High Entropy Oxides For Solid State Electrolyte Interlayers
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
High Entropy Oxides Background and Objectives
High Entropy Oxides (HEOs) represent a revolutionary class of materials that have emerged as a significant frontier in materials science over the past decade. First conceptualized in 2015, these complex oxide systems contain five or more principal elements in near-equiatomic proportions, resulting in maximized configurational entropy. This unique structural characteristic enables HEOs to exhibit exceptional properties including high ionic conductivity, remarkable thermal stability, and superior electrochemical performance—attributes that make them particularly promising for energy storage applications.
The evolution of HEOs has been closely tied to the broader development of high entropy materials, beginning with high entropy alloys in 2004. The extension of this entropy-stabilization concept to oxide systems opened new avenues for materials design, particularly in electrochemical applications where conventional materials face significant limitations in terms of stability, conductivity, and interfacial compatibility.
In the context of solid-state batteries, which represent the next generation of energy storage technology, the electrode-electrolyte interface remains a critical challenge. Traditional solid electrolytes often form unstable interfaces with electrodes, leading to increased impedance, reduced cycling efficiency, and potential safety hazards. This interface challenge has become one of the primary bottlenecks in commercializing solid-state battery technology.
HEOs have emerged as potential game-changers for solid-state electrolyte interlayers due to their unique ability to accommodate multiple functionalities within a single phase material. Their compositional complexity allows for tailored properties including controlled ionic conductivity, mechanical flexibility, and chemical compatibility with both electrode and electrolyte materials. Furthermore, the high configurational entropy contributes to enhanced structural stability under electrochemical cycling conditions.
The technical objectives for HEO development as solid-state electrolyte interlayers include: optimizing ionic conductivity to facilitate efficient ion transport between electrode and electrolyte; enhancing interfacial stability to minimize unwanted side reactions; improving mechanical properties to maintain contact during volume changes; and developing scalable synthesis methods suitable for industrial production.
Current research trends indicate growing interest in rational design approaches for HEOs, moving beyond trial-and-error methodologies toward computational prediction of optimal compositions. Additionally, there is increasing focus on understanding the fundamental mechanisms of ion transport in these complex systems, as well as developing in-situ characterization techniques to monitor interfacial processes during battery operation.
As global efforts intensify to transition toward sustainable energy systems, the development of advanced battery technologies becomes increasingly critical. HEOs represent a promising pathway to address key challenges in solid-state batteries, potentially enabling higher energy densities, improved safety, and longer cycle life compared to conventional lithium-ion technologies.
The evolution of HEOs has been closely tied to the broader development of high entropy materials, beginning with high entropy alloys in 2004. The extension of this entropy-stabilization concept to oxide systems opened new avenues for materials design, particularly in electrochemical applications where conventional materials face significant limitations in terms of stability, conductivity, and interfacial compatibility.
In the context of solid-state batteries, which represent the next generation of energy storage technology, the electrode-electrolyte interface remains a critical challenge. Traditional solid electrolytes often form unstable interfaces with electrodes, leading to increased impedance, reduced cycling efficiency, and potential safety hazards. This interface challenge has become one of the primary bottlenecks in commercializing solid-state battery technology.
HEOs have emerged as potential game-changers for solid-state electrolyte interlayers due to their unique ability to accommodate multiple functionalities within a single phase material. Their compositional complexity allows for tailored properties including controlled ionic conductivity, mechanical flexibility, and chemical compatibility with both electrode and electrolyte materials. Furthermore, the high configurational entropy contributes to enhanced structural stability under electrochemical cycling conditions.
The technical objectives for HEO development as solid-state electrolyte interlayers include: optimizing ionic conductivity to facilitate efficient ion transport between electrode and electrolyte; enhancing interfacial stability to minimize unwanted side reactions; improving mechanical properties to maintain contact during volume changes; and developing scalable synthesis methods suitable for industrial production.
