and prevention of cross-contamination in healthcare settings. Biocompatible materials used in laryngoscopes reduce the risk of adverse reactions and infections, making them increasingly preferred by healthcare professionals and patients alike.
The market is also influenced by technological advancements in laryngoscope design and materials. Manufacturers are investing in research and development to create innovative biocompatible materials that offer improved durability, flexibility, and optical properties. This trend is expected to continue, further stimulating market growth and product differentiation.
Geographically, North America and Europe currently dominate the biocompatible laryngoscope market, owing to well-established healthcare infrastructure and higher healthcare expenditure. However, the Asia-Pacific region is anticipated to witness the fastest growth in the coming years, driven by improving healthcare facilities, increasing medical tourism, and rising disposable incomes in emerging economies such as China and India.
The market landscape is characterized by the presence of both established medical device manufacturers and innovative start-ups. Key players are focusing on strategic collaborations, mergers, and acquisitions to expand their product portfolios and geographical presence. Additionally, there is a growing trend towards the development of disposable laryngoscopes made from biocompatible materials, addressing concerns related to sterilization and cross-contamination.
Despite the positive outlook, the market faces challenges such as stringent regulatory requirements for medical devices and the high cost associated with advanced biocompatible materials. However, ongoing research in material science and increasing healthcare investments are expected to mitigate these challenges and drive sustained market growth in the foreseeable future.
Current Challenges in Laryngoscope Material Biocompatibility
The biocompatibility of laryngoscope materials presents several significant challenges in the current medical device landscape. One of the primary concerns is the potential for allergic reactions or sensitivities to certain materials used in laryngoscope construction. Traditional materials such as stainless steel and rubber compounds have been associated with allergic responses in some patients, necessitating the exploration of alternative, hypoallergenic options.
Another challenge lies in the balance between material durability and biocompatibility. Laryngoscopes are subjected to frequent sterilization processes and mechanical stress during use, requiring materials that can withstand these conditions while maintaining their biocompatible properties. This dual requirement often leads to compromises in either durability or biocompatibility, as materials that excel in one area may fall short in the other.
The risk of microbial colonization on laryngoscope surfaces poses an additional biocompatibility challenge. Despite rigorous sterilization protocols, certain materials may be more prone to harboring bacteria or other microorganisms, potentially leading to healthcare-associated infections. Developing materials with inherent antimicrobial properties without compromising other aspects of biocompatibility remains a complex task.
Furthermore, the interaction between laryngoscope materials and various medications or bodily fluids encountered during intubation procedures can lead to material degradation or the release of potentially harmful substances. This chemical compatibility issue requires extensive testing and careful material selection to ensure long-term safety and efficacy.
The regulatory landscape surrounding biocompatible materials for medical devices adds another layer of complexity. Stringent approval processes and evolving standards necessitate continuous research and development efforts to meet or exceed regulatory requirements while addressing the aforementioned challenges.
Lastly, the cost-effectiveness of biocompatible materials presents a significant hurdle. Advanced materials that offer superior biocompatibility often come at a higher price point, potentially limiting their widespread adoption in healthcare settings with budget constraints. Striking a balance between cost and performance remains an ongoing challenge in the development of biocompatible laryngoscope materials.
Another challenge lies in the balance between material durability and biocompatibility. Laryngoscopes are subjected to frequent sterilization processes and mechanical stress during use, requiring materials that can withstand these conditions while maintaining their biocompatible properties. This dual requirement often leads to compromises in either durability or biocompatibility, as materials that excel in one area may fall short in the other.
The risk of microbial colonization on laryngoscope surfaces poses an additional biocompatibility challenge. Despite rigorous sterilization protocols, certain materials may be more prone to harboring bacteria or other microorganisms, potentially leading to healthcare-associated infections. Developing materials with inherent antimicrobial properties without compromising other aspects of biocompatibility remains a complex task.
Furthermore, the interaction between laryngoscope materials and various medications or bodily fluids encountered during intubation procedures can lead to material degradation or the release of potentially harmful substances. This chemical compatibility issue requires extensive testing and careful material selection to ensure long-term safety and efficacy.
The regulatory landscape surrounding biocompatible materials for medical devices adds another layer of complexity. Stringent approval processes and evolving standards necessitate continuous research and development efforts to meet or exceed regulatory requirements while addressing the aforementioned challenges.
Lastly, the cost-effectiveness of biocompatible materials presents a significant hurdle. Advanced materials that offer superior biocompatibility often come at a higher price point, potentially limiting their widespread adoption in healthcare settings with budget constraints. Striking a balance between cost and performance remains an ongoing challenge in the development of biocompatible laryngoscope materials.
