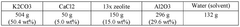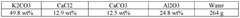How Does Material Composition Affect Carbon Capture Sorbent Efficiency?
OCT 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Carbon Capture Material Evolution and Objectives
Carbon capture technology has evolved significantly over the past several decades, transitioning from theoretical concepts to practical applications in various industrial settings. The earliest carbon capture methods emerged in the 1930s for natural gas purification, but dedicated research into materials specifically designed for carbon dioxide capture began in earnest during the 1970s energy crisis. This period marked the beginning of systematic exploration into how material composition fundamentally influences capture efficiency.
The evolution of carbon capture materials has followed several distinct phases. First-generation materials primarily consisted of aqueous amine solutions, notably monoethanolamine (MEA), which demonstrated good CO2 selectivity but suffered from high energy requirements for regeneration and corrosion issues. The second generation introduced solid sorbents including activated carbons, zeolites, and metal-organic frameworks (MOFs), which offered improved energy efficiency and stability.
Current third-generation materials represent a significant leap forward, incorporating engineered nanoporous structures, functionalized surfaces, and hybrid materials that combine the advantages of multiple capture mechanisms. These advanced materials demonstrate how precise control of chemical composition, pore architecture, and surface chemistry can dramatically enhance capture performance while reducing energy penalties.
The relationship between material composition and sorbent efficiency is governed by several key parameters: selectivity for CO2 over other gases, adsorption capacity under relevant conditions, kinetics of adsorption/desorption, stability over multiple cycles, and energy requirements for regeneration. Each of these factors is directly influenced by the material's chemical makeup, crystalline structure, and surface properties.
The primary objective of current research is to develop materials that achieve the U.S. Department of Energy's target of capturing CO2 at less than $40 per metric ton, representing a significant improvement over current costs ranging from $60-80 per ton. This requires materials that combine high capacity (>3 mmol CO2/g sorbent), excellent selectivity (CO2/N2 selectivity >100), fast kinetics (complete adsorption/desorption within minutes), and exceptional durability (thousands of cycles without significant degradation).
Looking forward, research objectives are increasingly focused on developing materials that can function effectively under real-world conditions, including tolerance to moisture, contaminants, and temperature fluctuations. Additionally, there is growing emphasis on materials that can be manufactured at scale using environmentally sustainable processes, aligning carbon capture technology with broader sustainability goals.
The evolution of carbon capture materials has followed several distinct phases. First-generation materials primarily consisted of aqueous amine solutions, notably monoethanolamine (MEA), which demonstrated good CO2 selectivity but suffered from high energy requirements for regeneration and corrosion issues. The second generation introduced solid sorbents including activated carbons, zeolites, and metal-organic frameworks (MOFs), which offered improved energy efficiency and stability.
Current third-generation materials represent a significant leap forward, incorporating engineered nanoporous structures, functionalized surfaces, and hybrid materials that combine the advantages of multiple capture mechanisms. These advanced materials demonstrate how precise control of chemical composition, pore architecture, and surface chemistry can dramatically enhance capture performance while reducing energy penalties.
The relationship between material composition and sorbent efficiency is governed by several key parameters: selectivity for CO2 over other gases, adsorption capacity under relevant conditions, kinetics of adsorption/desorption, stability over multiple cycles, and energy requirements for regeneration. Each of these factors is directly influenced by the material's chemical makeup, crystalline structure, and surface properties.
The primary objective of current research is to develop materials that achieve the U.S. Department of Energy's target of capturing CO2 at less than $40 per metric ton, representing a significant improvement over current costs ranging from $60-80 per ton. This requires materials that combine high capacity (>3 mmol CO2/g sorbent), excellent selectivity (CO2/N2 selectivity >100), fast kinetics (complete adsorption/desorption within minutes), and exceptional durability (thousands of cycles without significant degradation).
Looking forward, research objectives are increasingly focused on developing materials that can function effectively under real-world conditions, including tolerance to moisture, contaminants, and temperature fluctuations. Additionally, there is growing emphasis on materials that can be manufactured at scale using environmentally sustainable processes, aligning carbon capture technology with broader sustainability goals.
