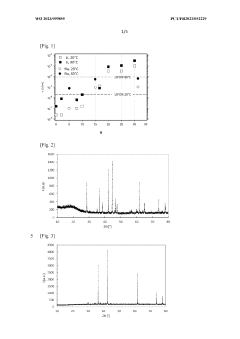
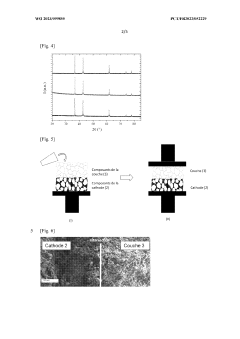
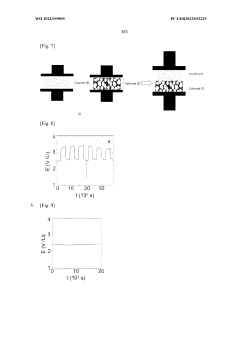
High Entropy Oxide Interfaces With Solid State Electrolytes
AUG 29, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
High Entropy Oxide Interface Development Background and Objectives
High entropy oxides (HEOs) have emerged as a revolutionary class of materials in the field of solid-state energy storage over the past decade. These materials, characterized by their incorporation of five or more cations in equimolar or near-equimolar proportions within a single crystallographic phase, represent a paradigm shift in materials design philosophy. The concept of high configurational entropy stabilization was first demonstrated in metallic alloys in 2004, but its extension to oxide systems only occurred in 2015, marking the beginning of intensive research into HEOs.
The development of HEO interfaces with solid-state electrolytes addresses critical challenges in next-generation energy storage technologies. Traditional lithium-ion batteries with liquid electrolytes face inherent limitations in energy density, safety, and form factor flexibility. Solid-state batteries promise to overcome these limitations but are hindered by interfacial issues between electrodes and electrolytes, including high resistance, chemical instability, and mechanical degradation during cycling.
HEO interfaces offer a promising solution pathway through their unique structural and compositional characteristics. The high entropy effect leads to exceptional properties including enhanced ionic conductivity, improved structural stability, and superior electrochemical performance. These attributes directly address the interfacial challenges that have limited solid-state battery commercialization.
The technical evolution in this field has accelerated significantly since 2018, with research groups worldwide exploring various compositional strategies and synthesis techniques. Initial work focused primarily on fundamental understanding of entropy stabilization mechanisms in oxides, while recent efforts have shifted toward tailored interface engineering for specific electrochemical applications.
Our technical objectives in this research domain are multifaceted. First, we aim to systematically investigate the relationship between compositional complexity and interfacial properties in HEO-electrolyte systems. Second, we seek to develop scalable synthesis methodologies that enable precise control over interface formation. Third, we intend to establish design principles for HEO interfaces that optimize ionic transport while maintaining mechanical integrity during electrochemical cycling.
The long-term vision encompasses the integration of optimized HEO interfaces into commercial solid-state battery architectures, potentially revolutionizing energy storage for applications ranging from portable electronics to electric vehicles and grid-scale storage. This technology aligns with global trends toward sustainable energy solutions and addresses the increasing demand for safer, higher-capacity energy storage systems with extended operational lifetimes.
The development of HEO interfaces with solid-state electrolytes addresses critical challenges in next-generation energy storage technologies. Traditional lithium-ion batteries with liquid electrolytes face inherent limitations in energy density, safety, and form factor flexibility. Solid-state batteries promise to overcome these limitations but are hindered by interfacial issues between electrodes and electrolytes, including high resistance, chemical instability, and mechanical degradation during cycling.
HEO interfaces offer a promising solution pathway through their unique structural and compositional characteristics. The high entropy effect leads to exceptional properties including enhanced ionic conductivity, improved structural stability, and superior electrochemical performance. These attributes directly address the interfacial challenges that have limited solid-state battery commercialization.