Existing Biocompatible Materials for Laryngoscopes
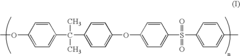
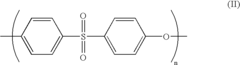
01 Biocompatible polymers for laryngoscope components
Laryngoscopes can be manufactured using biocompatible polymers to ensure safety during use. These materials are chosen for their non-toxic properties and ability to withstand sterilization processes. The use of such polymers helps reduce the risk of adverse reactions in patients and allows for the creation of disposable or reusable laryngoscope components.- Biocompatible polymers for laryngoscope components: Laryngoscopes can be constructed using biocompatible polymers to ensure safety and reduce the risk of adverse reactions. These materials are chosen for their compatibility with human tissue and their ability to withstand sterilization processes. Polymers such as medical-grade silicone, polyurethane, or polycarbonate may be used for various components of the laryngoscope, including the blade and handle.
- Metal alloys with biocompatible coatings: Metal alloys, such as stainless steel or titanium, are often used in laryngoscope construction due to their strength and durability. To enhance biocompatibility, these metals can be coated with biocompatible materials like titanium nitride or diamond-like carbon. These coatings provide a barrier between the metal and human tissue, reducing the risk of allergic reactions or corrosion.
- Disposable laryngoscope blades: Disposable laryngoscope blades made from biocompatible materials address concerns about cross-contamination and sterilization. These single-use blades are typically constructed from medical-grade plastics or reinforced polymers that are safe for patient contact and can be discarded after use, eliminating the need for reprocessing and reducing the risk of infection transmission.
- Antimicrobial materials and coatings: Incorporating antimicrobial materials or coatings into laryngoscope components can enhance their biocompatibility by reducing the risk of bacterial colonization and infection. These may include silver nanoparticles, copper alloys, or specialized polymer blends with inherent antimicrobial properties. Such materials can be used in the construction of blades, handles, or as surface treatments.
- Biocompatible electronic components: As laryngoscopes become more advanced, incorporating electronic components such as cameras or sensors, ensuring the biocompatibility of these elements is crucial. This includes using encapsulation techniques with biocompatible materials to isolate electronic components from direct tissue contact, as well as selecting low-toxicity materials for circuit boards and connectors that may come into proximity with the patient.
02 Metal alloys with enhanced biocompatibility
Certain metal alloys are utilized in laryngoscope construction due to their biocompatibility and durability. These alloys are carefully selected to minimize the risk of allergic reactions or tissue irritation. The use of such materials allows for the creation of sturdy, long-lasting laryngoscope blades and handles that can withstand repeated use and sterilization.Expand Specific Solutions03 Coatings to improve biocompatibility
Specialized coatings can be applied to laryngoscope surfaces to enhance their biocompatibility. These coatings may include antimicrobial agents or materials that reduce friction and tissue trauma during intubation. Such coatings can significantly improve the safety profile of the device and reduce the risk of infection or injury during use.Expand Specific Solutions04 Disposable covers for improved hygiene
Disposable, biocompatible covers or sheaths can be used with laryngoscopes to enhance hygiene and reduce the risk of cross-contamination. These covers are typically made from materials that do not interfere with the function of the laryngoscope while providing a barrier between the device and the patient's tissues.Expand Specific Solutions05 Novel materials for optical components
Advanced, biocompatible materials are being developed for use in the optical components of laryngoscopes. These materials offer improved light transmission and durability while maintaining a high level of biocompatibility. The use of such materials can enhance the performance of video laryngoscopes and other devices with integrated imaging systems.Expand Specific Solutions
Key Players in Laryngoscope Manufacturing and Biomaterials
The biocompatibility evaluation of laryngoscope materials is in a mature stage of development, with a significant market size due to the essential nature of these devices in medical procedures. The technology's maturity is evident from the involvement of established players like Bausch & Lomb and Koninklijke Philips NV, alongside specialized medical equipment manufacturers such as Zhejiang Youyi Medical Equipment Co Ltd. The competitive landscape is diverse, including both large multinational corporations and smaller, specialized firms. Research institutions like Johns Hopkins University and Kyushu University contribute to ongoing advancements, indicating a continuous focus on improving material biocompatibility and device performance.
Zhejiang Youyi Medical Equipment Co Ltd
Technical Solution: Zhejiang Youyi Medical Equipment Co Ltd has developed a novel approach to evaluating biocompatibility in laryngoscope materials. Their method involves a multi-step process including in vitro cytotoxicity testing, sensitization studies, and irritation tests[1]. The company utilizes advanced surface modification techniques to enhance the biocompatibility of their laryngoscope blades, incorporating antimicrobial coatings that reduce the risk of infection during intubation procedures[2]. They have also implemented a rigorous quality control system that includes regular biocompatibility assessments of their materials throughout the manufacturing process[3].
