How to Select ECM Electrolyte for Copper vs Stainless
MAY 5, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
ECM Electrolyte Technology Background and Objectives
Electrochemical machining (ECM) represents a non-traditional manufacturing process that utilizes controlled electrochemical dissolution to remove material from conductive workpieces. This technology emerged in the 1960s as an alternative to conventional machining methods, particularly for processing hard-to-machine materials and complex geometries. The fundamental principle involves the anodic dissolution of the workpiece material in an electrolytic solution, where the workpiece serves as the anode and the tool acts as the cathode.
The evolution of ECM technology has been closely tied to advancements in electrolyte chemistry and process control systems. Early implementations primarily focused on steel machining using sodium chloride solutions, but the expanding application scope has necessitated the development of specialized electrolytes for different material systems. The transition from simple salt solutions to sophisticated electrolyte formulations reflects the industry's growing understanding of electrochemical kinetics and material-specific dissolution mechanisms.
Contemporary ECM applications span aerospace, automotive, medical device manufacturing, and precision tooling industries. The technology's ability to machine complex internal geometries, maintain excellent surface finish, and avoid thermal damage has positioned it as a critical manufacturing process for high-value components. However, the selection of appropriate electrolytes remains a significant technical challenge, particularly when processing dissimilar materials such as copper and stainless steel within the same manufacturing environment.
The primary objective of ECM electrolyte technology development centers on achieving selective and controlled material removal while maintaining process stability and environmental compliance. For copper machining, the focus lies in managing the high dissolution rates and preventing copper ion accumulation that can lead to workpiece contamination and dimensional inaccuracies. Stainless steel processing presents different challenges, including passivation layer management and achieving uniform dissolution across varying alloy compositions.
Current research directions emphasize the development of adaptive electrolyte systems capable of optimizing performance for specific material combinations. This includes investigating pH-buffered solutions, chelating agents, and inhibitor systems that can modulate dissolution kinetics based on workpiece material properties. The ultimate goal involves creating intelligent electrolyte selection frameworks that consider material compatibility, process requirements, and environmental sustainability while maximizing machining efficiency and part quality.
The evolution of ECM technology has been closely tied to advancements in electrolyte chemistry and process control systems. Early implementations primarily focused on steel machining using sodium chloride solutions, but the expanding application scope has necessitated the development of specialized electrolytes for different material systems. The transition from simple salt solutions to sophisticated electrolyte formulations reflects the industry's growing understanding of electrochemical kinetics and material-specific dissolution mechanisms.
Contemporary ECM applications span aerospace, automotive, medical device manufacturing, and precision tooling industries. The technology's ability to machine complex internal geometries, maintain excellent surface finish, and avoid thermal damage has positioned it as a critical manufacturing process for high-value components. However, the selection of appropriate electrolytes remains a significant technical challenge, particularly when processing dissimilar materials such as copper and stainless steel within the same manufacturing environment.
The primary objective of ECM electrolyte technology development centers on achieving selective and controlled material removal while maintaining process stability and environmental compliance. For copper machining, the focus lies in managing the high dissolution rates and preventing copper ion accumulation that can lead to workpiece contamination and dimensional inaccuracies. Stainless steel processing presents different challenges, including passivation layer management and achieving uniform dissolution across varying alloy compositions.
Current research directions emphasize the development of adaptive electrolyte systems capable of optimizing performance for specific material combinations. This includes investigating pH-buffered solutions, chelating agents, and inhibitor systems that can modulate dissolution kinetics based on workpiece material properties. The ultimate goal involves creating intelligent electrolyte selection frameworks that consider material compatibility, process requirements, and environmental sustainability while maximizing machining efficiency and part quality.
Market Demand for Advanced ECM Processing Solutions
The global electrochemical machining market is experiencing significant growth driven by increasing demand for precision manufacturing across multiple industries. Aerospace and automotive sectors represent the largest consumer segments, requiring advanced ECM solutions for processing complex geometries in both copper and stainless steel components. The aerospace industry particularly demands high-precision machining of turbine blades, fuel injection systems, and heat exchangers where material-specific electrolyte selection becomes critical for achieving required surface finishes and dimensional tolerances.
Medical device manufacturing has emerged as a rapidly expanding market segment for ECM processing solutions. The production of surgical instruments, implants, and micro-components requires precise material removal capabilities that can handle both copper-based alloys and various stainless steel grades. The biocompatibility requirements and stringent quality standards in medical applications drive demand for specialized electrolyte formulations that can deliver consistent results across different material types.