The technical evolution in this field has accelerated significantly since 2018, with research groups worldwide exploring various compositional strategies and synthesis techniques. Initial work focused primarily on fundamental understanding of entropy stabilization mechanisms in oxides, while recent efforts have shifted toward tailored interface engineering for specific electrochemical applications.
Our technical objectives in this research domain are multifaceted. First, we aim to systematically investigate the relationship between compositional complexity and interfacial properties in HEO-electrolyte systems. Second, we seek to develop scalable synthesis methodologies that enable precise control over interface formation. Third, we intend to establish design principles for HEO interfaces that optimize ionic transport while maintaining mechanical integrity during electrochemical cycling.
The long-term vision encompasses the integration of optimized HEO interfaces into commercial solid-state battery architectures, potentially revolutionizing energy storage for applications ranging from portable electronics to electric vehicles and grid-scale storage. This technology aligns with global trends toward sustainable energy solutions and addresses the increasing demand for safer, higher-capacity energy storage systems with extended operational lifetimes.
Market Analysis for Solid State Battery Technologies
The solid-state battery market is experiencing unprecedented growth, driven by increasing demand for high-energy density, safe, and long-lasting energy storage solutions. Current market valuations place the global solid-state battery sector at approximately $0.5 billion in 2023, with projections indicating a compound annual growth rate (CAGR) of 34.2% through 2030, potentially reaching $3.3 billion by the end of the decade.
The automotive industry represents the largest potential market for solid-state battery technologies, particularly those incorporating high entropy oxide interfaces with solid-state electrolytes. Major automotive manufacturers including Toyota, Volkswagen, and BMW have announced significant investments in solid-state battery development, with commercialization targets set between 2025 and 2028. These investments reflect the industry's recognition of solid-state batteries as a critical enabling technology for next-generation electric vehicles.
Consumer electronics constitutes the second-largest market segment, with manufacturers seeking higher energy density and improved safety profiles for portable devices. This sector is expected to adopt solid-state technologies earlier than automotive applications due to less stringent requirements for power output and cycle life.
Market analysis reveals that high entropy oxide interfaces with solid-state electrolytes address several critical pain points in current battery technologies. The enhanced ionic conductivity at these interfaces potentially solves the persistent challenge of high internal resistance in solid-state systems. Additionally, the improved electrochemical stability at electrode-electrolyte interfaces addresses degradation issues that have limited commercial viability.
Regional market distribution shows Asia-Pacific leading in both manufacturing capacity and R&D investment, with Japan and South Korea at the forefront. North America follows with significant venture capital funding flowing into startups focused on novel interface engineering approaches. Europe has established strong government-backed research initiatives, particularly in Germany and France.
Customer willingness-to-pay analysis indicates premium pricing potential for solid-state batteries incorporating high entropy oxide interfaces, with automotive OEMs willing to pay up to 30% more compared to conventional lithium-ion technologies, provided performance metrics meet or exceed targets for energy density (>400 Wh/kg) and cycle life (>1,000 cycles).
Market barriers include scaling manufacturing processes for high entropy oxide interfaces, supply chain constraints for critical materials, and competition from incremental improvements in conventional lithium-ion technologies. However, the unique performance advantages offered by high entropy oxide interfaces, particularly in terms of thermal stability and fast charging capabilities, position this technology favorably against competing approaches.
The automotive industry represents the largest potential market for solid-state battery technologies, particularly those incorporating high entropy oxide interfaces with solid-state electrolytes. Major automotive manufacturers including Toyota, Volkswagen, and BMW have announced significant investments in solid-state battery development, with commercialization targets set between 2025 and 2028. These investments reflect the industry's recognition of solid-state batteries as a critical enabling technology for next-generation electric vehicles.
Consumer electronics constitutes the second-largest market segment, with manufacturers seeking higher energy density and improved safety profiles for portable devices. This sector is expected to adopt solid-state technologies earlier than automotive applications due to less stringent requirements for power output and cycle life.
