How to Mitigate Dendrite Growth in Sulfide Electrolyte Solid-State Cells
AUG 20, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Dendrite Growth Challenges in Solid-State Batteries
Dendrite growth poses a significant challenge in the development and commercialization of solid-state batteries, particularly those utilizing sulfide electrolytes. This issue stems from the uneven deposition of lithium metal during charging cycles, leading to the formation of needle-like structures known as dendrites. These dendrites can penetrate the solid electrolyte, causing short circuits and potentially catastrophic battery failure.
The primary factors contributing to dendrite growth in sulfide electrolyte solid-state cells include inhomogeneous current distribution, mechanical stress at the electrode-electrolyte interface, and the inherent properties of the sulfide electrolyte itself. The high ionic conductivity of sulfide electrolytes, while beneficial for overall battery performance, can paradoxically exacerbate dendrite formation due to rapid lithium ion transport.
Researchers have identified several key mechanisms that drive dendrite growth in these systems. These include the formation of space charges at the electrode-electrolyte interface, localized current hotspots, and the presence of defects or impurities in the electrolyte material. Additionally, the mechanical properties of the sulfide electrolyte, such as its relatively low shear modulus, can facilitate dendrite propagation.
Various strategies have been proposed to mitigate dendrite growth in sulfide electrolyte solid-state cells. One approach involves engineering the electrode-electrolyte interface to promote more uniform lithium deposition. This can be achieved through the use of interlayers or by modifying the surface chemistry of the electrodes. Another strategy focuses on enhancing the mechanical properties of the electrolyte to increase its resistance to dendrite penetration.
The development of novel electrolyte compositions and architectures also shows promise in addressing this challenge. For instance, the incorporation of nanostructured materials or the use of composite electrolytes combining sulfide and oxide components has demonstrated potential in suppressing dendrite growth. Furthermore, advanced characterization techniques, such as in situ electron microscopy and synchrotron-based imaging, are providing valuable insights into the mechanisms of dendrite formation and growth, enabling more targeted mitigation strategies.
Despite these efforts, dendrite growth remains a persistent obstacle in the path towards commercially viable sulfide electrolyte solid-state batteries. Overcoming this challenge requires a multidisciplinary approach, combining materials science, electrochemistry, and advanced manufacturing techniques. As research in this field progresses, it is likely that a combination of strategies will be necessary to effectively mitigate dendrite growth and unlock the full potential of solid-state battery technology.
The primary factors contributing to dendrite growth in sulfide electrolyte solid-state cells include inhomogeneous current distribution, mechanical stress at the electrode-electrolyte interface, and the inherent properties of the sulfide electrolyte itself. The high ionic conductivity of sulfide electrolytes, while beneficial for overall battery performance, can paradoxically exacerbate dendrite formation due to rapid lithium ion transport.
Researchers have identified several key mechanisms that drive dendrite growth in these systems. These include the formation of space charges at the electrode-electrolyte interface, localized current hotspots, and the presence of defects or impurities in the electrolyte material. Additionally, the mechanical properties of the sulfide electrolyte, such as its relatively low shear modulus, can facilitate dendrite propagation.
Various strategies have been proposed to mitigate dendrite growth in sulfide electrolyte solid-state cells. One approach involves engineering the electrode-electrolyte interface to promote more uniform lithium deposition. This can be achieved through the use of interlayers or by modifying the surface chemistry of the electrodes. Another strategy focuses on enhancing the mechanical properties of the electrolyte to increase its resistance to dendrite penetration.
The development of novel electrolyte compositions and architectures also shows promise in addressing this challenge. For instance, the incorporation of nanostructured materials or the use of composite electrolytes combining sulfide and oxide components has demonstrated potential in suppressing dendrite growth. Furthermore, advanced characterization techniques, such as in situ electron microscopy and synchrotron-based imaging, are providing valuable insights into the mechanisms of dendrite formation and growth, enabling more targeted mitigation strategies.
