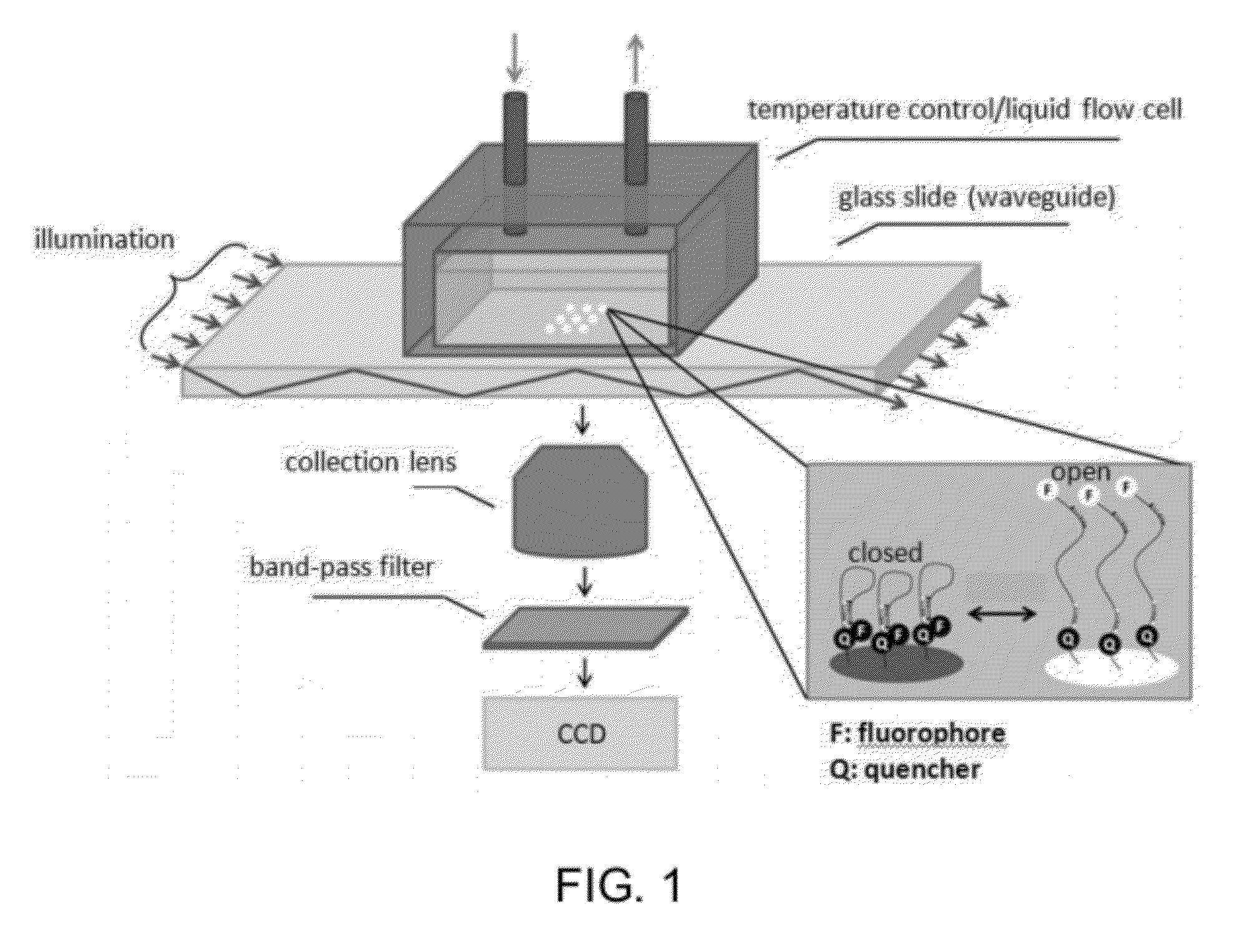
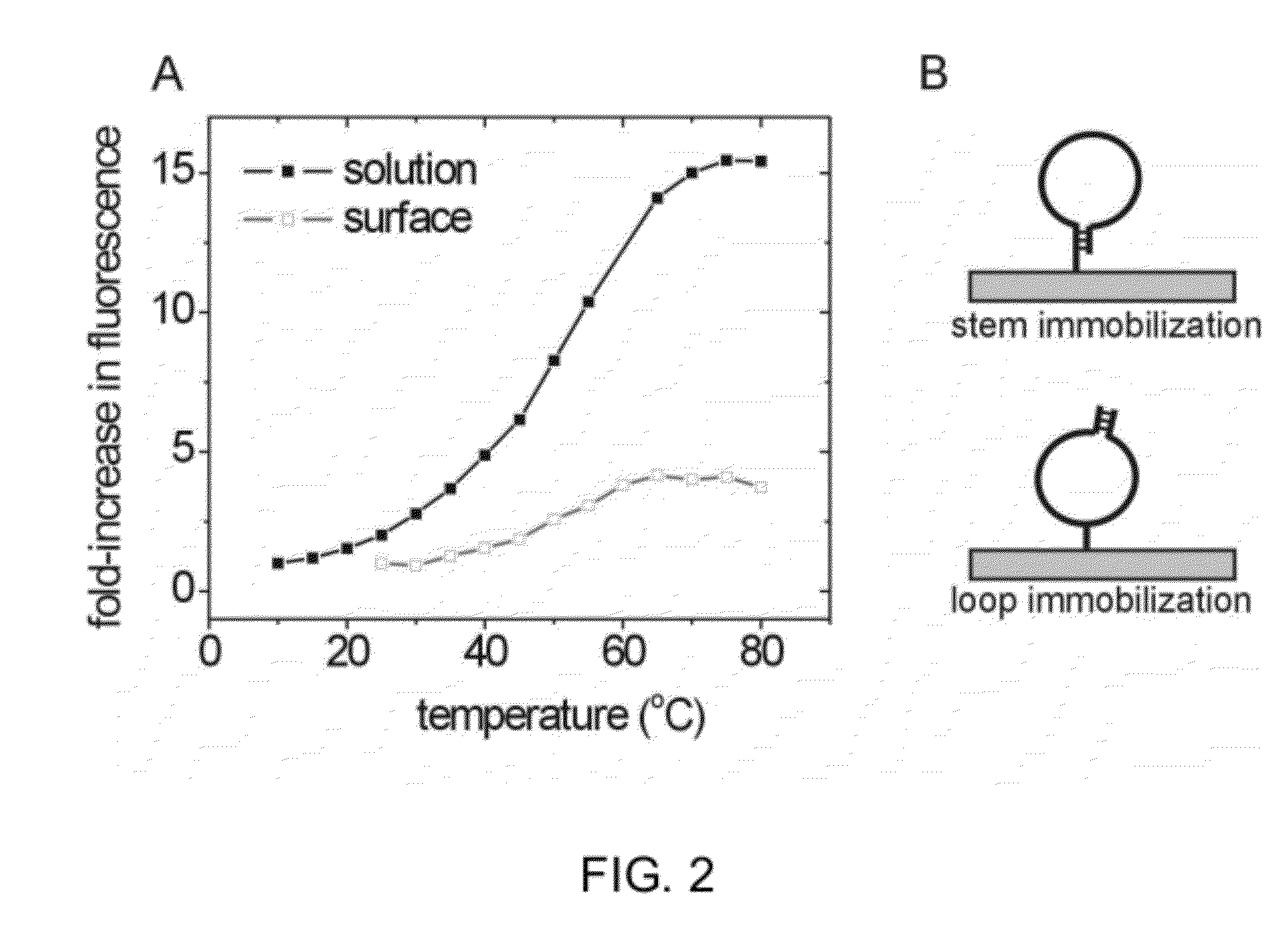
Measure Interaction between Nitrogenous Bases and Small Molecules
MAR 5, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Nitrogenous Base Interaction Research Background and Objectives
The study of interactions between nitrogenous bases and small molecules represents a fundamental area of molecular biology and biochemistry that has gained significant momentum over the past several decades. Nitrogenous bases, including purines (adenine and guanine) and pyrimidines (cytosine, thymine, and uracil), serve as the building blocks of nucleic acids and play crucial roles in genetic information storage, transcription, and translation processes. Understanding how these bases interact with various small molecules has become increasingly important for advancing our knowledge of cellular mechanisms and developing therapeutic interventions.
