How to Design Composite Cathodes Compatible with Sulfide Electrolytes
AUG 20, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Sulfide Electrolyte Cathode Design Background
The development of all-solid-state batteries has gained significant attention in recent years due to their potential to overcome the safety and energy density limitations of conventional lithium-ion batteries. At the heart of this technological advancement lies the challenge of designing composite cathodes compatible with sulfide electrolytes, a crucial component for achieving high-performance solid-state batteries.
Sulfide electrolytes have emerged as promising candidates for solid-state batteries due to their high ionic conductivity and favorable mechanical properties. However, the integration of these electrolytes with conventional cathode materials presents several challenges that need to be addressed to realize the full potential of all-solid-state batteries.
The evolution of cathode design for sulfide electrolyte-based batteries can be traced back to the early 2000s when researchers began exploring the potential of sulfide-based solid electrolytes. Initially, the focus was on adapting existing cathode materials used in liquid electrolyte systems to work with sulfide electrolytes. However, it quickly became apparent that this approach was insufficient due to the unique chemical and electrochemical properties of sulfide electrolytes.
As research progressed, the emphasis shifted towards developing composite cathodes specifically tailored for sulfide electrolytes. These composite cathodes typically consist of active cathode materials, sulfide solid electrolytes, and conductive additives. The goal is to create a cathode structure that maximizes the contact area between the active material and the electrolyte while maintaining good electronic conductivity throughout the electrode.
One of the primary challenges in designing compatible composite cathodes is addressing the chemical instability at the cathode-electrolyte interface. Many conventional cathode materials react unfavorably with sulfide electrolytes, leading to the formation of resistive interfacial layers that impede ion transport and degrade battery performance over time.
To overcome these challenges, researchers have explored various strategies, including the development of protective coatings for cathode particles, the use of buffer layers between the cathode and electrolyte, and the synthesis of novel cathode materials with improved compatibility with sulfide electrolytes.
The current technological landscape is characterized by a diverse range of approaches to composite cathode design, each aiming to optimize the delicate balance between electrochemical performance, interfacial stability, and manufacturability. As research in this field continues to advance, the focus is increasingly on developing scalable and cost-effective solutions that can bridge the gap between laboratory demonstrations and commercial viability.
Sulfide electrolytes have emerged as promising candidates for solid-state batteries due to their high ionic conductivity and favorable mechanical properties. However, the integration of these electrolytes with conventional cathode materials presents several challenges that need to be addressed to realize the full potential of all-solid-state batteries.
The evolution of cathode design for sulfide electrolyte-based batteries can be traced back to the early 2000s when researchers began exploring the potential of sulfide-based solid electrolytes. Initially, the focus was on adapting existing cathode materials used in liquid electrolyte systems to work with sulfide electrolytes. However, it quickly became apparent that this approach was insufficient due to the unique chemical and electrochemical properties of sulfide electrolytes.
As research progressed, the emphasis shifted towards developing composite cathodes specifically tailored for sulfide electrolytes. These composite cathodes typically consist of active cathode materials, sulfide solid electrolytes, and conductive additives. The goal is to create a cathode structure that maximizes the contact area between the active material and the electrolyte while maintaining good electronic conductivity throughout the electrode.
One of the primary challenges in designing compatible composite cathodes is addressing the chemical instability at the cathode-electrolyte interface. Many conventional cathode materials react unfavorably with sulfide electrolytes, leading to the formation of resistive interfacial layers that impede ion transport and degrade battery performance over time.
To overcome these challenges, researchers have explored various strategies, including the development of protective coatings for cathode particles, the use of buffer layers between the cathode and electrolyte, and the synthesis of novel cathode materials with improved compatibility with sulfide electrolytes.
The current technological landscape is characterized by a diverse range of approaches to composite cathode design, each aiming to optimize the delicate balance between electrochemical performance, interfacial stability, and manufacturability. As research in this field continues to advance, the focus is increasingly on developing scalable and cost-effective solutions that can bridge the gap between laboratory demonstrations and commercial viability.
