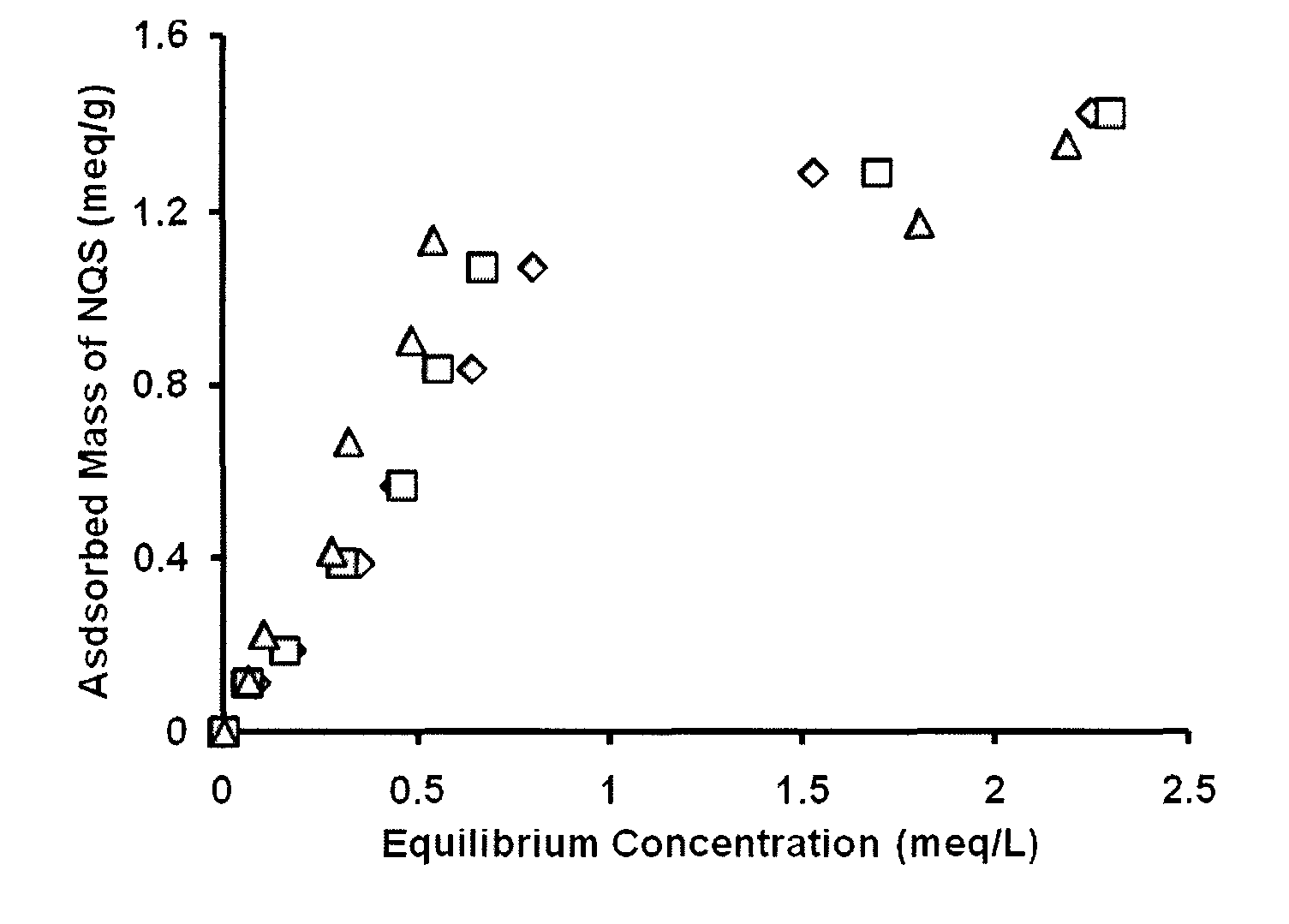
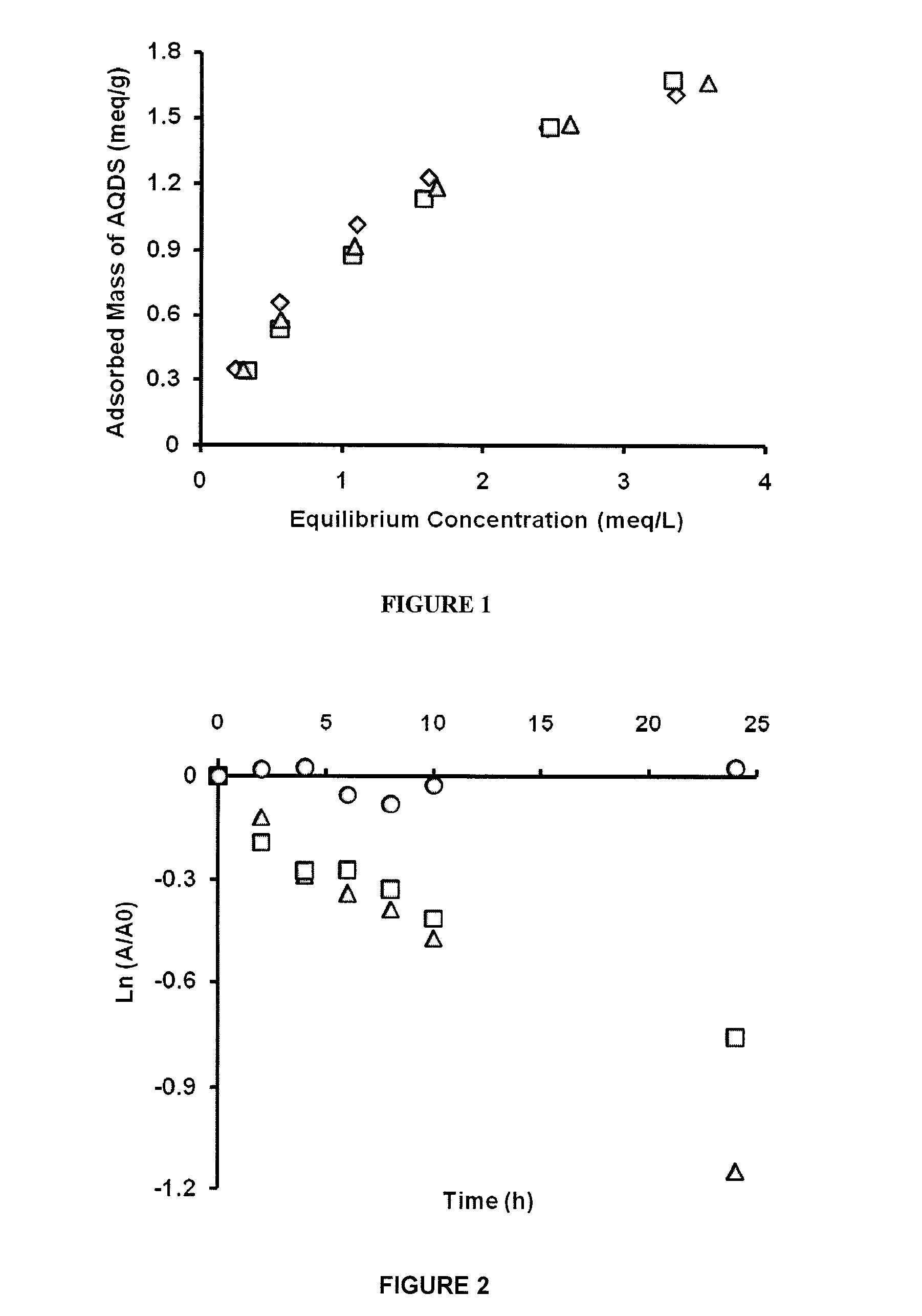
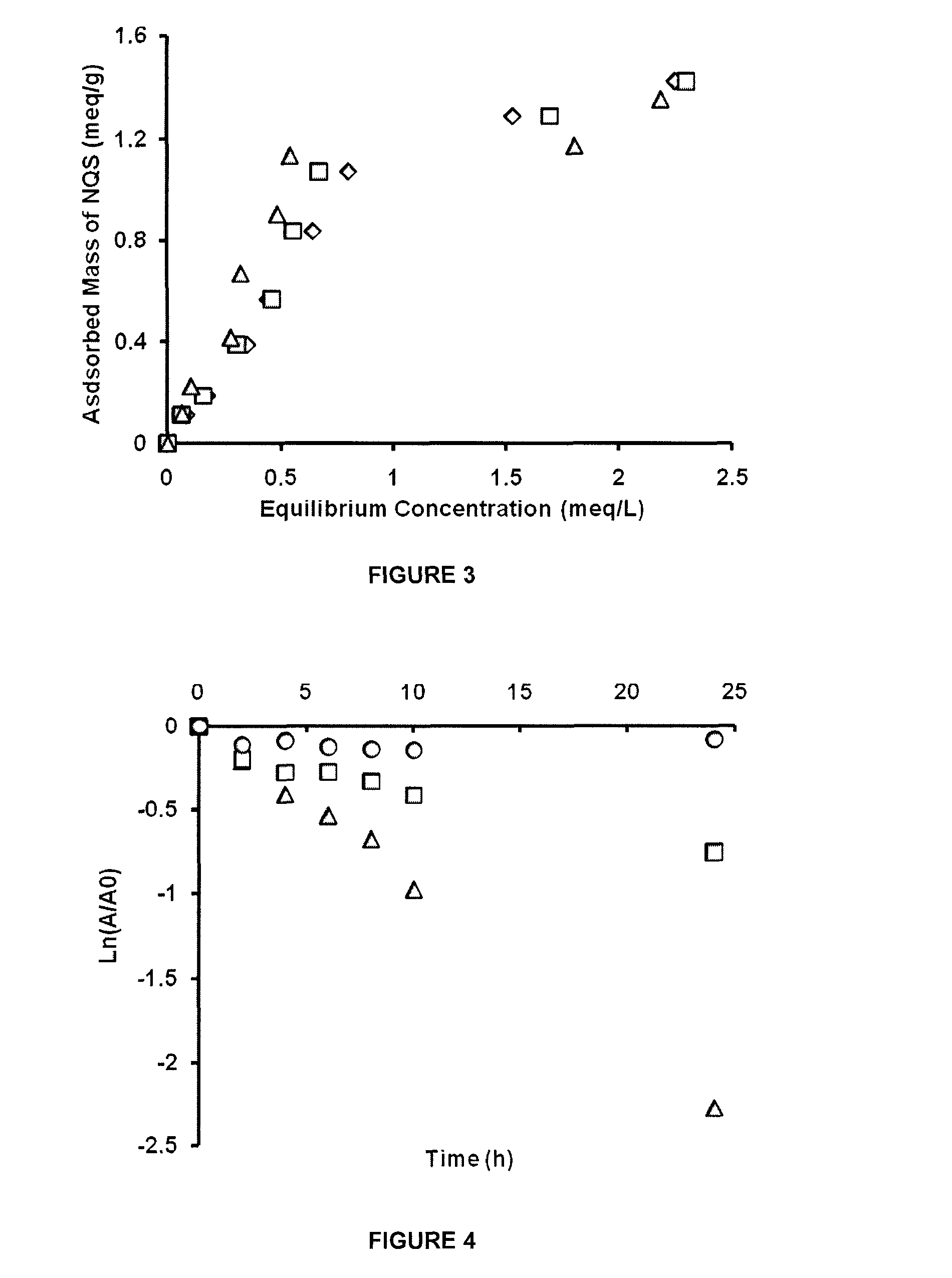
How to Mitigate Biodegradation Risks in Redox Mediators
APR 29, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Redox Mediator Biodegradation Background and Objectives
Redox mediators have emerged as critical components in various electrochemical applications, including microbial fuel cells, bioelectrochemical systems, and enzymatic biofuel cells. These synthetic or naturally occurring compounds facilitate electron transfer between biological systems and electrodes, significantly enhancing the efficiency of bioelectrochemical processes. However, the widespread deployment of redox mediator-based technologies faces a fundamental challenge: biodegradation susceptibility that compromises system performance and longevity.
The biodegradation of redox mediators represents a complex phenomenon where microorganisms metabolize these compounds as carbon or energy sources, leading to their structural modification or complete mineralization. This biological transformation not only reduces the effective concentration of active mediators but can also generate metabolic byproducts