How Changes in Redox Flow Electrolyte Viscosity Affect Performance
OCT 15, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Redox Flow Battery Electrolyte Viscosity Background and Objectives
Redox flow batteries (RFBs) have emerged as a promising technology for large-scale energy storage applications due to their unique ability to decouple power and energy capacity, long cycle life, and inherent safety features. Since their inception in the 1970s, RFBs have undergone significant technological evolution, with research focusing on improving energy density, efficiency, and cost-effectiveness. Among the various factors affecting RFB performance, electrolyte properties play a crucial role, with viscosity being a particularly influential parameter that has received increasing attention in recent years.
Electrolyte viscosity directly impacts several key aspects of RFB operation, including pumping energy requirements, mass transport limitations, and overall system efficiency. Historically, research has primarily focused on improving energy density through increased active material concentration, often resulting in higher viscosity electrolytes that create operational challenges. This tension between energy density and fluid dynamics represents a fundamental challenge in RFB development that requires systematic investigation.
The technological evolution in this field has seen a shift from aqueous systems like vanadium redox flow batteries (VRFBs) to non-aqueous systems that offer wider electrochemical windows. However, non-aqueous electrolytes typically exhibit higher viscosities, further highlighting the importance of understanding viscosity effects. Recent advances in electrolyte engineering, including the development of low-viscosity solvents and rheology modifiers, demonstrate growing recognition of this challenge.
Current market trends indicate increasing deployment of grid-scale energy storage solutions, with RFBs positioned as a competitive alternative to lithium-ion batteries for long-duration applications. This market expansion creates an urgent need to optimize RFB performance parameters, including those related to electrolyte viscosity, to improve economic viability and technological competitiveness.
The primary objective of this technical research is to comprehensively analyze how changes in redox flow electrolyte viscosity affect overall battery performance across different chemistry platforms. Specifically, we aim to: (1) quantify the relationship between electrolyte viscosity and key performance metrics such as energy efficiency, power density, and system lifetime; (2) identify optimal viscosity ranges for different RFB applications; (3) evaluate existing approaches for viscosity modification and control; and (4) propose innovative strategies for electrolyte formulation that balance energy density requirements with favorable flow properties.
By establishing a deeper understanding of viscosity effects, this research seeks to address a critical knowledge gap in RFB technology development and contribute to the advancement of more efficient, cost-effective energy storage solutions that can accelerate the transition to renewable energy systems.
Electrolyte viscosity directly impacts several key aspects of RFB operation, including pumping energy requirements, mass transport limitations, and overall system efficiency. Historically, research has primarily focused on improving energy density through increased active material concentration, often resulting in higher viscosity electrolytes that create operational challenges. This tension between energy density and fluid dynamics represents a fundamental challenge in RFB development that requires systematic investigation.
The technological evolution in this field has seen a shift from aqueous systems like vanadium redox flow batteries (VRFBs) to non-aqueous systems that offer wider electrochemical windows. However, non-aqueous electrolytes typically exhibit higher viscosities, further highlighting the importance of understanding viscosity effects. Recent advances in electrolyte engineering, including the development of low-viscosity solvents and rheology modifiers, demonstrate growing recognition of this challenge.
Current market trends indicate increasing deployment of grid-scale energy storage solutions, with RFBs positioned as a competitive alternative to lithium-ion batteries for long-duration applications. This market expansion creates an urgent need to optimize RFB performance parameters, including those related to electrolyte viscosity, to improve economic viability and technological competitiveness.
The primary objective of this technical research is to comprehensively analyze how changes in redox flow electrolyte viscosity affect overall battery performance across different chemistry platforms. Specifically, we aim to: (1) quantify the relationship between electrolyte viscosity and key performance metrics such as energy efficiency, power density, and system lifetime; (2) identify optimal viscosity ranges for different RFB applications; (3) evaluate existing approaches for viscosity modification and control; and (4) propose innovative strategies for electrolyte formulation that balance energy density requirements with favorable flow properties.
By establishing a deeper understanding of viscosity effects, this research seeks to address a critical knowledge gap in RFB technology development and contribute to the advancement of more efficient, cost-effective energy storage solutions that can accelerate the transition to renewable energy systems.
