How to Differentiate Isomer Efficiency in Reaction Pathways
MAR 16, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Isomer Differentiation Background and Research Objectives
Isomer differentiation in reaction pathways represents a fundamental challenge in modern chemical synthesis and catalysis, where the selective formation of specific molecular configurations directly impacts reaction efficiency, product yield, and economic viability. The ability to control and predict isomeric outcomes has become increasingly critical as industries demand higher precision in pharmaceutical manufacturing, materials science, and fine chemical production.
The historical development of isomer differentiation techniques traces back to early stereochemical studies in the 19th century, evolving through mechanistic organic chemistry breakthroughs in the mid-20th century to contemporary computational and analytical approaches. This evolution reflects the growing understanding that molecular geometry and electronic distribution fundamentally govern reaction selectivity and pathway preferences.
Current technological trends indicate a convergence of experimental and theoretical methodologies, with machine learning algorithms increasingly complementing traditional kinetic and thermodynamic analyses. Advanced spectroscopic techniques, coupled with real-time monitoring capabilities, are enabling unprecedented insights into transient intermediates and transition states that determine isomeric distributions.
The primary technical objectives center on developing robust methodologies for predicting, controlling, and optimizing isomeric selectivity across diverse reaction systems. This encompasses establishing quantitative structure-activity relationships that correlate molecular features with pathway preferences, developing computational models capable of accurately predicting isomeric ratios under varying conditions, and creating experimental frameworks for systematic evaluation of selectivity factors.
Secondary objectives include advancing mechanistic understanding of how catalyst design, reaction conditions, and substrate modifications influence isomeric outcomes. The integration of high-throughput screening methodologies with automated analysis systems represents another crucial goal, enabling rapid evaluation of multiple variables affecting isomer distribution.
Long-term strategic objectives focus on establishing predictive frameworks that can guide synthetic route design from the earliest planning stages, reducing the traditional trial-and-error approach that characterizes much of current synthetic chemistry. This includes developing standardized protocols for isomer differentiation assessment and creating comprehensive databases linking molecular structures to observed selectivity patterns across different reaction classes.
The historical development of isomer differentiation techniques traces back to early stereochemical studies in the 19th century, evolving through mechanistic organic chemistry breakthroughs in the mid-20th century to contemporary computational and analytical approaches. This evolution reflects the growing understanding that molecular geometry and electronic distribution fundamentally govern reaction selectivity and pathway preferences.
Current technological trends indicate a convergence of experimental and theoretical methodologies, with machine learning algorithms increasingly complementing traditional kinetic and thermodynamic analyses. Advanced spectroscopic techniques, coupled with real-time monitoring capabilities, are enabling unprecedented insights into transient intermediates and transition states that determine isomeric distributions.
The primary technical objectives center on developing robust methodologies for predicting, controlling, and optimizing isomeric selectivity across diverse reaction systems. This encompasses establishing quantitative structure-activity relationships that correlate molecular features with pathway preferences, developing computational models capable of accurately predicting isomeric ratios under varying conditions, and creating experimental frameworks for systematic evaluation of selectivity factors.
Secondary objectives include advancing mechanistic understanding of how catalyst design, reaction conditions, and substrate modifications influence isomeric outcomes. The integration of high-throughput screening methodologies with automated analysis systems represents another crucial goal, enabling rapid evaluation of multiple variables affecting isomer distribution.
Long-term strategic objectives focus on establishing predictive frameworks that can guide synthetic route design from the earliest planning stages, reducing the traditional trial-and-error approach that characterizes much of current synthetic chemistry. This includes developing standardized protocols for isomer differentiation assessment and creating comprehensive databases linking molecular structures to observed selectivity patterns across different reaction classes.
Market Demand for Selective Isomer Synthesis
The pharmaceutical industry represents the largest market segment driving demand for selective isomer synthesis technologies. Modern drug development increasingly requires the production of single enantiomers rather than racemic mixtures, as different isomers can exhibit vastly different pharmacological properties. Regulatory agencies worldwide have established stringent guidelines mandating the separate evaluation of each stereoisomer in drug candidates, creating substantial market pressure for efficient isomer differentiation and selective synthesis capabilities.
Fine chemicals and specialty chemicals sectors constitute another significant market driver, particularly in the production of agrochemicals, fragrances, and food additives. The agricultural industry specifically demands highly selective synthesis methods for pesticides and herbicides, where the active isomer determines efficacy while inactive isomers may contribute to environmental concerns or reduced performance. This sector's growth trajectory continues to expand as precision agriculture practices become more prevalent globally.