The electronics industry presents substantial opportunities for advanced ECM processing, particularly in manufacturing connectors, heat sinks, and precision components. As electronic devices become increasingly miniaturized, the need for precise machining of copper conductors and stainless steel housings has intensified. This trend creates demand for electrolyte solutions that can maintain processing accuracy while switching between different materials within the same manufacturing line.
Automotive electrification is reshaping market demand patterns, with electric vehicle components requiring specialized ECM processing for battery cooling systems, electrical connectors, and power management components. These applications often involve processing both copper and stainless steel materials, necessitating flexible electrolyte selection strategies that can optimize processing parameters for each material type.
The market is also witnessing increased demand from the energy sector, particularly in renewable energy applications. Wind turbine components, solar panel manufacturing equipment, and energy storage systems require precision machining of various metal components. The ability to efficiently process both copper and stainless steel using optimized electrolyte solutions has become a competitive advantage for manufacturers serving this sector.
Regional market dynamics show strong growth in Asia-Pacific manufacturing hubs, where automotive and electronics production drives demand for advanced ECM solutions. European markets focus on aerospace and medical applications, while North American demand centers on automotive electrification and advanced manufacturing initiatives.
Medical device manufacturing has emerged as a rapidly expanding market segment for ECM processing solutions. The production of surgical instruments, implants, and micro-components requires precise material removal capabilities that can handle both copper-based alloys and various stainless steel grades. The biocompatibility requirements and stringent quality standards in medical applications drive demand for specialized electrolyte formulations that can deliver consistent results across different material types.
The electronics industry presents substantial opportunities for advanced ECM processing, particularly in manufacturing connectors, heat sinks, and precision components. As electronic devices become increasingly miniaturized, the need for precise machining of copper conductors and stainless steel housings has intensified. This trend creates demand for electrolyte solutions that can maintain processing accuracy while switching between different materials within the same manufacturing line.
Automotive electrification is reshaping market demand patterns, with electric vehicle components requiring specialized ECM processing for battery cooling systems, electrical connectors, and power management components. These applications often involve processing both copper and stainless steel materials, necessitating flexible electrolyte selection strategies that can optimize processing parameters for each material type.
The market is also witnessing increased demand from the energy sector, particularly in renewable energy applications. Wind turbine components, solar panel manufacturing equipment, and energy storage systems require precision machining of various metal components. The ability to efficiently process both copper and stainless steel using optimized electrolyte solutions has become a competitive advantage for manufacturers serving this sector.
Regional market dynamics show strong growth in Asia-Pacific manufacturing hubs, where automotive and electronics production drives demand for advanced ECM solutions. European markets focus on aerospace and medical applications, while North American demand centers on automotive electrification and advanced manufacturing initiatives.
Current ECM Electrolyte Challenges for Metal Processing
Electrochemical machining (ECM) electrolyte selection presents significant challenges when processing different metallic materials, particularly when transitioning between copper and stainless steel substrates. The fundamental issue stems from the vastly different electrochemical properties and dissolution behaviors of these materials, requiring distinct electrolyte compositions and operating parameters for optimal processing results.
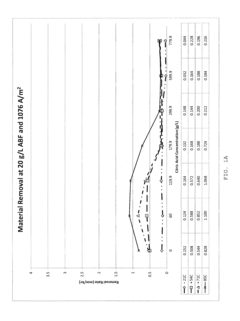
The primary challenge lies in the electrochemical potential differences between copper and stainless steel. Copper exhibits relatively low dissolution potentials and high conductivity, enabling efficient material removal with mild electrolytes such as sodium chloride or sodium nitrate solutions. Conversely, stainless steel's passive oxide layer and complex alloy composition demand more aggressive electrolytes, often requiring acidic solutions or specialized additives to achieve effective dissolution rates.
Electrolyte conductivity optimization represents another critical challenge. Copper processing typically benefits from moderate conductivity electrolytes that provide controlled dissolution without excessive heat generation. However, stainless steel machining requires higher conductivity solutions to overcome the material's inherent resistance to electrochemical attack, leading to increased power consumption and thermal management complexities.