that may inhibit electrochemical processes or create environmental concerns. Common redox mediators such as methylene blue, neutral red, and various quinone derivatives have demonstrated varying degrees of susceptibility to microbial degradation under different environmental conditions.
Current research indicates that biodegradation rates are influenced by multiple factors including mediator chemical structure, microbial community composition, environmental pH, temperature, and oxygen availability. Electron-rich aromatic compounds and those containing easily cleavable functional groups typically exhibit higher biodegradation susceptibility. The presence of specific microbial strains capable of utilizing these compounds as substrates further accelerates the degradation process, creating a significant operational challenge for long-term system stability.
The primary objective of addressing biodegradation risks is to develop comprehensive mitigation strategies that preserve redox mediator functionality while maintaining system performance over extended operational periods. This involves understanding the fundamental mechanisms governing microbial degradation pathways and identifying structural modifications or operational conditions that can minimize biodegradation rates without compromising electrochemical activity.
Secondary objectives include establishing predictive models for mediator stability assessment, developing biodegradation-resistant mediator designs, and creating operational protocols that balance biological activity with mediator preservation. These efforts aim to enable the commercial viability of redox mediator-based technologies across diverse applications ranging from wastewater treatment to renewable energy generation.
The biodegradation of redox mediators represents a complex phenomenon where microorganisms metabolize these compounds as carbon or energy sources, leading to their structural modification or complete mineralization. This biological transformation not only reduces the effective concentration of active mediators but can also generate metabolic byproducts that may inhibit electrochemical processes or create environmental concerns. Common redox mediators such as methylene blue, neutral red, and various quinone derivatives have demonstrated varying degrees of susceptibility to microbial degradation under different environmental conditions.
