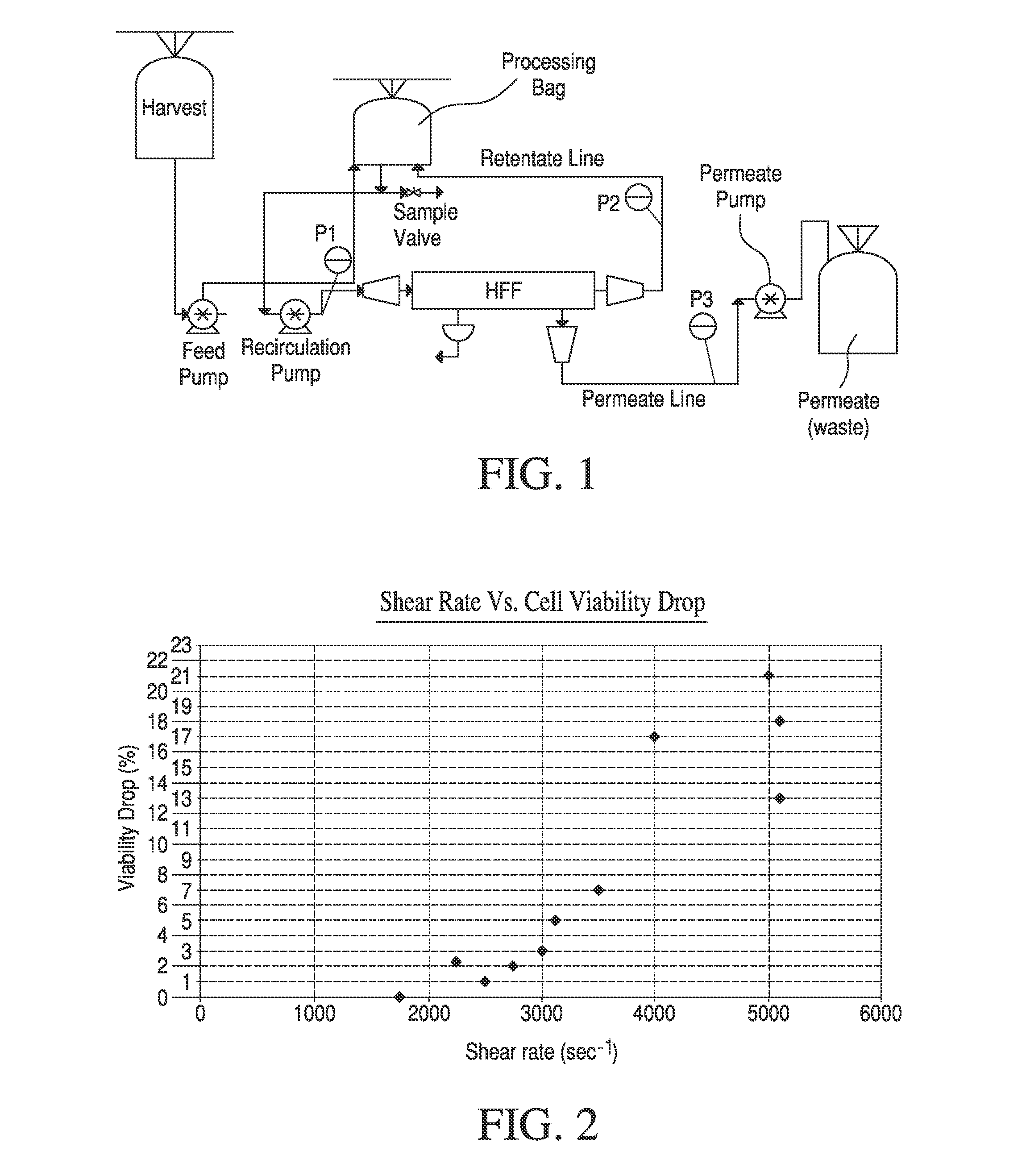
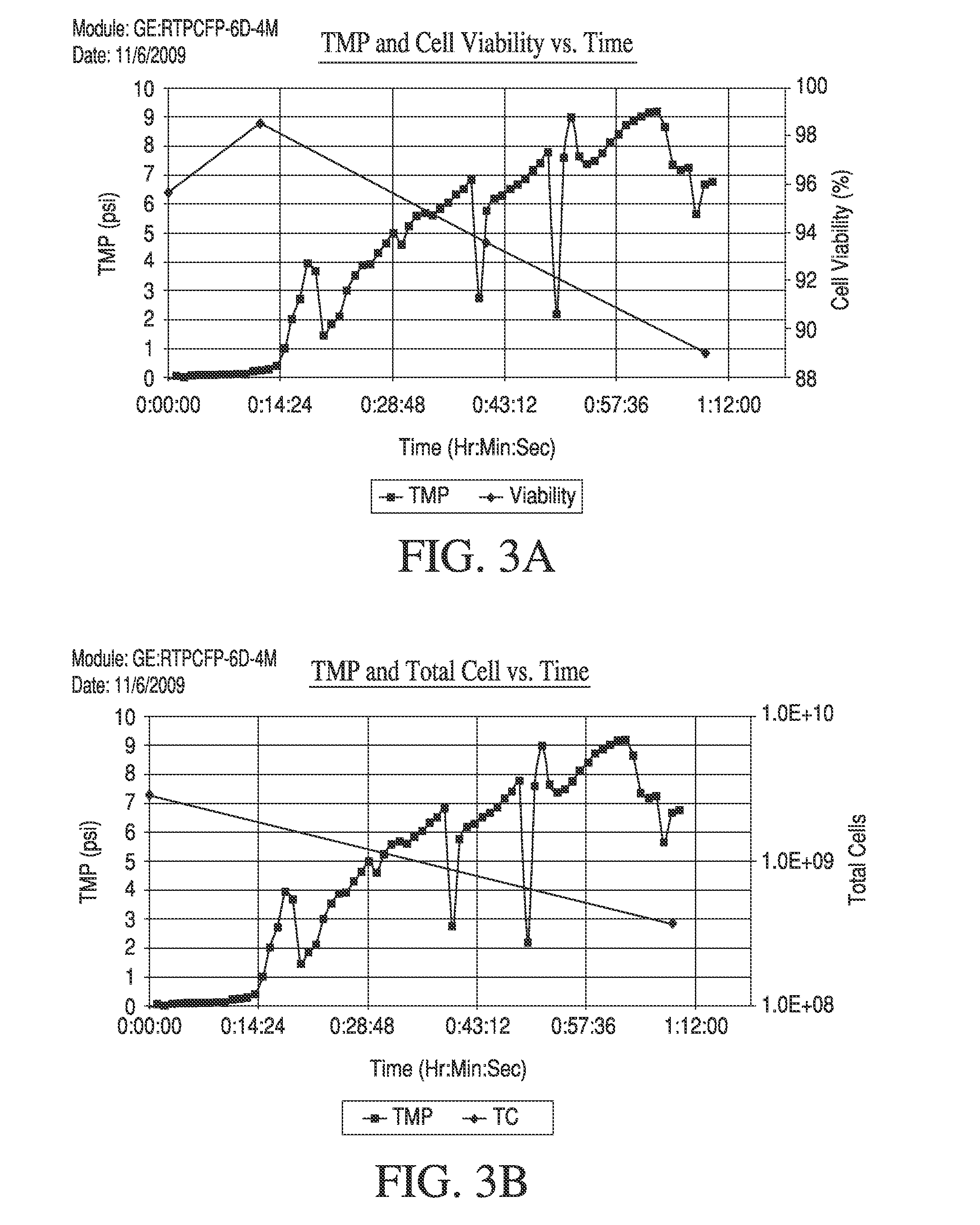
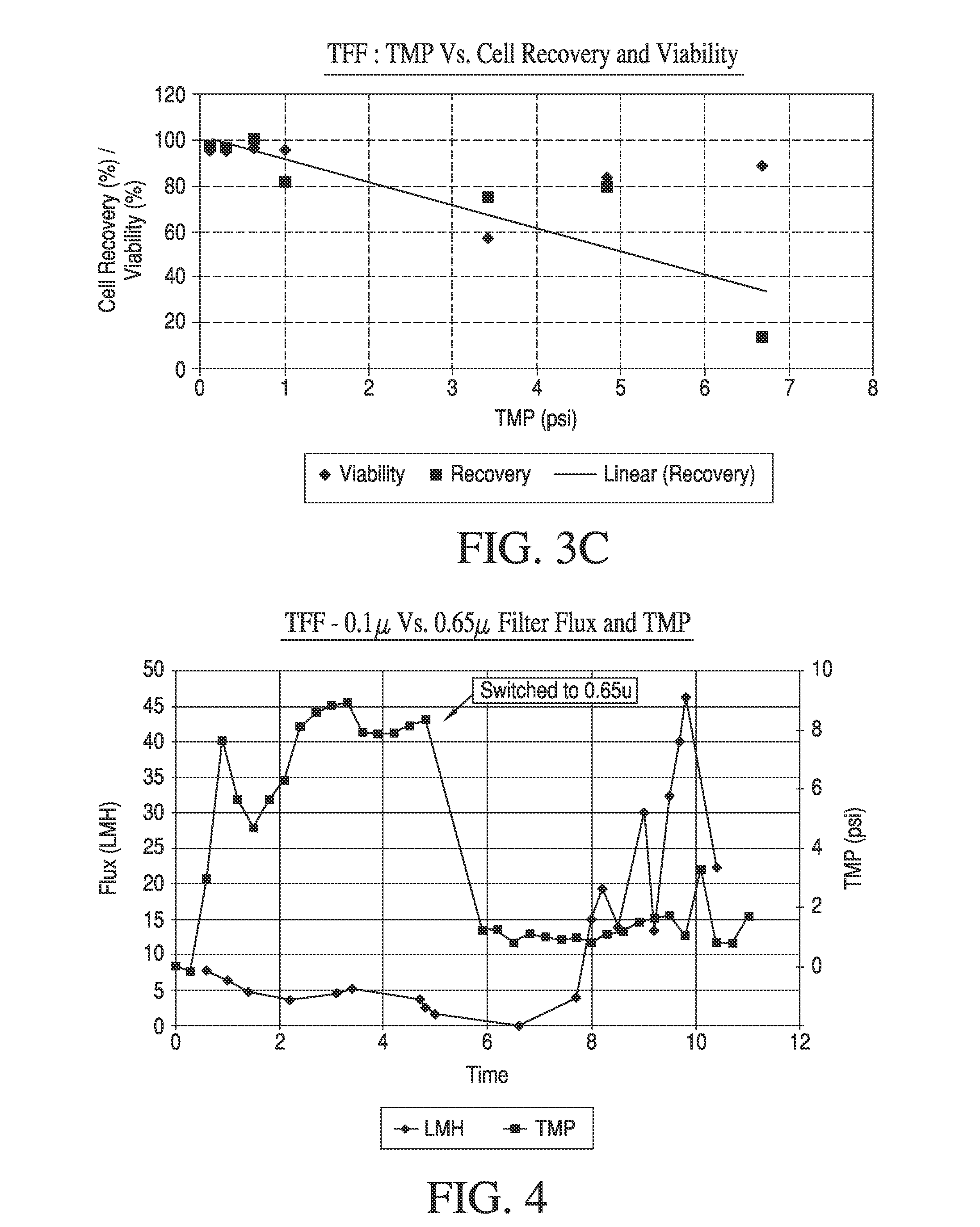
Maximizing Yield Consistency in Tangential Flow Filtration Setups
MAR 16, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
TFF Yield Optimization Background and Objectives
Tangential Flow Filtration has emerged as a critical separation technology in biotechnology and pharmaceutical manufacturing, fundamentally transforming how industries approach product purification and concentration processes. Unlike traditional dead-end filtration methods, TFF operates by flowing the feed solution parallel to the membrane surface, creating a cross-flow that minimizes membrane fouling and enables continuous processing. This technology has become indispensable in applications ranging from protein purification and virus concentration to cell harvesting and buffer exchange operations.
The evolution of TFF technology spans several decades, beginning with its initial development in the 1960s for water treatment applications and subsequently advancing into sophisticated bioprocessing systems. Early implementations focused primarily on basic separation functions, but modern TFF systems have evolved to incorporate advanced process control mechanisms, real-time monitoring