Market Analysis for Carbon Capture Technologies
The global carbon capture market is experiencing significant growth, driven by increasing environmental regulations and corporate sustainability commitments. As of 2023, the market was valued at approximately $7.3 billion, with projections indicating growth to reach $20 billion by 2030, representing a compound annual growth rate (CAGR) of 15.4%. This expansion is primarily fueled by governmental carbon reduction targets established under the Paris Agreement and subsequent climate accords.
North America currently dominates the carbon capture market, accounting for roughly 40% of global installations, followed by Europe at 30% and Asia-Pacific at 20%. The remaining 10% is distributed across other regions. This geographic distribution correlates strongly with regulatory frameworks and carbon pricing mechanisms in these regions.
The market segmentation reveals distinct categories based on capture technology types. Post-combustion capture technologies hold the largest market share at 45%, followed by pre-combustion (30%) and oxy-fuel combustion (15%), with emerging technologies comprising the remaining 10%. Within these segments, material-based solutions—particularly advanced sorbents—are experiencing the fastest growth due to their efficiency improvements and cost reduction potential.
Key industry verticals adopting carbon capture technologies include power generation (35%), oil and gas (25%), cement production (15%), chemical manufacturing (10%), and other industrial applications (15%). The power generation sector remains the primary adopter due to stringent emission regulations for coal and natural gas plants.
Customer demand patterns indicate a shift toward integrated solutions that offer not only capture capabilities but also utilization pathways for the captured carbon. This trend is creating new market opportunities in carbon utilization technologies, with the CO₂-to-products segment growing at 18% annually.
Price sensitivity remains a critical market factor, with current carbon capture costs ranging from $40-100 per ton of CO₂ depending on the technology and application. Market adoption accelerates significantly when costs fall below $50 per ton, highlighting the importance of material composition innovations that can improve efficiency while reducing costs.
Market barriers include high capital expenditure requirements, uncertain regulatory landscapes in developing economies, and limited infrastructure for carbon transport and storage. However, these challenges are being addressed through public-private partnerships and technology innovation consortia focused specifically on material science advancements for more efficient carbon capture sorbents.
North America currently dominates the carbon capture market, accounting for roughly 40% of global installations, followed by Europe at 30% and Asia-Pacific at 20%. The remaining 10% is distributed across other regions. This geographic distribution correlates strongly with regulatory frameworks and carbon pricing mechanisms in these regions.
The market segmentation reveals distinct categories based on capture technology types. Post-combustion capture technologies hold the largest market share at 45%, followed by pre-combustion (30%) and oxy-fuel combustion (15%), with emerging technologies comprising the remaining 10%. Within these segments, material-based solutions—particularly advanced sorbents—are experiencing the fastest growth due to their efficiency improvements and cost reduction potential.
Key industry verticals adopting carbon capture technologies include power generation (35%), oil and gas (25%), cement production (15%), chemical manufacturing (10%), and other industrial applications (15%). The power generation sector remains the primary adopter due to stringent emission regulations for coal and natural gas plants.
Customer demand patterns indicate a shift toward integrated solutions that offer not only capture capabilities but also utilization pathways for the captured carbon. This trend is creating new market opportunities in carbon utilization technologies, with the CO₂-to-products segment growing at 18% annually.
Price sensitivity remains a critical market factor, with current carbon capture costs ranging from $40-100 per ton of CO₂ depending on the technology and application. Market adoption accelerates significantly when costs fall below $50 per ton, highlighting the importance of material composition innovations that can improve efficiency while reducing costs.
Market barriers include high capital expenditure requirements, uncertain regulatory landscapes in developing economies, and limited infrastructure for carbon transport and storage. However, these challenges are being addressed through public-private partnerships and technology innovation consortia focused specifically on material science advancements for more efficient carbon capture sorbents.
Current Sorbent Materials and Technical Barriers
Carbon capture technology currently employs a diverse range of sorbent materials, each with distinct advantages and limitations. Amine-based sorbents, particularly monoethanolamine (MEA), remain the industry standard due to their high CO2 affinity and relatively mature implementation. These materials achieve 90% capture efficiency in optimal conditions but suffer from high regeneration energy requirements (3.5-4.2 GJ/tCO2) and degradation issues when exposed to oxygen and impurities.
