How Enol Formation Affects Catalytic Efficiency
MAR 6, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Enol Chemistry Background and Catalytic Targets
Enol chemistry represents a fundamental aspect of organic chemistry that has evolved significantly since its initial discovery in the late 19th century. The term "enol" derives from the combination of "alkene" and "alcohol," reflecting the dual nature of these compounds containing both C=C double bonds and hydroxyl groups. This tautomeric form exists in dynamic equilibrium with its corresponding keto form, a phenomenon known as keto-enol tautomerism, which was first systematically studied by Ludwig Claisen and others in the early 1900s.
The historical development of enol chemistry has been closely intertwined with advances in catalytic science. Early observations of enol intermediates in aldol condensations and other carbon-carbon bond forming reactions laid the groundwork for understanding their catalytic significance. The recognition that enol and enolate species serve as nucleophilic partners in numerous organic transformations has positioned them as central players in synthetic methodology.
Modern catalytic systems increasingly rely on controlled enol formation to achieve high selectivity and efficiency. The ability to generate and stabilize enol intermediates under mild conditions has become a cornerstone of contemporary organic synthesis. This evolution reflects broader trends toward more sustainable and atom-economical processes, where precise control over reactive intermediates translates directly to improved catalytic performance.
The primary technical objectives in enol-mediated catalysis center on achieving optimal balance between enol stability and reactivity. Current research focuses on developing catalytic systems that can selectively promote enol formation while maintaining sufficient reactivity for subsequent transformations. This involves fine-tuning electronic and steric factors that govern the keto-enol equilibrium position and the kinetics of enol-mediated reactions.
Advanced catalytic targets include the development of asymmetric enol-forming processes that can generate chiral products with high enantioselectivity. Additionally, there is growing interest in designing catalysts that can operate under environmentally benign conditions while maintaining high turnover frequencies and selectivities. The integration of computational modeling with experimental catalyst design has emerged as a powerful approach for predicting and optimizing enol formation pathways in complex catalytic cycles.
The historical development of enol chemistry has been closely intertwined with advances in catalytic science. Early observations of enol intermediates in aldol condensations and other carbon-carbon bond forming reactions laid the groundwork for understanding their catalytic significance. The recognition that enol and enolate species serve as nucleophilic partners in numerous organic transformations has positioned them as central players in synthetic methodology.
Modern catalytic systems increasingly rely on controlled enol formation to achieve high selectivity and efficiency. The ability to generate and stabilize enol intermediates under mild conditions has become a cornerstone of contemporary organic synthesis. This evolution reflects broader trends toward more sustainable and atom-economical processes, where precise control over reactive intermediates translates directly to improved catalytic performance.
The primary technical objectives in enol-mediated catalysis center on achieving optimal balance between enol stability and reactivity. Current research focuses on developing catalytic systems that can selectively promote enol formation while maintaining sufficient reactivity for subsequent transformations. This involves fine-tuning electronic and steric factors that govern the keto-enol equilibrium position and the kinetics of enol-mediated reactions.
Advanced catalytic targets include the development of asymmetric enol-forming processes that can generate chiral products with high enantioselectivity. Additionally, there is growing interest in designing catalysts that can operate under environmentally benign conditions while maintaining high turnover frequencies and selectivities. The integration of computational modeling with experimental catalyst design has emerged as a powerful approach for predicting and optimizing enol formation pathways in complex catalytic cycles.
Market Demand for Enol-Enhanced Catalytic Processes
The pharmaceutical industry represents the largest market segment for enol-enhanced catalytic processes, driven by the critical need for stereoselective synthesis and improved reaction efficiency. Enol intermediates play pivotal roles in the production of active pharmaceutical ingredients, particularly in asymmetric synthesis where precise control over molecular configuration directly impacts drug efficacy and safety profiles. The growing emphasis on green chemistry principles has intensified demand for catalytic processes that leverage enol formation to reduce waste generation and energy consumption.
Fine chemical manufacturing constitutes another substantial market driver, where enol-enhanced catalysis enables the production of high-value specialty chemicals with superior purity and yield. Industries producing agrochemicals, flavors, fragrances, and advanced materials increasingly rely on these processes to achieve complex molecular transformations that would otherwise require multiple synthetic steps or harsh reaction conditions.