The catalyst and process technology market has experienced substantial growth in response to these demands. Companies developing asymmetric catalysts, chiral auxiliaries, and enantioselective reaction methodologies are witnessing increased investment and market expansion. The transition from traditional resolution methods to direct asymmetric synthesis approaches has created new opportunities for technology providers specializing in reaction pathway optimization and isomer selectivity enhancement.
Biotechnology applications represent an emerging high-growth segment, particularly in the production of chiral building blocks and pharmaceutical intermediates through enzymatic processes. The demand for biocatalytic solutions that can achieve high enantioselectivity while operating under mild conditions has intensified, driven by sustainability considerations and cost-effectiveness requirements in large-scale manufacturing.
Market dynamics are further influenced by regulatory compliance costs and the economic advantages of single-isomer products. Companies investing in advanced isomer differentiation technologies can command premium pricing for their products while reducing waste streams and improving overall process efficiency. The convergence of environmental regulations, quality standards, and economic incentives continues to strengthen market demand for innovative selective synthesis solutions across multiple industrial sectors.
Fine chemicals and specialty chemicals sectors constitute another significant market driver, particularly in the production of agrochemicals, fragrances, and food additives. The agricultural industry specifically demands highly selective synthesis methods for pesticides and herbicides, where the active isomer determines efficacy while inactive isomers may contribute to environmental concerns or reduced performance. This sector's growth trajectory continues to expand as precision agriculture practices become more prevalent globally.
The catalyst and process technology market has experienced substantial growth in response to these demands. Companies developing asymmetric catalysts, chiral auxiliaries, and enantioselective reaction methodologies are witnessing increased investment and market expansion. The transition from traditional resolution methods to direct asymmetric synthesis approaches has created new opportunities for technology providers specializing in reaction pathway optimization and isomer selectivity enhancement.
Biotechnology applications represent an emerging high-growth segment, particularly in the production of chiral building blocks and pharmaceutical intermediates through enzymatic processes. The demand for biocatalytic solutions that can achieve high enantioselectivity while operating under mild conditions has intensified, driven by sustainability considerations and cost-effectiveness requirements in large-scale manufacturing.
Market dynamics are further influenced by regulatory compliance costs and the economic advantages of single-isomer products. Companies investing in advanced isomer differentiation technologies can command premium pricing for their products while reducing waste streams and improving overall process efficiency. The convergence of environmental regulations, quality standards, and economic incentives continues to strengthen market demand for innovative selective synthesis solutions across multiple industrial sectors.
Current Challenges in Isomer Pathway Analysis
The differentiation of isomer efficiency in reaction pathways faces several fundamental challenges that significantly impact both theoretical understanding and practical applications in chemical synthesis. These challenges stem from the complex interplay between molecular structure, thermodynamic stability, and kinetic accessibility of different isomeric forms.
One of the primary obstacles lies in the accurate prediction and measurement of activation energies for competing isomeric pathways. Traditional computational methods often struggle to capture the subtle energy differences between isomeric transition states, particularly when dealing with conformational isomers or stereoisomers. The energy barriers separating these pathways can be remarkably small, sometimes within the margin of error of current theoretical models, making reliable predictions extremely difficult.
Experimental characterization presents equally formidable challenges. Real-time monitoring of isomer formation during reactions requires sophisticated analytical techniques capable of distinguishing between structurally similar compounds. Many conventional analytical methods lack the resolution or sensitivity needed to track rapid isomeric interconversions or to quantify minor isomeric products that may play crucial roles in overall reaction efficiency.
The temporal dynamics of isomeric pathways add another layer of complexity. Isomers may interconvert during the reaction process, leading to kinetic versus thermodynamic product distributions that evolve over time. This dynamic behavior makes it challenging to establish clear structure-activity relationships and to predict which isomeric pathway will dominate under specific reaction conditions.
Catalyst selectivity represents a critical bottleneck in controlling isomeric outcomes. Designing catalysts that can selectively promote one isomeric pathway over another requires deep understanding of the molecular-level interactions between catalyst active sites and different isomeric intermediates. Current catalyst design approaches often rely on trial-and-error methodologies rather than predictive frameworks.
Environmental factors further complicate isomer pathway analysis. Temperature, pressure, solvent effects, and the presence of additives can dramatically alter the relative efficiencies of competing isomeric routes. The multidimensional parameter space created by these variables makes systematic optimization extremely challenging and often leads to contradictory results across different experimental conditions.
One of the primary obstacles lies in the accurate prediction and measurement of activation energies for competing isomeric pathways. Traditional computational methods often struggle to capture the subtle energy differences between isomeric transition states, particularly when dealing with conformational isomers or stereoisomers. The energy barriers separating these pathways can be remarkably small, sometimes within the margin of error of current theoretical models, making reliable predictions extremely difficult.