capabilities, and automated optimization features. The technology has progressed from simple laboratory-scale devices to complex industrial systems capable of processing thousands of liters while maintaining precise control over critical process parameters.
Current market demands in biopharmaceutical manufacturing have intensified the focus on yield consistency as a paramount objective. Regulatory agencies increasingly require manufacturers to demonstrate robust process control and reproducible outcomes across production batches. The growing complexity of biological products, including monoclonal antibodies, gene therapies, and vaccine components, has elevated the importance of maintaining consistent recovery rates while preserving product quality and biological activity.
The primary technical objective in maximizing yield consistency centers on achieving predictable and reproducible recovery rates across multiple processing cycles and different operational conditions. This involves establishing optimal transmembrane pressure profiles, maintaining consistent flux rates throughout the process duration, and minimizing product loss due to membrane interactions or process variability. Secondary objectives include reducing processing time variability, minimizing membrane fouling impacts, and ensuring scalable performance from development through commercial manufacturing.
Contemporary challenges in TFF yield optimization encompass membrane selection criteria, process parameter standardization, and real-time process monitoring implementation. The industry seeks to develop predictive models that can anticipate yield variations based on feed characteristics, environmental conditions, and equipment performance parameters. Additionally, there is growing emphasis on developing adaptive control strategies that can automatically adjust process conditions to maintain target yield levels despite variations in input materials or operating conditions.
The evolution of TFF technology spans several decades, beginning with its initial development in the 1960s for water treatment applications and subsequently advancing into sophisticated bioprocessing systems. Early implementations focused primarily on basic separation functions, but modern TFF systems have evolved to incorporate advanced process control mechanisms, real-time monitoring capabilities, and automated optimization features. The technology has progressed from simple laboratory-scale devices to complex industrial systems capable of processing thousands of liters while maintaining precise control over critical process parameters.