The petrochemical sector demonstrates growing interest in enol-enhanced catalytic processes for refining applications and the production of platform chemicals. These processes offer pathways to convert renewable feedstocks into valuable chemical intermediates, aligning with industry sustainability goals and regulatory pressures for reduced carbon footprints.
Market demand is particularly strong in regions with established chemical manufacturing infrastructure, including North America, Europe, and Asia-Pacific. The Asia-Pacific region shows the highest growth potential due to expanding pharmaceutical manufacturing capabilities and increasing investment in specialty chemical production facilities.
Emerging applications in polymer synthesis and materials science are creating new market opportunities. Enol-enhanced catalytic processes enable the development of novel polymer architectures and functional materials with tailored properties, addressing demands from electronics, automotive, and aerospace industries.
The market trajectory indicates sustained growth driven by regulatory requirements for cleaner production methods, cost pressures demanding higher process efficiency, and the continuous need for innovative synthetic routes to complex molecules. Industrial adoption rates correlate strongly with demonstrated improvements in selectivity, yield, and overall process economics compared to conventional catalytic approaches.
Fine chemical manufacturing constitutes another substantial market driver, where enol-enhanced catalysis enables the production of high-value specialty chemicals with superior purity and yield. Industries producing agrochemicals, flavors, fragrances, and advanced materials increasingly rely on these processes to achieve complex molecular transformations that would otherwise require multiple synthetic steps or harsh reaction conditions.
The petrochemical sector demonstrates growing interest in enol-enhanced catalytic processes for refining applications and the production of platform chemicals. These processes offer pathways to convert renewable feedstocks into valuable chemical intermediates, aligning with industry sustainability goals and regulatory pressures for reduced carbon footprints.
Market demand is particularly strong in regions with established chemical manufacturing infrastructure, including North America, Europe, and Asia-Pacific. The Asia-Pacific region shows the highest growth potential due to expanding pharmaceutical manufacturing capabilities and increasing investment in specialty chemical production facilities.
Emerging applications in polymer synthesis and materials science are creating new market opportunities. Enol-enhanced catalytic processes enable the development of novel polymer architectures and functional materials with tailored properties, addressing demands from electronics, automotive, and aerospace industries.
The market trajectory indicates sustained growth driven by regulatory requirements for cleaner production methods, cost pressures demanding higher process efficiency, and the continuous need for innovative synthetic routes to complex molecules. Industrial adoption rates correlate strongly with demonstrated improvements in selectivity, yield, and overall process economics compared to conventional catalytic approaches.
Current Enol Formation Challenges in Catalysis
Enol formation in catalytic systems faces several fundamental challenges that significantly impact overall catalytic efficiency. The primary obstacle lies in the thermodynamic instability of enol intermediates, which tend to rapidly tautomerize back to their more stable keto forms. This inherent instability creates a narrow window for catalytic interaction, requiring precise control of reaction conditions to maintain sufficient enol concentrations for effective catalysis.
The kinetic barriers associated with enol formation present another critical challenge. Many catalytic processes require specific activation energies to facilitate the initial enolization step, often necessitating elevated temperatures or specialized catalytic environments. These conditions can paradoxically destabilize the desired enol intermediates, creating a delicate balance between formation and decomposition rates that must be carefully optimized.
Selectivity issues compound these difficulties, particularly in complex molecular systems where multiple enolization sites exist. Controlling regioselectivity to favor the formation of specific enol isomers remains a significant technical hurdle. The presence of competing reaction pathways can lead to unwanted side products, reducing overall catalytic efficiency and complicating product purification processes.
Catalyst deactivation represents a persistent challenge in enol-mediated catalytic systems. The reactive nature of enol intermediates can lead to catalyst poisoning through irreversible binding or the formation of inactive catalyst-substrate complexes. This phenomenon is particularly problematic in heterogeneous catalytic systems where surface active sites become blocked by strongly adsorbed enol species.
Solvent effects and reaction medium compatibility pose additional constraints on enol formation efficiency. The choice of solvent significantly influences enol stability and formation kinetics, yet many solvents that favor enol formation may be incompatible with specific catalytic systems or downstream processing requirements. This limitation often forces compromises that reduce overall system performance.