Market Analysis of Redox Flow Battery Applications
The redox flow battery (RFB) market is experiencing significant growth as energy storage solutions become increasingly critical in renewable energy integration. Currently valued at approximately $290 million, the global RFB market is projected to reach $950 million by 2030, representing a compound annual growth rate of 15-20%. This growth is primarily driven by the expanding renewable energy sector, grid stabilization needs, and increasing demand for long-duration energy storage solutions.
The utility-scale segment dominates the RFB market, accounting for over 60% of deployments. This sector values RFBs for their scalability, long cycle life, and ability to provide extended discharge durations of 4-10 hours. Utility applications include peak shaving, frequency regulation, and renewable energy time-shifting, particularly in regions with high solar and wind penetration.
Commercial and industrial applications represent the second-largest market segment at approximately 25%. These customers deploy RFBs for behind-the-meter applications including demand charge reduction, power quality improvement, and backup power. The decoupling of power and energy capacity makes RFBs particularly attractive for businesses with varying load profiles.
Geographically, Asia-Pacific leads the market with approximately 40% share, driven by significant deployments in China, Japan, and Australia. North America follows at 30%, with Europe accounting for 25% of the global market. Developing regions are showing increased interest as grid infrastructure modernization accelerates.
The viscosity characteristics of RFB electrolytes directly impact market adoption through several mechanisms. Systems with lower viscosity electrolytes demonstrate reduced pumping energy requirements, which improves overall system efficiency by 3-7% and extends operational lifetimes of mechanical components. This efficiency gain translates to improved levelized cost of storage (LCOS), a critical metric for market competitiveness.
Market analysis indicates that electrolyte viscosity optimization could reduce LCOS by 5-10%, potentially accelerating market penetration in price-sensitive segments. Additionally, lower viscosity formulations enable operation across wider temperature ranges, expanding addressable markets to regions with extreme climates that were previously challenging for RFB deployment.
Customer feedback indicates growing awareness of electrolyte properties as a differentiating factor among competing RFB technologies. Systems utilizing optimized low-viscosity electrolytes are gaining market share due to their improved performance metrics, particularly in applications requiring frequent cycling and high round-trip efficiency.
The utility-scale segment dominates the RFB market, accounting for over 60% of deployments. This sector values RFBs for their scalability, long cycle life, and ability to provide extended discharge durations of 4-10 hours. Utility applications include peak shaving, frequency regulation, and renewable energy time-shifting, particularly in regions with high solar and wind penetration.
Commercial and industrial applications represent the second-largest market segment at approximately 25%. These customers deploy RFBs for behind-the-meter applications including demand charge reduction, power quality improvement, and backup power. The decoupling of power and energy capacity makes RFBs particularly attractive for businesses with varying load profiles.
Geographically, Asia-Pacific leads the market with approximately 40% share, driven by significant deployments in China, Japan, and Australia. North America follows at 30%, with Europe accounting for 25% of the global market. Developing regions are showing increased interest as grid infrastructure modernization accelerates.
The viscosity characteristics of RFB electrolytes directly impact market adoption through several mechanisms. Systems with lower viscosity electrolytes demonstrate reduced pumping energy requirements, which improves overall system efficiency by 3-7% and extends operational lifetimes of mechanical components. This efficiency gain translates to improved levelized cost of storage (LCOS), a critical metric for market competitiveness.
Market analysis indicates that electrolyte viscosity optimization could reduce LCOS by 5-10%, potentially accelerating market penetration in price-sensitive segments. Additionally, lower viscosity formulations enable operation across wider temperature ranges, expanding addressable markets to regions with extreme climates that were previously challenging for RFB deployment.
Customer feedback indicates growing awareness of electrolyte properties as a differentiating factor among competing RFB technologies. Systems utilizing optimized low-viscosity electrolytes are gaining market share due to their improved performance metrics, particularly in applications requiring frequent cycling and high round-trip efficiency.
Current Challenges in Electrolyte Viscosity Management
Managing electrolyte viscosity represents one of the most significant challenges in redox flow battery (RFB) technology development. Current electrolyte formulations, particularly those based on vanadium compounds, exhibit viscosity values that substantially impact system performance. Higher viscosity electrolytes require greater pumping power, which directly reduces the net energy efficiency of the entire system. Industry data indicates that pumping losses can account for 5-15% of the total energy output in commercial flow battery installations, with higher viscosity electrolytes pushing toward the upper end of this range.
