Quantifying Enol Presence in Multi-Component Systems
MAR 6, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Enol Quantification Background and Research Objectives
Enol tautomers represent a fundamental class of organic compounds characterized by the presence of a hydroxyl group attached to a carbon-carbon double bond. These species exist in dynamic equilibrium with their corresponding keto forms through a process known as keto-enol tautomerism, which plays a crucial role in numerous chemical and biological processes. The quantification of enol presence in multi-component systems has emerged as a critical analytical challenge due to the transient nature of these species and their typically low equilibrium concentrations under standard conditions.
The historical development of enol quantification techniques traces back to early 20th-century organic chemistry research, where scientists first recognized the importance of tautomeric equilibria in understanding reaction mechanisms. Initial approaches relied primarily on indirect methods such as chemical trapping and derivatization techniques. The evolution progressed through spectroscopic methods in the mid-20th century, with nuclear magnetic resonance and infrared spectroscopy providing direct observation capabilities for enol species.
Modern analytical chemistry has witnessed significant advancement in enol detection methodologies, driven by the increasing recognition of their importance in pharmaceutical development, catalysis, and biochemical processes. Contemporary techniques encompass real-time monitoring capabilities, enabling researchers to observe tautomeric equilibria under physiological conditions and in complex matrices. The integration of computational chemistry with experimental approaches has further enhanced our understanding of enol stability and behavior in various environments.
The primary research objectives in this field center on developing robust, sensitive, and selective analytical methods capable of accurately quantifying enol concentrations in the presence of multiple interfering species. Key technical goals include achieving detection limits in the micromolar to nanomolar range, establishing methods that can operate under diverse pH and temperature conditions, and creating standardized protocols for enol quantification across different analytical platforms.
Another critical objective involves understanding the kinetic aspects of enol formation and decay in multi-component systems, particularly in the presence of catalysts, enzymes, or other reactive species that may influence tautomeric equilibria. This knowledge is essential for predicting enol behavior in complex biological and industrial systems.
The development of real-time monitoring capabilities represents a significant technological target, enabling researchers to track enol concentrations during chemical reactions or biological processes. This objective requires overcoming challenges related to the rapid interconversion rates between tautomeric forms and the potential interference from other system components.
Standardization and validation of enol quantification methods across different analytical platforms constitute another fundamental objective, ensuring reproducibility and comparability of results across research institutions and industrial applications. This includes establishing reference materials and developing quality control protocols specific to enol analysis in complex matrices.
The historical development of enol quantification techniques traces back to early 20th-century organic chemistry research, where scientists first recognized the importance of tautomeric equilibria in understanding reaction mechanisms. Initial approaches relied primarily on indirect methods such as chemical trapping and derivatization techniques. The evolution progressed through spectroscopic methods in the mid-20th century, with nuclear magnetic resonance and infrared spectroscopy providing direct observation capabilities for enol species.
Modern analytical chemistry has witnessed significant advancement in enol detection methodologies, driven by the increasing recognition of their importance in pharmaceutical development, catalysis, and biochemical processes. Contemporary techniques encompass real-time monitoring capabilities, enabling researchers to observe tautomeric equilibria under physiological conditions and in complex matrices. The integration of computational chemistry with experimental approaches has further enhanced our understanding of enol stability and behavior in various environments.
The primary research objectives in this field center on developing robust, sensitive, and selective analytical methods capable of accurately quantifying enol concentrations in the presence of multiple interfering species. Key technical goals include achieving detection limits in the micromolar to nanomolar range, establishing methods that can operate under diverse pH and temperature conditions, and creating standardized protocols for enol quantification across different analytical platforms.
Another critical objective involves understanding the kinetic aspects of enol formation and decay in multi-component systems, particularly in the presence of catalysts, enzymes, or other reactive species that may influence tautomeric equilibria. This knowledge is essential for predicting enol behavior in complex biological and industrial systems.
The development of real-time monitoring capabilities represents a significant technological target, enabling researchers to track enol concentrations during chemical reactions or biological processes. This objective requires overcoming challenges related to the rapid interconversion rates between tautomeric forms and the potential interference from other system components.