Surface quality and dimensional accuracy present conflicting requirements between these materials. Copper's tendency toward uniform dissolution allows for precise control with neutral salt solutions, while stainless steel's heterogeneous microstructure and varying alloy element distribution can result in selective dissolution, requiring carefully balanced electrolyte compositions to maintain surface integrity.
The formation of insoluble precipitates poses significant operational challenges. Copper dissolution typically produces soluble ionic species that remain stable in most electrolyte systems. In contrast, stainless steel processing generates complex metal hydroxides and oxides that can precipitate within the electrolyte, leading to reduced conductivity, clogged flow systems, and compromised machining performance.
Temperature sensitivity varies dramatically between these materials during ECM processing. Copper machining can operate effectively across a wide temperature range with stable electrolyte performance. Stainless steel processing often requires elevated temperatures to enhance dissolution kinetics, but this increases electrolyte decomposition rates and necessitates more frequent solution replacement.
Corrosion management of ECM equipment presents material-specific challenges. Electrolytes suitable for copper processing are generally less corrosive to system components, while stainless steel electrolytes, particularly acidic formulations, can cause accelerated degradation of pumps, valves, and machining fixtures, increasing maintenance requirements and operational costs.
Environmental and safety considerations further complicate electrolyte selection. Copper-compatible electrolytes often utilize relatively benign salt solutions with manageable waste treatment requirements. Stainless steel electrolytes may contain hazardous acids or specialized additives that require enhanced safety protocols and more complex waste disposal procedures, impacting overall process economics and regulatory compliance.
The primary challenge lies in the electrochemical potential differences between copper and stainless steel. Copper exhibits relatively low dissolution potentials and high conductivity, enabling efficient material removal with mild electrolytes such as sodium chloride or sodium nitrate solutions. Conversely, stainless steel's passive oxide layer and complex alloy composition demand more aggressive electrolytes, often requiring acidic solutions or specialized additives to achieve effective dissolution rates.
Electrolyte conductivity optimization represents another critical challenge. Copper processing typically benefits from moderate conductivity electrolytes that provide controlled dissolution without excessive heat generation. However, stainless steel machining requires higher conductivity solutions to overcome the material's inherent resistance to electrochemical attack, leading to increased power consumption and thermal management complexities.
Surface quality and dimensional accuracy present conflicting requirements between these materials. Copper's tendency toward uniform dissolution allows for precise control with neutral salt solutions, while stainless steel's heterogeneous microstructure and varying alloy element distribution can result in selective dissolution, requiring carefully balanced electrolyte compositions to maintain surface integrity.
The formation of insoluble precipitates poses significant operational challenges. Copper dissolution typically produces soluble ionic species that remain stable in most electrolyte systems. In contrast, stainless steel processing generates complex metal hydroxides and oxides that can precipitate within the electrolyte, leading to reduced conductivity, clogged flow systems, and compromised machining performance.
Temperature sensitivity varies dramatically between these materials during ECM processing. Copper machining can operate effectively across a wide temperature range with stable electrolyte performance. Stainless steel processing often requires elevated temperatures to enhance dissolution kinetics, but this increases electrolyte decomposition rates and necessitates more frequent solution replacement.
Corrosion management of ECM equipment presents material-specific challenges. Electrolytes suitable for copper processing are generally less corrosive to system components, while stainless steel electrolytes, particularly acidic formulations, can cause accelerated degradation of pumps, valves, and machining fixtures, increasing maintenance requirements and operational costs.
Environmental and safety considerations further complicate electrolyte selection. Copper-compatible electrolytes often utilize relatively benign salt solutions with manageable waste treatment requirements. Stainless steel electrolytes may contain hazardous acids or specialized additives that require enhanced safety protocols and more complex waste disposal procedures, impacting overall process economics and regulatory compliance.