Current research indicates that biodegradation rates are influenced by multiple factors including mediator chemical structure, microbial community composition, environmental pH, temperature, and oxygen availability. Electron-rich aromatic compounds and those containing easily cleavable functional groups typically exhibit higher biodegradation susceptibility. The presence of specific microbial strains capable of utilizing these compounds as substrates further accelerates the degradation process, creating a significant operational challenge for long-term system stability.
The primary objective of addressing biodegradation risks is to develop comprehensive mitigation strategies that preserve redox mediator functionality while maintaining system performance over extended operational periods. This involves understanding the fundamental mechanisms governing microbial degradation pathways and identifying structural modifications or operational conditions that can minimize biodegradation rates without compromising electrochemical activity.
Secondary objectives include establishing predictive models for mediator stability assessment, developing biodegradation-resistant mediator designs, and creating operational protocols that balance biological activity with mediator preservation. These efforts aim to enable the commercial viability of redox mediator-based technologies across diverse applications ranging from wastewater treatment to renewable energy generation.
Market Demand for Stable Redox Mediator Systems
The global market for stable redox mediator systems is experiencing unprecedented growth driven by the expanding applications in energy storage, biotechnology, and environmental remediation sectors. Bioelectrochemical systems, including microbial fuel cells and bioelectrosynthesis platforms, represent the fastest-growing segment where redox mediators play critical roles in electron transfer processes. The increasing adoption of these technologies in wastewater treatment facilities and renewable energy integration projects has created substantial demand for mediators that can withstand biological degradation over extended operational periods.
Industrial biotechnology applications constitute another major demand driver, particularly in pharmaceutical manufacturing and fine chemical synthesis. Companies are increasingly seeking redox mediators that maintain catalytic efficiency while resisting enzymatic breakdown in complex biological environments. The pharmaceutical sector specifically requires mediators capable of functioning in sterile conditions without compromising product quality through degradation byproducts.
The renewable energy storage market presents significant opportunities for stable redox mediator systems, especially in flow battery technologies. Grid-scale energy storage projects demand mediators with exceptional longevity and resistance to microbial contamination, as system maintenance costs directly impact project economics. Utilities and energy developers are prioritizing mediator stability as a key selection criterion for large-scale deployments.
Environmental monitoring and biosensor applications represent an emerging market segment with stringent stability requirements. Continuous monitoring systems deployed in natural environments face constant exposure to diverse microbial communities, creating demand for mediators engineered to resist biodegradation while maintaining electrochemical performance over months or years of operation.
The agricultural biotechnology sector is driving demand for stable mediators in soil health monitoring and precision agriculture applications. These systems require mediators that function reliably in soil environments rich with degradative microorganisms while providing accurate electrochemical measurements for crop optimization and environmental assessment.
Market growth is further accelerated by regulatory pressures for sustainable technologies and the need for cost-effective solutions that minimize replacement frequency and maintenance requirements across all application sectors.
Industrial biotechnology applications constitute another major demand driver, particularly in pharmaceutical manufacturing and fine chemical synthesis. Companies are increasingly seeking redox mediators that maintain catalytic efficiency while resisting enzymatic breakdown in complex biological environments. The pharmaceutical sector specifically requires mediators capable of functioning in sterile conditions without compromising product quality through degradation byproducts.
The renewable energy storage market presents significant opportunities for stable redox mediator systems, especially in flow battery technologies. Grid-scale energy storage projects demand mediators with exceptional longevity and resistance to microbial contamination, as system maintenance costs directly impact project economics. Utilities and energy developers are prioritizing mediator stability as a key selection criterion for large-scale deployments.
Environmental monitoring and biosensor applications represent an emerging market segment with stringent stability requirements. Continuous monitoring systems deployed in natural environments face constant exposure to diverse microbial communities, creating demand for mediators engineered to resist biodegradation while maintaining electrochemical performance over months or years of operation.
The agricultural biotechnology sector is driving demand for stable mediators in soil health monitoring and precision agriculture applications. These systems require mediators that function reliably in soil environments rich with degradative microorganisms while providing accurate electrochemical measurements for crop optimization and environmental assessment.
Market growth is further accelerated by regulatory pressures for sustainable technologies and the need for cost-effective solutions that minimize replacement frequency and maintenance requirements across all application sectors.
Current Biodegradation Challenges in Redox Mediators
Redox mediators face significant biodegradation challenges that compromise their stability and performance in various applications, particularly in bioelectrochemical systems, environmental remediation, and energy storage technologies. The primary challenge stems from the inherent susceptibility of organic redox mediators to microbial attack, where naturally occurring microorganisms recognize these compounds as potential carbon and energy sources.