Experimental characterization presents equally formidable challenges. Real-time monitoring of isomer formation during reactions requires sophisticated analytical techniques capable of distinguishing between structurally similar compounds. Many conventional analytical methods lack the resolution or sensitivity needed to track rapid isomeric interconversions or to quantify minor isomeric products that may play crucial roles in overall reaction efficiency.
The temporal dynamics of isomeric pathways add another layer of complexity. Isomers may interconvert during the reaction process, leading to kinetic versus thermodynamic product distributions that evolve over time. This dynamic behavior makes it challenging to establish clear structure-activity relationships and to predict which isomeric pathway will dominate under specific reaction conditions.
Catalyst selectivity represents a critical bottleneck in controlling isomeric outcomes. Designing catalysts that can selectively promote one isomeric pathway over another requires deep understanding of the molecular-level interactions between catalyst active sites and different isomeric intermediates. Current catalyst design approaches often rely on trial-and-error methodologies rather than predictive frameworks.
Environmental factors further complicate isomer pathway analysis. Temperature, pressure, solvent effects, and the presence of additives can dramatically alter the relative efficiencies of competing isomeric routes. The multidimensional parameter space created by these variables makes systematic optimization extremely challenging and often leads to contradictory results across different experimental conditions.
Existing Methods for Isomer Efficiency Assessment
01 Isomer separation and purification methods
Various techniques can be employed to separate and purify isomers to improve efficiency. These methods include crystallization, distillation, chromatography, and membrane separation processes. The selection of appropriate separation methods depends on the physical and chemical properties of the isomers, such as boiling points, solubility differences, and molecular size. Efficient separation of isomers is crucial for obtaining high-purity products with desired properties and enhanced performance in various applications.- Isomer separation and purification methods: Various techniques can be employed to separate and purify specific isomers to improve efficiency. These methods include crystallization, distillation, chromatography, and selective adsorption processes. The separation of isomers is crucial for obtaining compounds with desired properties and enhanced performance. Efficient separation methods can significantly improve the yield and purity of target isomers, leading to better overall process efficiency.
- Catalytic isomerization processes: Catalytic processes can be utilized to convert one isomer form to another more desirable form, thereby improving efficiency. These processes employ various catalysts including zeolites, metal catalysts, and acid catalysts to facilitate isomerization reactions. The selection of appropriate catalysts and reaction conditions is critical for achieving high conversion rates and selectivity. Optimized catalytic isomerization can enhance product quality and process economics.
- Stereoselective synthesis of specific isomers: Stereoselective synthesis methods enable the direct production of desired isomers with high selectivity, eliminating the need for extensive separation processes. These approaches utilize chiral catalysts, asymmetric synthesis techniques, and enzymatic methods to control the stereochemistry of products. By producing the target isomer directly, these methods can significantly improve overall process efficiency and reduce waste generation.
- Isomer ratio optimization in product formulations: The efficiency of products can be enhanced by optimizing the ratio of different isomers in formulations. Specific isomer ratios can provide superior performance characteristics compared to single isomers or uncontrolled mixtures. This approach involves systematic evaluation of different isomer combinations to identify optimal compositions. The controlled blending of isomers can result in synergistic effects that improve product efficacy and stability.
- Analysis and characterization of isomer efficiency: Advanced analytical techniques are essential for evaluating the efficiency of different isomers and their impact on product performance. Methods such as spectroscopy, chromatography, and computational modeling can be used to assess isomer properties and behavior. Understanding the relationship between isomer structure and efficiency enables rational design and optimization. Comprehensive characterization helps in selecting the most efficient isomers for specific applications.