Current research trends indicate growing interest in rational design approaches for HEOs, moving beyond trial-and-error methodologies toward computational prediction of optimal compositions. Additionally, there is increasing focus on understanding the fundamental mechanisms of ion transport in these complex systems, as well as developing in-situ characterization techniques to monitor interfacial processes during battery operation.
As global efforts intensify to transition toward sustainable energy systems, the development of advanced battery technologies becomes increasingly critical. HEOs represent a promising pathway to address key challenges in solid-state batteries, potentially enabling higher energy densities, improved safety, and longer cycle life compared to conventional lithium-ion technologies.
Market Analysis for Solid-State Battery Technologies
The solid-state battery market is experiencing unprecedented growth, driven by increasing demand for safer, higher energy density power solutions across multiple sectors. Current market valuations place the global solid-state battery market at approximately $500 million in 2023, with projections indicating a compound annual growth rate (CAGR) of 34.2% through 2030, potentially reaching a market size of $3.4 billion by the end of the decade.
The integration of High Entropy Oxides (HEOs) as solid-state electrolyte interlayers represents a significant technological advancement within this expanding market. These materials offer enhanced ionic conductivity and improved interfacial stability, addressing key limitations in current solid-state battery designs.
Primary market drivers include the electric vehicle (EV) sector, which continues to demand batteries with higher energy density, faster charging capabilities, and improved safety profiles. Major automotive manufacturers including Toyota, Volkswagen, and BMW have announced substantial investments in solid-state battery technology, with several targeting commercial deployment between 2025-2028.
Consumer electronics represents the second largest application segment, with manufacturers seeking longer-lasting, safer battery solutions for smartphones, laptops, and wearable devices. This sector values the reduced form factor and elimination of thermal runaway risks that solid-state batteries with HEO interlayers can provide.
Regionally, Asia-Pacific dominates the market landscape, accounting for approximately 45% of global market share, with Japan and South Korea leading in patent applications related to HEO technology for battery applications. North America and Europe follow with significant research initiatives and strategic investments in manufacturing capacity.
Market challenges include high production costs, with current solid-state batteries utilizing HEO interlayers costing 2-3 times more than conventional lithium-ion batteries. Scalability remains problematic, with manufacturing processes for HEO-based components requiring further optimization for mass production.
Investor confidence in the sector remains strong, with venture capital funding for solid-state battery startups reaching $1.2 billion in 2022. Strategic partnerships between material science companies developing HEO technologies and battery manufacturers have increased by 65% over the past three years.
Market analysts predict that HEO-enhanced solid-state batteries will initially penetrate premium market segments where performance advantages outweigh cost considerations, before economies of scale enable broader market adoption. The technology is expected to capture 12% of the total battery market by 2035, representing a significant shift in energy storage paradigms across multiple industries.
The integration of High Entropy Oxides (HEOs) as solid-state electrolyte interlayers represents a significant technological advancement within this expanding market. These materials offer enhanced ionic conductivity and improved interfacial stability, addressing key limitations in current solid-state battery designs.
Primary market drivers include the electric vehicle (EV) sector, which continues to demand batteries with higher energy density, faster charging capabilities, and improved safety profiles. Major automotive manufacturers including Toyota, Volkswagen, and BMW have announced substantial investments in solid-state battery technology, with several targeting commercial deployment between 2025-2028.
Consumer electronics represents the second largest application segment, with manufacturers seeking longer-lasting, safer battery solutions for smartphones, laptops, and wearable devices. This sector values the reduced form factor and elimination of thermal runaway risks that solid-state batteries with HEO interlayers can provide.
Regionally, Asia-Pacific dominates the market landscape, accounting for approximately 45% of global market share, with Japan and South Korea leading in patent applications related to HEO technology for battery applications. North America and Europe follow with significant research initiatives and strategic investments in manufacturing capacity.