Strengths: Comprehensive testing methodology, innovative surface treatments, and stringent quality control. Weaknesses: Potential increased production costs due to extensive testing and specialized coatings.
Koninklijke Philips NV
Technical Solution: Koninklijke Philips NV has developed a sophisticated approach to evaluating biocompatibility in laryngoscope materials. Their method incorporates advanced imaging techniques, including high-resolution microscopy and spectroscopic analysis, to assess material-tissue interactions at the molecular level[4]. Philips has also pioneered the use of 3D-printed biocompatible polymers for laryngoscope components, allowing for customized designs that minimize tissue trauma during intubation[5]. The company employs machine learning algorithms to analyze biocompatibility data from clinical trials, enabling rapid identification of optimal material compositions for different patient populations[6].
Strengths: Cutting-edge imaging and analysis techniques, customizable 3D-printed components, and data-driven material optimization. Weaknesses: High development costs and potential regulatory hurdles for novel materials and manufacturing processes.
Innovative Biocompatibility Testing Methods for Laryngoscopes
Method of evaluating biocompatibility
PatentInactiveUS20040241690A1
Innovation
- A method involving in-vitro contact of leukocytes with medical materials, followed by RNA extraction and real-time PCR or DNA array analysis to quantify the expression levels of urokinase plasminogen activator receptor gene and protein, allowing for sensitive and quantitative evaluation of biocompatibility without requiring clinical blood samples and minimizing endotoxin contamination.
Tissue Augmentation Material and Method
PatentInactiveUS20100100179A1
Innovation
- A biocompatible material comprising smooth, rounded, substantially spherical ceramic particles, particularly calcium hydroxyapatite, is used in conjunction with a polysaccharide gel carrier for soft tissue augmentation, providing a scaffold for autogenous tissue growth and minimizing foreign body response.
Regulatory Framework for Medical Device Biocompatibility
The regulatory framework for medical device biocompatibility is a critical aspect of evaluating laryngoscope materials. This framework is designed to ensure the safety and efficacy of medical devices, including laryngoscopes, which come into direct contact with human tissues during use.
At the forefront of this regulatory landscape is the International Organization for Standardization (ISO) 10993 series, which provides a comprehensive set of standards for biological evaluation of medical devices. ISO 10993-1, in particular, outlines the general principles of biological assessment and the categorization of devices based on the nature and duration of their contact with the body.
For laryngoscopes, which are typically classified as surface devices with limited contact duration, the FDA recommends a subset of biocompatibility tests. These may include cytotoxicity, sensitization, and irritation tests. The specific tests required can vary depending on the materials used and the intended use of the device.
In the United States, the Food and Drug Administration (FDA) oversees the regulation of medical devices. The FDA's guidance document "Use of International Standard ISO 10993-1" provides recommendations for biological evaluation of medical devices as part of the overall risk management process.
The European Union's Medical Device Regulation (MDR) also incorporates biocompatibility requirements. Under the MDR, manufacturers must demonstrate compliance with the General Safety and Performance Requirements, which include biocompatibility considerations.
Manufacturers of laryngoscopes must navigate these regulatory requirements by conducting appropriate biocompatibility testing and risk assessments. This process typically involves selecting materials with a history of safe use in medical devices, performing the necessary biological evaluations, and documenting the results in a biological evaluation report.
It's important to note that regulatory requirements can evolve over time. For instance, there is an increasing focus on the potential long-term effects of materials, even for devices with limited contact duration. This has led to more stringent requirements for chemical characterization and toxicological risk assessments.
Compliance with these regulatory frameworks not only ensures patient safety but also facilitates market access. Many countries have adopted or recognize ISO 10993 standards, which can streamline the approval process for manufacturers seeking to market their devices globally.
At the forefront of this regulatory landscape is the International Organization for Standardization (ISO) 10993 series, which provides a comprehensive set of standards for biological evaluation of medical devices. ISO 10993-1, in particular, outlines the general principles of biological assessment and the categorization of devices based on the nature and duration of their contact with the body.
For laryngoscopes, which are typically classified as surface devices with limited contact duration, the FDA recommends a subset of biocompatibility tests. These may include cytotoxicity, sensitization, and irritation tests. The specific tests required can vary depending on the materials used and the intended use of the device.
In the United States, the Food and Drug Administration (FDA) oversees the regulation of medical devices. The FDA's guidance document "Use of International Standard ISO 10993-1" provides recommendations for biological evaluation of medical devices as part of the overall risk management process.
The European Union's Medical Device Regulation (MDR) also incorporates biocompatibility requirements. Under the MDR, manufacturers must demonstrate compliance with the General Safety and Performance Requirements, which include biocompatibility considerations.
Manufacturers of laryngoscopes must navigate these regulatory requirements by conducting appropriate biocompatibility testing and risk assessments. This process typically involves selecting materials with a history of safe use in medical devices, performing the necessary biological evaluations, and documenting the results in a biological evaluation report.