02 Catalytic isomerization processes
Catalytic isomerization is a key process for converting one isomer into another more valuable or efficient form. Various catalysts, including zeolites, metal oxides, and acid catalysts, can be used to facilitate isomerization reactions. The efficiency of isomerization can be enhanced by optimizing reaction conditions such as temperature, pressure, and catalyst composition. This approach is particularly important in petroleum refining and chemical synthesis where specific isomers are required for improved product performance.Expand Specific Solutions03 Selective synthesis of specific isomers
Controlling the synthesis process to selectively produce desired isomers can significantly improve efficiency. This can be achieved through the use of stereoselective catalysts, specific reaction conditions, or chiral auxiliaries. By directing the synthesis toward the formation of the most efficient isomer, waste can be minimized and product quality can be enhanced. This approach is particularly valuable in pharmaceutical and fine chemical industries where specific isomeric forms exhibit superior biological activity or physical properties.Expand Specific Solutions04 Isomer ratio optimization in product formulations
The ratio of different isomers in a product formulation can be optimized to achieve maximum efficiency. Different isomers may exhibit varying levels of activity, stability, or other desirable properties. By adjusting the isomer composition, the overall performance of the product can be enhanced. This optimization process may involve analytical techniques to determine the optimal isomer ratio and formulation strategies to maintain the desired composition throughout the product lifecycle.Expand Specific Solutions05 Isomer stability and conversion control
Managing the stability of isomers and controlling unwanted isomer conversion is essential for maintaining efficiency. Some isomers may undergo spontaneous conversion to less efficient forms under certain conditions. Stabilization techniques, such as the addition of stabilizers, control of storage conditions, or use of protective packaging, can prevent isomer degradation. Additionally, understanding the kinetics of isomer interconversion allows for the development of strategies to maintain the desired isomeric form throughout processing and storage.Expand Specific Solutions
Key Players in Stereochemistry and Catalysis Industry
The isomer efficiency differentiation in reaction pathways represents a mature yet rapidly evolving field within pharmaceutical and chemical industries. The market demonstrates significant scale, driven by major pharmaceutical companies like Pfizer, Novartis, and Astellas Pharma who require precise stereochemical control for drug development. Technology maturity varies across segments, with established analytical instrumentation companies such as Shimadzu Corp. and Micromass UK Ltd. providing sophisticated mass spectrometry and chromatography solutions for isomer analysis. Meanwhile, emerging players like Insilico Medicine Shanghai Ltd. are advancing AI-driven approaches for predictive isomer behavior modeling. The competitive landscape spans from traditional chemical manufacturers like Daicel Corp. and Nissan Chemical Corp., who focus on chiral separation materials, to research institutions like Swiss Federal Institute of Technology developing novel catalytic methods. This convergence of established analytical capabilities with emerging computational technologies positions the field at an inflection point toward more predictive and efficient isomer differentiation methodologies.
Pfizer Inc.
Technical Solution: Pfizer employs advanced chiral chromatography and computational modeling to differentiate isomer efficiency in drug development pathways. Their approach combines high-performance liquid chromatography (HPLC) with chiral stationary phases to separate enantiomers, coupled with molecular dynamics simulations to predict binding affinities of different isomers to target proteins. The company utilizes machine learning algorithms to analyze structure-activity relationships and optimize reaction conditions for selective isomer production. Their integrated platform includes automated screening systems that can evaluate thousands of isomeric compounds simultaneously, enabling rapid identification of the most efficient stereoisomers for therapeutic applications.
Strengths: Extensive pharmaceutical expertise and advanced analytical capabilities. Weaknesses: High costs and complex regulatory requirements for implementation.
Novartis AG
Technical Solution: Novartis utilizes a comprehensive approach combining crystallographic analysis with computational drug design to evaluate isomer efficiency in pharmaceutical development. Their methodology includes X-ray crystallography to determine precise three-dimensional structures of isomeric compounds bound to target proteins, enabling detailed understanding of binding modes and selectivity. The company employs artificial intelligence-driven molecular modeling to predict the pharmacokinetic and pharmacodynamic properties of different isomers. Their platform integrates high-throughput screening with structure-based drug design, allowing for systematic evaluation of isomeric libraries and optimization of lead compounds for enhanced therapeutic efficacy and reduced side effects.
Strengths: Advanced structural biology capabilities and AI-driven drug design expertise. Weaknesses: Focus primarily on pharmaceutical applications with limited broader chemical industry applicability.