Despite these efforts, dendrite growth remains a persistent obstacle in the path towards commercially viable sulfide electrolyte solid-state batteries. Overcoming this challenge requires a multidisciplinary approach, combining materials science, electrochemistry, and advanced manufacturing techniques. As research in this field progresses, it is likely that a combination of strategies will be necessary to effectively mitigate dendrite growth and unlock the full potential of solid-state battery technology.
Market Demand for Advanced Energy Storage Solutions
The demand for advanced energy storage solutions has been growing exponentially in recent years, driven by the increasing adoption of electric vehicles, renewable energy integration, and the need for grid stabilization. Solid-state batteries, particularly those utilizing sulfide electrolytes, have emerged as a promising technology to address the limitations of conventional lithium-ion batteries. These advanced batteries offer higher energy density, improved safety, and longer lifespan, making them attractive for various applications.
The automotive industry is a key driver of market demand for solid-state batteries. As electric vehicle adoption accelerates globally, manufacturers are seeking battery technologies that can provide longer driving ranges, faster charging times, and enhanced safety. Solid-state batteries with sulfide electrolytes have the potential to meet these requirements, potentially revolutionizing the electric vehicle market.
In the renewable energy sector, the intermittent nature of solar and wind power generation necessitates efficient and large-scale energy storage solutions. Solid-state batteries could play a crucial role in grid-scale energy storage, enabling better integration of renewable energy sources and improving grid stability. The ability to store excess energy during peak production periods and release it during high demand times is essential for the widespread adoption of renewable energy.
Consumer electronics is another significant market segment driving demand for advanced energy storage solutions. As devices become more powerful and energy-intensive, there is a growing need for batteries that can provide longer operating times and faster charging capabilities. Solid-state batteries with improved energy density and safety features could meet these requirements, potentially leading to a new generation of consumer electronics with enhanced performance.
The aerospace and defense sectors also present opportunities for solid-state battery technology. These industries require high-performance, lightweight, and safe energy storage solutions for various applications, including unmanned aerial vehicles, satellites, and portable military equipment. The improved energy density and safety characteristics of solid-state batteries make them particularly attractive for these demanding applications.
However, the widespread adoption of solid-state batteries, including those with sulfide electrolytes, faces challenges related to dendrite growth. Addressing this issue is crucial for realizing the full potential of this technology and meeting the growing market demand for advanced energy storage solutions. As research progresses and solutions to mitigate dendrite growth are developed, the market for solid-state batteries is expected to expand rapidly, potentially reshaping the energy storage landscape across multiple industries.
The automotive industry is a key driver of market demand for solid-state batteries. As electric vehicle adoption accelerates globally, manufacturers are seeking battery technologies that can provide longer driving ranges, faster charging times, and enhanced safety. Solid-state batteries with sulfide electrolytes have the potential to meet these requirements, potentially revolutionizing the electric vehicle market.
In the renewable energy sector, the intermittent nature of solar and wind power generation necessitates efficient and large-scale energy storage solutions. Solid-state batteries could play a crucial role in grid-scale energy storage, enabling better integration of renewable energy sources and improving grid stability. The ability to store excess energy during peak production periods and release it during high demand times is essential for the widespread adoption of renewable energy.
Consumer electronics is another significant market segment driving demand for advanced energy storage solutions. As devices become more powerful and energy-intensive, there is a growing need for batteries that can provide longer operating times and faster charging capabilities. Solid-state batteries with improved energy density and safety features could meet these requirements, potentially leading to a new generation of consumer electronics with enhanced performance.
The aerospace and defense sectors also present opportunities for solid-state battery technology. These industries require high-performance, lightweight, and safe energy storage solutions for various applications, including unmanned aerial vehicles, satellites, and portable military equipment. The improved energy density and safety characteristics of solid-state batteries make them particularly attractive for these demanding applications.