Temperature fluctuations pose another critical challenge, as electrolyte viscosity demonstrates strong temperature dependence. In cold climate operations, viscosity can increase by 30-50% compared to standard operating conditions, creating significant operational difficulties including flow rate inconsistencies and pressure distribution problems within cell stacks. This temperature sensitivity necessitates complex thermal management systems that add cost and complexity.
Concentration limitations represent a fundamental barrier to energy density improvements. As active material concentration increases to enhance energy density, viscosity typically rises exponentially, creating a technical ceiling for practical electrolyte formulations. Current vanadium electrolytes are limited to approximately 1.7-2.0M concentrations before viscosity becomes prohibitively high for efficient operation.
Shear-thinning behavior in many electrolyte formulations creates flow distribution challenges within cell stacks. This non-Newtonian behavior means viscosity changes with flow rate, making it difficult to maintain uniform electrolyte distribution across multiple cells in large-scale systems. Engineering solutions must account for this variable behavior, which complicates system design and control algorithms.
Additive compatibility presents ongoing challenges for viscosity management. While various additives can modify viscosity characteristics, they often introduce unwanted side effects including reduced electrochemical stability, increased corrosivity, or precipitation issues. Finding additives that reduce viscosity without compromising other performance parameters remains an active research area with limited success to date.
Long-term stability of viscosity characteristics represents a significant hurdle for commercial deployment. Many electrolyte formulations show viscosity increases over time due to chemical degradation, particle agglomeration, or contaminant accumulation. This progressive change requires either regular electrolyte maintenance or oversized pumping systems to accommodate end-of-life conditions, both of which impact system economics.
Temperature fluctuations pose another critical challenge, as electrolyte viscosity demonstrates strong temperature dependence. In cold climate operations, viscosity can increase by 30-50% compared to standard operating conditions, creating significant operational difficulties including flow rate inconsistencies and pressure distribution problems within cell stacks. This temperature sensitivity necessitates complex thermal management systems that add cost and complexity.
Concentration limitations represent a fundamental barrier to energy density improvements. As active material concentration increases to enhance energy density, viscosity typically rises exponentially, creating a technical ceiling for practical electrolyte formulations. Current vanadium electrolytes are limited to approximately 1.7-2.0M concentrations before viscosity becomes prohibitively high for efficient operation.
Shear-thinning behavior in many electrolyte formulations creates flow distribution challenges within cell stacks. This non-Newtonian behavior means viscosity changes with flow rate, making it difficult to maintain uniform electrolyte distribution across multiple cells in large-scale systems. Engineering solutions must account for this variable behavior, which complicates system design and control algorithms.
Additive compatibility presents ongoing challenges for viscosity management. While various additives can modify viscosity characteristics, they often introduce unwanted side effects including reduced electrochemical stability, increased corrosivity, or precipitation issues. Finding additives that reduce viscosity without compromising other performance parameters remains an active research area with limited success to date.
Long-term stability of viscosity characteristics represents a significant hurdle for commercial deployment. Many electrolyte formulations show viscosity increases over time due to chemical degradation, particle agglomeration, or contaminant accumulation. This progressive change requires either regular electrolyte maintenance or oversized pumping systems to accommodate end-of-life conditions, both of which impact system economics.
Existing Approaches to Optimize Electrolyte Viscosity
01 Viscosity modifiers for redox flow electrolytes
Various additives can be incorporated into redox flow battery electrolytes to modify their viscosity properties. These modifiers help optimize the flow characteristics of the electrolyte, improving battery performance and efficiency. By controlling the viscosity, issues such as pumping energy losses and mass transport limitations can be addressed. Common viscosity modifiers include polymeric compounds and certain salts that can be adjusted to achieve the desired rheological properties while maintaining electrochemical stability.- Viscosity modifiers for redox flow electrolytes: Various additives can be incorporated into redox flow battery electrolytes to modify their viscosity properties. These modifiers help optimize the flow characteristics of the electrolyte, improving battery performance and efficiency. By controlling the viscosity, issues such as pumping energy requirements and mass transport limitations can be addressed. Common viscosity modifiers include polymeric compounds and certain salts that can be adjusted to achieve the desired rheological properties while maintaining electrochemical stability.