Core Innovations in Chiral Recognition Technologies
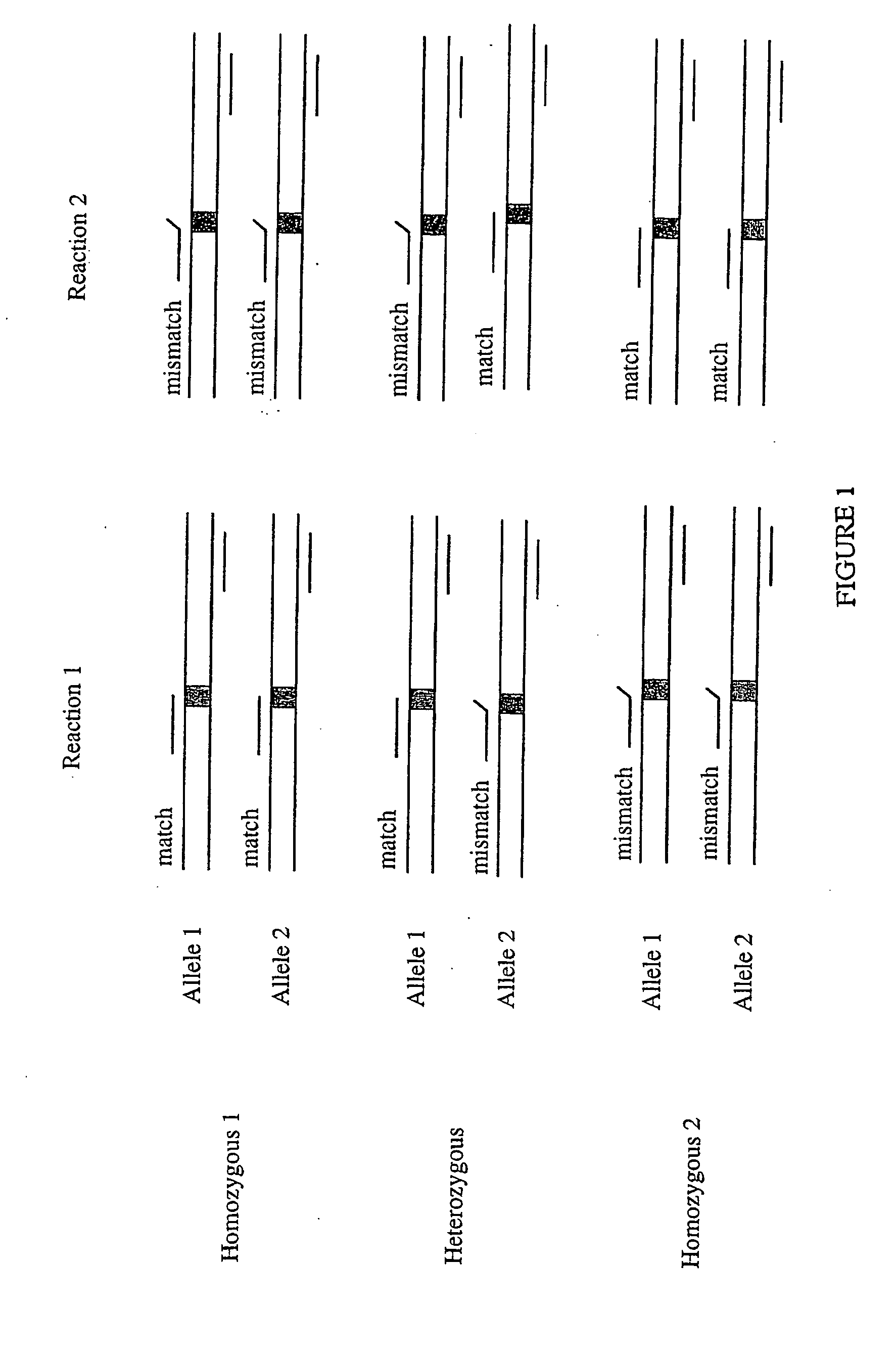
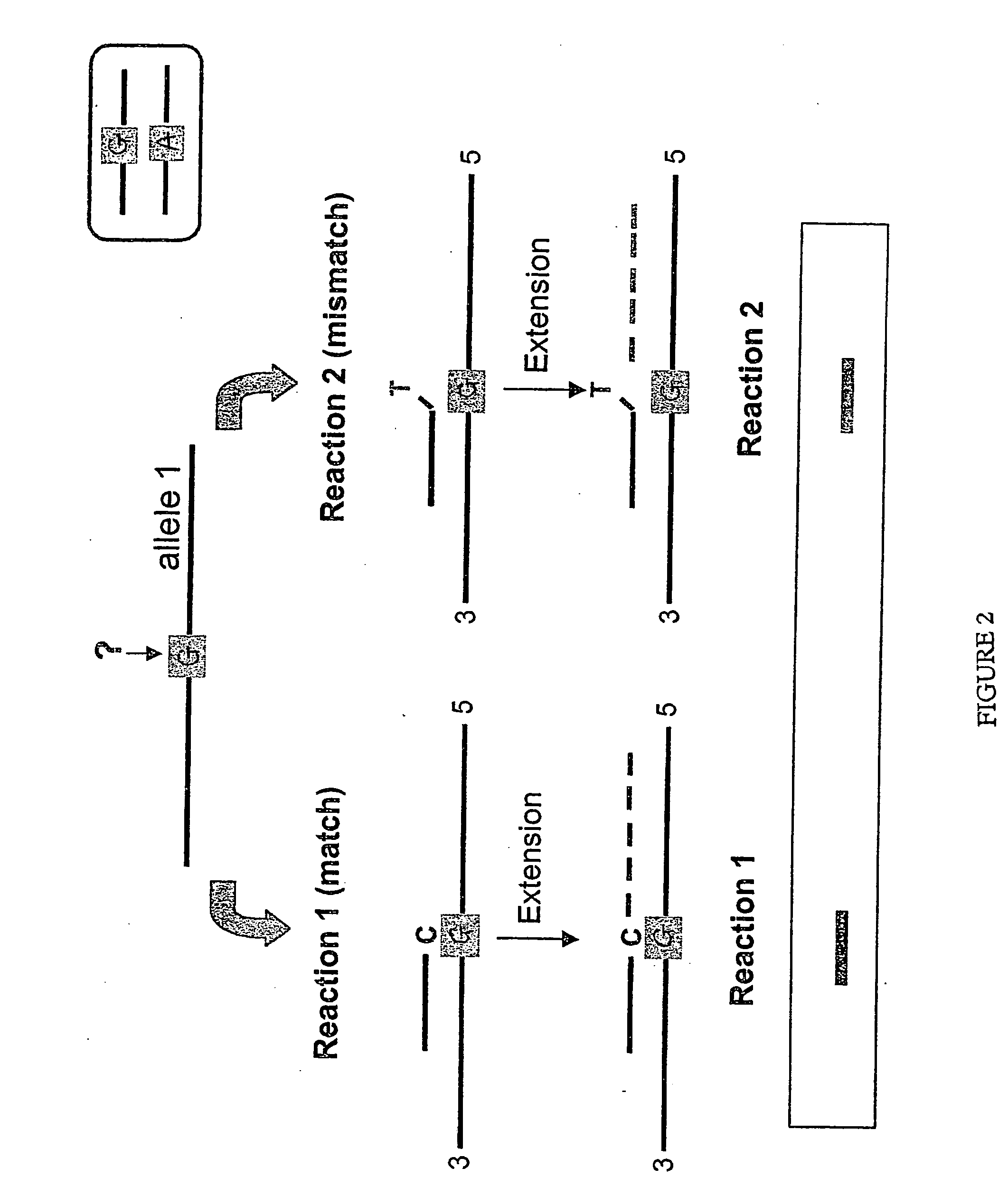
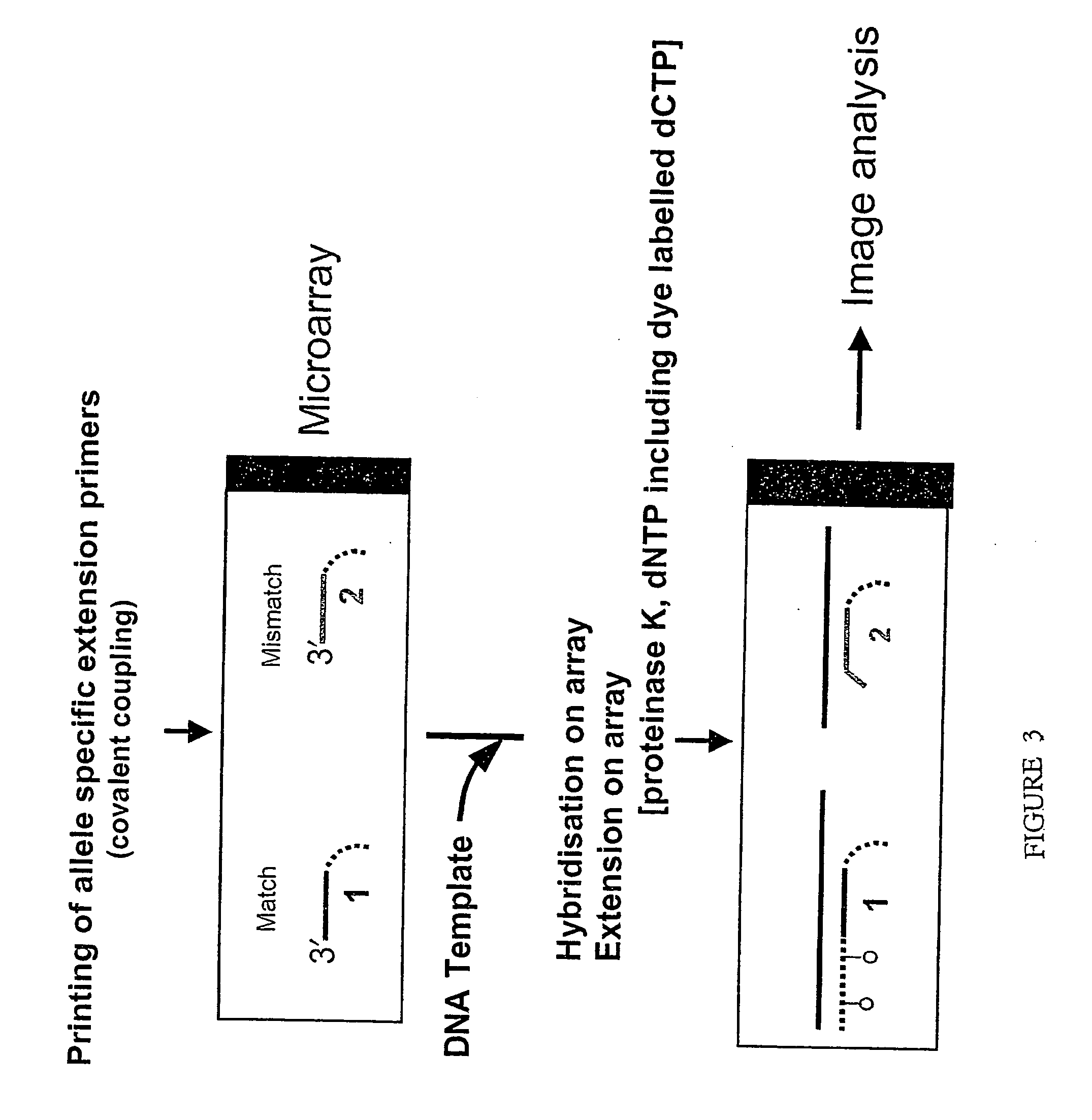
Allele-specific mutation detection assay
PatentInactiveUS20060088872A1
Innovation
- The method involves disabling the polymerase enzyme in mismatched primer-template configurations to prevent extension, allowing only matched configurations to proceed, thereby enhancing discrimination and reducing false positives through the use of enzyme-disabling agents like Proteinase K.
Computational Modeling for Isomer Prediction
Computational modeling has emerged as a transformative approach for predicting isomer behavior and efficiency in chemical reaction pathways. Advanced quantum mechanical calculations, particularly density functional theory (DT) methods, enable researchers to predict the formation, stability, and reactivity of different isomeric structures before experimental synthesis. These computational frameworks provide detailed insights into electronic structures, molecular geometries, and thermodynamic properties that directly influence isomer selectivity.
Machine learning algorithms have revolutionized isomer prediction capabilities by analyzing vast datasets of molecular structures and reaction outcomes. Neural networks trained on experimental data can identify subtle structural patterns that correlate with isomer formation