Standardization and validation of enol quantification methods across different analytical platforms constitute another fundamental objective, ensuring reproducibility and comparability of results across research institutions and industrial applications. This includes establishing reference materials and developing quality control protocols specific to enol analysis in complex matrices.
Market Demand for Multi-Component Enol Analysis
The pharmaceutical industry represents the largest market segment for multi-component enol analysis technologies, driven by stringent regulatory requirements for drug purity and quality control. Pharmaceutical manufacturers require precise quantification of enol tautomers in active pharmaceutical ingredients, as these forms can significantly impact drug efficacy, stability, and bioavailability. The increasing complexity of modern drug formulations, particularly those involving multiple active compounds, has intensified the demand for sophisticated analytical methods capable of distinguishing and quantifying enol species in complex matrices.
Chemical manufacturing sectors, including specialty chemicals, agrochemicals, and fine chemicals, constitute another substantial market driver. These industries face growing pressure to optimize reaction yields and product quality, necessitating real-time monitoring of enol-keto equilibria during synthesis processes. The ability to quantify enol presence in multi-component reaction mixtures enables manufacturers to fine-tune reaction conditions, reduce waste, and improve overall process efficiency.
Academic and research institutions contribute significantly to market demand through fundamental research in organic chemistry, biochemistry, and materials science. Universities and research centers require advanced analytical capabilities to study tautomeric equilibria, reaction mechanisms, and molecular interactions. This segment drives innovation in analytical methodologies and creates demand for cutting-edge instrumentation and software solutions.
The food and beverage industry presents an emerging market opportunity, particularly in quality control and flavor analysis applications. Enol compounds play crucial roles in flavor development and food stability, making their quantification essential for product consistency and shelf-life optimization. Regulatory compliance requirements for food additives and natural products further amplify this demand.
Environmental monitoring and forensic analysis sectors represent niche but growing markets. Environmental agencies require enol quantification capabilities for pollutant analysis and environmental impact assessments, while forensic laboratories need these technologies for trace evidence analysis and contamination studies.
Market growth is accelerated by increasing regulatory scrutiny across industries, technological advancements in analytical instrumentation, and growing awareness of the importance of tautomeric forms in chemical and biological systems. The trend toward personalized medicine and precision manufacturing further drives demand for more sophisticated analytical capabilities in multi-component systems.
Chemical manufacturing sectors, including specialty chemicals, agrochemicals, and fine chemicals, constitute another substantial market driver. These industries face growing pressure to optimize reaction yields and product quality, necessitating real-time monitoring of enol-keto equilibria during synthesis processes. The ability to quantify enol presence in multi-component reaction mixtures enables manufacturers to fine-tune reaction conditions, reduce waste, and improve overall process efficiency.
Academic and research institutions contribute significantly to market demand through fundamental research in organic chemistry, biochemistry, and materials science. Universities and research centers require advanced analytical capabilities to study tautomeric equilibria, reaction mechanisms, and molecular interactions. This segment drives innovation in analytical methodologies and creates demand for cutting-edge instrumentation and software solutions.
The food and beverage industry presents an emerging market opportunity, particularly in quality control and flavor analysis applications. Enol compounds play crucial roles in flavor development and food stability, making their quantification essential for product consistency and shelf-life optimization. Regulatory compliance requirements for food additives and natural products further amplify this demand.
Environmental monitoring and forensic analysis sectors represent niche but growing markets. Environmental agencies require enol quantification capabilities for pollutant analysis and environmental impact assessments, while forensic laboratories need these technologies for trace evidence analysis and contamination studies.
Market growth is accelerated by increasing regulatory scrutiny across industries, technological advancements in analytical instrumentation, and growing awareness of the importance of tautomeric forms in chemical and biological systems. The trend toward personalized medicine and precision manufacturing further drives demand for more sophisticated analytical capabilities in multi-component systems.
Current Challenges in Enol Detection and Quantification
The quantification of enol tautomers in multi-component systems presents significant analytical challenges that stem from the inherent instability and low concentrations of these species. Traditional spectroscopic methods often struggle with the rapid equilibrium dynamics between keto and enol forms, particularly when the equilibrium heavily favors the keto tautomer. This fundamental limitation creates substantial detection thresholds that may exceed the actual enol concentrations present in many systems.