Current ECM Electrolyte Solutions for Different Metals
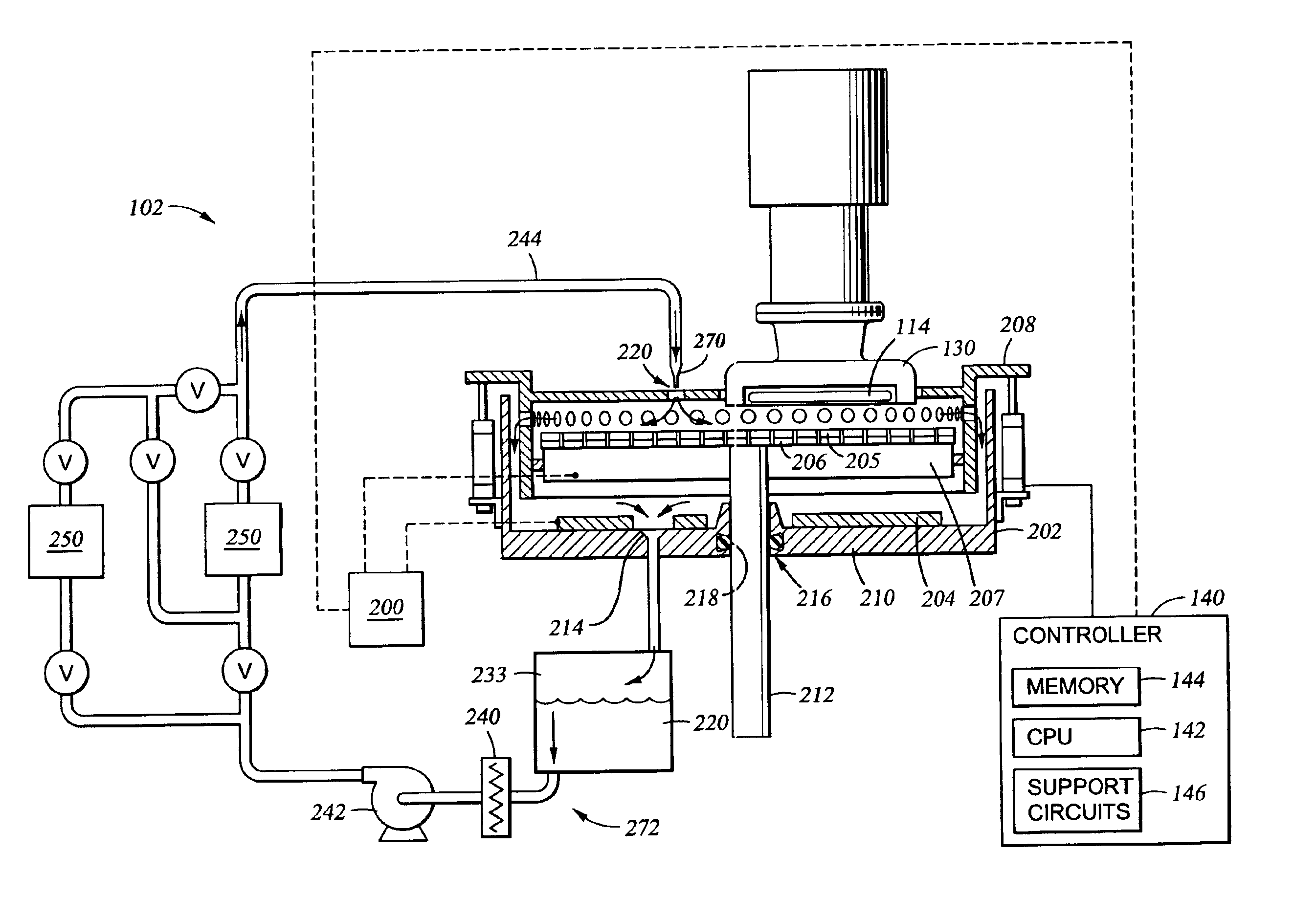
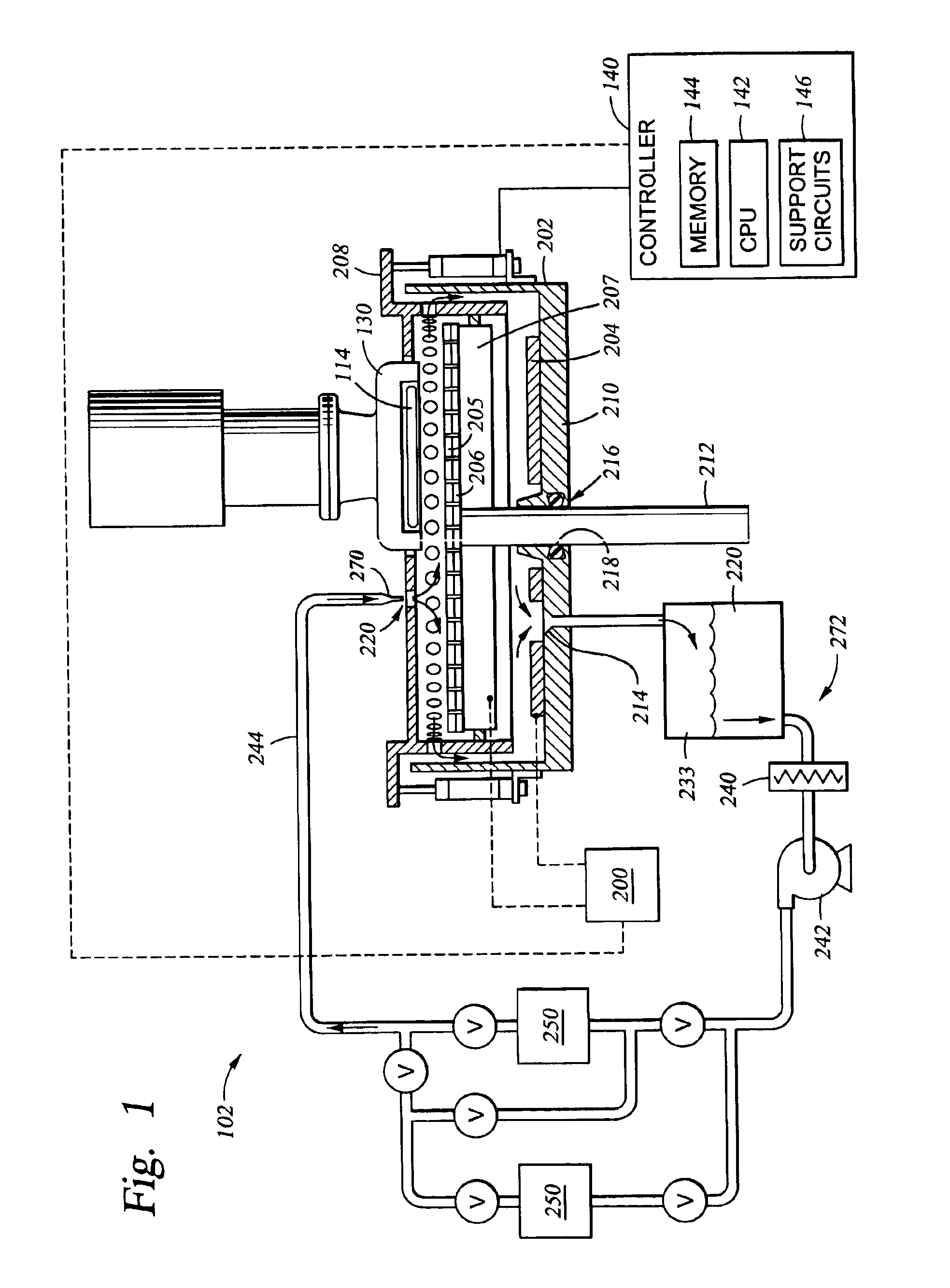
01 Electrolyte composition and chemical properties
The selection of electrolytes for ECM processes involves careful consideration of chemical composition, including ionic conductivity, chemical stability, and compatibility with electrode materials. Key factors include the electrolyte's ability to maintain consistent performance under varying operating conditions and its resistance to degradation over time.- Electrolyte composition and chemical properties: The selection of electrolytes for ECM processes involves careful consideration of chemical composition, including ionic conductivity, pH stability, and chemical compatibility with workpiece materials. The electrolyte must maintain consistent performance across varying operating conditions while providing optimal material removal rates and surface finish quality.
- Temperature and thermal management considerations: Thermal properties of electrolytes significantly impact ECM performance, requiring evaluation of temperature stability, thermal conductivity, and heat dissipation characteristics. Proper thermal management ensures consistent machining accuracy and prevents electrolyte degradation during high-current operations.
- Flow characteristics and hydrodynamic properties: Electrolyte flow behavior is critical for effective material removal and debris evacuation in ECM processes. Selection criteria include viscosity, flow rate optimization, pressure requirements, and the ability to maintain laminar flow conditions for precise machining control and uniform surface treatment.
- Environmental and safety compliance factors: Environmental impact and safety considerations play a crucial role in electrolyte selection, encompassing toxicity levels, biodegradability, waste disposal requirements, and worker safety protocols. Compliance with environmental regulations and minimization of hazardous waste generation are essential selection criteria.