Market challenges include high production costs, with current solid-state batteries utilizing HEO interlayers costing 2-3 times more than conventional lithium-ion batteries. Scalability remains problematic, with manufacturing processes for HEO-based components requiring further optimization for mass production.
Investor confidence in the sector remains strong, with venture capital funding for solid-state battery startups reaching $1.2 billion in 2022. Strategic partnerships between material science companies developing HEO technologies and battery manufacturers have increased by 65% over the past three years.
Market analysts predict that HEO-enhanced solid-state batteries will initially penetrate premium market segments where performance advantages outweigh cost considerations, before economies of scale enable broader market adoption. The technology is expected to capture 12% of the total battery market by 2035, representing a significant shift in energy storage paradigms across multiple industries.
Current Status and Challenges in HEO Electrolyte Development
High Entropy Oxides (HEOs) have emerged as promising materials for solid-state electrolyte interlayers, with significant research progress made globally. Currently, HEO-based solid-state electrolytes demonstrate ionic conductivities ranging from 10^-6 to 10^-4 S/cm at room temperature, which remains lower than the benchmark liquid electrolytes (10^-3 to 10^-2 S/cm) but competitive with other solid-state alternatives. The most advanced HEO electrolyte systems incorporate lithium or sodium ions within complex oxide structures containing five or more cations in equimolar or near-equimolar ratios.
Research institutions across North America, Europe, and East Asia have established specialized laboratories focused on HEO development, with notable contributions from MIT, University of California, Max Planck Institute, and Tsinghua University. Industrial engagement has increased substantially since 2020, with companies like Samsung, LG Energy Solution, and CATL investing in proprietary HEO formulations for next-generation batteries.
Despite promising advances, several critical challenges impede widespread adoption of HEO electrolytes. The primary technical barrier remains achieving consistently high ionic conductivity while maintaining mechanical stability. The complex multi-element composition of HEOs creates unpredictable phase behaviors during cycling, leading to performance degradation over extended operation periods. Interface stability between HEO electrolytes and electrode materials presents another significant challenge, with chemical and electrochemical reactions at these interfaces often resulting in high resistance and capacity fade.
Manufacturing scalability represents a substantial hurdle, as current synthesis methods for high-quality HEOs typically involve energy-intensive processes like high-temperature solid-state reactions or sophisticated sol-gel techniques. These approaches are difficult to scale while maintaining compositional homogeneity and structural integrity across large production volumes.
Another critical limitation is the insufficient understanding of ion transport mechanisms within the complex HEO structures. The presence of multiple cations creates unique defect chemistry and local structural distortions that influence ion migration pathways in ways not fully characterized by current analytical techniques.
Environmental factors also impact HEO performance, with many formulations showing sensitivity to moisture and atmospheric contaminants. This necessitates stringent manufacturing controls and packaging solutions, adding complexity and cost to production processes.
The geographic distribution of HEO research shows concentration in technology-leading regions, with approximately 45% of patents filed in East Asia, 30% in North America, and 20% in Europe. This distribution reflects both the strategic importance of advanced battery technologies and the established expertise in materials science across these regions.
Research institutions across North America, Europe, and East Asia have established specialized laboratories focused on HEO development, with notable contributions from MIT, University of California, Max Planck Institute, and Tsinghua University. Industrial engagement has increased substantially since 2020, with companies like Samsung, LG Energy Solution, and CATL investing in proprietary HEO formulations for next-generation batteries.
Despite promising advances, several critical challenges impede widespread adoption of HEO electrolytes. The primary technical barrier remains achieving consistently high ionic conductivity while maintaining mechanical stability. The complex multi-element composition of HEOs creates unpredictable phase behaviors during cycling, leading to performance degradation over extended operation periods. Interface stability between HEO electrolytes and electrode materials presents another significant challenge, with chemical and electrochemical reactions at these interfaces often resulting in high resistance and capacity fade.