Market Analysis for Solid-State Batteries
The solid-state battery market is experiencing rapid growth and attracting significant attention from both established automotive manufacturers and emerging technology companies. This surge in interest is primarily driven by the potential advantages of solid-state batteries over traditional lithium-ion batteries, including higher energy density, improved safety, and faster charging capabilities.
Market projections for solid-state batteries are highly optimistic, with estimates suggesting a compound annual growth rate (CAGR) of over 30% in the coming years. The automotive sector is expected to be the primary driver of this growth, as major car manufacturers are investing heavily in solid-state battery technology to power their next-generation electric vehicles.
The demand for solid-state batteries is also expanding beyond the automotive industry. Consumer electronics, aerospace, and energy storage sectors are showing increasing interest in this technology due to its potential to offer longer-lasting and more compact power solutions.
However, the market faces several challenges that need to be addressed before widespread adoption can occur. The most significant hurdle is the high cost of production, which currently makes solid-state batteries considerably more expensive than traditional lithium-ion batteries. Additionally, there are technical challenges related to scaling up production and ensuring long-term stability and performance of these batteries in real-world conditions.
Despite these challenges, the market outlook remains positive. Major players in the battery industry are investing heavily in research and development to overcome technical barriers and reduce production costs. Governments worldwide are also supporting the development of solid-state battery technology through funding initiatives and policy support, recognizing its potential to revolutionize energy storage and contribute to sustainability goals.
The market for sulfide-based solid electrolytes, which are crucial for designing composite cathodes compatible with sulfide electrolytes, is a subset of the broader solid-state battery market. These materials are gaining traction due to their high ionic conductivity and potential for improved battery performance. However, the development of composite cathodes that are fully compatible with sulfide electrolytes remains a key technical challenge that researchers and companies are actively working to solve.
As the technology matures and production scales up, analysts expect the cost of solid-state batteries to decrease significantly, potentially reaching price parity with lithium-ion batteries within the next decade. This cost reduction, coupled with the performance advantages of solid-state technology, is likely to drive widespread adoption across various industries, reshaping the global energy storage landscape.
Market projections for solid-state batteries are highly optimistic, with estimates suggesting a compound annual growth rate (CAGR) of over 30% in the coming years. The automotive sector is expected to be the primary driver of this growth, as major car manufacturers are investing heavily in solid-state battery technology to power their next-generation electric vehicles.
The demand for solid-state batteries is also expanding beyond the automotive industry. Consumer electronics, aerospace, and energy storage sectors are showing increasing interest in this technology due to its potential to offer longer-lasting and more compact power solutions.
However, the market faces several challenges that need to be addressed before widespread adoption can occur. The most significant hurdle is the high cost of production, which currently makes solid-state batteries considerably more expensive than traditional lithium-ion batteries. Additionally, there are technical challenges related to scaling up production and ensuring long-term stability and performance of these batteries in real-world conditions.
Despite these challenges, the market outlook remains positive. Major players in the battery industry are investing heavily in research and development to overcome technical barriers and reduce production costs. Governments worldwide are also supporting the development of solid-state battery technology through funding initiatives and policy support, recognizing its potential to revolutionize energy storage and contribute to sustainability goals.
The market for sulfide-based solid electrolytes, which are crucial for designing composite cathodes compatible with sulfide electrolytes, is a subset of the broader solid-state battery market. These materials are gaining traction due to their high ionic conductivity and potential for improved battery performance. However, the development of composite cathodes that are fully compatible with sulfide electrolytes remains a key technical challenge that researchers and companies are actively working to solve.
As the technology matures and production scales up, analysts expect the cost of solid-state batteries to decrease significantly, potentially reaching price parity with lithium-ion batteries within the next decade. This cost reduction, coupled with the performance advantages of solid-state technology, is likely to drive widespread adoption across various industries, reshaping the global energy storage landscape.