- Economic and operational efficiency parameters: Cost-effectiveness and operational efficiency factors include electrolyte lifespan, recycling potential, maintenance requirements, and overall process economics. Selection involves balancing initial costs with long-term operational expenses, including electrolyte replacement frequency and system maintenance needs.
02 Concentration and molarity optimization
Optimal electrolyte concentration is critical for achieving desired machining rates and surface quality. The selection criteria focus on balancing conductivity with viscosity, ensuring adequate ion mobility while maintaining proper flow characteristics. Concentration levels must be tailored to specific machining requirements and workpiece materials.Expand Specific Solutions03 Temperature stability and thermal properties
Electrolyte selection must account for thermal stability across operating temperature ranges. Critical considerations include thermal expansion coefficients, boiling points, and temperature-dependent conductivity changes. The electrolyte must maintain consistent performance characteristics despite heat generation during the machining process.Expand Specific Solutions04 Environmental and safety considerations
Selection criteria encompass environmental impact, toxicity levels, disposal requirements, and worker safety protocols. Electrolytes must comply with environmental regulations while providing effective machining performance. Biodegradability, corrosiveness, and handling safety are key evaluation parameters.Expand Specific Solutions05 Machining performance and surface quality factors
Electrolyte selection directly impacts material removal rates, surface finish quality, and dimensional accuracy. Key criteria include the electrolyte's ability to facilitate uniform current distribution, minimize electrode wear, and produce desired surface characteristics. Performance metrics include machining speed, precision, and repeatability.Expand Specific Solutions
Key Players in ECM and Electrolyte Industry
The ECM electrolyte selection for copper versus stainless steel represents a mature industrial technology in the growth phase, with significant market expansion driven by precision manufacturing demands across aerospace, automotive, and electronics sectors. The market demonstrates substantial scale, particularly in semiconductor fabrication and automotive component production. Technology maturity varies significantly among key players: Taiwan Semiconductor Manufacturing Co. and Applied Materials lead in advanced semiconductor applications, while BASF Corp. and Johnson Matthey Plc dominate chemical formulation expertise. Asian manufacturers like SK Hynix and Sumitomo Electric Industries excel in high-volume production applications, whereas European players such as Aurubis AG focus on specialized metallurgical solutions. The competitive landscape shows clear segmentation between equipment manufacturers, chemical suppliers, and end-users, with increasing convergence toward integrated solutions combining optimized electrolyte chemistry with advanced process control systems.
Taiwan Semiconductor Manufacturing Co., Ltd.
Technical Solution: TSMC has developed sophisticated ECM electrolyte selection protocols for advanced semiconductor manufacturing processes involving both copper interconnects and stainless steel tooling. Their copper ECM processes utilize carefully balanced acidic electrolytes with specific copper ion concentrations to achieve uniform material removal while preventing dendrite formation. For stainless steel components, they employ neutral to slightly alkaline electrolytes with specialized additives to prevent passivation and ensure consistent machining rates. TSMC's approach includes comprehensive electrolyte characterization using advanced analytical techniques to optimize composition for specific alloy grades. They have implemented closed-loop electrolyte management systems with real-time monitoring and automated adjustment capabilities to maintain process stability and repeatability across high-volume production environments.
Strengths: Extensive semiconductor manufacturing experience, high-volume production capabilities, advanced quality control systems. Weaknesses: Focus primarily on semiconductor applications, may not be suitable for general industrial ECM applications, proprietary processes may limit accessibility.
Novellus Systems, Inc.
Technical Solution: Novellus Systems specializes in ECM electrolyte selection through their advanced electrochemical processing platforms. For copper applications, they employ acidic electrolytes containing copper ions to maintain electrochemical equilibrium and prevent copper redeposition. Their stainless steel ECM processes utilize specially formulated neutral electrolytes with passivation inhibitors to ensure consistent material removal without surface oxidation. The company has developed proprietary electrolyte circulation systems that maintain optimal temperature and concentration levels. Their approach includes real-time monitoring of electrolyte conductivity and pH levels, with automated dosing systems to maintain electrolyte chemistry within specified parameters. They also incorporate advanced filtration systems to remove machining debris and maintain electrolyte purity throughout the process.
Strengths: Specialized semiconductor processing expertise, advanced automation capabilities, precise process control. Weaknesses: Limited to specific applications, high maintenance requirements, expensive consumables.