It's important to note that regulatory requirements can evolve over time. For instance, there is an increasing focus on the potential long-term effects of materials, even for devices with limited contact duration. This has led to more stringent requirements for chemical characterization and toxicological risk assessments.
Compliance with these regulatory frameworks not only ensures patient safety but also facilitates market access. Many countries have adopted or recognize ISO 10993 standards, which can streamline the approval process for manufacturers seeking to market their devices globally.
Patient Safety and Infection Control Considerations
Patient safety and infection control are paramount considerations when evaluating biocompatibility in laryngoscope materials. The materials used in laryngoscopes must not only be effective for their intended purpose but also ensure the well-being of patients and healthcare providers.
One of the primary concerns in laryngoscope design is the prevention of cross-contamination between patients. Materials that are resistant to microbial colonization and can withstand rigorous sterilization processes are essential. Stainless steel, for instance, has been a popular choice due to its durability and ease of cleaning. However, newer materials such as medical-grade polymers are gaining traction for their lightweight properties and ability to be disposable, reducing the risk of pathogen transmission.
The biocompatibility of laryngoscope materials also extends to their potential for causing allergic reactions or tissue irritation. Nickel allergy, for example, has been a concern with some metal components. As a result, there is a growing trend towards using hypoallergenic materials or applying specialized coatings to minimize adverse reactions.
Another critical aspect is the material's resistance to degradation during use and sterilization. Repeated exposure to disinfectants and high-temperature sterilization methods can compromise the integrity of certain materials, potentially leading to the release of harmful substances or the formation of microcracks that harbor pathogens.
The choice of materials also impacts the efficacy of infection control protocols. Materials that allow for easy cleaning and disinfection without retaining residues are preferred. This has led to the development of smooth, non-porous surfaces that resist bacterial adhesion and facilitate thorough decontamination.
In recent years, there has been increased interest in incorporating antimicrobial properties directly into laryngoscope materials. This approach aims to provide an additional layer of protection against pathogen transmission. Technologies such as silver ion-infused polymers or copper-alloy coatings are being explored for their potential to actively reduce microbial loads on device surfaces.
The environmental impact of laryngoscope materials is also becoming a consideration in patient safety discussions. There is a growing awareness of the need to balance single-use, disposable designs that minimize infection risks with the environmental concerns of medical waste generation. This has spurred research into biodegradable materials that maintain the necessary biocompatibility and performance characteristics.
As healthcare-associated infections remain a significant concern, regulatory bodies are increasingly scrutinizing the materials used in medical devices. Manufacturers must now provide comprehensive data on the biocompatibility and infection control properties of their chosen materials, including long-term safety profiles and potential interactions with common sterilization methods.
One of the primary concerns in laryngoscope design is the prevention of cross-contamination between patients. Materials that are resistant to microbial colonization and can withstand rigorous sterilization processes are essential. Stainless steel, for instance, has been a popular choice due to its durability and ease of cleaning. However, newer materials such as medical-grade polymers are gaining traction for their lightweight properties and ability to be disposable, reducing the risk of pathogen transmission.
The biocompatibility of laryngoscope materials also extends to their potential for causing allergic reactions or tissue irritation. Nickel allergy, for example, has been a concern with some metal components. As a result, there is a growing trend towards using hypoallergenic materials or applying specialized coatings to minimize adverse reactions.
Another critical aspect is the material's resistance to degradation during use and sterilization. Repeated exposure to disinfectants and high-temperature sterilization methods can compromise the integrity of certain materials, potentially leading to the release of harmful substances or the formation of microcracks that harbor pathogens.
The choice of materials also impacts the efficacy of infection control protocols. Materials that allow for easy cleaning and disinfection without retaining residues are preferred. This has led to the development of smooth, non-porous surfaces that resist bacterial adhesion and facilitate thorough decontamination.
In recent years, there has been increased interest in incorporating antimicrobial properties directly into laryngoscope materials. This approach aims to provide an additional layer of protection against pathogen transmission. Technologies such as silver ion-infused polymers or copper-alloy coatings are being explored for their potential to actively reduce microbial loads on device surfaces.
The environmental impact of laryngoscope materials is also becoming a consideration in patient safety discussions. There is a growing awareness of the need to balance single-use, disposable designs that minimize infection risks with the environmental concerns of medical waste generation. This has spurred research into biodegradable materials that maintain the necessary biocompatibility and performance characteristics.
As healthcare-associated infections remain a significant concern, regulatory bodies are increasingly scrutinizing the materials used in medical devices. Manufacturers must now provide comprehensive data on the biocompatibility and infection control properties of their chosen materials, including long-term safety profiles and potential interactions with common sterilization methods.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!