Challenges in Sulfide Electrolyte Cathode Interfaces
The interface between sulfide electrolytes and cathode materials presents significant challenges in the development of all-solid-state batteries. One of the primary issues is the high reactivity of sulfide electrolytes with common cathode materials, leading to the formation of interfacial layers that can impede ion transport and increase cell resistance. This reactivity is particularly problematic with high-voltage cathodes, as the sulfide electrolytes tend to decompose at potentials above 2.5 V vs. Li/Li+.
Another major challenge is the mechanical instability at the cathode-electrolyte interface. During cycling, the volume changes in the cathode material can lead to contact loss with the solid electrolyte, resulting in increased interfacial resistance and capacity fade. This issue is exacerbated by the brittle nature of sulfide electrolytes, which are prone to cracking under mechanical stress.
The formation of space-charge layers at the cathode-electrolyte interface also poses a significant challenge. These layers can arise due to the redistribution of mobile ions near the interface, leading to a depletion region that hinders ion transport. This phenomenon is particularly pronounced in sulfide systems due to the high ionic conductivity of the electrolyte and the large difference in chemical potentials between the cathode and electrolyte materials.
Furthermore, the chemical compatibility between sulfide electrolytes and cathode materials is a critical issue. Many high-performance cathode materials, such as layered oxides, react with sulfide electrolytes to form interfacial phases that are often ionically and electronically insulating. These reactions can consume active material, deplete lithium inventory, and create barriers to ion transport, all of which contribute to capacity loss and increased cell impedance.
The challenge of maintaining intimate contact between the cathode and electrolyte is also significant. Unlike liquid electrolytes, which can easily wet the cathode surface, solid sulfide electrolytes require careful engineering to ensure good interfacial contact. This is particularly challenging given the volume changes that occur during cycling and the differences in thermal expansion coefficients between the cathode and electrolyte materials.
Addressing these challenges requires a multifaceted approach, including the development of novel cathode materials with enhanced stability against sulfide electrolytes, the design of protective coatings or buffer layers to mitigate interfacial reactions, and the optimization of cathode microstructures to improve mechanical stability and maintain good contact with the electrolyte. Additionally, advanced characterization techniques are needed to better understand the complex interfacial phenomena occurring at the cathode-electrolyte interface in sulfide-based solid-state batteries.
Another major challenge is the mechanical instability at the cathode-electrolyte interface. During cycling, the volume changes in the cathode material can lead to contact loss with the solid electrolyte, resulting in increased interfacial resistance and capacity fade. This issue is exacerbated by the brittle nature of sulfide electrolytes, which are prone to cracking under mechanical stress.
The formation of space-charge layers at the cathode-electrolyte interface also poses a significant challenge. These layers can arise due to the redistribution of mobile ions near the interface, leading to a depletion region that hinders ion transport. This phenomenon is particularly pronounced in sulfide systems due to the high ionic conductivity of the electrolyte and the large difference in chemical potentials between the cathode and electrolyte materials.
Furthermore, the chemical compatibility between sulfide electrolytes and cathode materials is a critical issue. Many high-performance cathode materials, such as layered oxides, react with sulfide electrolytes to form interfacial phases that are often ionically and electronically insulating. These reactions can consume active material, deplete lithium inventory, and create barriers to ion transport, all of which contribute to capacity loss and increased cell impedance.
The challenge of maintaining intimate contact between the cathode and electrolyte is also significant. Unlike liquid electrolytes, which can easily wet the cathode surface, solid sulfide electrolytes require careful engineering to ensure good interfacial contact. This is particularly challenging given the volume changes that occur during cycling and the differences in thermal expansion coefficients between the cathode and electrolyte materials.