Current market demands in biopharmaceutical manufacturing have intensified the focus on yield consistency as a paramount objective. Regulatory agencies increasingly require manufacturers to demonstrate robust process control and reproducible outcomes across production batches. The growing complexity of biological products, including monoclonal antibodies, gene therapies, and vaccine components, has elevated the importance of maintaining consistent recovery rates while preserving product quality and biological activity.
The primary technical objective in maximizing yield consistency centers on achieving predictable and reproducible recovery rates across multiple processing cycles and different operational conditions. This involves establishing optimal transmembrane pressure profiles, maintaining consistent flux rates throughout the process duration, and minimizing product loss due to membrane interactions or process variability. Secondary objectives include reducing processing time variability, minimizing membrane fouling impacts, and ensuring scalable performance from development through commercial manufacturing.
Contemporary challenges in TFF yield optimization encompass membrane selection criteria, process parameter standardization, and real-time process monitoring implementation. The industry seeks to develop predictive models that can anticipate yield variations based on feed characteristics, environmental conditions, and equipment performance parameters. Additionally, there is growing emphasis on developing adaptive control strategies that can automatically adjust process conditions to maintain target yield levels despite variations in input materials or operating conditions.
Market Demand for Consistent TFF Performance
The biopharmaceutical industry faces mounting pressure to deliver consistent, high-quality products while managing escalating production costs and regulatory compliance requirements. Tangential Flow Filtration has emerged as a critical unit operation in downstream processing, particularly for monoclonal antibodies, vaccines, and cell and gene therapies. The demand for consistent TFF performance stems from the technology's direct impact on product yield, quality, and overall manufacturing economics.
Market drivers for enhanced TFF consistency are multifaceted. Regulatory agencies worldwide have intensified their focus on process consistency and control, requiring manufacturers to demonstrate robust process understanding and predictable outcomes. The FDA's Process Validation Guidance and ICH Q8-Q12 guidelines emphasize the importance of process consistency throughout the product lifecycle. This regulatory landscape creates substantial demand for TFF systems that can deliver reproducible results across multiple batches and manufacturing sites.
The growing complexity of biopharmaceutical products amplifies the need for consistent TFF performance. Next-generation biologics, including bispecific antibodies, antibody-drug conjugates, and viral vectors for gene therapy, present unique filtration challenges. These products often exhibit variable aggregation behavior, concentration-dependent viscosity changes, and sensitivity to shear stress, making consistent processing outcomes more difficult to achieve but increasingly valuable.
Economic factors significantly influence market demand for TFF consistency solutions. Product losses due to inconsistent filtration performance can result in substantial financial impact, particularly for high-value biologics where raw materials and upstream processing costs are considerable. Manufacturing facilities seek TFF technologies that minimize batch-to-batch variability to reduce the risk of product loss and associated regulatory investigations.
The trend toward continuous manufacturing and integrated bioprocessing platforms creates additional demand for consistent TFF performance. As manufacturers move away from traditional batch processing, the need for predictable and stable filtration performance becomes paramount. Continuous TFF systems must maintain consistent separation efficiency and flux rates over extended operating periods to ensure overall process stability.
Contract manufacturing organizations represent a significant market segment driving demand for consistent TFF solutions. These organizations must demonstrate process robustness across diverse product portfolios and varying operating conditions. The ability to achieve consistent performance regardless of product characteristics or operator variability provides competitive advantages in securing manufacturing contracts.
Emerging markets in Asia-Pacific and Latin America are experiencing rapid growth in biopharmaceutical manufacturing capacity. These regions demand TFF technologies that can deliver consistent performance with potentially less experienced operators and varying infrastructure conditions, further expanding the market for robust, consistent TFF solutions.
Market drivers for enhanced TFF consistency are multifaceted. Regulatory agencies worldwide have intensified their focus on process consistency and control, requiring manufacturers to demonstrate robust process understanding and predictable outcomes. The FDA's Process Validation Guidance and ICH Q8-Q12 guidelines emphasize the importance of process consistency throughout the product lifecycle. This regulatory landscape creates substantial demand for TFF systems that can deliver reproducible results across multiple batches and manufacturing sites.
The growing complexity of biopharmaceutical products amplifies the need for consistent TFF performance. Next-generation biologics, including bispecific antibodies, antibody-drug conjugates, and viral vectors for gene therapy, present unique filtration challenges. These products often exhibit variable aggregation behavior, concentration-dependent viscosity changes, and sensitivity to shear stress, making consistent processing outcomes more difficult to achieve but increasingly valuable.
Economic factors significantly influence market demand for TFF consistency solutions. Product losses due to inconsistent filtration performance can result in substantial financial impact, particularly for high-value biologics where raw materials and upstream processing costs are considerable. Manufacturing facilities seek TFF technologies that minimize batch-to-batch variability to reduce the risk of product loss and associated regulatory investigations.
The trend toward continuous manufacturing and integrated bioprocessing platforms creates additional demand for consistent TFF performance. As manufacturers move away from traditional batch processing, the need for predictable and stable filtration performance becomes paramount. Continuous TFF systems must maintain consistent separation efficiency and flux rates over extended operating periods to ensure overall process stability.