Market analysis reveals that high entropy oxide interfaces with solid-state electrolytes address several critical pain points in current battery technologies. The enhanced ionic conductivity at these interfaces potentially solves the persistent challenge of high internal resistance in solid-state systems. Additionally, the improved electrochemical stability at electrode-electrolyte interfaces addresses degradation issues that have limited commercial viability.
Regional market distribution shows Asia-Pacific leading in both manufacturing capacity and R&D investment, with Japan and South Korea at the forefront. North America follows with significant venture capital funding flowing into startups focused on novel interface engineering approaches. Europe has established strong government-backed research initiatives, particularly in Germany and France.
Customer willingness-to-pay analysis indicates premium pricing potential for solid-state batteries incorporating high entropy oxide interfaces, with automotive OEMs willing to pay up to 30% more compared to conventional lithium-ion technologies, provided performance metrics meet or exceed targets for energy density (>400 Wh/kg) and cycle life (>1,000 cycles).
Market barriers include scaling manufacturing processes for high entropy oxide interfaces, supply chain constraints for critical materials, and competition from incremental improvements in conventional lithium-ion technologies. However, the unique performance advantages offered by high entropy oxide interfaces, particularly in terms of thermal stability and fast charging capabilities, position this technology favorably against competing approaches.
Current Challenges in High Entropy Oxide-Electrolyte Interfaces
Despite significant advancements in high entropy oxide (HEO) materials and solid-state electrolytes (SSEs), their interfaces present formidable challenges that impede practical applications in energy storage devices. The primary obstacle lies in the interfacial resistance between HEOs and electrolytes, which significantly reduces ion transport efficiency and overall device performance. This resistance stems from chemical incompatibilities, lattice mismatches, and space charge effects that create barriers to ion migration across the interface.
Chemical instability at HEO-electrolyte interfaces represents another critical challenge. Many high entropy oxides undergo undesirable side reactions with electrolyte components, forming resistive interphases that evolve over time. These reactions can lead to capacity fading, increased impedance, and shortened device lifetimes. The multi-elemental nature of HEOs complicates this issue, as different constituent elements may exhibit varying reactivity with the electrolyte.
Mechanical stress at interfaces poses additional difficulties. During cycling, volume changes in electrode materials create mechanical strain at the HEO-electrolyte interface, potentially leading to delamination, crack formation, and loss of electrical contact. This mechanical degradation is particularly problematic in solid-state systems where maintaining intimate contact between components is essential for efficient ion transfer.
The complex microstructure of HEO interfaces presents characterization challenges that hinder fundamental understanding. Traditional analytical techniques often lack the spatial resolution and chemical sensitivity required to fully elucidate interfacial phenomena in these multi-element systems. This knowledge gap impedes rational design approaches for interface engineering.
Manufacturing scalable, high-quality interfaces between HEOs and solid electrolytes remains technically demanding. Current fabrication methods often fail to create uniform, defect-free interfaces at scales relevant for commercial applications. Processing parameters must be precisely controlled to avoid elemental segregation, phase separation, or introduction of impurities that could compromise interfacial properties.
Temperature sensitivity further complicates interface management. Many promising HEO-electrolyte combinations exhibit accelerated degradation at elevated temperatures, limiting operational windows for devices. Conversely, low temperatures can significantly reduce ionic conductivity across interfaces, affecting cold-start capabilities in applications like electric vehicles.
Addressing these challenges requires interdisciplinary approaches combining materials science, electrochemistry, and advanced manufacturing. Recent research has begun exploring interface engineering strategies including buffer layers, gradient compositions, and surface modifications to mitigate these issues, though comprehensive solutions remain elusive.
Chemical instability at HEO-electrolyte interfaces represents another critical challenge. Many high entropy oxides undergo undesirable side reactions with electrolyte components, forming resistive interphases that evolve over time. These reactions can lead to capacity fading, increased impedance, and shortened device lifetimes. The multi-elemental nature of HEOs complicates this issue, as different constituent elements may exhibit varying reactivity with the electrolyte.