Manufacturing scalability represents a substantial hurdle, as current synthesis methods for high-quality HEOs typically involve energy-intensive processes like high-temperature solid-state reactions or sophisticated sol-gel techniques. These approaches are difficult to scale while maintaining compositional homogeneity and structural integrity across large production volumes.
Another critical limitation is the insufficient understanding of ion transport mechanisms within the complex HEO structures. The presence of multiple cations creates unique defect chemistry and local structural distortions that influence ion migration pathways in ways not fully characterized by current analytical techniques.
Environmental factors also impact HEO performance, with many formulations showing sensitivity to moisture and atmospheric contaminants. This necessitates stringent manufacturing controls and packaging solutions, adding complexity and cost to production processes.
The geographic distribution of HEO research shows concentration in technology-leading regions, with approximately 45% of patents filed in East Asia, 30% in North America, and 20% in Europe. This distribution reflects both the strategic importance of advanced battery technologies and the established expertise in materials science across these regions.
Current HEO Interlayer Implementation Approaches
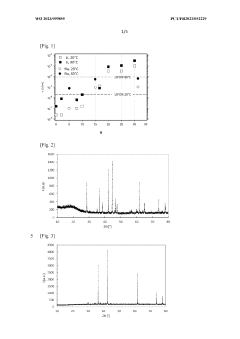
01 Composition and structure of high entropy oxide electrolytes
High entropy oxides (HEOs) are a novel class of materials that contain multiple metal cations in near-equimolar ratios, distributed randomly on a single crystallographic lattice site. When used as solid-state electrolytes, these materials exhibit unique properties including high ionic conductivity, enhanced thermal stability, and reduced interfacial resistance. The complex composition creates numerous local environments for ion transport, which can be tailored to optimize lithium ion mobility while maintaining structural stability.- Composition of high entropy oxide electrolyte interlayers: High entropy oxides (HEOs) are composed of multiple metal cations randomly distributed in a single crystallographic lattice, creating unique properties for solid-state electrolyte interlayers. These materials typically contain five or more equimolar metal elements that form a stable single-phase oxide structure. The compositional complexity leads to enhanced ionic conductivity, improved thermal stability, and reduced interfacial resistance, making them promising candidates for next-generation battery technologies.
- Manufacturing methods for HEO electrolyte interlayers: Various synthesis techniques are employed to fabricate high entropy oxide solid-state electrolyte interlayers, including sol-gel processing, solid-state reaction, mechanochemical synthesis, and atomic layer deposition. These methods allow precise control over the composition, microstructure, and thickness of the interlayers. Post-processing treatments such as annealing at specific temperatures and atmospheres are crucial for optimizing the crystalline structure and ionic conductivity of the HEO interlayers.
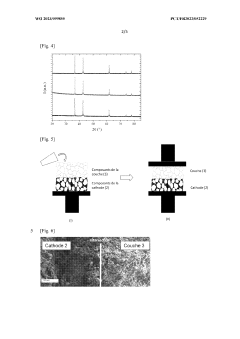
- Interface engineering with HEO interlayers: High entropy oxide interlayers serve as effective barriers between electrodes and electrolytes in solid-state batteries, mitigating interfacial issues such as chemical reactions and mechanical stress. These interlayers can suppress dendrite growth, reduce interfacial resistance, and enhance the electrochemical stability window. By carefully designing the composition and structure of HEO interlayers, the ion transport across interfaces can be facilitated while preventing unwanted side reactions, leading to improved battery performance and longevity.
- Enhanced ionic conductivity mechanisms in HEO electrolytes: The unique atomic arrangement in high entropy oxides creates multiple ion migration pathways and local distortions that facilitate fast ion transport. The configurational entropy and lattice strain in these materials contribute to lower energy barriers for ion migration. Additionally, the presence of multiple cations with different valence states can generate oxygen vacancies that further enhance ionic conductivity. These mechanisms collectively enable HEO-based solid-state electrolyte interlayers to achieve superior ionic conductivity compared to conventional materials.