Core Innovations in Copper vs Stainless ECM Electrolytes
Electrolyte with good planarization capability, high removal rate and smooth surface finish for electrochemically controlled copper CMP
PatentInactiveUS6863797B2
Innovation
- The use of electrolyte compositions containing chelating agents, corrosion inhibitors, and pH adjusting agents, including organic compounds with azole groups and polymeric inhibitors, to minimize substrate damage and enhance planarization efficiency during ECMP.
Electrolyte solution and electrochemical surface modification methods
PatentInactiveUS20170051428A1
Innovation
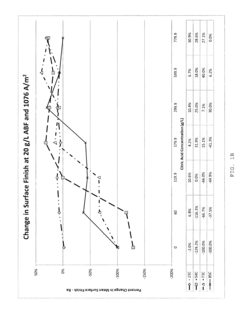
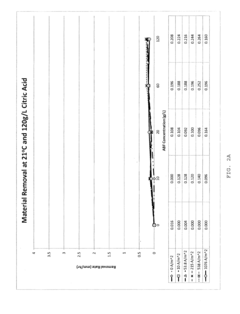
- Development of novel electropolishing bath chemistries using aqueous solutions with carboxylic acids, such as citric acid, and fluoride salts like ammonium bifluoride, substantially free of strong acids, to facilitate controlled alpha case removal and crack modulation through electropolishing.
Environmental Regulations for ECM Electrolyte Usage
The environmental regulatory landscape for ECM electrolyte usage has become increasingly stringent across major manufacturing regions, directly impacting the selection criteria for copper and stainless steel processing applications. In the United States, the Environmental Protection Agency (EPA) enforces strict discharge limits under the Clean Water Act, particularly for heavy metal concentrations in wastewater streams. European Union regulations under REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) impose comprehensive chemical safety assessments for electrolyte components, while RoHS directives restrict hazardous substances in manufacturing processes.
Copper ECM operations face specific regulatory challenges due to the inherent toxicity of copper ions in aquatic environments. The EPA's effluent guidelines for metal finishing operations set copper discharge limits as low as 2.07 mg/L for daily maximum concentrations. This necessitates robust wastewater treatment systems and careful electrolyte formulation to minimize copper leaching. Additionally, copper-containing electrolytes often require specialized disposal protocols under hazardous waste regulations, increasing operational costs and compliance complexity.
Stainless steel ECM electrolytes encounter different regulatory pressures, primarily related to chromium and nickel content management. Hexavalent chromium, a potential byproduct in certain electrolyte formulations, is subject to extremely strict regulations with workplace exposure limits of 5 μg/m³ under OSHA standards. The European Chemicals Agency has classified several nickel compounds as carcinogenic, requiring enhanced safety protocols and worker protection measures during electrolyte handling and processing.
Regional variations in environmental standards significantly influence electrolyte selection strategies. Asian markets, particularly China and Japan, have implemented increasingly stringent environmental protection laws that mirror Western standards. China's revised Environmental Protection Law imposes severe penalties for non-compliance, while Japan's Chemical Substances Control Law requires extensive testing for new electrolyte formulations.
Emerging regulatory trends focus on sustainability metrics and lifecycle assessments of electrolyte systems. The EU's Green Deal initiatives promote circular economy principles, encouraging the development of recyclable and biodegradable electrolyte components. These evolving standards are driving innovation toward environmentally benign electrolyte formulations that maintain processing effectiveness while meeting stringent environmental compliance requirements for both copper and stainless steel ECM applications.
Copper ECM operations face specific regulatory challenges due to the inherent toxicity of copper ions in aquatic environments. The EPA's effluent guidelines for metal finishing operations set copper discharge limits as low as 2.07 mg/L for daily maximum concentrations. This necessitates robust wastewater treatment systems and careful electrolyte formulation to minimize copper leaching. Additionally, copper-containing electrolytes often require specialized disposal protocols under hazardous waste regulations, increasing operational costs and compliance complexity.
Stainless steel ECM electrolytes encounter different regulatory pressures, primarily related to chromium and nickel content management. Hexavalent chromium, a potential byproduct in certain electrolyte formulations, is subject to extremely strict regulations with workplace exposure limits of 5 μg/m³ under OSHA standards. The European Chemicals Agency has classified several nickel compounds as carcinogenic, requiring enhanced safety protocols and worker protection measures during electrolyte handling and processing.