Addressing these challenges requires a multifaceted approach, including the development of novel cathode materials with enhanced stability against sulfide electrolytes, the design of protective coatings or buffer layers to mitigate interfacial reactions, and the optimization of cathode microstructures to improve mechanical stability and maintain good contact with the electrolyte. Additionally, advanced characterization techniques are needed to better understand the complex interfacial phenomena occurring at the cathode-electrolyte interface in sulfide-based solid-state batteries.
Current Composite Cathode Design Strategies
01 Composite cathode materials for improved compatibility
Composite cathode materials are developed to enhance compatibility and performance in batteries. These materials often combine different active components, binders, and conductive additives to achieve optimal electrochemical properties and stability. The composite structure can improve ion diffusion, electron conductivity, and structural integrity during charge-discharge cycles.- Composite cathode materials for improved compatibility: Composite cathode materials are developed to enhance compatibility and performance in battery systems. These materials often combine different active components to achieve synergistic effects, improving energy density, cycling stability, and overall battery performance.
- Binder systems for composite cathodes: Specialized binder systems are utilized to improve the compatibility of composite cathodes. These binders help to maintain the structural integrity of the cathode, enhance adhesion between components, and facilitate better ion transport, leading to improved battery performance and longevity.
- Electrolyte compatibility with composite cathodes: The development of electrolytes that are compatible with composite cathodes is crucial for optimal battery performance. These electrolytes are designed to minimize unwanted side reactions, enhance ion transport, and maintain stability at the cathode-electrolyte interface, contributing to improved battery efficiency and lifespan.
- Surface modification of composite cathodes: Surface modification techniques are employed to enhance the compatibility of composite cathodes with other battery components. These modifications can improve the cathode's stability, reduce unwanted reactions, and optimize the interface between the cathode and electrolyte, leading to better overall battery performance.
- Nanostructured composite cathodes for enhanced compatibility: Nanostructured composite cathodes are developed to improve compatibility and performance in battery systems. These cathodes leverage nanoscale engineering to enhance surface area, facilitate better ion transport, and improve overall electrochemical properties, resulting in batteries with higher energy density and improved cycling stability.
02 Binder selection for composite cathodes
The choice of binder plays a crucial role in the compatibility of composite cathodes. Appropriate binders ensure good adhesion between active materials and current collectors, maintain electrode integrity, and contribute to the overall electrochemical performance. Research focuses on developing binders that are chemically stable, flexible, and compatible with various cathode materials.Expand Specific Solutions03 Electrolyte compatibility with composite cathodes
Ensuring compatibility between the electrolyte and composite cathodes is essential for long-term battery performance. This involves selecting electrolyte compositions that are stable against the cathode materials, prevent unwanted side reactions, and maintain good ionic conductivity. Additives in the electrolyte can also be used to form protective layers on the cathode surface, enhancing compatibility and cycle life.Expand Specific Solutions04 Surface modification of composite cathodes
Surface modification techniques are employed to improve the compatibility of composite cathodes with electrolytes and other battery components. These modifications can include coatings, doping, or surface treatments that enhance the cathode's chemical stability, reduce unwanted reactions, and improve the interface between the cathode and electrolyte. Such modifications can lead to better cycling performance and increased battery lifespan.Expand Specific Solutions05 Nanostructured composite cathodes for enhanced compatibility
Nanostructured composite cathodes are designed to improve compatibility and performance in batteries. These cathodes often incorporate nanomaterials or nanostructured components that enhance ion diffusion, electron transport, and structural stability. The increased surface area and unique properties of nanomaterials can lead to better electrochemical performance and improved compatibility with other battery components.Expand Specific Solutions
Key Players in Solid-State Battery Industry
The development of composite cathodes compatible with sulfide electrolytes is in a nascent stage, with significant potential for growth in the solid-state battery market. The technology's maturity is still evolving, as evidenced by ongoing research efforts from major players like LG Energy Solution, Sion Power, and CATL. These companies are investing heavily in R&D to overcome challenges related to interfacial stability and ion conductivity. The market size is expected to expand rapidly, driven by the automotive industry's push towards higher energy density batteries. While established firms lead the charge, emerging players and research institutions are also making notable contributions, indicating a competitive and dynamic landscape in this critical area of battery technology.