However, the widespread adoption of solid-state batteries, including those with sulfide electrolytes, faces challenges related to dendrite growth. Addressing this issue is crucial for realizing the full potential of this technology and meeting the growing market demand for advanced energy storage solutions. As research progresses and solutions to mitigate dendrite growth are developed, the market for solid-state batteries is expected to expand rapidly, potentially reshaping the energy storage landscape across multiple industries.
Current State of Sulfide Electrolyte Technology
Sulfide electrolytes have emerged as promising candidates for solid-state batteries due to their high ionic conductivity and favorable mechanical properties. The current state of sulfide electrolyte technology reflects significant advancements in material synthesis, composition optimization, and interface engineering. Researchers have successfully developed various sulfide-based solid electrolytes, including Li10GeP2S12 (LGPS), Li6PS5Cl, and Li3PS4, which exhibit ionic conductivities comparable to or even surpassing those of liquid electrolytes.
Recent progress in sulfide electrolyte technology has focused on addressing key challenges such as chemical and electrochemical stability, interfacial resistance, and mechanical properties. Advanced synthesis methods, including solution-based processes and mechanochemical techniques, have been developed to produce high-quality sulfide electrolytes with controlled composition and microstructure. These methods have enabled the production of sulfide electrolytes with improved purity, reduced impurities, and enhanced ionic conductivity.
Efforts to enhance the chemical stability of sulfide electrolytes have led to the development of composite electrolytes and protective coatings. Researchers have explored the incorporation of oxide additives, such as Li3BO3 or Li4SiO4, to improve the chemical stability and mechanical properties of sulfide electrolytes. Additionally, the use of protective coatings, such as LiNbO3 or Li3PO4, has shown promise in mitigating undesirable reactions at the electrode-electrolyte interface.
Interfacial engineering has been a key focus area in sulfide electrolyte technology. Researchers have developed various strategies to reduce interfacial resistance and improve the compatibility between sulfide electrolytes and electrode materials. These approaches include the use of buffer layers, surface modifications, and gradient electrolyte compositions to facilitate smooth ion transport across interfaces and minimize side reactions.
The mechanical properties of sulfide electrolytes have also been a subject of intensive research. While sulfide electrolytes generally exhibit higher ductility compared to oxide-based solid electrolytes, efforts have been made to further improve their mechanical stability and reduce the formation of voids or cracks during cycling. Strategies such as particle size optimization, pressure application during cell assembly, and the incorporation of reinforcing agents have been explored to enhance the mechanical robustness of sulfide electrolyte-based cells.
Despite these advancements, challenges remain in the practical implementation of sulfide electrolytes in commercial solid-state batteries. Issues such as air sensitivity, scalability of production processes, and long-term cycling stability continue to be areas of active research and development. Ongoing efforts are focused on addressing these challenges through innovative material design, advanced manufacturing techniques, and improved cell architectures.
Recent progress in sulfide electrolyte technology has focused on addressing key challenges such as chemical and electrochemical stability, interfacial resistance, and mechanical properties. Advanced synthesis methods, including solution-based processes and mechanochemical techniques, have been developed to produce high-quality sulfide electrolytes with controlled composition and microstructure. These methods have enabled the production of sulfide electrolytes with improved purity, reduced impurities, and enhanced ionic conductivity.
Efforts to enhance the chemical stability of sulfide electrolytes have led to the development of composite electrolytes and protective coatings. Researchers have explored the incorporation of oxide additives, such as Li3BO3 or Li4SiO4, to improve the chemical stability and mechanical properties of sulfide electrolytes. Additionally, the use of protective coatings, such as LiNbO3 or Li3PO4, has shown promise in mitigating undesirable reactions at the electrode-electrolyte interface.
Interfacial engineering has been a key focus area in sulfide electrolyte technology. Researchers have developed various strategies to reduce interfacial resistance and improve the compatibility between sulfide electrolytes and electrode materials. These approaches include the use of buffer layers, surface modifications, and gradient electrolyte compositions to facilitate smooth ion transport across interfaces and minimize side reactions.