Enzymatic degradation represents the most prevalent biodegradation pathway affecting redox mediators. Microorganisms produce specific enzymes such as oxidases, reductases, and hydrolases that can cleave chemical bonds within mediator molecules. This enzymatic activity is particularly problematic for quinone-based mediators, phenolic compounds, and aromatic heterocycles commonly used in redox applications. The degradation process often results in the formation of metabolites that lack redox activity, effectively reducing the mediator concentration and system efficiency.
Structural vulnerability constitutes another critical challenge, as many effective redox mediators contain functional groups that are inherently biodegradable. Ester linkages, ether bonds, and aromatic rings with electron-donating substituents are particularly susceptible to microbial metabolism. The presence of hydroxyl groups, amino groups, and carboxyl functionalities further increases biodegradation susceptibility by providing recognition sites for microbial enzymes.
Environmental factors significantly exacerbate biodegradation risks. Temperature fluctuations, pH variations, and oxygen availability create conditions that either accelerate microbial growth or enhance enzymatic activity. In aqueous environments, the presence of nutrients and trace metals can stimulate microbial populations, leading to increased biodegradation rates. Additionally, biofilm formation on electrode surfaces or reactor components creates localized environments with concentrated microbial activity.
The temporal aspect of biodegradation presents ongoing operational challenges. Unlike chemical degradation that may follow predictable kinetics, biodegradation rates can vary dramatically based on microbial adaptation, seasonal changes, and substrate availability. This variability makes it difficult to predict mediator lifetime and system performance, complicating maintenance schedules and replacement strategies.
Concentration-dependent biodegradation effects add complexity to system design and operation. At low concentrations, mediators may be completely metabolized by microorganisms, while higher concentrations might exhibit toxicity effects that inhibit microbial growth but also potentially impact system biocompatibility. This concentration dependency requires careful optimization to balance mediator effectiveness with biodegradation resistance.
Enzymatic degradation represents the most prevalent biodegradation pathway affecting redox mediators. Microorganisms produce specific enzymes such as oxidases, reductases, and hydrolases that can cleave chemical bonds within mediator molecules. This enzymatic activity is particularly problematic for quinone-based mediators, phenolic compounds, and aromatic heterocycles commonly used in redox applications. The degradation process often results in the formation of metabolites that lack redox activity, effectively reducing the mediator concentration and system efficiency.
Structural vulnerability constitutes another critical challenge, as many effective redox mediators contain functional groups that are inherently biodegradable. Ester linkages, ether bonds, and aromatic rings with electron-donating substituents are particularly susceptible to microbial metabolism. The presence of hydroxyl groups, amino groups, and carboxyl functionalities further increases biodegradation susceptibility by providing recognition sites for microbial enzymes.
Environmental factors significantly exacerbate biodegradation risks. Temperature fluctuations, pH variations, and oxygen availability create conditions that either accelerate microbial growth or enhance enzymatic activity. In aqueous environments, the presence of nutrients and trace metals can stimulate microbial populations, leading to increased biodegradation rates. Additionally, biofilm formation on electrode surfaces or reactor components creates localized environments with concentrated microbial activity.
The temporal aspect of biodegradation presents ongoing operational challenges. Unlike chemical degradation that may follow predictable kinetics, biodegradation rates can vary dramatically based on microbial adaptation, seasonal changes, and substrate availability. This variability makes it difficult to predict mediator lifetime and system performance, complicating maintenance schedules and replacement strategies.
Concentration-dependent biodegradation effects add complexity to system design and operation. At low concentrations, mediators may be completely metabolized by microorganisms, while higher concentrations might exhibit toxicity effects that inhibit microbial growth but also potentially impact system biocompatibility. This concentration dependency requires careful optimization to balance mediator effectiveness with biodegradation resistance.
Existing Biodegradation Prevention Solutions
01 Redox mediator stability and degradation pathways
Research focuses on understanding the chemical stability of redox mediators under various environmental conditions and identifying specific degradation pathways. Studies examine how factors such as pH, temperature, oxygen exposure, and microbial activity affect the breakdown of these compounds. The degradation mechanisms include oxidative processes, hydrolysis reactions, and enzymatic transformations that can lead to the formation of potentially harmful metabolites.- Redox mediator stability and degradation pathways: Research focuses on understanding the chemical stability of redox mediators under various environmental conditions and identifying specific degradation pathways. Studies examine how factors such as pH, temperature, oxygen exposure, and microbial activity affect the breakdown of these compounds. The degradation mechanisms include oxidative processes, hydrolysis reactions, and enzymatic transformations that can lead to the formation of potentially harmful metabolites.