Contract manufacturing organizations represent a significant market segment driving demand for consistent TFF solutions. These organizations must demonstrate process robustness across diverse product portfolios and varying operating conditions. The ability to achieve consistent performance regardless of product characteristics or operator variability provides competitive advantages in securing manufacturing contracts.
Emerging markets in Asia-Pacific and Latin America are experiencing rapid growth in biopharmaceutical manufacturing capacity. These regions demand TFF technologies that can deliver consistent performance with potentially less experienced operators and varying infrastructure conditions, further expanding the market for robust, consistent TFF solutions.
Current TFF Yield Variability Challenges
Tangential Flow Filtration systems face significant yield variability challenges that directly impact process economics and product quality consistency. Current industry data indicates yield variations ranging from 15-35% across different batches, even when operating under seemingly identical conditions. This variability stems from multiple interconnected factors that create a complex web of process uncertainties.
Membrane fouling represents the most critical challenge affecting yield consistency. Protein aggregation, particulate accumulation, and concentration polarization create dynamic resistance patterns that vary unpredictably between runs. The heterogeneous nature of biological feedstocks introduces compositional variations that interact differently with membrane surfaces, leading to inconsistent filtration performance. Even minor changes in feed stream characteristics can trigger cascade effects that dramatically alter permeate flux and product recovery rates.
Temperature fluctuations during processing create another layer of complexity. Viscosity changes, protein stability variations, and altered membrane permeability characteristics all contribute to yield inconsistencies. Current temperature control systems often lack the precision required to maintain optimal conditions throughout extended filtration cycles, particularly in large-scale operations where thermal gradients become more pronounced.
Transmembrane pressure management presents ongoing difficulties in maintaining consistent driving forces. Pressure decay due to membrane compaction, pump performance variations, and system hydraulics create dynamic conditions that operators struggle to control effectively. The relationship between pressure, flux, and yield is non-linear and highly sensitive to process conditions, making predictive control challenging.
Crossflow velocity optimization remains problematic across different scales and applications. Insufficient velocity leads to concentration polarization and reduced mass transfer, while excessive velocity can cause shear-induced product degradation. The optimal velocity window is narrow and varies with feed composition, concentration factors, and membrane characteristics, making standardization difficult.
Concentration factor determination lacks standardized approaches, with many operations relying on empirical methods rather than systematic optimization. The relationship between concentration endpoints and final yield is complex and product-specific, leading to conservative processing approaches that sacrifice potential yield for consistency assurance.
System cleaning and sanitization protocols introduce additional variability sources. Incomplete cleaning can leave residual fouling layers that affect subsequent runs, while aggressive cleaning may alter membrane properties. The lack of real-time cleaning validation methods means operators often rely on time-based protocols that may be insufficient or excessive for actual conditions.
Membrane fouling represents the most critical challenge affecting yield consistency. Protein aggregation, particulate accumulation, and concentration polarization create dynamic resistance patterns that vary unpredictably between runs. The heterogeneous nature of biological feedstocks introduces compositional variations that interact differently with membrane surfaces, leading to inconsistent filtration performance. Even minor changes in feed stream characteristics can trigger cascade effects that dramatically alter permeate flux and product recovery rates.
Temperature fluctuations during processing create another layer of complexity. Viscosity changes, protein stability variations, and altered membrane permeability characteristics all contribute to yield inconsistencies. Current temperature control systems often lack the precision required to maintain optimal conditions throughout extended filtration cycles, particularly in large-scale operations where thermal gradients become more pronounced.
Transmembrane pressure management presents ongoing difficulties in maintaining consistent driving forces. Pressure decay due to membrane compaction, pump performance variations, and system hydraulics create dynamic conditions that operators struggle to control effectively. The relationship between pressure, flux, and yield is non-linear and highly sensitive to process conditions, making predictive control challenging.
Crossflow velocity optimization remains problematic across different scales and applications. Insufficient velocity leads to concentration polarization and reduced mass transfer, while excessive velocity can cause shear-induced product degradation. The optimal velocity window is narrow and varies with feed composition, concentration factors, and membrane characteristics, making standardization difficult.
Concentration factor determination lacks standardized approaches, with many operations relying on empirical methods rather than systematic optimization. The relationship between concentration endpoints and final yield is complex and product-specific, leading to conservative processing approaches that sacrifice potential yield for consistency assurance.
System cleaning and sanitization protocols introduce additional variability sources. Incomplete cleaning can leave residual fouling layers that affect subsequent runs, while aggressive cleaning may alter membrane properties. The lack of real-time cleaning validation methods means operators often rely on time-based protocols that may be insufficient or excessive for actual conditions.