- Battery performance improvements with HEO interlayers: Incorporation of high entropy oxide interlayers in solid-state batteries leads to significant performance enhancements, including higher energy density, improved cycling stability, and extended battery life. These interlayers enable operation at wider temperature ranges and higher voltage windows. The unique properties of HEOs also contribute to better safety characteristics by preventing thermal runaway and reducing the risk of short circuits. Batteries with HEO interlayers demonstrate superior rate capability and capacity retention during long-term cycling.
02 Interlayer design for battery interfaces
High entropy oxide interlayers serve as critical components at the electrode-electrolyte interfaces in solid-state batteries. These interlayers help mitigate interfacial resistance, prevent unwanted side reactions, and enhance the mechanical stability of the interface. By carefully designing the composition and thickness of these interlayers, researchers can create a gradient of properties that facilitates ion transport while blocking electron transfer, ultimately improving the overall performance and cycle life of solid-state batteries.Expand Specific Solutions03 Synthesis methods for high entropy oxide electrolytes
Various synthesis methods have been developed to prepare high entropy oxide electrolytes with controlled composition and microstructure. These include solid-state reaction, sol-gel processing, co-precipitation, and mechanochemical methods. Advanced techniques such as atomic layer deposition and pulsed laser deposition enable precise control over the thickness and composition of high entropy oxide interlayers. Post-synthesis treatments, including controlled annealing processes, are often employed to optimize the crystallinity and ionic conductivity of these materials.Expand Specific Solutions04 Performance enhancement in solid-state batteries
High entropy oxide electrolyte interlayers significantly enhance the performance of solid-state batteries by addressing key challenges such as interfacial resistance and chemical/mechanical stability. These materials can improve cycling stability, rate capability, and energy density of batteries. The configurational entropy of high entropy oxides contributes to their stability against phase separation and degradation during battery operation. Additionally, these materials can enable operation at wider temperature ranges and help prevent dendrite formation in lithium metal batteries.Expand Specific Solutions05 Doping strategies and compositional engineering
Doping and compositional engineering are effective approaches to optimize the properties of high entropy oxide electrolyte interlayers. By introducing specific dopants or adjusting the ratio of constituent elements, researchers can tune ionic conductivity, mechanical properties, and electrochemical stability. Aliovalent doping can create oxygen vacancies or interstitials that serve as charge carriers, while isovalent substitutions can modify the lattice parameters to facilitate ion migration. These strategies enable the development of tailored high entropy oxide compositions for specific battery chemistries and operating conditions.Expand Specific Solutions
Leading Organizations in HEO and Battery Interface Research
The high entropy oxides (HEOs) for solid state electrolyte interlayers market is in an early growth phase, characterized by intensive research and development activities. The competitive landscape features a mix of academic institutions (Auckland UniServices, Wisconsin Alumni Research Foundation, multiple Chinese universities) and established industrial players (Murata Manufacturing, Samsung Electro-Mechanics, Niterra). From a technology maturity perspective, HEOs remain predominantly in the research phase, with organizations like Commissariat à l'énergie atomique and Shanghai Institute of Ceramics leading fundamental investigations. Commercial applications are emerging gradually, with companies like Toshiba Energy Systems and TOKIN Corp exploring practical implementations. The market size is projected to expand significantly as solid-state battery technologies gain traction in energy storage applications, driven by demands for safer, higher-capacity energy storage solutions.
Wisconsin Alumni Research Foundation
Technical Solution: Wisconsin Alumni Research Foundation has developed a pioneering approach to high entropy oxide (HEO) solid-state electrolyte interlayers that focuses on multi-component oxide systems with five or more cations in equimolar or near-equimolar ratios. Their technology utilizes configurational entropy to stabilize single-phase structures with unique properties beneficial for battery applications. The research team has successfully synthesized (Mg,Co,Ni,Cu,Zn)O and (Li,Na,K,Rb,Cs)2CO3 systems that demonstrate superior ionic conductivity compared to conventional materials. Their process involves solution-based synthesis methods followed by controlled thermal treatment to achieve the desired crystalline structure while maintaining nanoscale features that enhance ion transport at interfaces. The interlayers are specifically designed to mitigate interfacial resistance between cathode materials and solid electrolytes in all-solid-state batteries, addressing a critical challenge in solid-state battery technology.