Regional variations in environmental standards significantly influence electrolyte selection strategies. Asian markets, particularly China and Japan, have implemented increasingly stringent environmental protection laws that mirror Western standards. China's revised Environmental Protection Law imposes severe penalties for non-compliance, while Japan's Chemical Substances Control Law requires extensive testing for new electrolyte formulations.
Emerging regulatory trends focus on sustainability metrics and lifecycle assessments of electrolyte systems. The EU's Green Deal initiatives promote circular economy principles, encouraging the development of recyclable and biodegradable electrolyte components. These evolving standards are driving innovation toward environmentally benign electrolyte formulations that maintain processing effectiveness while meeting stringent environmental compliance requirements for both copper and stainless steel ECM applications.
Safety Standards in ECM Electrolyte Handling
The handling of ECM electrolytes for copper and stainless steel processing requires strict adherence to established safety standards due to the corrosive and potentially hazardous nature of these chemical solutions. International safety frameworks such as OSHA regulations, ISO 45001 occupational health standards, and NFPA chemical handling guidelines form the foundation for safe electrolyte management protocols.
Personal protective equipment requirements vary significantly between copper and stainless steel ECM operations. Copper electrolyte handling typically involves acidic solutions containing sulfuric acid or phosphoric acid, necessitating chemical-resistant gloves, face shields, and acid-proof aprons. Stainless steel electrolytes often contain more aggressive chemicals including nitric acid mixtures, requiring enhanced respiratory protection and specialized ventilation systems to manage toxic vapor emissions.
Containment and spill response protocols must be tailored to the specific electrolyte chemistry. Secondary containment systems should accommodate at least 110% of the primary container volume, with chemical-compatible materials selected based on electrolyte composition. Emergency response procedures require immediate access to appropriate neutralizing agents - sodium bicarbonate solutions for acidic copper electrolytes and specialized alkaline neutralizers for stainless steel processing chemicals.
Storage requirements mandate temperature-controlled environments with appropriate segregation between incompatible chemicals. Copper electrolytes typically require storage temperatures between 15-25°C, while certain stainless steel electrolytes may need refrigerated conditions to prevent decomposition. Automated monitoring systems should track temperature, humidity, and chemical vapor concentrations continuously.
Waste disposal protocols must comply with environmental regulations including EPA hazardous waste classifications and local water treatment requirements. Spent copper electrolytes often contain dissolved metal ions requiring specialized treatment before disposal, while stainless steel electrolyte waste may contain chromium compounds subject to strict environmental controls.
Training and certification programs should encompass chemical hazard recognition, proper handling techniques, emergency response procedures, and regular safety audits. Documentation requirements include material safety data sheets, handling logs, and incident reporting systems to ensure continuous improvement in safety performance and regulatory compliance.
Personal protective equipment requirements vary significantly between copper and stainless steel ECM operations. Copper electrolyte handling typically involves acidic solutions containing sulfuric acid or phosphoric acid, necessitating chemical-resistant gloves, face shields, and acid-proof aprons. Stainless steel electrolytes often contain more aggressive chemicals including nitric acid mixtures, requiring enhanced respiratory protection and specialized ventilation systems to manage toxic vapor emissions.
Containment and spill response protocols must be tailored to the specific electrolyte chemistry. Secondary containment systems should accommodate at least 110% of the primary container volume, with chemical-compatible materials selected based on electrolyte composition. Emergency response procedures require immediate access to appropriate neutralizing agents - sodium bicarbonate solutions for acidic copper electrolytes and specialized alkaline neutralizers for stainless steel processing chemicals.
Storage requirements mandate temperature-controlled environments with appropriate segregation between incompatible chemicals. Copper electrolytes typically require storage temperatures between 15-25°C, while certain stainless steel electrolytes may need refrigerated conditions to prevent decomposition. Automated monitoring systems should track temperature, humidity, and chemical vapor concentrations continuously.
Waste disposal protocols must comply with environmental regulations including EPA hazardous waste classifications and local water treatment requirements. Spent copper electrolytes often contain dissolved metal ions requiring specialized treatment before disposal, while stainless steel electrolyte waste may contain chromium compounds subject to strict environmental controls.
Training and certification programs should encompass chemical hazard recognition, proper handling techniques, emergency response procedures, and regular safety audits. Documentation requirements include material safety data sheets, handling logs, and incident reporting systems to ensure continuous improvement in safety performance and regulatory compliance.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!