Metal-organic frameworks (MOFs) represent a promising alternative with exceptional surface areas exceeding 7,000 m²/g and tunable pore structures. Notable examples include Mg-MOF-74 and HKUST-1, which demonstrate CO2 capacities of 8.61 and 4.5 mmol/g respectively. However, MOFs face stability challenges in humid conditions and high manufacturing costs that currently limit industrial deployment.
Zeolites, particularly 13X and 5A variants, offer robust thermal stability and moderate CO2 capacities (3-5 mmol/g). Their well-established synthesis processes make them commercially viable, though their performance deteriorates significantly in the presence of moisture, requiring energy-intensive pre-drying of flue gases.
Activated carbons provide cost-effective solutions with good stability but exhibit lower CO2 selectivity compared to other sorbents. Their primary advantages lie in low regeneration energy and resistance to contaminants, making them suitable for specific applications despite capacity limitations.
Several technical barriers persist across these material classes. Thermal management during adsorption-desorption cycles remains challenging, with temperature swings causing material degradation and efficiency losses. Most materials face a fundamental trade-off between adsorption capacity and regeneration energy requirements – stronger CO2 binding typically demands more energy for sorbent regeneration.
Moisture sensitivity presents another significant barrier, particularly for zeolites and certain MOFs, necessitating additional process steps that increase operational complexity and costs. Material durability under repeated cycling conditions remains problematic, with performance degradation of 0.5-2% per cycle observed in many systems.
Manufacturing scalability constitutes a critical challenge, especially for advanced materials like MOFs and functionalized sorbents. Current synthesis methods often involve expensive precursors and complex procedures that hinder industrial-scale production. The cost gap between conventional amine systems ($40-60/tCO2) and emerging materials (often exceeding $100/tCO2) must be addressed through material innovations and process optimizations to enable widespread commercial adoption.
Metal-organic frameworks (MOFs) represent a promising alternative with exceptional surface areas exceeding 7,000 m²/g and tunable pore structures. Notable examples include Mg-MOF-74 and HKUST-1, which demonstrate CO2 capacities of 8.61 and 4.5 mmol/g respectively. However, MOFs face stability challenges in humid conditions and high manufacturing costs that currently limit industrial deployment.
Zeolites, particularly 13X and 5A variants, offer robust thermal stability and moderate CO2 capacities (3-5 mmol/g). Their well-established synthesis processes make them commercially viable, though their performance deteriorates significantly in the presence of moisture, requiring energy-intensive pre-drying of flue gases.
Activated carbons provide cost-effective solutions with good stability but exhibit lower CO2 selectivity compared to other sorbents. Their primary advantages lie in low regeneration energy and resistance to contaminants, making them suitable for specific applications despite capacity limitations.
Several technical barriers persist across these material classes. Thermal management during adsorption-desorption cycles remains challenging, with temperature swings causing material degradation and efficiency losses. Most materials face a fundamental trade-off between adsorption capacity and regeneration energy requirements – stronger CO2 binding typically demands more energy for sorbent regeneration.
Moisture sensitivity presents another significant barrier, particularly for zeolites and certain MOFs, necessitating additional process steps that increase operational complexity and costs. Material durability under repeated cycling conditions remains problematic, with performance degradation of 0.5-2% per cycle observed in many systems.
Manufacturing scalability constitutes a critical challenge, especially for advanced materials like MOFs and functionalized sorbents. Current synthesis methods often involve expensive precursors and complex procedures that hinder industrial-scale production. The cost gap between conventional amine systems ($40-60/tCO2) and emerging materials (often exceeding $100/tCO2) must be addressed through material innovations and process optimizations to enable widespread commercial adoption.
Existing Sorbent Composition Optimization Approaches
01 Metal-organic frameworks (MOFs) for carbon capture
Metal-organic frameworks (MOFs) are highly porous materials that have shown exceptional efficiency for carbon capture applications. These materials offer high surface area, tunable pore sizes, and customizable chemical functionality that can be optimized for CO2 adsorption. MOFs can be designed with specific metal centers and organic linkers to enhance selectivity and capacity for carbon dioxide, making them promising sorbents for industrial carbon capture systems.- Metal-organic frameworks (MOFs) for carbon capture: Metal-organic frameworks are advanced porous materials with high surface area that can efficiently capture carbon dioxide. These crystalline structures consist of metal ions coordinated with organic ligands, creating a framework with tunable pore sizes and functionalities. MOFs demonstrate exceptional CO2 selectivity and adsorption capacity under various conditions, making them promising sorbents for carbon capture applications. Their efficiency can be further enhanced through post-synthetic modifications and incorporation of specific functional groups.