Existing TFF Yield Maximization Solutions
01 Optimization of filtration membrane configuration and materials
Tangential flow filtration yield consistency can be improved by optimizing the membrane configuration, including membrane material selection, pore size distribution, and surface area. The use of specific membrane materials with controlled porosity and chemical compatibility ensures consistent filtration performance across batches. Proper membrane selection based on the target molecule size and process requirements helps maintain uniform yield and product quality.- Optimization of filtration membrane configuration and design: Tangential flow filtration yield consistency can be improved through optimized membrane configuration, including selection of appropriate membrane materials, pore sizes, and surface areas. The design of filtration cassettes and modules with specific flow patterns and pressure distributions helps maintain consistent product recovery rates. Advanced membrane geometries and arrangements enable uniform filtration performance across multiple batches.
- Process parameter control and monitoring systems: Maintaining consistent yield in tangential flow filtration requires precise control of operational parameters such as transmembrane pressure, crossflow velocity, temperature, and feed concentration. Real-time monitoring systems and automated feedback controls help maintain optimal conditions throughout the filtration process. Implementation of standardized operating procedures and parameter ranges ensures reproducible results across different production runs.
- Buffer composition and formulation optimization: The consistency of tangential flow filtration yield is significantly influenced by buffer systems and solution formulations used during the process. Optimized buffer compositions help prevent protein aggregation, maintain product stability, and reduce membrane fouling. Proper pH control, ionic strength adjustment, and addition of stabilizing agents contribute to reproducible filtration performance and product recovery.
- Membrane cleaning and regeneration protocols: Consistent filtration yields depend on effective membrane maintenance strategies including cleaning-in-place procedures and regeneration protocols. Regular cleaning cycles using appropriate chemical agents remove fouling materials and restore membrane permeability. Validated cleaning procedures and membrane lifetime management ensure consistent performance across multiple use cycles and prevent yield variability due to membrane degradation.
- Scale-up and manufacturing consistency strategies: Achieving consistent yields during scale-up from laboratory to manufacturing scale requires careful consideration of equipment design, process scaling factors, and validation protocols. Implementation of quality-by-design principles and process analytical technology enables consistent performance across different scales. Standardized equipment qualification, process validation, and batch-to-batch monitoring ensure reproducible yields in commercial production.
02 Process parameter control and monitoring
Maintaining consistent yield in tangential flow filtration requires precise control of process parameters such as transmembrane pressure, crossflow velocity, temperature, and feed concentration. Real-time monitoring systems and automated control mechanisms help maintain these parameters within optimal ranges throughout the filtration process. Standardized operating procedures and parameter setpoints ensure reproducible results across different production runs.Expand Specific Solutions03 Membrane fouling prevention and cleaning strategies
Consistent filtration yield depends on effective strategies to prevent and manage membrane fouling. This includes implementing appropriate pre-filtration steps, optimizing feed solution conditions, and establishing validated cleaning protocols. Regular membrane cleaning and sanitization procedures using specific cleaning agents help restore membrane performance and maintain consistent permeability and selectivity over multiple cycles.Expand Specific Solutions04 System design and equipment standardization
Achieving yield consistency requires standardized tangential flow filtration system design with appropriate equipment sizing, flow path configuration, and component selection. This includes proper pump selection, pressure sensor placement, and flow distribution systems that ensure uniform conditions across the membrane surface. Modular system designs and scalable configurations enable consistent performance from laboratory to production scale.Expand Specific Solutions05 Quality control and validation methods
Ensuring tangential flow filtration yield consistency requires comprehensive quality control measures and validation protocols. This includes establishing critical quality attributes, implementing in-process testing, and conducting regular system qualification. Statistical process control methods and batch-to-batch comparison help identify variations and maintain consistent product recovery and purity across manufacturing campaigns.Expand Specific Solutions
Key Players in TFF Equipment and Membrane Industry
The tangential flow filtration (TFF) market for yield consistency optimization represents a mature but rapidly evolving sector within bioprocessing, driven by increasing biopharmaceutical manufacturing demands. The industry has reached commercial maturity with established players like EMD Millipore Corp. and Repligen Corp. leading through their comprehensive filtration portfolios and ATF systems. Market growth is substantial, fueled by expanding biologics production and regulatory emphasis on process consistency. Technology maturity varies significantly across the competitive landscape - while traditional filtration leaders like Repligen and EMD Millipore demonstrate advanced capabilities with proven commercial systems, emerging players such as Suzhou Yingsaisi and PendoTECH are introducing innovative monitoring and control technologies. Major pharmaceutical companies including Eli Lilly, Bristol Myers Squibb, and AstraZeneca are driving demand through their manufacturing requirements, while specialized equipment providers like Hangzhou Cobetter and InnovaPrep focus on niche filtration solutions, creating a diverse ecosystem spanning from established multinational corporations to innovative technology startups.
EMD Millipore Corp.