LG Chem Ltd.
Technical Solution: LG Chem has developed a composite cathode compatible with sulfide electrolytes by utilizing a protective coating strategy. They employ a thin layer of lithium niobium oxide (LiNbO3) as a coating material on the cathode surface. This coating acts as a buffer layer between the cathode and the sulfide electrolyte, preventing direct contact and mitigating undesirable side reactions[1]. The company has also explored the use of lithium phosphorus oxynitride (LiPON) as an alternative coating material, which has shown promising results in improving the interface stability[2]. Additionally, LG Chem has implemented a gradient concentration approach in their composite cathodes, where the concentration of active materials varies from the surface to the bulk, further enhancing the compatibility with sulfide electrolytes[3].
Strengths: Improved interface stability, reduced side reactions, and enhanced cycling performance. Weaknesses: Potential increase in production costs due to additional coating processes and complexity in maintaining uniform coating thickness.
LG Energy Solution Ltd.
Technical Solution: LG Energy Solution has developed a novel approach to designing composite cathodes compatible with sulfide electrolytes by focusing on the optimization of particle morphology and surface chemistry. They have implemented a hierarchical structure in their cathode materials, consisting of primary nanoparticles aggregated into secondary microparticles[4]. This structure allows for better ion diffusion and electron transport while maintaining good contact with the sulfide electrolyte. The company has also incorporated dopants such as aluminum and magnesium into the cathode material to stabilize the crystal structure and improve the interfacial compatibility[5]. Furthermore, LG Energy Solution has developed a proprietary surface modification technique that involves the in-situ formation of a lithium-rich layer on the cathode surface, which acts as a protective barrier against sulfide electrolyte degradation[6].
Strengths: Enhanced ion and electron transport, improved structural stability, and better interfacial compatibility. Weaknesses: Potential challenges in scaling up the production of hierarchical structures and maintaining consistent doping levels.
Innovations in Cathode-Electrolyte Interfaces
Solid-state cathode composition
PatentWO2025111401A1
Innovation
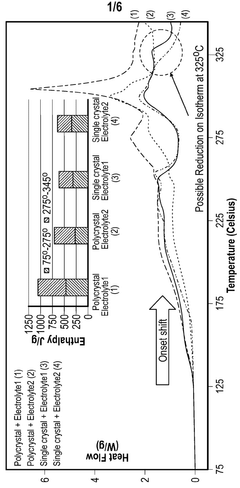
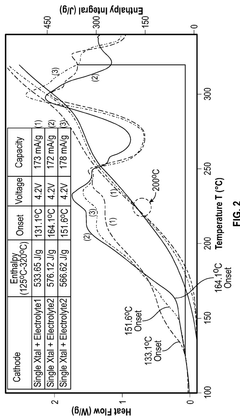
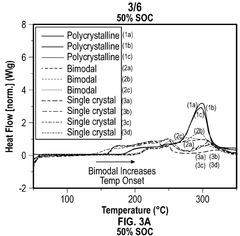
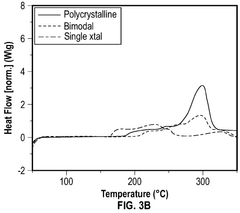
- A cathode composite composition comprising a blend of single-crystal and polycrystalline cathode active materials combined with solid sulfide electrolytes, such as Li7-yPS6-yXy, where 0≤y≤2 and X is a halogen, to reduce exothermic reactions and enhance safety.
Composite cathodes for solid-state lithium sulfur batteries and methods of manufacturing thereof
PatentActiveUS11682792B2
Innovation
- A composite cathode is formed using carbonized cotton fiber as a conductive skeleton with a sulfide electrolyte, such as Li7P3S11, coated onto the carbon fiber via a liquid-phase process, allowing for close contact with elemental sulfur without requiring high external pressures, enabling efficient electron and ion conduction.