- Amine-functionalized sorbents for CO2 capture: Amine-functionalized materials represent a significant class of carbon capture sorbents with high efficiency. These materials incorporate various amine groups that chemically bind with CO2 through acid-base interactions. The amine functionalization can be applied to different support materials including silica, polymers, and porous carbons. These sorbents demonstrate high CO2 selectivity even in humid conditions and can be designed for specific operating temperatures and pressures, offering versatile solutions for carbon capture applications.
- Zeolite-based carbon capture materials: Zeolites are aluminosilicate minerals with well-defined microporous structures that show promising performance as carbon capture sorbents. Their crystalline framework contains uniform pores and channels that can selectively adsorb CO2 molecules. The efficiency of zeolite-based sorbents can be enhanced through ion exchange, framework modification, and optimization of the silicon-to-aluminum ratio. These materials offer advantages including thermal stability, regenerability, and resistance to contaminants in flue gas streams.
- Novel composite and hybrid sorbent materials: Composite and hybrid materials combine the advantages of different sorbent types to achieve enhanced carbon capture efficiency. These innovative materials integrate multiple components such as polymers with inorganic particles, layered structures, or combinations of different functional materials. The synergistic effects between components can lead to improved adsorption capacity, selectivity, and mechanical stability. These hybrid approaches often address limitations of single-component sorbents while maintaining regenerability and operational durability under industrial conditions.
- Process optimization for sorbent efficiency enhancement: Various process optimization techniques can significantly improve the efficiency of carbon capture sorbents. These approaches include temperature swing adsorption, pressure swing processes, vacuum swing adsorption, and moisture control strategies. Advanced regeneration methods, fluidized bed technologies, and innovative reactor designs can maximize sorbent performance while minimizing energy penalties. Process intensification and integration with existing industrial systems can further enhance overall carbon capture efficiency while reducing operational costs and environmental impact.
02 Amine-functionalized sorbents for enhanced CO2 capture
Amine-functionalized materials represent a significant advancement in carbon capture technology. These sorbents incorporate amine groups that chemically bind with CO2 molecules, resulting in higher capture efficiency even at low CO2 concentrations. The amine functionality can be incorporated into various support materials including silica, polymers, and porous frameworks. These materials demonstrate improved selectivity, capacity, and regeneration properties compared to conventional sorbents, making them suitable for post-combustion carbon capture applications.Expand Specific Solutions03 Novel porous materials with optimized adsorption kinetics
Advanced porous materials with optimized adsorption kinetics have been developed to improve carbon capture efficiency. These materials feature engineered pore structures that facilitate rapid CO2 diffusion and adsorption while minimizing mass transfer limitations. By controlling the pore size distribution, surface chemistry, and material morphology, these sorbents achieve faster adsorption rates and higher working capacities. The improved kinetics allow for more efficient pressure or temperature swing adsorption processes in industrial carbon capture applications.Expand Specific Solutions04 Composite and hybrid sorbent materials
Composite and hybrid sorbent materials combine multiple components to achieve superior carbon capture performance. These materials integrate the advantages of different sorbent types, such as the high capacity of chemical sorbents with the fast kinetics of physical adsorbents. Examples include polymer-inorganic composites, layered double hydroxides with functional additives, and hierarchical structured materials. The synergistic effects between components result in enhanced CO2 selectivity, improved stability during cycling, and better regeneration properties, leading to more efficient carbon capture processes.Expand Specific Solutions05 Regeneration methods for improved sorbent lifecycle efficiency
Advanced regeneration methods have been developed to improve the lifecycle efficiency of carbon capture sorbents. These techniques focus on reducing the energy requirements for sorbent regeneration while maintaining capture capacity over multiple cycles. Innovations include temperature-swing processes with optimized heating profiles, pressure-swing systems with reduced pressure differentials, and hybrid approaches combining multiple driving forces. Some methods incorporate novel heating techniques such as microwave or electrical swing adsorption to achieve more targeted and energy-efficient regeneration, significantly improving the overall efficiency of carbon capture systems.Expand Specific Solutions
Leading Organizations in Carbon Capture Material Research
The carbon capture sorbent efficiency market is currently in a growth phase, with increasing demand driven by global decarbonization efforts. The competitive landscape features established industrial players like Shell, GE, Siemens Energy, and Volkswagen alongside specialized carbon capture innovators such as Climeworks AG, Susteon, and DACMa GmbH. Material composition research represents a critical differentiation factor, with companies exploring diverse sorbent technologies including amine-based materials, metal-organic frameworks, and novel graphene structures (as developed by 3DC). Academic-industry partnerships are accelerating innovation, with institutions like Shanghai Advanced Research Institute, Arizona State University, and USC collaborating with commercial entities. The market is expected to reach significant scale as technical improvements in sorbent efficiency drive down costs toward the critical $100/ton threshold for commercial viability.