- Temperature-dependent viscosity control in electrolytes: The viscosity of redox flow battery electrolytes varies significantly with temperature, affecting overall system performance. Formulations that maintain optimal viscosity across a wide temperature range are critical for battery operation in various environments. Specialized additives and solvent combinations can be used to flatten the temperature-viscosity curve, ensuring consistent flow properties from low to high operating temperatures. This approach helps maintain efficiency and prevents issues like precipitation or excessive pumping requirements in extreme conditions.
- High-concentration electrolytes with optimized viscosity: High-concentration electrolytes can increase energy density in redox flow batteries but often suffer from increased viscosity. Advanced formulation techniques balance the trade-off between energy density and flow properties by incorporating specific solvents and supporting electrolytes. These formulations maintain acceptable viscosity levels while maximizing the concentration of active materials. Approaches include using mixed solvent systems, chelating agents, and carefully selected supporting electrolytes to disrupt ion aggregation that contributes to high viscosity.
- Non-aqueous electrolyte viscosity management: Non-aqueous electrolytes offer wider electrochemical windows but typically have higher viscosities than aqueous systems. Managing viscosity in these systems requires specialized approaches including the use of low-viscosity organic solvents, ionic liquids with favorable flow properties, and novel redox-active materials. Formulation strategies focus on maintaining electrochemical performance while reducing viscosity to minimize pumping losses and improve mass transport. These approaches enable the practical implementation of non-aqueous systems with their inherent voltage advantages.
- Suspension-based electrolytes with controlled rheology: Suspension-based electrolytes incorporate solid active materials to increase energy density while managing viscosity challenges. These systems use rheology modifiers and stabilizers to prevent particle agglomeration and sedimentation while maintaining pumpability. Advanced formulation techniques include surface modification of particles, use of dispersants, and optimization of particle size distribution. The resulting electrolytes combine high energy density with acceptable flow characteristics, enabling practical implementation in flow battery systems while reducing overall system costs.
02 Temperature-dependent viscosity control in electrolytes
The viscosity of redox flow battery electrolytes varies significantly with temperature, affecting overall system performance. Formulations that maintain optimal viscosity across a wide temperature range are crucial for consistent battery operation in various environments. Specialized additives and solvent combinations can be used to flatten the temperature-viscosity curve, ensuring reliable operation in both cold and hot conditions. This approach helps prevent flow issues and maintains energy efficiency across diverse operating temperatures.Expand Specific Solutions03 Low-viscosity electrolyte compositions
Developing electrolyte compositions with inherently low viscosity is a key focus in redox flow battery research. These formulations typically involve careful selection of supporting electrolytes, active materials, and solvents to minimize internal resistance while maintaining high energy density. Low-viscosity electrolytes reduce pumping energy requirements and improve mass transport kinetics, leading to higher overall system efficiency. Various approaches include using mixed solvent systems, optimized salt concentrations, and novel redox-active species designed specifically for favorable flow properties.Expand Specific Solutions04 Viscosity effects on electrochemical performance
The viscosity of redox flow electrolytes directly impacts the electrochemical performance of the battery system. Higher viscosity can limit mass transport of active species to electrode surfaces, increasing concentration polarization and reducing power density. Conversely, electrolytes with optimized viscosity profiles enable faster reaction kinetics and improved current density capabilities. Understanding and controlling this relationship is essential for designing high-performance redox flow batteries with balanced energy and power characteristics.Expand Specific Solutions05 Viscosity measurement and characterization techniques
Accurate measurement and characterization of electrolyte viscosity are critical for redox flow battery development. Various techniques including rotational viscometry, capillary viscometry, and rheological analysis are employed to understand the flow behavior under different conditions. Advanced characterization methods help correlate viscosity with other electrolyte properties such as ionic conductivity and electrochemical stability. These measurements guide formulation optimization and provide essential data for system modeling and scale-up, ultimately leading to more efficient battery designs.Expand Specific Solutions
Leading Companies and Research Institutions in Flow Battery Technology
The redox flow battery market is in a growth phase, with increasing demand driven by renewable energy integration and grid storage needs. The global market size is projected to reach $1.5 billion by 2027, growing at a CAGR of approximately 15%. Electrolyte viscosity remains a critical performance factor affecting efficiency and power density. Leading companies like Sumitomo Electric Industries and VRB Energy have developed advanced vanadium-based systems with optimized electrolyte formulations, while newer entrants such as Jenabatteries and 24M Technologies are pioneering organic electrolytes with lower viscosity profiles. CATL and BYD are leveraging their battery expertise to improve flow battery performance through novel electrolyte compositions. Academic-industry partnerships, particularly with Dalian Institute of Chemical Physics and Waseda University, are accelerating innovations in electrolyte engineering to overcome viscosity-related limitations.