The mechanical properties of sulfide electrolytes have also been a subject of intensive research. While sulfide electrolytes generally exhibit higher ductility compared to oxide-based solid electrolytes, efforts have been made to further improve their mechanical stability and reduce the formation of voids or cracks during cycling. Strategies such as particle size optimization, pressure application during cell assembly, and the incorporation of reinforcing agents have been explored to enhance the mechanical robustness of sulfide electrolyte-based cells.
Despite these advancements, challenges remain in the practical implementation of sulfide electrolytes in commercial solid-state batteries. Issues such as air sensitivity, scalability of production processes, and long-term cycling stability continue to be areas of active research and development. Ongoing efforts are focused on addressing these challenges through innovative material design, advanced manufacturing techniques, and improved cell architectures.
Existing Dendrite Mitigation Strategies
01 Composition of sulfide solid electrolytes
Sulfide solid electrolytes are being developed with specific compositions to improve ionic conductivity and suppress dendrite growth. These electrolytes often include combinations of lithium, sulfur, and other elements like phosphorus or germanium. The composition is crucial in determining the electrolyte's performance and its ability to prevent dendrite formation in solid-state cells.- Composition of sulfide solid electrolytes: Sulfide solid electrolytes are being developed with specific compositions to enhance ionic conductivity and suppress dendrite growth. These electrolytes often include combinations of lithium, sulfur, and other elements like phosphorus or germanium. The composition is crucial in determining the electrolyte's performance and its ability to prevent dendrite formation in solid-state cells.
- Interface engineering for dendrite suppression: Researchers are focusing on engineering the interface between the sulfide electrolyte and electrodes to mitigate dendrite growth. This includes developing protective coatings, buffer layers, or modifying the surface properties of the electrolyte. These interface modifications aim to improve the stability of the solid-state cell and prevent lithium dendrite penetration.
- Structural design of solid-state cells: The structural design of solid-state cells plays a crucial role in preventing dendrite growth. This includes optimizing the thickness and uniformity of the electrolyte layer, as well as developing novel cell architectures that can mechanically suppress dendrite formation. Some designs incorporate pressure application or specific electrode configurations to enhance dendrite resistance.
- Additives and dopants for electrolyte enhancement: Various additives and dopants are being explored to enhance the properties of sulfide electrolytes. These additives can improve ionic conductivity, mechanical strength, or chemical stability of the electrolyte. Some additives are specifically designed to create a more uniform lithium deposition, thereby reducing the likelihood of dendrite formation.
- In-situ monitoring and control of dendrite growth: Advanced techniques for in-situ monitoring of dendrite growth in sulfide electrolyte solid-state cells are being developed. These methods allow for real-time observation and control of dendrite formation, enabling researchers to better understand the mechanisms of growth and develop more effective prevention strategies. Some approaches involve integrating sensors or using advanced imaging techniques to detect early signs of dendrite formation.