Scale-up challenges emerge when transitioning from laboratory-scale enol formation studies to industrial applications. The precise control required for optimal enol formation becomes increasingly difficult to maintain in larger reactor systems, where heat and mass transfer limitations can create non-uniform reaction conditions that negatively impact enol formation rates and catalytic efficiency.
The kinetic barriers associated with enol formation present another critical challenge. Many catalytic processes require specific activation energies to facilitate the initial enolization step, often necessitating elevated temperatures or specialized catalytic environments. These conditions can paradoxically destabilize the desired enol intermediates, creating a delicate balance between formation and decomposition rates that must be carefully optimized.
Selectivity issues compound these difficulties, particularly in complex molecular systems where multiple enolization sites exist. Controlling regioselectivity to favor the formation of specific enol isomers remains a significant technical hurdle. The presence of competing reaction pathways can lead to unwanted side products, reducing overall catalytic efficiency and complicating product purification processes.
Catalyst deactivation represents a persistent challenge in enol-mediated catalytic systems. The reactive nature of enol intermediates can lead to catalyst poisoning through irreversible binding or the formation of inactive catalyst-substrate complexes. This phenomenon is particularly problematic in heterogeneous catalytic systems where surface active sites become blocked by strongly adsorbed enol species.
Solvent effects and reaction medium compatibility pose additional constraints on enol formation efficiency. The choice of solvent significantly influences enol stability and formation kinetics, yet many solvents that favor enol formation may be incompatible with specific catalytic systems or downstream processing requirements. This limitation often forces compromises that reduce overall system performance.
Scale-up challenges emerge when transitioning from laboratory-scale enol formation studies to industrial applications. The precise control required for optimal enol formation becomes increasingly difficult to maintain in larger reactor systems, where heat and mass transfer limitations can create non-uniform reaction conditions that negatively impact enol formation rates and catalytic efficiency.
Existing Enol Formation Enhancement Solutions
01 Use of metal catalysts for enol formation
Metal catalysts, particularly transition metals and their complexes, can be employed to enhance the efficiency of enol formation reactions. These catalysts facilitate the tautomerization process by lowering the activation energy required for the keto-enol conversion. The catalytic systems can include various metal ions and organometallic compounds that promote the formation of enol intermediates in organic synthesis reactions.- Use of metal catalysts for enol formation: Metal catalysts, particularly transition metals and their complexes, can be employed to enhance the efficiency of enol formation reactions. These catalysts facilitate the tautomerization process by lowering the activation energy required for the keto-enol conversion. The catalytic systems can include various metal ions and organometallic compounds that promote the formation of enol intermediates in organic synthesis reactions.
- Acid-base catalysis for enol formation: Acid or base catalysts can be utilized to improve the rate and selectivity of enol formation. These catalysts work by either protonating the carbonyl oxygen or deprotonating the alpha-carbon, thereby facilitating the formation of the enol tautomer. The choice between acidic or basic conditions depends on the substrate structure and desired reaction pathway. Various organic and inorganic acids or bases can be employed to optimize the catalytic efficiency.
- Enzyme-catalyzed enol formation: Enzymatic catalysts can provide highly selective and efficient pathways for enol formation under mild conditions. Specific enzymes can catalyze the tautomerization process with excellent stereoselectivity and regioselectivity. These biocatalytic approaches offer advantages in terms of environmental friendliness and specificity compared to traditional chemical catalysts.
- Heterogeneous catalysts for enol formation: Solid-phase heterogeneous catalysts can be employed to facilitate enol formation with advantages in catalyst recovery and reusability. These catalysts include supported metal catalysts, zeolites, and other porous materials that provide active sites for the tautomerization reaction. The heterogeneous nature allows for easier separation from reaction mixtures and potential for continuous flow processes.
- Optimization of reaction conditions for enol formation: The catalytic efficiency of enol formation can be significantly enhanced by optimizing various reaction parameters including temperature, pressure, solvent selection, and catalyst concentration. Specific reaction conditions can be tailored to favor the enol form over the keto form, improving both yield and selectivity. The use of appropriate additives and co-catalysts can further enhance the overall catalytic performance.