preferences. These models incorporate molecular descriptors, reaction conditions, and catalyst properties to predict which isomeric products will predominate under specific circumstances, significantly reducing the need for extensive experimental screening.
Molecular dynamics simulations offer dynamic perspectives on isomer formation mechanisms by modeling real-time molecular interactions during reaction processes. These simulations reveal how reaction intermediates evolve and which pathways lead to specific isomeric products. By incorporating temperature effects, solvent interactions, and catalyst surfaces, molecular dynamics provides comprehensive understanding of the kinetic factors governing isomer selectivity.
Integrated computational platforms now combine multiple modeling approaches to enhance prediction accuracy. These systems merge quantum calculations with machine learning predictions and molecular dynamics insights to create robust models for isomer efficiency assessment. Such platforms enable researchers to optimize reaction conditions computationally, predict novel isomeric structures, and design selective synthetic pathways with unprecedented precision, ultimately accelerating the development of more efficient and selective chemical processes.
Machine learning algorithms have revolutionized isomer prediction capabilities by analyzing vast datasets of molecular structures and reaction outcomes. Neural networks trained on experimental data can identify subtle structural patterns that correlate with isomer formation preferences. These models incorporate molecular descriptors, reaction conditions, and catalyst properties to predict which isomeric products will predominate under specific circumstances, significantly reducing the need for extensive experimental screening.
Molecular dynamics simulations offer dynamic perspectives on isomer formation mechanisms by modeling real-time molecular interactions during reaction processes. These simulations reveal how reaction intermediates evolve and which pathways lead to specific isomeric products. By incorporating temperature effects, solvent interactions, and catalyst surfaces, molecular dynamics provides comprehensive understanding of the kinetic factors governing isomer selectivity.
Integrated computational platforms now combine multiple modeling approaches to enhance prediction accuracy. These systems merge quantum calculations with machine learning predictions and molecular dynamics insights to create robust models for isomer efficiency assessment. Such platforms enable researchers to optimize reaction conditions computationally, predict novel isomeric structures, and design selective synthetic pathways with unprecedented precision, ultimately accelerating the development of more efficient and selective chemical processes.