Spectral interference represents another critical obstacle in multi-component analysis. The presence of multiple chemical species with overlapping absorption bands, fluorescence emissions, or NMR signals can mask the characteristic signatures of enol tautomers. This interference becomes particularly problematic in complex matrices where solvent effects, hydrogen bonding networks, and intermolecular interactions can shift spectral features unpredictably.
Temperature and pH sensitivity of tautomeric equilibria introduce additional complexity to quantification efforts. Small variations in experimental conditions can dramatically alter enol populations, making reproducible measurements extremely difficult. The challenge intensifies when attempting to study enol behavior under physiologically relevant conditions where precise environmental control becomes paramount.
Current analytical techniques face temporal resolution limitations when monitoring dynamic tautomeric processes. Conventional methods often require measurement times that exceed the timescale of tautomeric interconversion, resulting in averaged signals that obscure true enol concentrations. This temporal mismatch prevents accurate real-time monitoring of enol populations during chemical reactions or biological processes.
Matrix effects in complex systems further complicate quantification efforts. The presence of proteins, lipids, salts, and other biomolecules can alter tautomeric equilibria through specific interactions, making it difficult to establish reliable calibration standards. These matrix-dependent variations necessitate system-specific analytical approaches that limit the transferability of quantification methods across different applications.
The lack of standardized reference materials and validated analytical protocols represents a significant methodological challenge. Without established benchmarks for enol quantification, inter-laboratory comparisons become unreliable, hindering the development of robust analytical frameworks for multi-component systems.
Spectral interference represents another critical obstacle in multi-component analysis. The presence of multiple chemical species with overlapping absorption bands, fluorescence emissions, or NMR signals can mask the characteristic signatures of enol tautomers. This interference becomes particularly problematic in complex matrices where solvent effects, hydrogen bonding networks, and intermolecular interactions can shift spectral features unpredictably.
Temperature and pH sensitivity of tautomeric equilibria introduce additional complexity to quantification efforts. Small variations in experimental conditions can dramatically alter enol populations, making reproducible measurements extremely difficult. The challenge intensifies when attempting to study enol behavior under physiologically relevant conditions where precise environmental control becomes paramount.
Current analytical techniques face temporal resolution limitations when monitoring dynamic tautomeric processes. Conventional methods often require measurement times that exceed the timescale of tautomeric interconversion, resulting in averaged signals that obscure true enol concentrations. This temporal mismatch prevents accurate real-time monitoring of enol populations during chemical reactions or biological processes.
Matrix effects in complex systems further complicate quantification efforts. The presence of proteins, lipids, salts, and other biomolecules can alter tautomeric equilibria through specific interactions, making it difficult to establish reliable calibration standards. These matrix-dependent variations necessitate system-specific analytical approaches that limit the transferability of quantification methods across different applications.
The lack of standardized reference materials and validated analytical protocols represents a significant methodological challenge. Without established benchmarks for enol quantification, inter-laboratory comparisons become unreliable, hindering the development of robust analytical frameworks for multi-component systems.
Existing Enol Quantification Techniques and Approaches
01 Detection and identification methods for enol forms
Various analytical techniques and methods have been developed to detect and identify the presence of enol forms in chemical compounds. These methods include spectroscopic analysis, chromatographic techniques, and chemical derivatization approaches that can distinguish between keto and enol tautomers. The detection methods are crucial for understanding the tautomeric equilibrium and structural characteristics of compounds exhibiting keto-enol tautomerism.- Detection and identification methods for enol forms: Various analytical techniques and methods have been developed to detect and identify the presence of enol tautomers in chemical compounds. These methods include spectroscopic analysis, chromatographic techniques, and chemical derivatization approaches that can distinguish between keto and enol forms. The detection methods are particularly useful in quality control and structural characterization of organic compounds.
- Enol stabilization in pharmaceutical compounds: Certain pharmaceutical formulations utilize enol forms or enolizable compounds as active ingredients or intermediates. The stabilization of enol tautomers can be achieved through specific formulation techniques, pH control, and the use of stabilizing agents. These approaches are important for maintaining the efficacy and shelf-life of pharmaceutical products containing enol-containing compounds.