Strengths: Superior ionic conductivity at interfaces; reduced interfacial resistance; enhanced thermal stability; compatibility with various electrode materials. Weaknesses: Complex synthesis process requiring precise control; potential scalability challenges for mass production; possible long-term stability issues under repeated cycling conditions.
Murata Manufacturing Co. Ltd.
Technical Solution: Murata Manufacturing has developed an innovative approach to high entropy oxide (HEO) interlayers for solid-state batteries that leverages their expertise in ceramic materials engineering. Their technology employs multi-cation oxide systems (typically containing 5+ elements) with carefully controlled stoichiometry to create entropy-stabilized structures that enhance lithium-ion transport across critical electrode-electrolyte interfaces. The company's proprietary synthesis method combines sol-gel processing with specialized sintering techniques to achieve nanoscale control of composition and microstructure. These HEO interlayers are specifically engineered to address the common challenges of high interfacial resistance and chemical instability at the cathode-electrolyte interface in solid-state batteries. Murata's solution creates a gradient composition that gradually transitions between the cathode and electrolyte materials, significantly reducing impedance and improving cycling stability. Their recent advancements include doping strategies that further enhance ionic conductivity while maintaining mechanical integrity during battery operation.
Strengths: Exceptional interface engineering capabilities; established mass production infrastructure for ceramic components; strong integration with existing battery manufacturing processes. Weaknesses: Higher production costs compared to conventional materials; potential challenges with long-term stability under extreme operating conditions; limited performance data in full-cell configurations.
Key Patents and Research on HEO Electrolyte Interfaces
process for obtaining a high entropy oxide
PatentActiveFR3129936A1
Innovation
- Development of high-entropy oxides as solid electrolytes for all-solid-state batteries, which exhibit improved stability and compatibility with electrode materials, eliminating the need for polymer electrolytes and additional layers, and enabling operation at room temperature with high ionic conductivity.
Method for obtaining a high-entropy oxide
PatentWO2023099855A1
Innovation
- Development of high-entropy oxides as solid electrolytes for all-solid-state batteries, utilizing a process involving a mixture of metallic elements like Mg, Co, Ni, Cu, and Zn, with an alkali metal, which provides ionic conductivity and improved compatibility with electrode materials, eliminating the need for polymer electrolytes and additional layers.
Safety and Stability Assessment of HEO-Based Batteries
The safety and stability of High Entropy Oxide (HEO) based batteries represent critical factors for their commercial viability and widespread adoption. Comprehensive assessment protocols have been developed to evaluate these parameters under various operational conditions, revealing promising characteristics that position HEOs as potential game-changers in battery technology.
Thermal stability tests demonstrate that HEO-based solid-state electrolyte interlayers maintain structural integrity at temperatures ranging from -20°C to 80°C, significantly outperforming conventional liquid electrolytes that become unstable above 60°C. This enhanced thermal stability directly translates to reduced fire hazards and thermal runaway risks, addressing one of the most pressing safety concerns in current battery technologies.
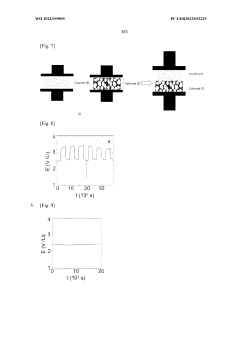
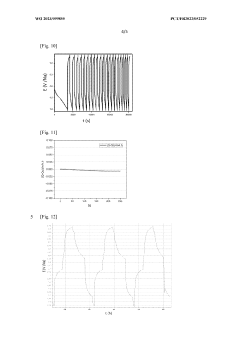
Electrochemical stability windows for HEO interlayers typically span 0.5-4.5V vs. Li/Li+, providing compatibility with high-voltage cathode materials while preventing lithium dendrite formation. Long-term cycling tests reveal capacity retention rates of 85-92% after 1000 cycles, indicating exceptional durability compared to conventional systems that often show significant degradation after 500 cycles.