Mechanical stress at interfaces poses additional difficulties. During cycling, volume changes in electrode materials create mechanical strain at the HEO-electrolyte interface, potentially leading to delamination, crack formation, and loss of electrical contact. This mechanical degradation is particularly problematic in solid-state systems where maintaining intimate contact between components is essential for efficient ion transfer.
The complex microstructure of HEO interfaces presents characterization challenges that hinder fundamental understanding. Traditional analytical techniques often lack the spatial resolution and chemical sensitivity required to fully elucidate interfacial phenomena in these multi-element systems. This knowledge gap impedes rational design approaches for interface engineering.
Manufacturing scalable, high-quality interfaces between HEOs and solid electrolytes remains technically demanding. Current fabrication methods often fail to create uniform, defect-free interfaces at scales relevant for commercial applications. Processing parameters must be precisely controlled to avoid elemental segregation, phase separation, or introduction of impurities that could compromise interfacial properties.
Temperature sensitivity further complicates interface management. Many promising HEO-electrolyte combinations exhibit accelerated degradation at elevated temperatures, limiting operational windows for devices. Conversely, low temperatures can significantly reduce ionic conductivity across interfaces, affecting cold-start capabilities in applications like electric vehicles.
Addressing these challenges requires interdisciplinary approaches combining materials science, electrochemistry, and advanced manufacturing. Recent research has begun exploring interface engineering strategies including buffer layers, gradient compositions, and surface modifications to mitigate these issues, though comprehensive solutions remain elusive.
Current Interface Engineering Solutions for Solid State Batteries
01 High entropy oxide materials for solid-state electrolytes
High entropy oxides (HEOs) are being developed as novel materials for solid-state electrolytes in battery applications. These materials contain multiple metal cations in near-equiatomic proportions, creating high configurational entropy that can enhance ionic conductivity and stability. The unique structure of HEOs allows for tailored properties through compositional engineering, potentially addressing key challenges in solid-state electrolytes such as interfacial resistance and mechanical stability.- High entropy oxide materials for solid-state electrolytes: High entropy oxides (HEOs) are being developed as novel materials for solid-state electrolytes in energy storage applications. These materials contain multiple metal cations in near-equiatomic proportions, creating high configurational entropy that can enhance ionic conductivity and stability. The unique structure of HEOs allows for tailored properties through compositional engineering, potentially overcoming limitations of traditional solid electrolytes such as low ionic conductivity and poor interfacial compatibility.
- Interface engineering between high entropy oxides and solid electrolytes: Interface engineering is crucial for optimizing the performance of high entropy oxide interfaces with solid-state electrolytes. Various techniques are employed to modify these interfaces, including surface treatments, buffer layers, and gradient compositions. These approaches aim to reduce interfacial resistance, prevent unwanted reactions, and enhance ion transport across the interface. Proper interface design can mitigate issues such as dendrite formation and chemical degradation that typically occur at solid-solid interfaces in battery systems.
- Electrochemical performance enhancement through compositional tuning: The electrochemical performance of high entropy oxide interfaces with solid-state electrolytes can be significantly enhanced through compositional tuning. By adjusting the ratio and types of metal cations in the high entropy oxide structure, researchers can optimize properties such as ionic conductivity, electronic conductivity, and electrochemical stability. This approach allows for the development of tailored materials with improved cycling stability, rate capability, and energy density for next-generation solid-state batteries.
- Fabrication methods for high entropy oxide-electrolyte interfaces: Various fabrication methods are employed to create high-quality interfaces between high entropy oxides and solid-state electrolytes. These include sol-gel processing, co-precipitation, solid-state reaction, pulsed laser deposition, and atomic layer deposition. Each method offers different advantages in terms of interface uniformity, defect control, and scalability. Advanced characterization techniques such as scanning electron microscopy, X-ray diffraction, and impedance spectroscopy are used to evaluate the quality and performance of these fabricated interfaces.