Safety Considerations for Sulfide-Based Batteries
Safety considerations are paramount in the development and implementation of sulfide-based batteries, particularly those utilizing composite cathodes compatible with sulfide electrolytes. The reactive nature of sulfide materials presents unique challenges that must be addressed to ensure the safe operation and handling of these energy storage systems.
One of the primary safety concerns is the potential for hydrogen sulfide (H2S) gas formation. Sulfide electrolytes can react with moisture in the air, producing this toxic and flammable gas. To mitigate this risk, stringent moisture control measures must be implemented throughout the manufacturing, assembly, and usage processes. This includes the use of dry rooms, inert atmospheres, and hermetically sealed battery enclosures.
The thermal stability of sulfide-based batteries is another critical safety aspect. These batteries may be susceptible to thermal runaway under certain conditions, such as overcharging or physical damage. To address this, advanced thermal management systems and robust battery management systems (BMS) are essential. These systems should continuously monitor temperature, voltage, and current, implementing safeguards to prevent overheating and potential thermal events.
Mechanical integrity is crucial for sulfide-based batteries, as physical damage can lead to internal short circuits and safety hazards. The design of composite cathodes and overall battery structure must account for mechanical stress and potential impacts. This may involve the use of reinforced separators, protective coatings, and shock-absorbing materials to enhance the battery's resilience to physical stress.
The reactivity of sulfide electrolytes with conventional current collectors, particularly those made of aluminum, poses another safety challenge. This can lead to corrosion and the formation of an insulating layer, potentially causing performance degradation and safety issues. To address this, research into alternative current collector materials or protective coatings is necessary to ensure long-term stability and safety.
Environmental considerations are also vital in the context of sulfide-based batteries. Proper disposal and recycling protocols must be established to prevent environmental contamination and potential health hazards. This includes developing safe methods for disassembling and recycling spent batteries, as well as implementing strict guidelines for handling and transporting these materials.
In conclusion, while sulfide-based batteries offer promising advantages in terms of energy density and performance, their widespread adoption hinges on addressing these critical safety considerations. Ongoing research and development efforts must focus on enhancing the inherent safety of these systems, developing robust protective measures, and establishing comprehensive safety protocols for all stages of the battery lifecycle.
One of the primary safety concerns is the potential for hydrogen sulfide (H2S) gas formation. Sulfide electrolytes can react with moisture in the air, producing this toxic and flammable gas. To mitigate this risk, stringent moisture control measures must be implemented throughout the manufacturing, assembly, and usage processes. This includes the use of dry rooms, inert atmospheres, and hermetically sealed battery enclosures.
The thermal stability of sulfide-based batteries is another critical safety aspect. These batteries may be susceptible to thermal runaway under certain conditions, such as overcharging or physical damage. To address this, advanced thermal management systems and robust battery management systems (BMS) are essential. These systems should continuously monitor temperature, voltage, and current, implementing safeguards to prevent overheating and potential thermal events.
Mechanical integrity is crucial for sulfide-based batteries, as physical damage can lead to internal short circuits and safety hazards. The design of composite cathodes and overall battery structure must account for mechanical stress and potential impacts. This may involve the use of reinforced separators, protective coatings, and shock-absorbing materials to enhance the battery's resilience to physical stress.
The reactivity of sulfide electrolytes with conventional current collectors, particularly those made of aluminum, poses another safety challenge. This can lead to corrosion and the formation of an insulating layer, potentially causing performance degradation and safety issues. To address this, research into alternative current collector materials or protective coatings is necessary to ensure long-term stability and safety.
Environmental considerations are also vital in the context of sulfide-based batteries. Proper disposal and recycling protocols must be established to prevent environmental contamination and potential health hazards. This includes developing safe methods for disassembling and recycling spent batteries, as well as implementing strict guidelines for handling and transporting these materials.