- Enol-keto tautomerism in synthetic processes: The enol-keto equilibrium plays a crucial role in various synthetic chemical processes and reactions. Control of tautomeric forms can be achieved through reaction conditions, catalysts, and solvent selection. Understanding and manipulating this equilibrium is essential for optimizing yields and selectivity in organic synthesis, particularly in the production of fine chemicals and intermediates.
- Enol derivatives in polymer and material applications: Enol-containing compounds and their derivatives are utilized in polymer chemistry and material science applications. These compounds can serve as monomers, crosslinking agents, or functional additives that impart specific properties to polymeric materials. The enol functionality provides reactive sites for polymerization and modification reactions, enabling the development of materials with tailored characteristics.
- Enol forms in agricultural and chemical formulations: Enol tautomers and enolizable compounds find applications in agricultural chemicals and industrial formulations. The presence of enol forms can influence the biological activity, stability, and delivery characteristics of active ingredients. Formulation strategies may involve controlling the enol-keto ratio to optimize performance, enhance solubility, or improve compatibility with other components in the mixture.
02 Enol stabilization through chemical modifications
Chemical modifications and structural designs can be employed to stabilize enol forms of compounds. This includes the use of specific substituents, protective groups, or molecular frameworks that favor the enol tautomer over the keto form. Stabilization techniques are important in synthetic chemistry and pharmaceutical applications where the enol form exhibits desired properties or reactivity patterns.Expand Specific Solutions03 Enol-based synthetic intermediates and reactions
Enol forms serve as important intermediates in various synthetic reactions and chemical transformations. The reactivity of enol species can be exploited in condensation reactions, alkylation processes, and other organic synthesis pathways. Understanding and controlling enol formation is essential for developing efficient synthetic routes and improving reaction yields in chemical manufacturing.Expand Specific Solutions04 Pharmaceutical applications involving enol compounds
Enol-containing compounds and their derivatives have significant applications in pharmaceutical chemistry. The enol functionality can contribute to biological activity, drug metabolism, and pharmacokinetic properties. Various therapeutic agents utilize enol structures or enol-forming capabilities as part of their mechanism of action or as prodrug strategies for improved drug delivery.Expand Specific Solutions05 Industrial processes utilizing enol chemistry
Industrial chemical processes leverage enol chemistry for the production of various commercial products including polymers, dyes, and specialty chemicals. The enol form plays a role in polymerization reactions, coupling reactions, and other large-scale manufacturing processes. Control of enol-keto equilibrium is important for optimizing process efficiency and product quality in industrial applications.Expand Specific Solutions
Key Players in Analytical Chemistry and Instrumentation
The quantification of enol presence in multi-component systems represents a mature analytical chemistry field experiencing steady growth driven by pharmaceutical, petrochemical, and materials science applications. The market demonstrates moderate expansion with established analytical techniques including NMR spectroscopy, mass spectrometry, and chromatographic methods. Technology maturity varies significantly across industry players, with chemical giants like Dow Global Technologies LLC, ExxonMobil Chemical Patents Inc., and Mitsui Chemicals Inc. leading advanced analytical capabilities for polymer and petrochemical applications. Automotive manufacturers including Honda Motor Co. Ltd. and GM Global Technology Operations LLC focus on fuel system enol analysis, while energy companies such as Halliburton Energy Services Inc., Schlumberger Technologies Inc., and TotalEnergies SE emphasize oilfield chemistry applications. Academic institutions like City University of Hong Kong and University of Iowa Research Foundation contribute fundamental research, creating a competitive landscape characterized by specialized applications rather than broad technological disruption.
Schlumberger Technologies, Inc.
Technical Solution: Schlumberger has pioneered downhole analytical technologies for real-time quantification of enol compounds in complex reservoir fluids and drilling environments. Their system integrates miniaturized spectroscopic sensors with advanced signal processing algorithms to detect and quantify enol species under extreme pressure and temperature conditions. The technology employs machine learning approaches to distinguish enol signatures from interfering compounds in multi-phase fluid systems. Their platform provides continuous monitoring capabilities essential for optimizing extraction processes and ensuring fluid quality assessment in challenging operational environments.