02 Interface engineering for dendrite suppression
Researchers are focusing on engineering the interface between the sulfide electrolyte and electrodes to prevent dendrite growth. This includes developing protective coatings, buffer layers, or modifying the surface properties of the electrolyte. These interface modifications aim to improve the stability of the solid-state cell and reduce the likelihood of dendrite formation.Expand Specific Solutions03 Additives and dopants for enhanced performance
Various additives and dopants are being incorporated into sulfide electrolytes to enhance their performance and suppress dendrite growth. These additives can improve the mechanical properties, ionic conductivity, or chemical stability of the electrolyte. Some additives also help in forming a stable solid electrolyte interphase (SEI) layer, which is crucial for preventing dendrite formation.Expand Specific Solutions04 Structural design of solid-state cells
The structural design of solid-state cells using sulfide electrolytes is being optimized to minimize dendrite growth. This includes innovations in electrode architecture, electrolyte layer thickness, and overall cell configuration. Researchers are exploring various cell designs that can effectively distribute current and stress, reducing the likelihood of dendrite nucleation and growth.Expand Specific Solutions05 Advanced characterization and modeling techniques
Advanced characterization and modeling techniques are being employed to understand and predict dendrite growth in sulfide electrolyte solid-state cells. These include in-situ and operando imaging methods, computational simulations, and machine learning approaches. These techniques help in identifying the mechanisms of dendrite formation and in developing strategies to mitigate their growth.Expand Specific Solutions
Key Players in Solid-State Battery Development
The competition to mitigate dendrite growth in sulfide electrolyte solid-state cells is intensifying as the technology approaches commercialization. The market is in its early growth stage, with significant potential for expansion due to the increasing demand for safer and higher-energy-density batteries. Major players like LG Chem, Samsung SDI, and Toyota are investing heavily in research and development, while startups such as SES Holdings and SVOLT Energy are also making strides. The technology's maturity varies among companies, with some, like FUJIFILM and Panasonic, focusing on material innovations, while others, such as Nissan and Hyundai, are integrating these advancements into their electric vehicle strategies. Overall, the field remains highly competitive, with no clear market leader yet established.
LG Chem Ltd.
Technical Solution: LG Chem has developed a novel approach to mitigate dendrite growth in sulfide electrolyte solid-state cells by implementing a multi-layered electrolyte structure. This design incorporates a thin, high-ionic conductivity layer adjacent to the lithium metal anode, followed by a thicker, more stable layer. The high-conductivity layer promotes uniform lithium deposition, while the stable layer acts as a physical barrier against dendrite propagation. Additionally, LG Chem has introduced nano-engineered interfaces between the electrolyte layers, which further impede dendrite growth by creating tortuous paths for lithium ion transport[1][3]. The company has also developed proprietary sulfide-based solid electrolytes with optimized compositions to enhance mechanical strength and electrochemical stability, crucial for preventing dendrite formation and growth[2].
Strengths: Advanced multi-layer design enhances dendrite resistance; proprietary sulfide electrolytes offer improved mechanical and electrochemical properties. Weaknesses: Potential challenges in large-scale manufacturing of complex multi-layer structures; possible increased production costs.
Samsung SDI Co., Ltd.
Technical Solution: Samsung SDI has focused on developing a composite sulfide electrolyte system to address dendrite growth in solid-state cells. Their approach involves incorporating nano-sized ceramic particles into the sulfide electrolyte matrix, creating a hybrid structure that combines the high ionic conductivity of sulfides with the mechanical strength of ceramics. This composite design helps to suppress dendrite formation by providing a more uniform lithium ion distribution and increasing the overall mechanical robustness of the electrolyte[4]. Samsung SDI has also implemented advanced surface modification techniques for both the electrolyte and electrode interfaces, using thin coatings of lithium-conducting materials to promote stable and uniform lithium deposition[5]. Furthermore, the company has developed a gradient electrolyte structure, where the composition and properties of the electrolyte vary across its thickness, providing enhanced resistance to dendrite penetration[6].
Strengths: Innovative composite electrolyte design combines high conductivity with mechanical strength; advanced interface engineering improves lithium deposition. Weaknesses: Potential challenges in achieving uniform dispersion of nano-particles in large-scale production; possible increased material costs for composite electrolytes.
Core Innovations in Sulfide Electrolyte Design
Method for charging secondary battery
PatentPendingEP4459738A1
Innovation
- A multi-stage charging method is employed, where the battery is charged at a first current density to deposit metallic lithium on the solid electrolyte layer, and then at a higher current density to increase the deposited Li layer thickness, with the first charging step including pausing or discharging to prevent SOC from exceeding 4.5%, thereby controlling dendrite growth.