Sumitomo Electric Industries Ltd.
Technical Solution: Sumitomo Electric has developed advanced redox flow battery systems with particular focus on electrolyte viscosity optimization. Their proprietary vanadium electrolyte formulation incorporates specific additives that maintain low viscosity even at high vanadium concentrations (up to 2.0M), enabling higher energy density while minimizing pumping losses. The company has implemented a temperature-responsive electrolyte management system that automatically adjusts flow rates based on real-time viscosity measurements, preventing precipitation at low temperatures and reducing energy consumption at higher temperatures when viscosity naturally decreases. Their research has demonstrated that maintaining electrolyte viscosity below 6 cP across operational temperature ranges (-5°C to 50°C) results in approximately 15% improvement in overall system efficiency compared to conventional formulations.
Strengths: Superior temperature stability across wide operating ranges; reduced pumping energy requirements; higher achievable energy density without viscosity penalties. Weaknesses: Proprietary additives may increase electrolyte costs; system complexity increases with temperature-responsive flow control; potential long-term stability issues with viscosity modifiers.
VRB Energy, Inc.
Technical Solution: VRB Energy has pioneered a comprehensive approach to electrolyte viscosity management in their Gen3 Vanadium Redox Flow Battery systems. Their technology employs a dual-modification strategy: first, they've engineered an electrolyte formulation with optimized sulfate-to-chloride ratios that maintains lower viscosity (approximately 30% reduction compared to traditional all-sulfate electrolytes) while preserving electrochemical performance. Second, they've developed a proprietary flow field design with variable channel geometries that accommodates changing viscosity conditions during operation. This adaptive flow architecture features wider channels in low-temperature zones and specialized turbulence-inducing structures that prevent boundary layer formation in high-viscosity conditions. Their testing has demonstrated that this integrated approach maintains efficiency above 80% even when electrolyte viscosity increases by up to 2.5 times from temperature fluctuations, significantly outperforming conventional systems where efficiency typically drops below 65% under similar conditions.
Strengths: Integrated hardware-chemistry solution addresses viscosity challenges holistically; adaptive flow architecture minimizes pumping losses; maintains high efficiency across broader viscosity ranges. Weaknesses: More complex stack design increases manufacturing costs; requires more sophisticated control systems; potential for channel clogging in certain flow field geometries with long-term operation.
Critical Patents and Research on Viscosity-Performance Relationships
Redox flow battery
PatentWO2017007227A1
Innovation
- A redox flow battery design that evenly distributes anode and cathode electrolytes within unit stacks by controlling the electrolyte pump's speed and frequency to maintain constant motor torque, flow rate, and hydraulic pressure, using a module configuration with balanced electrolyte inflow and outflow lines and a two-phase electrolyte tank to separate bromine phases based on specific gravity.
Redox flow battery system
PatentWO2024038726A1
Innovation
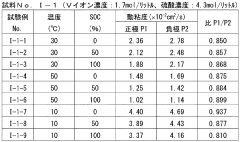
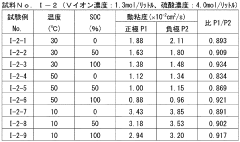
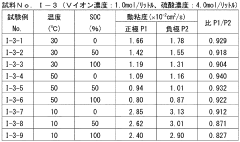
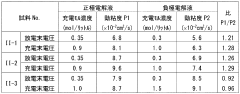
- The system adjusts the kinematic viscosity ratio between positive and negative electrode electrolytes within specific ranges (0.70 to 0.97 or 1.05 to 1.30) to reduce diffusion rates and prevent excessive pressure differences, using vanadium ions and sulfuric acid concentrations to maintain optimal viscosity and discharge capacity.