Technical Solution: EMD Millipore has developed advanced tangential flow filtration systems with integrated process analytical technology (PAT) for real-time monitoring and control. Their Pellicon cassettes feature optimized membrane chemistry and uniform flow distribution designs that minimize fouling and maintain consistent permeate flux throughout extended filtration runs. The company's Mobius FlexReady solutions incorporate single-use TFF systems with pre-validated scalability from laboratory to manufacturing scale, ensuring reproducible yield performance. Their proprietary membrane surface modifications and controlled pore size distribution technology help maintain consistent separation efficiency across different batches and operating conditions.
Strengths: Market-leading membrane technology with proven scalability and regulatory acceptance. Weaknesses: Higher cost compared to traditional systems and dependency on single-use components.
Repligen Corp.
Technical Solution: Repligen has developed the TangenX TFF platform featuring advanced hollow fiber membrane technology specifically designed for biopharmaceutical applications. Their system incorporates automated backpulsing capabilities and real-time turbidity monitoring to prevent membrane fouling and maintain consistent filtration performance. The company's FlowVPE (Variable Pathlength Extension) technology enables precise control of transmembrane pressure and crossflow velocity, optimizing yield consistency across different product types. Their integrated control systems use machine learning algorithms to predict and adjust operating parameters in real-time, minimizing batch-to-batch variability and maximizing product recovery rates throughout the filtration process.
Strengths: Advanced automation and predictive control capabilities with strong biopharmaceutical focus. Weaknesses: Limited to specific membrane configurations and requires specialized training for operation.
Core Patents in TFF Process Optimization
High yield method and apparatus for volume reduction and washing of therapeutic cells using tangential flow filtration
PatentInactiveUS20120294836A1
Innovation
- The use of Tangential Flow Filtration (TFF) with optimized parameters such as low trans-membrane pressure and shear rates to concentrate and wash live mammalian cells, achieving high yields and viability while minimizing processing time and residual contaminants, using a completely closed and scalable system.
High yield method and apparatus for volume reduction and washing of therapeutic cells using tangential flow filtration
PatentInactiveEP2525899A1
Innovation
- A high-yield Tangential Flow Filtration (TFF) process that reduces the volume of mammalian cell suspensions by maintaining low transmembrane pressure and shear rates, achieving high cell concentrations and viability, and incorporating a diafiltration step to minimize residual components, using either hollow fiber or flat sheet filters, and a completely closed, disposable TFF system for efficient processing.
Quality Standards for TFF Manufacturing
Quality standards for TFF manufacturing represent a critical framework that ensures consistent performance and regulatory compliance across tangential flow filtration operations. These standards encompass multiple dimensions including equipment qualification, process validation, documentation requirements, and continuous monitoring protocols that collectively support yield consistency objectives.
The foundation of TFF quality standards begins with equipment design qualification and installation qualification protocols. Manufacturing facilities must establish comprehensive specifications for membrane materials, housing integrity, pressure ratings, and flow distribution systems. These specifications directly impact filtration performance and yield predictability, requiring rigorous testing procedures to verify that equipment meets predetermined operational parameters before deployment.
Process validation standards form another essential component, mandating systematic approaches to demonstrate that TFF processes consistently produce results meeting predetermined specifications. This includes establishing critical process parameters such as transmembrane pressure, crossflow velocity, temperature control, and concentration factors. Validation protocols must demonstrate process robustness across expected operating ranges while maintaining yield consistency targets.
Documentation and traceability requirements constitute fundamental quality standards that enable effective process control and continuous improvement. Manufacturing operations must maintain detailed records of membrane lot information, cleaning validation data, process parameter logs, and yield performance metrics. These documentation standards facilitate root cause analysis when yield variations occur and support regulatory compliance requirements.
Quality control testing standards define specific analytical methods and acceptance criteria for both in-process monitoring and final product release. These standards typically include turbidity measurements, bioburden testing, endotoxin levels, and product recovery calculations. Establishing clear testing protocols ensures that yield consistency issues are detected promptly and addressed systematically.
Environmental control standards address factors that can significantly impact TFF performance, including cleanroom classifications, temperature and humidity controls, and contamination prevention measures. These standards recognize that environmental variations can introduce process variability that ultimately affects yield consistency, requiring careful monitoring and control systems.
Continuous improvement standards mandate regular review of quality metrics, trend analysis of yield performance data, and implementation of corrective and preventive actions. These standards ensure that quality systems evolve to address emerging challenges and incorporate technological advances that enhance yield consistency in TFF manufacturing operations.
The foundation of TFF quality standards begins with equipment design qualification and installation qualification protocols. Manufacturing facilities must establish comprehensive specifications for membrane materials, housing integrity, pressure ratings, and flow distribution systems. These specifications directly impact filtration performance and yield predictability, requiring rigorous testing procedures to verify that equipment meets predetermined operational parameters before deployment.