Strengths: Exceptional performance under extreme conditions with real-time monitoring capabilities and robust field deployment. Weaknesses: High complexity and cost, limited to specialized oilfield applications.
Dow Global Technologies LLC
Technical Solution: Dow has developed advanced analytical methodologies for quantifying enol content in complex polymer systems and chemical formulations. Their approach combines high-resolution NMR spectroscopy with computational modeling to accurately determine enol-keto tautomeric equilibria in multi-component mixtures. The company utilizes proprietary algorithms that account for intermolecular interactions and solvent effects, enabling precise quantification even in the presence of multiple chemical species. Their technology platform integrates real-time monitoring capabilities with predictive modeling to optimize reaction conditions and product quality in industrial applications.
Strengths: Comprehensive analytical platform with strong computational support and industrial scalability. Weaknesses: High equipment costs and requires specialized expertise for operation.
Core Innovations in Enol Detection Technologies
Photometric system and methodology for easy detection and quantification of components from binary mixtures
PatentWO2023058052A1
Innovation
- A photometric system utilizing Raman spectroscopy with a mathematical model that predicts component concentrations based on the intensity ratios of characteristic Raman peaks, independent of experimental conditions such as laser intensity and integration time, enabling automated detection and quantification of binary mixtures without the need for trained professionals.
Multicomponent analysis of volatile organic compositions in vapor samples
PatentInactiveUS20050214169A1
Innovation
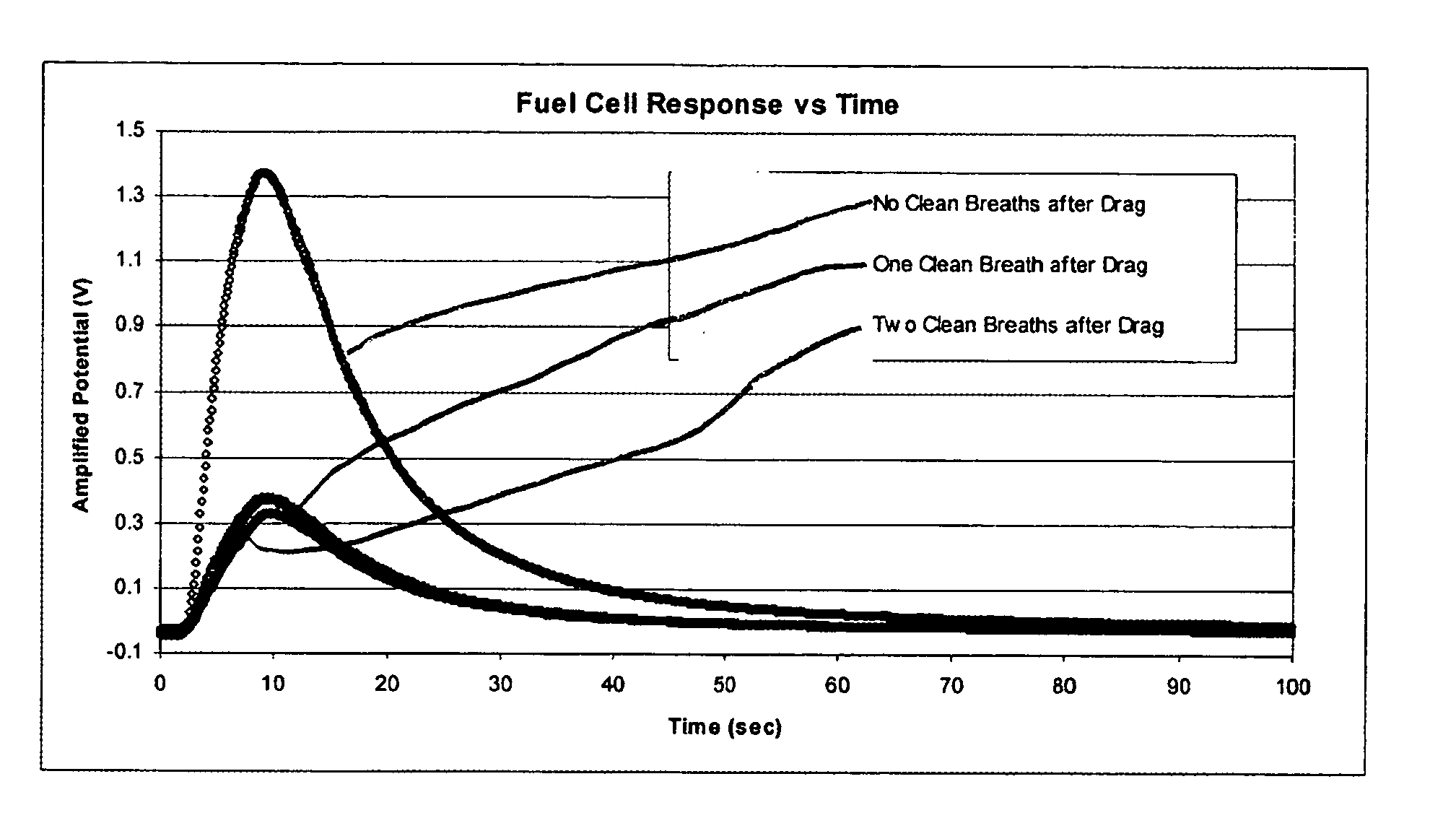
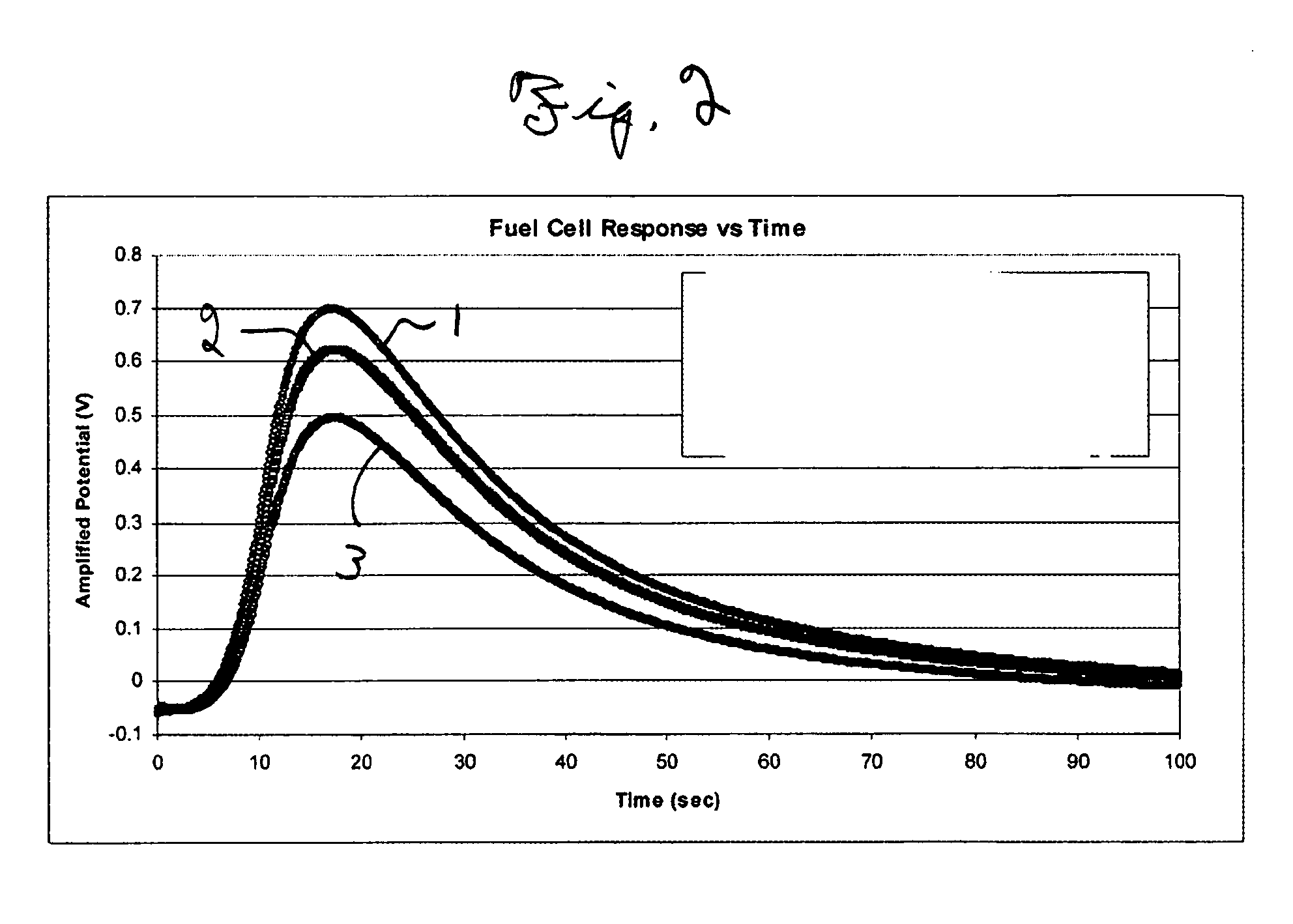
- A method using a fuel cell with a time response curve analysis to differentiate and quantify VOCs in breath samples by de-convoluting the fuel cell signal based on standard time response curves, allowing for the discrimination of alcohol consumption and smoking, and potentially evaluating individual health and environmental pollutants.