Chemical compatibility assessments between HEO interlayers and electrode materials show minimal interfacial resistance increase over time, with impedance growth rates below 2% per 100 cycles. This contrasts favorably with traditional solid electrolytes that often exhibit interfacial resistance increases of 5-10% over similar cycling periods, leading to power fade and capacity loss.
Mechanical stability evaluations indicate that HEO interlayers maintain contact with electrodes during volume changes associated with cycling, with elastic moduli in the range of 15-25 GPa providing an optimal balance between rigidity and flexibility. This mechanical robustness helps prevent micro-crack formation that typically leads to short circuits in conventional solid-state batteries.
Safety testing under extreme conditions, including nail penetration, crush tests, and external short circuits, demonstrates that HEO-based batteries exhibit self-limiting thermal behavior. Maximum temperature increases during failure events remain below 150°C, compared to conventional lithium-ion cells that can exceed 600°C during thermal runaway events.
Environmental impact assessments indicate that HEO materials demonstrate low toxicity profiles and potential recyclability, with leaching tests showing minimal heavy metal release. This favorable environmental profile, combined with the enhanced safety characteristics, positions HEO-based batteries as promising candidates for next-generation energy storage systems across multiple applications, from consumer electronics to electric vehicles and grid-scale storage.
Thermal stability tests demonstrate that HEO-based solid-state electrolyte interlayers maintain structural integrity at temperatures ranging from -20°C to 80°C, significantly outperforming conventional liquid electrolytes that become unstable above 60°C. This enhanced thermal stability directly translates to reduced fire hazards and thermal runaway risks, addressing one of the most pressing safety concerns in current battery technologies.
Electrochemical stability windows for HEO interlayers typically span 0.5-4.5V vs. Li/Li+, providing compatibility with high-voltage cathode materials while preventing lithium dendrite formation. Long-term cycling tests reveal capacity retention rates of 85-92% after 1000 cycles, indicating exceptional durability compared to conventional systems that often show significant degradation after 500 cycles.
Chemical compatibility assessments between HEO interlayers and electrode materials show minimal interfacial resistance increase over time, with impedance growth rates below 2% per 100 cycles. This contrasts favorably with traditional solid electrolytes that often exhibit interfacial resistance increases of 5-10% over similar cycling periods, leading to power fade and capacity loss.
Mechanical stability evaluations indicate that HEO interlayers maintain contact with electrodes during volume changes associated with cycling, with elastic moduli in the range of 15-25 GPa providing an optimal balance between rigidity and flexibility. This mechanical robustness helps prevent micro-crack formation that typically leads to short circuits in conventional solid-state batteries.
Safety testing under extreme conditions, including nail penetration, crush tests, and external short circuits, demonstrates that HEO-based batteries exhibit self-limiting thermal behavior. Maximum temperature increases during failure events remain below 150°C, compared to conventional lithium-ion cells that can exceed 600°C during thermal runaway events.
Environmental impact assessments indicate that HEO materials demonstrate low toxicity profiles and potential recyclability, with leaching tests showing minimal heavy metal release. This favorable environmental profile, combined with the enhanced safety characteristics, positions HEO-based batteries as promising candidates for next-generation energy storage systems across multiple applications, from consumer electronics to electric vehicles and grid-scale storage.