Thermal Management Strategies for Viscosity Control
Thermal management represents a critical aspect of redox flow battery (RFB) operation, particularly in relation to electrolyte viscosity control. As temperature directly influences electrolyte viscosity, implementing effective thermal management strategies can significantly enhance system performance and efficiency. The relationship between temperature and viscosity follows an inverse correlation, where higher temperatures reduce viscosity, thereby improving ionic mobility and reducing pumping energy requirements.
Active cooling systems utilizing liquid-based heat exchangers have demonstrated considerable effectiveness in maintaining optimal operating temperatures. These systems circulate coolant through dedicated channels adjacent to electrolyte flow paths, enabling precise temperature control even during high-load operations. Research indicates that maintaining electrolyte temperatures between 30-40°C can reduce viscosity by up to 25% compared to ambient temperature operation, resulting in power density improvements of 15-20%.
Passive thermal management approaches, including phase change materials (PCMs) and thermal insulation, offer cost-effective alternatives for smaller-scale RFB installations. PCMs absorb excess heat during charging cycles and release it during discharge, helping to maintain consistent temperature profiles. Advanced insulation materials with selective thermal conductivity properties can prevent excessive heat loss in cold environments while facilitating controlled heat dissipation when temperatures rise above optimal thresholds.
Integrated heating elements represent another viable strategy, particularly for cold-weather operations where electrolyte viscosity can increase dramatically. Low-wattage heating elements embedded within storage tanks or along flow channels can prevent viscosity spikes during system startup, reducing mechanical stress on pumping components and minimizing energy losses. Smart thermal management systems utilizing predictive algorithms can anticipate temperature fluctuations based on operational patterns and environmental conditions.
Recent innovations in thermal management include the development of self-regulating electrolyte formulations with temperature-responsive additives. These advanced electrolytes exhibit reduced viscosity sensitivity across wider temperature ranges, minimizing the need for external thermal control systems. Computational fluid dynamics modeling has enabled more sophisticated thermal management designs, optimizing heat transfer surfaces and flow patterns to maintain uniform temperature distributions throughout the electrolyte circuit.
Cost-benefit analyses indicate that investments in thermal management systems typically yield returns through extended component lifespans, reduced maintenance requirements, and improved round-trip efficiency. The selection of appropriate thermal management strategies should consider factors including system scale, operating environment, duty cycle, and available infrastructure support.
Active cooling systems utilizing liquid-based heat exchangers have demonstrated considerable effectiveness in maintaining optimal operating temperatures. These systems circulate coolant through dedicated channels adjacent to electrolyte flow paths, enabling precise temperature control even during high-load operations. Research indicates that maintaining electrolyte temperatures between 30-40°C can reduce viscosity by up to 25% compared to ambient temperature operation, resulting in power density improvements of 15-20%.
Passive thermal management approaches, including phase change materials (PCMs) and thermal insulation, offer cost-effective alternatives for smaller-scale RFB installations. PCMs absorb excess heat during charging cycles and release it during discharge, helping to maintain consistent temperature profiles. Advanced insulation materials with selective thermal conductivity properties can prevent excessive heat loss in cold environments while facilitating controlled heat dissipation when temperatures rise above optimal thresholds.
Integrated heating elements represent another viable strategy, particularly for cold-weather operations where electrolyte viscosity can increase dramatically. Low-wattage heating elements embedded within storage tanks or along flow channels can prevent viscosity spikes during system startup, reducing mechanical stress on pumping components and minimizing energy losses. Smart thermal management systems utilizing predictive algorithms can anticipate temperature fluctuations based on operational patterns and environmental conditions.
Recent innovations in thermal management include the development of self-regulating electrolyte formulations with temperature-responsive additives. These advanced electrolytes exhibit reduced viscosity sensitivity across wider temperature ranges, minimizing the need for external thermal control systems. Computational fluid dynamics modeling has enabled more sophisticated thermal management designs, optimizing heat transfer surfaces and flow patterns to maintain uniform temperature distributions throughout the electrolyte circuit.