- Environmental fate and transport of redox mediators: Investigation of how redox mediators behave in natural environments, including their mobility in soil and water systems. Research examines adsorption characteristics, leaching potential, and bioaccumulation tendencies. Studies focus on the persistence of these compounds in different environmental matrices and their potential for long-range transport through air, water, and biological systems.
- Toxicological assessment and risk evaluation methods: Development of comprehensive testing protocols to evaluate the potential health and environmental risks associated with redox mediator exposure. Research includes acute and chronic toxicity studies, genotoxicity assessments, and ecological impact evaluations. Methods focus on establishing safe exposure limits and developing standardized risk assessment frameworks for regulatory purposes.
- Mitigation strategies and containment technologies: Development of technologies and methods to prevent or minimize the release of redox mediators into the environment. Research includes containment systems, treatment technologies for contaminated media, and engineering controls to reduce exposure risks. Studies focus on remediation techniques, barrier systems, and monitoring approaches to manage potential contamination events.
- Biodegradation enhancement and bioremediation approaches: Investigation of biological methods to accelerate the breakdown of redox mediators using microorganisms, enzymes, or enhanced biological processes. Research examines the use of specific bacterial strains, fungal systems, and engineered biological pathways to promote safe degradation. Studies focus on optimizing conditions for biological treatment and developing sustainable remediation solutions.
02 Environmental fate and transport of redox mediators
Investigation of how redox mediators behave in natural environments, including their mobility in soil and water systems. Research examines adsorption characteristics, leaching potential, and bioaccumulation tendencies. Studies focus on predicting the environmental distribution and persistence of these compounds, considering factors such as soil composition, groundwater flow, and seasonal variations that influence their environmental fate.Expand Specific Solutions03 Microbial degradation mechanisms and pathways
Analysis of how microorganisms break down redox mediators through various biochemical processes. Research identifies specific bacterial and fungal species capable of metabolizing these compounds and characterizes the enzymatic pathways involved. Studies examine the role of microbial communities in natural attenuation processes and investigate factors that enhance or inhibit biodegradation rates in different environmental matrices.Expand Specific Solutions04 Toxicity assessment and ecological risk evaluation
Comprehensive evaluation of the potential adverse effects of redox mediators and their degradation products on various organisms and ecosystems. Research includes acute and chronic toxicity testing on aquatic organisms, terrestrial species, and microbial communities. Studies assess biomarkers of exposure and effect, establish dose-response relationships, and evaluate the potential for endocrine disruption or other sublethal effects on wildlife populations.Expand Specific Solutions05 Risk mitigation strategies and remediation technologies
Development of methods to reduce or eliminate the environmental risks associated with redox mediator contamination. Research focuses on advanced treatment technologies including bioremediation, chemical oxidation, and adsorption processes. Studies evaluate the effectiveness of containment strategies, natural attenuation enhancement techniques, and engineered systems designed to prevent or minimize the release of these compounds into the environment.Expand Specific Solutions
Key Players in Redox Mediator and Bioelectrochemistry
The biodegradation mitigation in redox mediators field represents an emerging technology sector in early development stages with significant growth potential. The market remains relatively nascent, driven by increasing demand for stable electrochemical systems in biosensors, energy storage, and biotechnology applications. Technology maturity varies considerably across players, with established chemical giants like Bayer AG, BASF Corp., and China Petroleum & Chemical Corp. leveraging extensive R&D capabilities and manufacturing expertise to develop advanced stabilization solutions. Academic institutions including Nanjing University, Dalian University of Technology, and Indian Institutes of Technology contribute fundamental research on molecular design and degradation mechanisms. Specialized biotechnology companies such as BeFC SAS and PBS Biotech focus on application-specific solutions, while healthcare-oriented firms like Abbott Diabetes Care address biosensor stability challenges. The competitive landscape shows a convergence of traditional chemical manufacturers, emerging biotech companies, and research institutions, indicating technology transition from laboratory research toward commercial viability, though widespread market adoption remains limited by technical complexity and cost considerations.
Abbott Diabetes Care, Inc.
Technical Solution: Abbott has developed advanced glucose biosensor technology that addresses redox mediator biodegradation through the use of stabilized osmium-based mediators and protective polymer matrices. Their FreeStyle glucose sensors employ osmium complexes with enhanced stability against enzymatic degradation and oxidative stress. The company implements multi-layered sensor architectures with biocompatible coatings that create barriers against microbial contamination while maintaining electrochemical performance. Abbott's approach includes the use of antimicrobial agents integrated into the sensor matrix and optimized pH buffering systems to prevent mediator degradation in physiological environments.
Strengths: Proven commercial success in glucose monitoring with stable mediator systems, extensive clinical validation data. Weaknesses: Limited to glucose sensing applications, mediator systems may not be directly transferable to other redox applications.