Climeworks AG
Technical Solution: Climeworks has developed a proprietary Direct Air Capture (DAC) technology using solid sorbent materials based on amine-functionalized filter materials. Their approach involves a cyclic adsorption-desorption process where ambient air passes through a collector with the sorbent material inside. The CO2 selectively binds to the amine groups on the sorbent surface at ambient conditions. Once saturated, the collector is closed and heated to approximately 100°C, which releases concentrated CO2 that can be collected for storage or utilization. Their latest generation sorbents have improved capacity and durability through optimized pore structure and amine distribution, allowing for thousands of adsorption-desorption cycles without significant degradation. Climeworks has also engineered their sorbents to minimize energy requirements during the regeneration phase, which is critical for overall process efficiency.
Strengths: High selectivity for CO2 even at low atmospheric concentrations (400ppm); modular design allows for scalable implementation; relatively low regeneration temperature reduces energy requirements. Weaknesses: Higher cost per ton of CO2 captured compared to point-source capture; sorbent degradation over time still requires periodic replacement; energy-intensive regeneration process impacts overall carbon balance.
Shell Internationale Research Maatschappij BV
Technical Solution: Shell has developed advanced metal-organic frameworks (MOFs) and amine-modified silica materials for carbon capture applications. Their research focuses on optimizing the pore structure and surface chemistry of these materials to enhance CO2 adsorption capacity and selectivity. Shell's proprietary CANSOLV technology, while primarily a solvent-based system, incorporates specialized solid sorbents in hybrid configurations to improve efficiency. Their latest research involves developing hierarchical porous structures that minimize mass transfer limitations and reduce pressure drop across adsorption beds. Shell has also pioneered temperature-swing adsorption (TSA) processes that utilize waste heat from industrial operations to regenerate sorbents, significantly improving energy efficiency. Their materials are designed to maintain stability under the harsh conditions found in flue gas streams, including resistance to SOx and NOx contaminants that typically degrade conventional sorbents.
Strengths: Extensive industrial implementation experience; materials designed for real-world conditions including contaminant resistance; integration with existing infrastructure. Weaknesses: Higher focus on point-source capture than direct air capture; some materials require significant energy for regeneration; complex synthesis procedures may limit large-scale production.
Key Innovations in Material Science for Carbon Capture
Solid sorbent materials functionalized with polyamines having oxygen-containing units selected from carbonyl units, hydroxyl units, and combinations thereof
PatentPendingUS20250288970A1
Innovation
- Development of solid sorbent materials functionalized with polyamines having oxygen-containing units such as carbonyl and hydroxyl units, which exhibit high adsorption capacity for carbon dioxide and low adsorption for water, with low desorption residuals under mild conditions.
Carbon capture sorbents with moisture control additives
PatentWO2025165762A1
Innovation
- Sorbent compositions incorporating moisture control additives and a support material, produced via mixing, drying, crushing, and sieving, enable stable carbon capture in humid conditions using a modified temperature swing adsorption process with reduced energy consumption.