02 Acid-base catalysis for enol formation
Acid and base catalysts play a crucial role in promoting enol formation through proton transfer mechanisms. Both Brønsted and Lewis acids or bases can be utilized to facilitate the tautomerization equilibrium. The catalytic efficiency can be optimized by selecting appropriate acid or base strength and reaction conditions to favor enol formation over the keto form.Expand Specific Solutions03 Enzyme-catalyzed enol formation
Enzymatic catalysis offers a biocatalytic approach to enol formation with high selectivity and efficiency. Specific enzymes can catalyze the conversion between keto and enol forms under mild conditions. These biocatalysts provide advantages in terms of stereoselectivity and environmental compatibility, making them suitable for pharmaceutical and fine chemical synthesis applications.Expand Specific Solutions04 Solvent effects on enol formation catalysis
The choice of solvent significantly impacts the catalytic efficiency of enol formation reactions. Polar and non-polar solvents can influence the tautomeric equilibrium and reaction kinetics differently. Solvent systems can be optimized to stabilize enol intermediates and enhance catalyst performance, with considerations for solvent polarity, hydrogen bonding capability, and coordination properties affecting the overall catalytic efficiency.Expand Specific Solutions05 Temperature and pressure optimization for enol formation
Reaction conditions such as temperature and pressure are critical parameters for optimizing enol formation catalytic efficiency. Elevated temperatures can increase reaction rates but may also affect the equilibrium position between keto and enol forms. Pressure adjustments can influence the reaction pathway and selectivity. Systematic optimization of these parameters enables improved catalytic performance and higher yields of desired enol products.Expand Specific Solutions
Key Players in Enol Chemistry and Catalysis Industry
The enol formation catalysis field represents a mature industrial sector with substantial market presence, dominated by established petrochemical giants and specialty chemical manufacturers. Major players include China Petroleum & Chemical Corp. (Sinopec), BASF Corp., Eastman Chemical Co., and DuPont de Nemours, alongside Asian leaders like Asahi Kasei Corp. and Wanhua Chemical Group. The competitive landscape spans from integrated oil companies like Saudi Arabian Oil Co. and ExxonMobil Chemical Patents to specialized catalyst developers such as Viridis Chemical LLC. Technology maturity varies significantly across applications, with traditional petrochemical processes being well-established while emerging sustainable catalytic routes remain in development phases. Research institutions like IFP Energies Nouvelles and King Fahd University contribute to advancing next-generation enol formation technologies, indicating ongoing innovation despite the sector's overall technological maturity.
China Petroleum & Chemical Corp.
Technical Solution: Sinopec has developed catalyst technologies that utilize enol formation principles to enhance efficiency in large-scale petrochemical processes. Their research focuses on creating bifunctional catalysts that can simultaneously promote enol formation and subsequent chemical transformations in integrated reaction systems. The company has implemented these catalysts in their refineries and chemical plants to improve conversion rates in processes such as alkylation, isomerization, and cracking reactions. Their catalyst designs incorporate novel support materials and active metal combinations that optimize enol-keto equilibria for maximum catalytic performance.
Strengths: Massive production scale and integrated petrochemical operations provide extensive testing and implementation opportunities. Weaknesses: Technology development primarily focused on traditional petrochemical processes with limited innovation in emerging green chemistry applications.
BASF Corp.
Technical Solution: BASF has developed advanced catalyst systems that leverage enol formation mechanisms to enhance catalytic efficiency in various chemical processes. Their proprietary catalyst designs incorporate metal-organic frameworks and zeolite-based materials that facilitate controlled enol-keto tautomerization, leading to improved reaction selectivity and conversion rates. The company's research focuses on understanding how enol intermediates can be stabilized through specific active site geometries and electronic environments, resulting in catalysts with enhanced turnover frequencies and reduced energy barriers for key industrial transformations.
Strengths: Global leader in catalyst technology with extensive R&D capabilities and industrial-scale manufacturing expertise. Weaknesses: High development costs and complex catalyst synthesis processes may limit rapid deployment.