Analytical Method Validation Standards
Analytical method validation for quantifying enol presence in multi-component systems requires adherence to internationally recognized standards that ensure measurement reliability and regulatory compliance. The International Conference on Harmonisation (ICH) Q2(R1) guidelines serve as the primary framework, supplemented by FDA, EMA, and USP standards that collectively define validation parameters essential for analytical accuracy.
Specificity represents a critical validation parameter, demanding demonstration that the analytical method can unambiguously identify and quantify enol tautomers in the presence of other chemical species, degradation products, and matrix components. This requirement becomes particularly challenging in multi-component systems where spectral overlap and chemical interference may compromise measurement integrity.
Linearity validation must establish a direct proportional relationship between enol concentration and analytical response across the expected concentration range. ICH guidelines typically require correlation coefficients exceeding 0.999, with residual analysis confirming homoscedasticity and absence of systematic bias throughout the calibration range.
Precision validation encompasses both repeatability and intermediate precision assessments. Repeatability studies evaluate method performance under identical conditions within short time intervals, while intermediate precision examines variability across different days, analysts, and equipment. Relative standard deviation values should remain below 2% for quantitative methods at therapeutic concentration levels.
Accuracy validation requires comparison against certified reference materials or alternative validated methods, with recovery studies demonstrating bias within ±2% of theoretical values. This parameter proves particularly crucial for enol quantification, where tautomeric equilibrium shifts may introduce systematic errors if not properly controlled.
Robustness testing evaluates method stability against deliberate variations in analytical conditions, including temperature fluctuations, pH changes, mobile phase composition alterations, and column aging effects. These studies identify critical method parameters requiring strict control during routine analysis.
Detection and quantitation limits must be scientifically justified based on signal-to-noise ratios, with LOD typically set at 3:1 and LOQ at 10:1 ratios. For enol quantification in pharmaceutical applications, these limits should align with ICH Q3A impurity thresholds, ensuring adequate sensitivity for regulatory compliance and quality control purposes.
Specificity represents a critical validation parameter, demanding demonstration that the analytical method can unambiguously identify and quantify enol tautomers in the presence of other chemical species, degradation products, and matrix components. This requirement becomes particularly challenging in multi-component systems where spectral overlap and chemical interference may compromise measurement integrity.
Linearity validation must establish a direct proportional relationship between enol concentration and analytical response across the expected concentration range. ICH guidelines typically require correlation coefficients exceeding 0.999, with residual analysis confirming homoscedasticity and absence of systematic bias throughout the calibration range.
Precision validation encompasses both repeatability and intermediate precision assessments. Repeatability studies evaluate method performance under identical conditions within short time intervals, while intermediate precision examines variability across different days, analysts, and equipment. Relative standard deviation values should remain below 2% for quantitative methods at therapeutic concentration levels.
Accuracy validation requires comparison against certified reference materials or alternative validated methods, with recovery studies demonstrating bias within ±2% of theoretical values. This parameter proves particularly crucial for enol quantification, where tautomeric equilibrium shifts may introduce systematic errors if not properly controlled.