Environmental Impact Assessment of Sorbent Materials
The environmental impact of carbon capture sorbent materials extends far beyond their primary function of CO2 sequestration. When evaluating these materials, a comprehensive life cycle assessment (LCA) becomes essential to understand their true environmental footprint. The production of high-performance sorbents often involves energy-intensive processes and potentially hazardous chemicals, creating a complex environmental trade-off equation that must be carefully balanced against their carbon capture benefits.
Material composition directly influences environmental impact through several pathways. Amine-based sorbents, while effective for carbon capture, may involve toxic precursors and generate harmful byproducts during synthesis and regeneration cycles. Metal-organic frameworks (MOFs), despite their exceptional capture capacity, frequently require solvents and metal precursors that pose ecological concerns if improperly managed. Conversely, naturally derived sorbents such as biochar present lower production impacts but may require land use changes that affect biodiversity.
Water consumption represents another critical environmental consideration. Zeolites and certain MOFs demand substantial water volumes during synthesis and activation processes. In water-stressed regions, this requirement creates additional environmental pressure that must be factored into sorbent selection decisions. Similarly, the energy requirements for sorbent regeneration—particularly for temperature swing adsorption systems—can potentially offset carbon benefits if powered by fossil fuel sources.
Durability and degradation characteristics significantly affect long-term environmental performance. Materials requiring frequent replacement generate additional waste streams and manufacturing impacts. Advanced composite sorbents may offer enhanced durability but often incorporate nanomaterials or polymers with uncertain environmental fate and potential ecotoxicological concerns. Recent studies indicate that microplastic shedding from polymer-supported sorbents may present emerging environmental risks in aquatic ecosystems.
End-of-life management presents perhaps the most overlooked environmental dimension. Spent sorbents may contain concentrated contaminants, heavy metals, or degradation products requiring specialized disposal protocols. Materials designed with circular economy principles—allowing for recycling, regeneration, or biodegradation—demonstrate superior environmental profiles compared to single-use alternatives. Innovative approaches incorporating biodegradable support structures or bio-based capture components show promise for minimizing disposal impacts.
Regulatory frameworks increasingly recognize these complex environmental dimensions, with several jurisdictions now requiring comprehensive impact assessments before large-scale deployment of carbon capture technologies. The development of standardized environmental impact metrics specific to sorbent materials would greatly enhance comparative evaluations and guide more sustainable material design approaches in this rapidly evolving field.
Material composition directly influences environmental impact through several pathways. Amine-based sorbents, while effective for carbon capture, may involve toxic precursors and generate harmful byproducts during synthesis and regeneration cycles. Metal-organic frameworks (MOFs), despite their exceptional capture capacity, frequently require solvents and metal precursors that pose ecological concerns if improperly managed. Conversely, naturally derived sorbents such as biochar present lower production impacts but may require land use changes that affect biodiversity.
Water consumption represents another critical environmental consideration. Zeolites and certain MOFs demand substantial water volumes during synthesis and activation processes. In water-stressed regions, this requirement creates additional environmental pressure that must be factored into sorbent selection decisions. Similarly, the energy requirements for sorbent regeneration—particularly for temperature swing adsorption systems—can potentially offset carbon benefits if powered by fossil fuel sources.
Durability and degradation characteristics significantly affect long-term environmental performance. Materials requiring frequent replacement generate additional waste streams and manufacturing impacts. Advanced composite sorbents may offer enhanced durability but often incorporate nanomaterials or polymers with uncertain environmental fate and potential ecotoxicological concerns. Recent studies indicate that microplastic shedding from polymer-supported sorbents may present emerging environmental risks in aquatic ecosystems.
End-of-life management presents perhaps the most overlooked environmental dimension. Spent sorbents may contain concentrated contaminants, heavy metals, or degradation products requiring specialized disposal protocols. Materials designed with circular economy principles—allowing for recycling, regeneration, or biodegradation—demonstrate superior environmental profiles compared to single-use alternatives. Innovative approaches incorporating biodegradable support structures or bio-based capture components show promise for minimizing disposal impacts.
Regulatory frameworks increasingly recognize these complex environmental dimensions, with several jurisdictions now requiring comprehensive impact assessments before large-scale deployment of carbon capture technologies. The development of standardized environmental impact metrics specific to sorbent materials would greatly enhance comparative evaluations and guide more sustainable material design approaches in this rapidly evolving field.