Core Patents in Enol-Catalysis Mechanisms
A process for enhancing the catalytic efficiency of oer
PatentPendingUS20220389597A1
Innovation
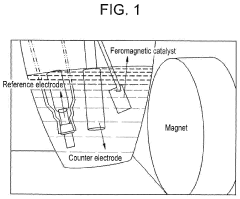
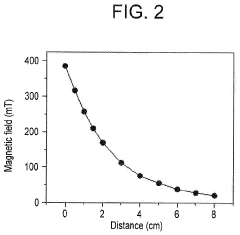
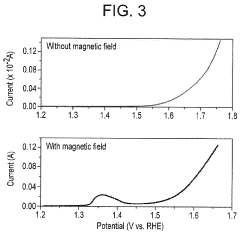
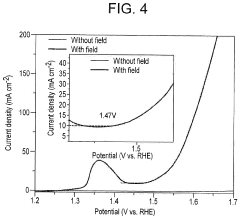
- Applying a small constant external magnetic field, greater than the Earth's magnetic field but ≤200 mT, to OER catalysts with a non-zero Berry phase, such as metals like Co, Cr, Mn, and Ni, or their alloys and compounds, to increase catalytic efficiency by aligning electron spins and modifying the Berry phase.
Apparatus for increasing catalytic efficiency
PatentInactiveUS5501846A
Innovation
- Mechanical and electromagnetic methods are employed to break up large crystallites into smaller domains, including compression, tension, torque, shock, and electromagnetic pulses, to enhance catalyst efficiency and maintain reactant mobility and surface area, thereby minimizing the need for manual intervention.
Green Chemistry Regulations for Enol Processes
The regulatory landscape for enol-based chemical processes has evolved significantly in response to growing environmental concerns and the principles of green chemistry. Current regulations primarily focus on minimizing waste generation, reducing hazardous substance usage, and promoting atom economy in enol formation reactions. The Environmental Protection Agency and European Chemicals Agency have established specific guidelines that directly impact how enol processes are designed and implemented in industrial settings.
Solvent selection represents a critical regulatory consideration for enol processes. Traditional organic solvents used in enol formation, such as chlorinated compounds and aromatic hydrocarbons, face increasing restrictions due to their environmental persistence and toxicity profiles. Regulatory frameworks now favor aqueous systems, ionic liquids, and supercritical fluids as alternative reaction media. These regulations have prompted significant research into water-compatible catalysts and solvent-free enol formation methodologies.
Catalyst composition and lifecycle management have become subject to stringent regulatory oversight. Heavy metal catalysts commonly employed in enol chemistry, including palladium, platinum, and rhodium complexes, must comply with strict leaching limits and recovery requirements. Regulations mandate comprehensive catalyst recycling protocols and encourage the development of earth-abundant metal alternatives. This has accelerated research into iron, copper, and nickel-based catalytic systems for enol formation.
Energy efficiency standards increasingly influence enol process design and operation. Regulatory bodies have established energy consumption benchmarks that favor mild reaction conditions and eliminate high-temperature requirements traditionally associated with enol formation. These standards promote the development of photocatalytic and electrochemical approaches that operate under ambient conditions while maintaining high catalytic efficiency.
Waste minimization regulations specifically target byproduct formation in enol processes. Current frameworks require detailed mass balance accounting and mandate waste reduction targets of at least 50% compared to conventional methods. This has driven innovation in cascade reactions and tandem processes that integrate enol formation with subsequent transformations, thereby eliminating isolation and purification steps that generate significant waste streams.
Emerging regulations address the entire process lifecycle, from raw material sourcing to product disposal. These comprehensive frameworks evaluate the environmental impact of feedstock production, transportation, and end-of-life considerations. For enol processes, this means prioritizing renewable feedstocks and designing products with enhanced biodegradability profiles to meet evolving regulatory requirements.
Solvent selection represents a critical regulatory consideration for enol processes. Traditional organic solvents used in enol formation, such as chlorinated compounds and aromatic hydrocarbons, face increasing restrictions due to their environmental persistence and toxicity profiles. Regulatory frameworks now favor aqueous systems, ionic liquids, and supercritical fluids as alternative reaction media. These regulations have prompted significant research into water-compatible catalysts and solvent-free enol formation methodologies.
Catalyst composition and lifecycle management have become subject to stringent regulatory oversight. Heavy metal catalysts commonly employed in enol chemistry, including palladium, platinum, and rhodium complexes, must comply with strict leaching limits and recovery requirements. Regulations mandate comprehensive catalyst recycling protocols and encourage the development of earth-abundant metal alternatives. This has accelerated research into iron, copper, and nickel-based catalytic systems for enol formation.