Solid electrolyte film, preparation method and use thereof, and solid state battery
PatentPendingUS20250219132A1
Innovation
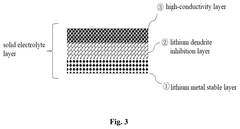
- A solid electrolyte film composed of a lithium metal stable layer, lithium dendrite inhibition layer, and high-conductivity layer, all made of sulfide solid electrolytes, with specific coatings and crystallinity indices to enhance stability and conductivity.
Safety Regulations for Solid-State Batteries
The development of solid-state batteries has brought about new challenges in terms of safety regulations. As these batteries utilize different materials and structures compared to traditional lithium-ion batteries, existing safety standards need to be adapted and new ones established to ensure their safe operation and widespread adoption.
One of the primary safety concerns for solid-state batteries is the potential for dendrite growth, particularly in sulfide electrolyte systems. Regulatory bodies are focusing on developing specific guidelines to address this issue. These regulations aim to establish testing protocols that can accurately assess the likelihood of dendrite formation and growth under various operating conditions.
Safety standards for solid-state batteries are also addressing thermal runaway risks. While solid-state batteries generally have a lower risk of thermal runaway compared to liquid electrolyte batteries, the potential still exists. Regulations are being developed to define acceptable temperature ranges during operation and charging, as well as requirements for thermal management systems.
Mechanical integrity is another crucial aspect covered by safety regulations. Solid-state batteries must withstand mechanical stresses without compromising their performance or safety. Standards are being established to define testing procedures for evaluating the mechanical robustness of solid-state cells, including vibration, shock, and crush tests.
Electrical safety is also a key focus of regulatory efforts. Guidelines are being developed to address issues such as short-circuit prevention, overcharge protection, and safe voltage ranges for solid-state batteries. These regulations aim to ensure that the unique electrical characteristics of solid-state systems are properly managed to prevent safety hazards.
Manufacturing processes for solid-state batteries are subject to increasing scrutiny from a safety perspective. Regulations are being formulated to establish quality control measures and manufacturing standards that minimize defects and ensure consistent production of safe, high-quality cells.
As solid-state battery technology continues to evolve, safety regulations are being designed with flexibility in mind. Regulatory bodies are working to create frameworks that can adapt to new materials, designs, and manufacturing techniques while maintaining rigorous safety standards. This approach aims to support innovation while ensuring that safety remains a top priority in the development and commercialization of solid-state battery technology.
One of the primary safety concerns for solid-state batteries is the potential for dendrite growth, particularly in sulfide electrolyte systems. Regulatory bodies are focusing on developing specific guidelines to address this issue. These regulations aim to establish testing protocols that can accurately assess the likelihood of dendrite formation and growth under various operating conditions.
Safety standards for solid-state batteries are also addressing thermal runaway risks. While solid-state batteries generally have a lower risk of thermal runaway compared to liquid electrolyte batteries, the potential still exists. Regulations are being developed to define acceptable temperature ranges during operation and charging, as well as requirements for thermal management systems.
Mechanical integrity is another crucial aspect covered by safety regulations. Solid-state batteries must withstand mechanical stresses without compromising their performance or safety. Standards are being established to define testing procedures for evaluating the mechanical robustness of solid-state cells, including vibration, shock, and crush tests.
Electrical safety is also a key focus of regulatory efforts. Guidelines are being developed to address issues such as short-circuit prevention, overcharge protection, and safe voltage ranges for solid-state batteries. These regulations aim to ensure that the unique electrical characteristics of solid-state systems are properly managed to prevent safety hazards.
Manufacturing processes for solid-state batteries are subject to increasing scrutiny from a safety perspective. Regulations are being formulated to establish quality control measures and manufacturing standards that minimize defects and ensure consistent production of safe, high-quality cells.