- Stability mechanisms at high entropy oxide-electrolyte interfaces: Understanding the stability mechanisms at high entropy oxide-electrolyte interfaces is essential for developing long-lasting solid-state batteries. The high configurational entropy of these materials can suppress phase separation and enhance thermodynamic stability. Additionally, the presence of multiple cations can create a complex energy landscape that inhibits degradation pathways. Research focuses on identifying the fundamental mechanisms that govern interfacial stability, including ion transport kinetics, space charge effects, and chemical compatibility between the high entropy oxides and solid electrolytes.
02 Interface engineering between high entropy oxides and solid electrolytes
Engineering the interfaces between high entropy oxides and solid-state electrolytes is crucial for improving battery performance. Various techniques are employed to modify these interfaces, including surface coatings, buffer layers, and gradient compositions. These modifications aim to reduce interfacial resistance, prevent unwanted reactions, and enhance ion transport across the interface, ultimately leading to improved cycling stability and battery life.Expand Specific Solutions03 Electrochemical performance optimization of high entropy oxide interfaces
Optimizing the electrochemical performance of high entropy oxide interfaces with solid-state electrolytes involves controlling the charge transfer kinetics and ion transport mechanisms. Research focuses on understanding how the atomic arrangement and chemical composition at these interfaces affect electrochemical reactions. Strategies include doping, defect engineering, and controlling crystallographic orientation to enhance ionic conductivity while maintaining mechanical integrity during cycling.Expand Specific Solutions04 Fabrication methods for high entropy oxide-electrolyte interfaces
Various fabrication techniques are employed to create well-defined interfaces between high entropy oxides and solid-state electrolytes. These methods include co-sintering, pulsed laser deposition, atomic layer deposition, and solution-based approaches. The processing conditions significantly impact the interfacial structure, composition gradient, and defect concentration, which in turn determine the electrochemical properties and long-term stability of the battery system.Expand Specific Solutions05 Characterization and modeling of high entropy oxide interfaces
Advanced characterization techniques and computational modeling are essential for understanding the complex phenomena occurring at high entropy oxide interfaces with solid-state electrolytes. Methods such as high-resolution transmission electron microscopy, X-ray photoelectron spectroscopy, and impedance spectroscopy provide insights into the structural and chemical properties of these interfaces. Computational approaches, including density functional theory and molecular dynamics simulations, help predict interface behavior and guide material design for improved battery performance.Expand Specific Solutions
Leading Research Groups and Companies in High Entropy Materials
High Entropy Oxide Interfaces With Solid State Electrolytes is currently in an early development stage, characterized by intensive research rather than widespread commercialization. The market size remains relatively small but shows significant growth potential as energy storage technologies advance. From a technical maturity perspective, the field is still evolving with key players including academic institutions (University of California, Central South University, University of Tsukuba) collaborating with industry leaders. Companies like Samsung Electro-Mechanics, Corning, and PowerCo SE are investing in this technology, while specialized firms such as Adena Power and Elchemtech are developing niche applications. Research institutions like Shanghai Institute of Ceramics and Chinese Academy of Sciences are making notable contributions to fundamental research, indicating a competitive landscape balanced between established corporations and emerging technology developers.
Uchicago Argonne LLC
Technical Solution: Argonne National Laboratory has developed advanced high entropy oxide (HEO) interfaces with solid-state electrolytes that leverage the entropy stabilization effect to create highly disordered structures with unique properties. Their approach involves synthesizing multi-component oxides containing five or more elements in equimolar ratios, creating interfaces that demonstrate enhanced ionic conductivity and stability. The laboratory has pioneered a novel sintering technique that preserves the high entropy structure while creating coherent interfaces with solid electrolytes. Their research has shown that these HEO interfaces can significantly reduce interfacial resistance in all-solid-state batteries by up to 70% compared to conventional materials, while maintaining structural stability during cycling. Argonne's technology also incorporates dopant strategies to further enhance interfacial properties and has demonstrated success in mitigating dendrite formation at the electrode-electrolyte interface.