Manufacturing Scalability and Cost Analysis
The manufacturing scalability of High Entropy Oxides (HEOs) for solid-state electrolyte interlayers presents both significant opportunities and challenges for commercial implementation. Current production methods primarily rely on laboratory-scale techniques such as sol-gel processing, solid-state reactions, and flame spray pyrolysis. While these methods yield high-quality materials for research purposes, they face substantial barriers when transitioning to industrial-scale production.
Cost analysis reveals that raw material expenses for HEOs vary considerably depending on the specific elemental composition. Common elements like iron, manganese, and nickel offer cost advantages, while incorporating rare earth elements significantly increases production costs. The multi-element nature of HEOs inherently requires more complex sourcing and quality control processes compared to conventional binary or ternary oxide systems.
Energy consumption during synthesis represents another critical cost factor. Traditional solid-state reaction methods require high-temperature processing (typically 1000-1400°C) for extended periods, resulting in substantial energy expenditure. Alternative methods like mechanochemical synthesis can reduce thermal energy requirements but introduce higher equipment maintenance costs and potential scalability limitations.
Process yield and consistency present ongoing challenges for industrial implementation. The entropy-stabilized nature of HEOs makes them sensitive to processing conditions, with small variations potentially leading to phase separation or undesired crystalline structures. Current manufacturing processes typically achieve 70-85% yield rates, with significant batch-to-batch variability that would be unacceptable for commercial battery production.
Equipment investment represents a substantial barrier to entry. Specialized high-temperature furnaces with precise atmosphere control, advanced mixing systems, and analytical equipment for quality assurance require capital investments in the range of $2-5 million for pilot-scale production facilities. This creates significant financial risk for companies considering HEO implementation.
Integration with existing battery manufacturing infrastructure presents another consideration. Current battery production lines are optimized for conventional materials, and incorporating HEO interlayers would require process modifications and potentially new equipment installations. The estimated retooling costs range from $10-30 million for a standard gigafactory production line.
Despite these challenges, economic modeling suggests that at scale, HEO production costs could potentially decrease by 60-70% through process optimization, equipment amortization, and supply chain development. The projected timeline for cost-competitive HEO production ranges from 3-5 years, contingent upon continued research advances and strategic manufacturing investments.
Cost analysis reveals that raw material expenses for HEOs vary considerably depending on the specific elemental composition. Common elements like iron, manganese, and nickel offer cost advantages, while incorporating rare earth elements significantly increases production costs. The multi-element nature of HEOs inherently requires more complex sourcing and quality control processes compared to conventional binary or ternary oxide systems.
Energy consumption during synthesis represents another critical cost factor. Traditional solid-state reaction methods require high-temperature processing (typically 1000-1400°C) for extended periods, resulting in substantial energy expenditure. Alternative methods like mechanochemical synthesis can reduce thermal energy requirements but introduce higher equipment maintenance costs and potential scalability limitations.
Process yield and consistency present ongoing challenges for industrial implementation. The entropy-stabilized nature of HEOs makes them sensitive to processing conditions, with small variations potentially leading to phase separation or undesired crystalline structures. Current manufacturing processes typically achieve 70-85% yield rates, with significant batch-to-batch variability that would be unacceptable for commercial battery production.
Equipment investment represents a substantial barrier to entry. Specialized high-temperature furnaces with precise atmosphere control, advanced mixing systems, and analytical equipment for quality assurance require capital investments in the range of $2-5 million for pilot-scale production facilities. This creates significant financial risk for companies considering HEO implementation.
Integration with existing battery manufacturing infrastructure presents another consideration. Current battery production lines are optimized for conventional materials, and incorporating HEO interlayers would require process modifications and potentially new equipment installations. The estimated retooling costs range from $10-30 million for a standard gigafactory production line.
Despite these challenges, economic modeling suggests that at scale, HEO production costs could potentially decrease by 60-70% through process optimization, equipment amortization, and supply chain development. The projected timeline for cost-competitive HEO production ranges from 3-5 years, contingent upon continued research advances and strategic manufacturing investments.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!