In conclusion, while sulfide-based batteries offer promising advantages in terms of energy density and performance, their widespread adoption hinges on addressing these critical safety considerations. Ongoing research and development efforts must focus on enhancing the inherent safety of these systems, developing robust protective measures, and establishing comprehensive safety protocols for all stages of the battery lifecycle.
Environmental Impact of Composite Cathodes
The environmental impact of composite cathodes compatible with sulfide electrolytes is a crucial consideration in the development of next-generation solid-state batteries. These cathodes, while offering improved performance and safety, also present unique environmental challenges throughout their lifecycle.
During the production phase, the synthesis of composite cathodes often involves energy-intensive processes and the use of potentially hazardous materials. The incorporation of sulfide electrolytes requires careful handling due to their reactivity with moisture, which can lead to the release of toxic hydrogen sulfide gas. This necessitates stringent safety measures and specialized manufacturing environments, potentially increasing the overall environmental footprint of production.
The use of rare earth elements and transition metals in many composite cathodes raises concerns about resource depletion and the environmental impact of mining activities. The extraction and processing of these materials can lead to habitat destruction, water pollution, and greenhouse gas emissions. However, the improved energy density and longer lifespan of batteries using these cathodes may offset some of these impacts by reducing the overall number of batteries required over time.
In the operational phase, the compatibility of composite cathodes with sulfide electrolytes contributes to enhanced battery performance and safety. This can lead to reduced energy consumption and lower risks of thermal runaway events, potentially decreasing the environmental impact associated with battery usage and accidents.
End-of-life considerations for these advanced cathodes present both challenges and opportunities. The complex composition of composite cathodes can make recycling more difficult, potentially leading to increased e-waste if not properly managed. However, the valuable materials contained within these cathodes provide a strong economic incentive for developing efficient recycling processes. Advanced recycling technologies, such as hydrometallurgical and pyrometallurgical methods, are being developed to recover and reuse the constituent materials, potentially creating a more circular economy for battery components.
The environmental impact of composite cathodes also extends to their potential role in enabling wider adoption of renewable energy sources. By improving the performance and reliability of energy storage systems, these cathodes could facilitate greater integration of intermittent renewable energy into the grid, contributing to a reduction in fossil fuel dependence and associated carbon emissions.
During the production phase, the synthesis of composite cathodes often involves energy-intensive processes and the use of potentially hazardous materials. The incorporation of sulfide electrolytes requires careful handling due to their reactivity with moisture, which can lead to the release of toxic hydrogen sulfide gas. This necessitates stringent safety measures and specialized manufacturing environments, potentially increasing the overall environmental footprint of production.
The use of rare earth elements and transition metals in many composite cathodes raises concerns about resource depletion and the environmental impact of mining activities. The extraction and processing of these materials can lead to habitat destruction, water pollution, and greenhouse gas emissions. However, the improved energy density and longer lifespan of batteries using these cathodes may offset some of these impacts by reducing the overall number of batteries required over time.
In the operational phase, the compatibility of composite cathodes with sulfide electrolytes contributes to enhanced battery performance and safety. This can lead to reduced energy consumption and lower risks of thermal runaway events, potentially decreasing the environmental impact associated with battery usage and accidents.
End-of-life considerations for these advanced cathodes present both challenges and opportunities. The complex composition of composite cathodes can make recycling more difficult, potentially leading to increased e-waste if not properly managed. However, the valuable materials contained within these cathodes provide a strong economic incentive for developing efficient recycling processes. Advanced recycling technologies, such as hydrometallurgical and pyrometallurgical methods, are being developed to recover and reuse the constituent materials, potentially creating a more circular economy for battery components.
The environmental impact of composite cathodes also extends to their potential role in enabling wider adoption of renewable energy sources. By improving the performance and reliability of energy storage systems, these cathodes could facilitate greater integration of intermittent renewable energy into the grid, contributing to a reduction in fossil fuel dependence and associated carbon emissions.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!