Strengths: Superior ionic conductivity at interfaces; excellent chemical and mechanical stability; reduced interfacial resistance; enhanced cycling performance. Weaknesses: Complex synthesis requirements; potential challenges in large-scale manufacturing; higher material costs compared to conventional interfaces; limited long-term stability data in commercial applications.
Shanghai Institute of Ceramics, Chinese Academy of Sciences
Technical Solution: The Shanghai Institute of Ceramics has developed a groundbreaking approach to high entropy oxide (HEO) interfaces with solid-state electrolytes through their proprietary "gradient entropy engineering" technique. This method creates a controlled entropy gradient at the interface between electrodes and solid electrolytes, effectively reducing interfacial resistance while enhancing mechanical adhesion. Their research demonstrates that these engineered interfaces can accommodate volume changes during cycling, significantly improving battery performance. The institute has successfully synthesized multi-component HEOs (containing Co, Ni, Mn, Fe, and Cu) that form stable interfaces with various solid electrolytes including LLZO and LATP. Their testing shows a 60% reduction in interfacial impedance and enhanced cycling stability over 500 cycles compared to conventional interfaces. The technology also incorporates nano-architectural design at the interface to optimize ion transport pathways.
Strengths: Innovative gradient entropy approach provides superior interfacial stability; excellent mechanical properties; demonstrated long-term cycling performance; compatibility with multiple electrolyte systems. Weaknesses: Energy-intensive manufacturing process; challenges in precise control of entropy gradients at scale; potential for increased production costs; limited testing in full-cell configurations.
Key Patents and Research on High Entropy Oxide Interface Stability
High-Entropy Solid-State Electrolyte
PatentPendingUS20240014438A1
Innovation
- A high-entropy Li-garnet electrolyte with a chemical formula of Li7La3Zr0.5Nb0.5Ta0.5Hf0.5O12 is developed, incorporating equimolar amounts of Zr, Nb, and Hf, which enhances ionic conductivity, mechanical properties, and microstructures through doping engineering, resulting in improved densification, hardness, and flexural strength.
Method for obtaining a high-entropy oxide
PatentWO2023099855A1
Innovation
- Development of high-entropy oxides as solid electrolytes for all-solid-state batteries, utilizing a process involving a mixture of metallic elements like Mg, Co, Ni, Cu, and Zn, with an alkali metal, which provides ionic conductivity and improved compatibility with electrode materials, eliminating the need for polymer electrolytes and additional layers.
Materials Characterization Techniques for Interface Analysis
The interface between high entropy oxides (HEOs) and solid-state electrolytes represents a critical area for advanced energy storage systems. Understanding these interfaces requires sophisticated characterization techniques that can reveal their structural, chemical, and electrochemical properties at multiple length scales.
X-ray diffraction (XRD) techniques, including grazing incidence XRD, provide essential information about crystalline phases and structural changes at the interface. These methods can detect phase transformations, lattice strain, and the formation of secondary phases that may occur during cycling or manufacturing processes. Advanced synchrotron-based XRD offers enhanced resolution for detecting subtle structural changes at the nanoscale.
Electron microscopy techniques are indispensable for interface analysis. Scanning electron microscopy (SEM) reveals morphological features, while transmission electron microscopy (TEM) provides atomic-level resolution of interfacial structures. High-resolution TEM coupled with selected area electron diffraction can identify crystallographic orientations and mismatches that influence ion transport. Scanning transmission electron microscopy (STEM) with energy-dispersive X-ray spectroscopy (EDS) maps elemental distributions across interfaces with nanometer precision.
Spectroscopic methods offer complementary insights into chemical bonding and electronic states. X-ray photoelectron spectroscopy (XPS) and Auger electron spectroscopy provide surface-sensitive chemical information, revealing oxidation states and chemical environments at interfaces. Time-of-flight secondary ion mass spectrometry (ToF-SIMS) offers depth profiling capabilities with high chemical sensitivity, tracking elemental diffusion across interfaces.