Process validation standards form another essential component, mandating systematic approaches to demonstrate that TFF processes consistently produce results meeting predetermined specifications. This includes establishing critical process parameters such as transmembrane pressure, crossflow velocity, temperature control, and concentration factors. Validation protocols must demonstrate process robustness across expected operating ranges while maintaining yield consistency targets.
Documentation and traceability requirements constitute fundamental quality standards that enable effective process control and continuous improvement. Manufacturing operations must maintain detailed records of membrane lot information, cleaning validation data, process parameter logs, and yield performance metrics. These documentation standards facilitate root cause analysis when yield variations occur and support regulatory compliance requirements.
Quality control testing standards define specific analytical methods and acceptance criteria for both in-process monitoring and final product release. These standards typically include turbidity measurements, bioburden testing, endotoxin levels, and product recovery calculations. Establishing clear testing protocols ensures that yield consistency issues are detected promptly and addressed systematically.
Environmental control standards address factors that can significantly impact TFF performance, including cleanroom classifications, temperature and humidity controls, and contamination prevention measures. These standards recognize that environmental variations can introduce process variability that ultimately affects yield consistency, requiring careful monitoring and control systems.
Continuous improvement standards mandate regular review of quality metrics, trend analysis of yield performance data, and implementation of corrective and preventive actions. These standards ensure that quality systems evolve to address emerging challenges and incorporate technological advances that enhance yield consistency in TFF manufacturing operations.
Process Analytics for TFF Yield Monitoring
Process analytics represents a critical component in achieving consistent yields within tangential flow filtration systems. Real-time monitoring capabilities enable operators to track key performance indicators throughout the filtration process, providing immediate feedback on system performance and product recovery rates. Advanced sensor technologies, including pressure transducers, flow meters, and conductivity sensors, form the foundation of comprehensive TFF monitoring systems.
Modern process analytical technology integrates multiple data streams to create a holistic view of filtration performance. Transmembrane pressure monitoring provides insights into membrane fouling patterns, while permeate flux measurements indicate filtration efficiency trends. Concentration polarization effects can be detected through real-time analysis of feed and retentate compositions, enabling proactive adjustments to maintain optimal operating conditions.
Data acquisition systems designed for TFF applications typically employ high-frequency sampling rates to capture rapid process variations. These systems utilize industrial-grade hardware capable of withstanding biopharmaceutical manufacturing environments while maintaining measurement accuracy. Integration with distributed control systems allows for automated response protocols when process parameters deviate from predetermined ranges.
Statistical process control methodologies enhance yield monitoring by establishing control limits based on historical performance data. Control charts track critical quality attributes such as product concentration, recovery percentage, and processing time, enabling early detection of process drift. Multivariate analysis techniques correlate multiple process variables to identify subtle relationships affecting yield consistency.
Machine learning algorithms increasingly support predictive analytics in TFF operations. These systems analyze historical process data to identify patterns associated with optimal yield performance, enabling predictive maintenance scheduling and process optimization recommendations. Neural networks can detect complex interactions between operating parameters that traditional statistical methods might overlook.
Cloud-based analytics platforms facilitate remote monitoring and cross-site performance comparisons. These systems aggregate data from multiple TFF installations, enabling benchmarking studies and best practice identification. Advanced visualization tools present complex process data in intuitive formats, supporting rapid decision-making during critical manufacturing operations.
Modern process analytical technology integrates multiple data streams to create a holistic view of filtration performance. Transmembrane pressure monitoring provides insights into membrane fouling patterns, while permeate flux measurements indicate filtration efficiency trends. Concentration polarization effects can be detected through real-time analysis of feed and retentate compositions, enabling proactive adjustments to maintain optimal operating conditions.
Data acquisition systems designed for TFF applications typically employ high-frequency sampling rates to capture rapid process variations. These systems utilize industrial-grade hardware capable of withstanding biopharmaceutical manufacturing environments while maintaining measurement accuracy. Integration with distributed control systems allows for automated response protocols when process parameters deviate from predetermined ranges.
Statistical process control methodologies enhance yield monitoring by establishing control limits based on historical performance data. Control charts track critical quality attributes such as product concentration, recovery percentage, and processing time, enabling early detection of process drift. Multivariate analysis techniques correlate multiple process variables to identify subtle relationships affecting yield consistency.
Machine learning algorithms increasingly support predictive analytics in TFF operations. These systems analyze historical process data to identify patterns associated with optimal yield performance, enabling predictive maintenance scheduling and process optimization recommendations. Neural networks can detect complex interactions between operating parameters that traditional statistical methods might overlook.
Cloud-based analytics platforms facilitate remote monitoring and cross-site performance comparisons. These systems aggregate data from multiple TFF installations, enabling benchmarking studies and best practice identification. Advanced visualization tools present complex process data in intuitive formats, supporting rapid decision-making during critical manufacturing operations.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!