Bayer AG
Technical Solution: Bayer has developed innovative approaches to mitigate biodegradation risks in redox mediators through their pharmaceutical and materials science divisions. Their strategy involves the design of bio-stable mediator molecules with enhanced resistance to enzymatic degradation and microbial attack. Bayer employs advanced drug delivery principles to create protective microenvironments around redox mediators, including the use of biodegradable polymer matrices that provide controlled release while protecting against degradation. The company has developed antimicrobial coating technologies and sterilization-compatible mediator systems for medical device applications. Their research includes the use of cyclodextrin complexation and liposomal encapsulation to shield sensitive redox compounds from biological degradation pathways.
Strengths: Strong pharmaceutical expertise in drug stability and biocompatibility, established regulatory pathways for medical applications. Weaknesses: Primary focus on pharmaceutical applications may limit broader industrial applicability, higher development costs due to regulatory requirements.
Core Innovations in Mediator Stability Enhancement
Non-dissolved redox mediator biofilm carrier and its preparation method
PatentActiveUS20190144316A1
Innovation
- A non-dissolved redox mediator biofilm carrier is developed by mixing graphene oxide or carbonylation modified graphene oxide with extrusion grade polyethylene/polypropylene particles, using a surface carbonylation modification process and screw extrusion method to create a carrier with enhanced electron transfer sites.
Immobilized redox mediators for the treatment of contaminated waters and gas emissions
PatentInactiveUS8147701B2
Innovation
- Immobilization of quinoid redox mediators on ion exchange resins, which are selected for their physical-chemical properties and stability, allowing stable attraction and retention of redox active groups, reducing mass transfer limitations and wash-out, and enabling effective electron transfer to pollutants.
Environmental Regulations for Redox Mediator Applications
The regulatory landscape for redox mediator applications is rapidly evolving as environmental agencies worldwide recognize the need to address potential biodegradation risks and ecological impacts. Current environmental regulations primarily focus on chemical registration requirements, environmental fate assessments, and risk evaluation protocols that directly influence how redox mediators can be developed, tested, and deployed in various applications.
In the United States, the Environmental Protection Agency (EPA) regulates redox mediators under the Toxic Substances Control Act (TSCA), requiring comprehensive pre-manufacture notifications for new chemical substances. These regulations mandate detailed biodegradation studies, ecotoxicity assessments, and environmental fate modeling to evaluate potential risks before market entry. The EPA's New Chemicals Program specifically requires manufacturers to demonstrate that redox mediators will not pose unreasonable environmental risks, including assessment of their persistence, bioaccumulation potential, and toxicity profiles.
European Union regulations under REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) impose even more stringent requirements for redox mediator applications. The regulation demands extensive dossiers containing biodegradation data, environmental monitoring protocols, and risk mitigation strategies. Particularly relevant are the requirements for persistent, bioaccumulative, and toxic (PBT) substance evaluations, which directly address biodegradation concerns by establishing clear criteria for environmental persistence and biological effects.
Emerging regulatory frameworks are increasingly incorporating life-cycle assessment requirements and extended producer responsibility principles. These regulations mandate that manufacturers of redox mediators implement comprehensive monitoring systems throughout the product lifecycle, from synthesis to disposal. Recent regulatory updates have introduced specific guidelines for biodegradation testing protocols, requiring standardized methodologies such as OECD guidelines for ready biodegradability and inherent biodegradability assessments.
International harmonization efforts through organizations like the OECD are establishing global standards for redox mediator environmental assessment. These initiatives focus on developing consistent testing protocols, risk assessment methodologies, and regulatory approval processes that address biodegradation risks while facilitating innovation in sustainable technologies.
In the United States, the Environmental Protection Agency (EPA) regulates redox mediators under the Toxic Substances Control Act (TSCA), requiring comprehensive pre-manufacture notifications for new chemical substances. These regulations mandate detailed biodegradation studies, ecotoxicity assessments, and environmental fate modeling to evaluate potential risks before market entry. The EPA's New Chemicals Program specifically requires manufacturers to demonstrate that redox mediators will not pose unreasonable environmental risks, including assessment of their persistence, bioaccumulation potential, and toxicity profiles.
European Union regulations under REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) impose even more stringent requirements for redox mediator applications. The regulation demands extensive dossiers containing biodegradation data, environmental monitoring protocols, and risk mitigation strategies. Particularly relevant are the requirements for persistent, bioaccumulative, and toxic (PBT) substance evaluations, which directly address biodegradation concerns by establishing clear criteria for environmental persistence and biological effects.