Scalability and Cost Analysis of Advanced Sorbents
The economic viability of carbon capture technologies hinges significantly on the scalability and cost-effectiveness of sorbent materials. Current advanced sorbents demonstrate promising capture efficiencies in laboratory settings, but their implementation at industrial scales presents substantial challenges. The capital expenditure for deploying these materials in large-scale carbon capture units remains prohibitively high, with estimates ranging from $400-800 per ton of CO2 captured, depending on the specific sorbent composition and system design.
Material composition directly impacts production costs, with highly engineered materials like metal-organic frameworks (MOFs) and functionalized porous polymers commanding premium prices due to complex synthesis procedures and expensive precursors. For instance, amine-functionalized silica sorbents cost approximately $20-50 per kilogram, while more advanced MOFs can exceed $100 per kilogram at current production scales. These costs must decrease by at least an order of magnitude to achieve economic viability in commercial applications.
Scalability challenges are equally significant. Many high-performance sorbents that demonstrate excellent CO2 selectivity and capacity in laboratory conditions face manufacturing bottlenecks when scaled to industrial production. Synthesis methods that work efficiently at gram-scale often encounter issues with batch consistency, reaction kinetics, and purification when scaled to kilogram or ton quantities. Materials requiring precise nanostructure control, such as hierarchical porous carbons or precisely engineered MOFs, face particularly severe scaling limitations.
Lifecycle considerations further complicate the economic equation. Sorbent degradation rates vary dramatically based on material composition, with some advanced materials losing 5-15% capacity per 100 cycles under industrial conditions. More stable materials typically trade performance for longevity, necessitating larger quantities of sorbent and consequently larger equipment footprints. The environmental footprint of sorbent production itself must also be considered, as energy-intensive synthesis processes can potentially offset carbon reduction benefits.
Recent techno-economic analyses suggest that breakthrough materials combining low-cost precursors with simplified synthesis routes offer the most promising path forward. Waste-derived sorbents, including those synthesized from industrial by-products or biomass, demonstrate particularly favorable economics with production costs potentially below $5 per kilogram while maintaining acceptable performance metrics. These materials, though often exhibiting lower per-gram capacities than highly engineered alternatives, may prove more economically viable at scale due to dramatically lower production costs and sufficient durability.
Material composition directly impacts production costs, with highly engineered materials like metal-organic frameworks (MOFs) and functionalized porous polymers commanding premium prices due to complex synthesis procedures and expensive precursors. For instance, amine-functionalized silica sorbents cost approximately $20-50 per kilogram, while more advanced MOFs can exceed $100 per kilogram at current production scales. These costs must decrease by at least an order of magnitude to achieve economic viability in commercial applications.
Scalability challenges are equally significant. Many high-performance sorbents that demonstrate excellent CO2 selectivity and capacity in laboratory conditions face manufacturing bottlenecks when scaled to industrial production. Synthesis methods that work efficiently at gram-scale often encounter issues with batch consistency, reaction kinetics, and purification when scaled to kilogram or ton quantities. Materials requiring precise nanostructure control, such as hierarchical porous carbons or precisely engineered MOFs, face particularly severe scaling limitations.
Lifecycle considerations further complicate the economic equation. Sorbent degradation rates vary dramatically based on material composition, with some advanced materials losing 5-15% capacity per 100 cycles under industrial conditions. More stable materials typically trade performance for longevity, necessitating larger quantities of sorbent and consequently larger equipment footprints. The environmental footprint of sorbent production itself must also be considered, as energy-intensive synthesis processes can potentially offset carbon reduction benefits.
Recent techno-economic analyses suggest that breakthrough materials combining low-cost precursors with simplified synthesis routes offer the most promising path forward. Waste-derived sorbents, including those synthesized from industrial by-products or biomass, demonstrate particularly favorable economics with production costs potentially below $5 per kilogram while maintaining acceptable performance metrics. These materials, though often exhibiting lower per-gram capacities than highly engineered alternatives, may prove more economically viable at scale due to dramatically lower production costs and sufficient durability.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!