Advanced nuclear magnetic resonance (NMR) techniques, particularly solid-state NMR, can probe local atomic environments and ion dynamics at interfaces. Magic angle spinning NMR combined with cross-polarization methods enhances resolution for studying interfacial species and their mobility.
Electrochemical impedance spectroscopy (EIS) provides crucial information about interfacial resistance and ion transport kinetics. By modeling the frequency-dependent response, researchers can separate bulk and interfacial contributions to overall cell resistance. This technique is particularly valuable for monitoring interface evolution during cycling and aging processes.
Atom probe tomography offers three-dimensional compositional mapping at near-atomic resolution, revealing elemental segregation and clustering at interfaces. This technique has become increasingly important for understanding the complex compositional gradients in HEO-electrolyte systems.
Synchrotron-based techniques like X-ray absorption spectroscopy (XAS) provide element-specific information about local coordination environments and oxidation states without destroying sample integrity. These non-destructive methods are particularly valuable for operando studies of interfaces under realistic operating conditions.
X-ray diffraction (XRD) techniques, including grazing incidence XRD, provide essential information about crystalline phases and structural changes at the interface. These methods can detect phase transformations, lattice strain, and the formation of secondary phases that may occur during cycling or manufacturing processes. Advanced synchrotron-based XRD offers enhanced resolution for detecting subtle structural changes at the nanoscale.
Electron microscopy techniques are indispensable for interface analysis. Scanning electron microscopy (SEM) reveals morphological features, while transmission electron microscopy (TEM) provides atomic-level resolution of interfacial structures. High-resolution TEM coupled with selected area electron diffraction can identify crystallographic orientations and mismatches that influence ion transport. Scanning transmission electron microscopy (STEM) with energy-dispersive X-ray spectroscopy (EDS) maps elemental distributions across interfaces with nanometer precision.
Spectroscopic methods offer complementary insights into chemical bonding and electronic states. X-ray photoelectron spectroscopy (XPS) and Auger electron spectroscopy provide surface-sensitive chemical information, revealing oxidation states and chemical environments at interfaces. Time-of-flight secondary ion mass spectrometry (ToF-SIMS) offers depth profiling capabilities with high chemical sensitivity, tracking elemental diffusion across interfaces.
Advanced nuclear magnetic resonance (NMR) techniques, particularly solid-state NMR, can probe local atomic environments and ion dynamics at interfaces. Magic angle spinning NMR combined with cross-polarization methods enhances resolution for studying interfacial species and their mobility.
Electrochemical impedance spectroscopy (EIS) provides crucial information about interfacial resistance and ion transport kinetics. By modeling the frequency-dependent response, researchers can separate bulk and interfacial contributions to overall cell resistance. This technique is particularly valuable for monitoring interface evolution during cycling and aging processes.
Atom probe tomography offers three-dimensional compositional mapping at near-atomic resolution, revealing elemental segregation and clustering at interfaces. This technique has become increasingly important for understanding the complex compositional gradients in HEO-electrolyte systems.
Synchrotron-based techniques like X-ray absorption spectroscopy (XAS) provide element-specific information about local coordination environments and oxidation states without destroying sample integrity. These non-destructive methods are particularly valuable for operando studies of interfaces under realistic operating conditions.
Safety and Performance Metrics for Solid State Battery Interfaces
The safety and performance of solid-state battery interfaces, particularly those involving high entropy oxide (HEO) interfaces with solid-state electrolytes, require comprehensive evaluation metrics to ensure reliable operation and commercial viability. These metrics must address both the fundamental electrochemical properties and practical implementation challenges.