As solid-state battery technology continues to evolve, safety regulations are being designed with flexibility in mind. Regulatory bodies are working to create frameworks that can adapt to new materials, designs, and manufacturing techniques while maintaining rigorous safety standards. This approach aims to support innovation while ensuring that safety remains a top priority in the development and commercialization of solid-state battery technology.
Environmental Impact of Sulfide-Based Batteries
The environmental impact of sulfide-based batteries is a crucial consideration in the development and adoption of solid-state battery technology. These batteries, which utilize sulfide electrolytes, offer promising advantages in terms of energy density and safety. However, their potential environmental implications must be carefully evaluated throughout their lifecycle.
During the production phase, the synthesis of sulfide-based electrolytes often involves energy-intensive processes and the use of potentially hazardous materials. The extraction and processing of raw materials, such as lithium and sulfur, can have significant environmental consequences, including habitat disruption and water pollution. Additionally, the manufacturing of sulfide electrolytes may generate toxic byproducts that require proper handling and disposal.
In the operational phase, sulfide-based batteries demonstrate improved safety characteristics compared to conventional lithium-ion batteries, reducing the risk of thermal runaway and fire incidents. This enhanced safety profile can potentially decrease the environmental impact associated with battery-related accidents and fires. Furthermore, the higher energy density of sulfide-based batteries may lead to reduced material consumption and improved overall efficiency in energy storage applications.
End-of-life considerations for sulfide-based batteries present both challenges and opportunities. The recycling of these batteries is essential to recover valuable materials and minimize environmental impact. However, the complex composition of sulfide electrolytes may require the development of specialized recycling processes. Proper recycling techniques can help recover critical materials, reduce the demand for raw material extraction, and mitigate potential environmental contamination from improper disposal.
The long-term environmental effects of sulfide-based batteries are still being studied. While they offer potential benefits in terms of energy efficiency and reduced reliance on fossil fuels, the cumulative impact of large-scale production and deployment must be carefully assessed. This includes evaluating the potential for sulfur emissions during battery degradation and the long-term stability of sulfide electrolytes in various environmental conditions.
As research and development in sulfide-based batteries progress, it is crucial to prioritize sustainable manufacturing practices, efficient resource utilization, and effective recycling strategies. By addressing these environmental considerations, the technology can contribute to a more sustainable energy future while minimizing its ecological footprint.
During the production phase, the synthesis of sulfide-based electrolytes often involves energy-intensive processes and the use of potentially hazardous materials. The extraction and processing of raw materials, such as lithium and sulfur, can have significant environmental consequences, including habitat disruption and water pollution. Additionally, the manufacturing of sulfide electrolytes may generate toxic byproducts that require proper handling and disposal.
In the operational phase, sulfide-based batteries demonstrate improved safety characteristics compared to conventional lithium-ion batteries, reducing the risk of thermal runaway and fire incidents. This enhanced safety profile can potentially decrease the environmental impact associated with battery-related accidents and fires. Furthermore, the higher energy density of sulfide-based batteries may lead to reduced material consumption and improved overall efficiency in energy storage applications.
End-of-life considerations for sulfide-based batteries present both challenges and opportunities. The recycling of these batteries is essential to recover valuable materials and minimize environmental impact. However, the complex composition of sulfide electrolytes may require the development of specialized recycling processes. Proper recycling techniques can help recover critical materials, reduce the demand for raw material extraction, and mitigate potential environmental contamination from improper disposal.
The long-term environmental effects of sulfide-based batteries are still being studied. While they offer potential benefits in terms of energy efficiency and reduced reliance on fossil fuels, the cumulative impact of large-scale production and deployment must be carefully assessed. This includes evaluating the potential for sulfur emissions during battery degradation and the long-term stability of sulfide electrolytes in various environmental conditions.
As research and development in sulfide-based batteries progress, it is crucial to prioritize sustainable manufacturing practices, efficient resource utilization, and effective recycling strategies. By addressing these environmental considerations, the technology can contribute to a more sustainable energy future while minimizing its ecological footprint.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!