Energy efficiency standards increasingly influence enol process design and operation. Regulatory bodies have established energy consumption benchmarks that favor mild reaction conditions and eliminate high-temperature requirements traditionally associated with enol formation. These standards promote the development of photocatalytic and electrochemical approaches that operate under ambient conditions while maintaining high catalytic efficiency.
Waste minimization regulations specifically target byproduct formation in enol processes. Current frameworks require detailed mass balance accounting and mandate waste reduction targets of at least 50% compared to conventional methods. This has driven innovation in cascade reactions and tandem processes that integrate enol formation with subsequent transformations, thereby eliminating isolation and purification steps that generate significant waste streams.
Emerging regulations address the entire process lifecycle, from raw material sourcing to product disposal. These comprehensive frameworks evaluate the environmental impact of feedstock production, transportation, and end-of-life considerations. For enol processes, this means prioritizing renewable feedstocks and designing products with enhanced biodegradability profiles to meet evolving regulatory requirements.
Safety Considerations in Enol-Based Catalysis
Enol-based catalytic processes present unique safety challenges that require comprehensive risk assessment and mitigation strategies. The inherent reactivity of enol intermediates, characterized by their nucleophilic carbon centers and acidic hydrogen atoms, creates potential hazards including uncontrolled polymerization, thermal runaway reactions, and formation of explosive peroxides under oxidative conditions. These reactive species can undergo rapid decomposition or side reactions when exposed to trace metals, light, or elevated temperatures.
The tautomeric equilibrium between keto and enol forms introduces additional complexity in safety protocols. Shifts in this equilibrium due to temperature fluctuations, pH changes, or solvent effects can lead to unexpected concentration spikes of highly reactive enol species. This variability necessitates continuous monitoring systems and adaptive control mechanisms to maintain safe operating conditions throughout the catalytic process.
Solvent selection plays a critical role in enol-based catalysis safety. Protic solvents can stabilize enol intermediates through hydrogen bonding but may also facilitate unwanted proton transfer reactions. Aprotic solvents, while reducing side reactions, can increase the concentration of reactive enol species, potentially leading to higher reaction rates and associated thermal risks. The choice of solvent system must balance catalytic efficiency with thermal stability and vapor pressure considerations.
Catalyst design and handling procedures require special attention in enol-based systems. Metal catalysts that promote enol formation may also catalyze decomposition pathways, particularly under oxygen exposure or in the presence of moisture. Proper storage under inert atmospheres, temperature control during catalyst preparation, and implementation of quenching protocols are essential safety measures.
Emergency response protocols must account for the rapid kinetics of enol chemistry. Standard fire suppression methods may be ineffective or counterproductive with certain enol-containing systems. Specialized extinguishing agents and containment procedures should be developed based on the specific enol substrates and reaction conditions employed. Personnel training must emphasize recognition of early warning signs of thermal instability and proper shutdown procedures to prevent escalation of safety incidents.
The tautomeric equilibrium between keto and enol forms introduces additional complexity in safety protocols. Shifts in this equilibrium due to temperature fluctuations, pH changes, or solvent effects can lead to unexpected concentration spikes of highly reactive enol species. This variability necessitates continuous monitoring systems and adaptive control mechanisms to maintain safe operating conditions throughout the catalytic process.
Solvent selection plays a critical role in enol-based catalysis safety. Protic solvents can stabilize enol intermediates through hydrogen bonding but may also facilitate unwanted proton transfer reactions. Aprotic solvents, while reducing side reactions, can increase the concentration of reactive enol species, potentially leading to higher reaction rates and associated thermal risks. The choice of solvent system must balance catalytic efficiency with thermal stability and vapor pressure considerations.
Catalyst design and handling procedures require special attention in enol-based systems. Metal catalysts that promote enol formation may also catalyze decomposition pathways, particularly under oxygen exposure or in the presence of moisture. Proper storage under inert atmospheres, temperature control during catalyst preparation, and implementation of quenching protocols are essential safety measures.
Emergency response protocols must account for the rapid kinetics of enol chemistry. Standard fire suppression methods may be ineffective or counterproductive with certain enol-containing systems. Specialized extinguishing agents and containment procedures should be developed based on the specific enol substrates and reaction conditions employed. Personnel training must emphasize recognition of early warning signs of thermal instability and proper shutdown procedures to prevent escalation of safety incidents.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!