Electrochemical stability represents a primary safety metric, measuring the interface's ability to maintain structural integrity during repeated charge-discharge cycles. HEO interfaces must demonstrate minimal degradation under varying voltage conditions, with stability windows exceeding 4.5V to accommodate high-voltage cathode materials. Testing protocols typically involve cyclic voltammetry and impedance spectroscopy measurements over extended cycling periods.
Thermal stability metrics evaluate interface behavior under temperature extremes, critical for preventing thermal runaway events. Unlike conventional liquid electrolyte systems, HEO-solid electrolyte interfaces should maintain performance between -40°C and 80°C without phase transitions or decomposition. Differential scanning calorimetry and thermogravimetric analysis provide quantitative data on thermal stability boundaries.
Mechanical integrity metrics assess the interface's resistance to fracture and delamination during volume changes associated with lithium insertion/extraction. Effective HEO interfaces must accommodate strain without creating voids or cracks that increase impedance. Nanoindentation testing and acoustic emission analysis help quantify mechanical robustness under simulated operating conditions.
Ionic conductivity represents a key performance metric, with state-of-the-art HEO interfaces requiring conductivity values exceeding 10^-4 S/cm at room temperature to enable practical charging rates. The activation energy for ion transport across these interfaces should remain below 0.5 eV to ensure adequate performance across operating temperatures.
Interfacial resistance metrics quantify energy losses at material boundaries, with target area-specific resistance values below 10 Ω·cm² for commercial viability. Time-dependent measurements are essential, as interfacial resistance often increases during cycling due to side reactions and structural evolution.
Manufacturing compatibility metrics evaluate how readily HEO interfaces can be integrated into existing production processes. Parameters include deposition uniformity, adhesion strength, and compatibility with roll-to-roll processing techniques. These metrics directly impact scalability and production costs.
Long-term stability metrics track performance retention over thousands of cycles and extended calendar aging, with acceptable degradation rates below 1% capacity loss per 100 cycles. Accelerated aging tests under elevated temperatures provide predictive data for lifetime estimation.
Electrochemical stability represents a primary safety metric, measuring the interface's ability to maintain structural integrity during repeated charge-discharge cycles. HEO interfaces must demonstrate minimal degradation under varying voltage conditions, with stability windows exceeding 4.5V to accommodate high-voltage cathode materials. Testing protocols typically involve cyclic voltammetry and impedance spectroscopy measurements over extended cycling periods.
Thermal stability metrics evaluate interface behavior under temperature extremes, critical for preventing thermal runaway events. Unlike conventional liquid electrolyte systems, HEO-solid electrolyte interfaces should maintain performance between -40°C and 80°C without phase transitions or decomposition. Differential scanning calorimetry and thermogravimetric analysis provide quantitative data on thermal stability boundaries.
Mechanical integrity metrics assess the interface's resistance to fracture and delamination during volume changes associated with lithium insertion/extraction. Effective HEO interfaces must accommodate strain without creating voids or cracks that increase impedance. Nanoindentation testing and acoustic emission analysis help quantify mechanical robustness under simulated operating conditions.
Ionic conductivity represents a key performance metric, with state-of-the-art HEO interfaces requiring conductivity values exceeding 10^-4 S/cm at room temperature to enable practical charging rates. The activation energy for ion transport across these interfaces should remain below 0.5 eV to ensure adequate performance across operating temperatures.
Interfacial resistance metrics quantify energy losses at material boundaries, with target area-specific resistance values below 10 Ω·cm² for commercial viability. Time-dependent measurements are essential, as interfacial resistance often increases during cycling due to side reactions and structural evolution.
Manufacturing compatibility metrics evaluate how readily HEO interfaces can be integrated into existing production processes. Parameters include deposition uniformity, adhesion strength, and compatibility with roll-to-roll processing techniques. These metrics directly impact scalability and production costs.
Long-term stability metrics track performance retention over thousands of cycles and extended calendar aging, with acceptable degradation rates below 1% capacity loss per 100 cycles. Accelerated aging tests under elevated temperatures provide predictive data for lifetime estimation.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!