Emerging regulatory frameworks are increasingly incorporating life-cycle assessment requirements and extended producer responsibility principles. These regulations mandate that manufacturers of redox mediators implement comprehensive monitoring systems throughout the product lifecycle, from synthesis to disposal. Recent regulatory updates have introduced specific guidelines for biodegradation testing protocols, requiring standardized methodologies such as OECD guidelines for ready biodegradability and inherent biodegradability assessments.
International harmonization efforts through organizations like the OECD are establishing global standards for redox mediator environmental assessment. These initiatives focus on developing consistent testing protocols, risk assessment methodologies, and regulatory approval processes that address biodegradation risks while facilitating innovation in sustainable technologies.
Biocompatibility and Safety Assessment Frameworks
The establishment of comprehensive biocompatibility and safety assessment frameworks for redox mediators represents a critical foundation for mitigating biodegradation risks in bioelectrochemical systems. These frameworks must encompass standardized protocols that evaluate both acute and chronic toxicity effects of redox mediators on target organisms and surrounding ecosystems. Current assessment methodologies primarily rely on established toxicological testing procedures adapted from pharmaceutical and chemical industries, including cytotoxicity assays, genotoxicity studies, and environmental fate assessments.
Regulatory compliance frameworks vary significantly across different jurisdictions, with the European Union's REACH regulation providing the most comprehensive guidance for chemical safety assessment, while the United States relies on EPA guidelines under the Toxic Substances Control Act. These regulatory landscapes necessitate multi-tiered testing approaches that begin with in vitro screening methods and progress through increasingly complex biological systems. The integration of computational toxicology models, including QSAR analysis and molecular docking studies, has emerged as a valuable tool for preliminary risk assessment and prioritization of testing protocols.
Standardized biocompatibility testing protocols specifically designed for redox mediators must address unique challenges related to their electrochemical properties and potential for generating reactive intermediates. The development of specialized assays that can evaluate mediator interactions under both oxidized and reduced states represents a significant advancement in safety assessment methodologies. These protocols typically incorporate multiple endpoints, including cell viability, membrane integrity, oxidative stress markers, and DNA damage indicators.
Environmental safety assessment frameworks require particular attention to biodegradation pathways and metabolite formation, as degradation products may exhibit different toxicological profiles compared to parent compounds. The implementation of tiered testing strategies, beginning with standardized organisms such as Daphnia magna and Vibrio fischeri, followed by more complex ecosystem-level studies, provides a systematic approach to environmental risk characterization. Advanced analytical techniques, including LC-MS/MS and NMR spectroscopy, enable comprehensive identification and quantification of degradation products throughout the assessment process.
The integration of real-time monitoring systems within biocompatibility frameworks allows for continuous assessment of mediator stability and safety during operational conditions. These systems incorporate biosensors and automated analytical platforms that can detect early indicators of biodegradation or toxicity, enabling proactive risk management strategies. The development of predictive models based on machine learning algorithms further enhances the capability to anticipate potential safety issues before they manifest in operational systems.
Regulatory compliance frameworks vary significantly across different jurisdictions, with the European Union's REACH regulation providing the most comprehensive guidance for chemical safety assessment, while the United States relies on EPA guidelines under the Toxic Substances Control Act. These regulatory landscapes necessitate multi-tiered testing approaches that begin with in vitro screening methods and progress through increasingly complex biological systems. The integration of computational toxicology models, including QSAR analysis and molecular docking studies, has emerged as a valuable tool for preliminary risk assessment and prioritization of testing protocols.
Standardized biocompatibility testing protocols specifically designed for redox mediators must address unique challenges related to their electrochemical properties and potential for generating reactive intermediates. The development of specialized assays that can evaluate mediator interactions under both oxidized and reduced states represents a significant advancement in safety assessment methodologies. These protocols typically incorporate multiple endpoints, including cell viability, membrane integrity, oxidative stress markers, and DNA damage indicators.
Environmental safety assessment frameworks require particular attention to biodegradation pathways and metabolite formation, as degradation products may exhibit different toxicological profiles compared to parent compounds. The implementation of tiered testing strategies, beginning with standardized organisms such as Daphnia magna and Vibrio fischeri, followed by more complex ecosystem-level studies, provides a systematic approach to environmental risk characterization. Advanced analytical techniques, including LC-MS/MS and NMR spectroscopy, enable comprehensive identification and quantification of degradation products throughout the assessment process.
The integration of real-time monitoring systems within biocompatibility frameworks allows for continuous assessment of mediator stability and safety during operational conditions. These systems incorporate biosensors and automated analytical platforms that can detect early indicators of biodegradation or toxicity, enabling proactive risk management strategies. The development of predictive models based on machine learning algorithms further enhances the capability to anticipate potential safety issues before they manifest in operational systems.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!