Robustness testing evaluates method stability against deliberate variations in analytical conditions, including temperature fluctuations, pH changes, mobile phase composition alterations, and column aging effects. These studies identify critical method parameters requiring strict control during routine analysis.
Detection and quantitation limits must be scientifically justified based on signal-to-noise ratios, with LOD typically set at 3:1 and LOQ at 10:1 ratios. For enol quantification in pharmaceutical applications, these limits should align with ICH Q3A impurity thresholds, ensuring adequate sensitivity for regulatory compliance and quality control purposes.
Quality Control in Complex Chemical Analysis
Quality control in complex chemical analysis of multi-component systems containing enol tautomers presents unique challenges that require sophisticated analytical protocols and stringent validation procedures. The dynamic equilibrium between keto and enol forms creates inherent variability that must be carefully managed through standardized sampling, storage, and measurement protocols to ensure reproducible and accurate quantification results.
Sample preparation represents a critical control point in enol quantification workflows. Temperature fluctuations during sample handling can significantly shift tautomeric equilibria, necessitating strict thermal management protocols. Standardized procedures must specify exact temperature ranges, equilibration times, and storage conditions to minimize variability. Additionally, pH control becomes paramount as even minor variations can dramatically alter enol-keto ratios, requiring buffered systems and continuous monitoring throughout the analytical process.
Analytical method validation for enol quantification demands comprehensive assessment of precision, accuracy, linearity, and robustness across the expected concentration ranges. Inter-laboratory studies reveal significant variability in enol measurements when standardized protocols are not rigorously followed, emphasizing the need for detailed standard operating procedures. Method validation must account for matrix effects in multi-component systems, where interfering compounds can affect both chromatographic separation and spectroscopic detection.
Reference standard preparation and stability assessment constitute fundamental quality control elements. Pure enol standards often exhibit limited stability, requiring frequent preparation and rigorous characterization. Certified reference materials specifically designed for enol quantification remain limited, necessitating in-house standard preparation with comprehensive purity assessment and stability studies under various storage conditions.
Statistical process control implementation enables real-time monitoring of analytical performance through control charts tracking key parameters such as retention times, peak areas, and calibration curve parameters. Trend analysis helps identify systematic drift before it impacts measurement quality, while control limits establish acceptable performance boundaries for routine operations.
Documentation and traceability systems ensure complete analytical chain custody from sample collection through final reporting. Electronic laboratory information management systems facilitate automated data capture, reduce transcription errors, and maintain comprehensive audit trails essential for regulatory compliance and quality assurance in pharmaceutical and industrial applications.
Sample preparation represents a critical control point in enol quantification workflows. Temperature fluctuations during sample handling can significantly shift tautomeric equilibria, necessitating strict thermal management protocols. Standardized procedures must specify exact temperature ranges, equilibration times, and storage conditions to minimize variability. Additionally, pH control becomes paramount as even minor variations can dramatically alter enol-keto ratios, requiring buffered systems and continuous monitoring throughout the analytical process.
Analytical method validation for enol quantification demands comprehensive assessment of precision, accuracy, linearity, and robustness across the expected concentration ranges. Inter-laboratory studies reveal significant variability in enol measurements when standardized protocols are not rigorously followed, emphasizing the need for detailed standard operating procedures. Method validation must account for matrix effects in multi-component systems, where interfering compounds can affect both chromatographic separation and spectroscopic detection.
Reference standard preparation and stability assessment constitute fundamental quality control elements. Pure enol standards often exhibit limited stability, requiring frequent preparation and rigorous characterization. Certified reference materials specifically designed for enol quantification remain limited, necessitating in-house standard preparation with comprehensive purity assessment and stability studies under various storage conditions.
Statistical process control implementation enables real-time monitoring of analytical performance through control charts tracking key parameters such as retention times, peak areas, and calibration curve parameters. Trend analysis helps identify systematic drift before it impacts measurement quality, while control limits establish acceptable performance boundaries for routine operations.
Documentation and traceability systems ensure complete analytical chain custody from sample collection through final reporting. Electronic laboratory information management systems facilitate automated data capture, reduce transcription errors, and maintain comprehensive audit trails essential for regulatory compliance and quality assurance in pharmaceutical and industrial applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!