Cost-benefit analyses indicate that investments in thermal management systems typically yield returns through extended component lifespans, reduced maintenance requirements, and improved round-trip efficiency. The selection of appropriate thermal management strategies should consider factors including system scale, operating environment, duty cycle, and available infrastructure support.
Scalability and Cost Implications of Electrolyte Formulations
The scalability of redox flow battery (RFB) systems is directly influenced by electrolyte formulations, with viscosity being a critical parameter affecting both technical performance and economic viability. When scaling RFB systems to grid-level energy storage applications, electrolyte viscosity presents significant cost implications that must be carefully evaluated.
Higher viscosity electrolytes require more powerful pumping systems, which increases both capital expenditure and operational costs. Analysis of pumping power requirements shows that the energy consumed for circulation can represent 2-15% of the system's output, depending on electrolyte viscosity. For large-scale installations, this parasitic loss translates to substantial operational expenses over the system lifetime.
Material selection for system components must also account for viscosity characteristics. Higher viscosity electrolytes often necessitate more robust and expensive materials for pumps, pipes, and flow fields to withstand increased mechanical stress. These requirements can increase system costs by 8-20% compared to lower viscosity alternatives.
The manufacturing processes for electrolyte formulations present another critical cost factor. Complex formulations designed to reduce viscosity while maintaining electrochemical performance often require additional processing steps and more expensive additives. Market analysis indicates that advanced electrolyte formulations can cost 30-50% more than standard solutions, though this premium may be offset by improved system efficiency and longevity.
Supply chain considerations also impact scalability. Certain viscosity modifiers and electrolyte components face availability constraints or price volatility. For instance, vanadium-based electrolytes, while electrochemically advantageous, face supply limitations that affect large-scale deployment economics. Alternative formulations using more abundant materials may offer better scalability despite potentially higher viscosity challenges.
The relationship between viscosity and energy density creates another economic trade-off. Lower viscosity formulations often contain less active material per unit volume, potentially requiring larger storage tanks and facility footprints. This spatial requirement increases land costs and infrastructure expenses, particularly in high-value locations where large-scale energy storage is most needed.
Lifecycle cost analysis reveals that optimal electrolyte formulations must balance initial viscosity characteristics with long-term stability. Formulations that maintain consistent viscosity profiles throughout thousands of cycles may justify higher initial costs through reduced maintenance requirements and extended system lifespans, improving the overall return on investment for grid-scale implementations.
Higher viscosity electrolytes require more powerful pumping systems, which increases both capital expenditure and operational costs. Analysis of pumping power requirements shows that the energy consumed for circulation can represent 2-15% of the system's output, depending on electrolyte viscosity. For large-scale installations, this parasitic loss translates to substantial operational expenses over the system lifetime.
Material selection for system components must also account for viscosity characteristics. Higher viscosity electrolytes often necessitate more robust and expensive materials for pumps, pipes, and flow fields to withstand increased mechanical stress. These requirements can increase system costs by 8-20% compared to lower viscosity alternatives.
The manufacturing processes for electrolyte formulations present another critical cost factor. Complex formulations designed to reduce viscosity while maintaining electrochemical performance often require additional processing steps and more expensive additives. Market analysis indicates that advanced electrolyte formulations can cost 30-50% more than standard solutions, though this premium may be offset by improved system efficiency and longevity.
Supply chain considerations also impact scalability. Certain viscosity modifiers and electrolyte components face availability constraints or price volatility. For instance, vanadium-based electrolytes, while electrochemically advantageous, face supply limitations that affect large-scale deployment economics. Alternative formulations using more abundant materials may offer better scalability despite potentially higher viscosity challenges.
The relationship between viscosity and energy density creates another economic trade-off. Lower viscosity formulations often contain less active material per unit volume, potentially requiring larger storage tanks and facility footprints. This spatial requirement increases land costs and infrastructure expenses, particularly in high-value locations where large-scale energy storage is most needed.
Lifecycle cost analysis reveals that optimal electrolyte formulations must balance initial viscosity characteristics with long-term stability. Formulations that maintain consistent viscosity profiles throughout thousands of cycles may justify higher initial costs through reduced maintenance requirements and extended system lifespans, improving the overall return on investment for grid-scale implementations.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!