Green Chemistry Impact on Isomer Synthesis
Green chemistry principles have fundamentally transformed the landscape of isomer synthesis, introducing sustainability considerations that directly impact the efficiency evaluation of reaction pathways. The integration of environmental consciousness into synthetic chemistry has created new paradigms for assessing isomeric differentiation, where traditional efficiency metrics must now be balanced against ecological footprint and resource conservation.
The adoption of atom economy principles has revolutionized how chemists approach isomer-selective reactions. This concept, pioneered by Barry Trost, emphasizes maximizing the incorporation of starting materials into final products, thereby minimizing waste generation. In isomer synthesis, this translates to developing reaction pathways that not only achieve high selectivity but also utilize every atom from reactants in the desired isomeric product. Catalytic processes have emerged as particularly valuable tools, enabling precise control over stereochemistry while maintaining excellent atom efficiency.
Solvent selection represents another critical aspect where green chemistry intersects with isomer differentiation. Traditional organic solvents, often toxic and environmentally persistent, are being replaced with greener alternatives such as water, ionic liquids, and supercritical fluids. These alternative media can dramatically influence reaction selectivity and isomer distribution. Water-based reactions, for instance, often exhibit unique selectivity patterns due to hydrophobic effects and hydrogen bonding interactions that can favor specific isomeric outcomes.
The development of biocatalytic approaches has opened unprecedented opportunities for sustainable isomer synthesis. Enzymes naturally exhibit exceptional selectivity, often producing single isomers with near-perfect efficiency. Directed evolution techniques have expanded the scope of biocatalysis, enabling the creation of engineered enzymes capable of producing desired isomers under mild, environmentally benign conditions. This biological approach eliminates the need for harsh reagents and extreme reaction conditions typically associated with traditional synthetic methods.
Renewable feedstock utilization has become increasingly important in green isomer synthesis. The shift from petroleum-based starting materials to bio-derived precursors not only reduces environmental impact but can also influence the inherent selectivity of synthetic pathways. Biomass-derived compounds often possess unique structural features that can be leveraged to achieve specific isomeric outcomes through carefully designed reaction sequences.
The implementation of continuous flow chemistry represents a significant advancement in green isomer synthesis. Flow reactors enable precise control over reaction parameters, leading to improved selectivity and reduced waste generation. The enhanced heat and mass transfer characteristics of flow systems often result in cleaner reaction profiles with fewer side products, thereby improving the overall efficiency of isomer differentiation processes while minimizing environmental impact.
The adoption of atom economy principles has revolutionized how chemists approach isomer-selective reactions. This concept, pioneered by Barry Trost, emphasizes maximizing the incorporation of starting materials into final products, thereby minimizing waste generation. In isomer synthesis, this translates to developing reaction pathways that not only achieve high selectivity but also utilize every atom from reactants in the desired isomeric product. Catalytic processes have emerged as particularly valuable tools, enabling precise control over stereochemistry while maintaining excellent atom efficiency.
Solvent selection represents another critical aspect where green chemistry intersects with isomer differentiation. Traditional organic solvents, often toxic and environmentally persistent, are being replaced with greener alternatives such as water, ionic liquids, and supercritical fluids. These alternative media can dramatically influence reaction selectivity and isomer distribution. Water-based reactions, for instance, often exhibit unique selectivity patterns due to hydrophobic effects and hydrogen bonding interactions that can favor specific isomeric outcomes.
The development of biocatalytic approaches has opened unprecedented opportunities for sustainable isomer synthesis. Enzymes naturally exhibit exceptional selectivity, often producing single isomers with near-perfect efficiency. Directed evolution techniques have expanded the scope of biocatalysis, enabling the creation of engineered enzymes capable of producing desired isomers under mild, environmentally benign conditions. This biological approach eliminates the need for harsh reagents and extreme reaction conditions typically associated with traditional synthetic methods.
Renewable feedstock utilization has become increasingly important in green isomer synthesis. The shift from petroleum-based starting materials to bio-derived precursors not only reduces environmental impact but can also influence the inherent selectivity of synthetic pathways. Biomass-derived compounds often possess unique structural features that can be leveraged to achieve specific isomeric outcomes through carefully designed reaction sequences.
The implementation of continuous flow chemistry represents a significant advancement in green isomer synthesis. Flow reactors enable precise control over reaction parameters, leading to improved selectivity and reduced waste generation. The enhanced heat and mass transfer characteristics of flow systems often result in cleaner reaction profiles with fewer side products, thereby improving the overall efficiency of isomer differentiation processes while minimizing environmental impact.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!