The historical development of this research field can be traced back to the early structural studies of DNA and RNA in the 1950s, which laid the foundation for understanding base-pairing mechanisms. As analytical techniques evolved, researchers began investigating more complex interactions involving small molecules such as drugs, metabolites, cofactors, and synthetic compounds. The advent of sophisticated spectroscopic methods, computational modeling, and high-resolution imaging technologies has dramatically expanded our ability to characterize these molecular interactions at unprecedented levels of detail.
Current research objectives in this field encompass multiple dimensions of scientific inquiry. Primary goals include developing accurate and reliable methods for quantifying binding affinities between nitrogenous bases and small molecules, elucidating the structural basis of these interactions, and understanding their thermodynamic and kinetic properties. Researchers aim to establish comprehensive databases of interaction parameters that can inform drug design efforts and predict biological outcomes.
The technological evolution in this domain has progressed from basic chemical assays to sophisticated biophysical techniques including surface plasmon resonance, isothermal titration calorimetry, nuclear magnetic resonance spectroscopy, and molecular dynamics simulations. These advances have enabled researchers to probe interactions with increasing precision and to investigate previously inaccessible aspects of molecular recognition processes.
Contemporary research objectives also focus on translational applications, particularly in pharmaceutical development where understanding base-small molecule interactions is critical for designing nucleic acid-targeting therapeutics, optimizing drug delivery systems, and minimizing off-target effects. The field continues to evolve toward more integrated approaches that combine experimental validation with computational predictions to accelerate discovery processes.
The historical development of this research field can be traced back to the early structural studies of DNA and RNA in the 1950s, which laid the foundation for understanding base-pairing mechanisms. As analytical techniques evolved, researchers began investigating more complex interactions involving small molecules such as drugs, metabolites, cofactors, and synthetic compounds. The advent of sophisticated spectroscopic methods, computational modeling, and high-resolution imaging technologies has dramatically expanded our ability to characterize these molecular interactions at unprecedented levels of detail.
Current research objectives in this field encompass multiple dimensions of scientific inquiry. Primary goals include developing accurate and reliable methods for quantifying binding affinities between nitrogenous bases and small molecules, elucidating the structural basis of these interactions, and understanding their thermodynamic and kinetic properties. Researchers aim to establish comprehensive databases of interaction parameters that can inform drug design efforts and predict biological outcomes.
The technological evolution in this domain has progressed from basic chemical assays to sophisticated biophysical techniques including surface plasmon resonance, isothermal titration calorimetry, nuclear magnetic resonance spectroscopy, and molecular dynamics simulations. These advances have enabled researchers to probe interactions with increasing precision and to investigate previously inaccessible aspects of molecular recognition processes.
Contemporary research objectives also focus on translational applications, particularly in pharmaceutical development where understanding base-small molecule interactions is critical for designing nucleic acid-targeting therapeutics, optimizing drug delivery systems, and minimizing off-target effects. The field continues to evolve toward more integrated approaches that combine experimental validation with computational predictions to accelerate discovery processes.
Market Demand for Small Molecule-DNA Interaction Analysis
The pharmaceutical industry represents the largest market segment driving demand for small molecule-DNA interaction analysis technologies. Drug discovery processes increasingly rely on understanding how potential therapeutic compounds interact with genetic material, particularly in developing targeted therapies for cancer, genetic disorders, and infectious diseases. Major pharmaceutical companies are investing heavily in screening platforms that can rapidly assess binding affinities, selectivity profiles, and potential off-target effects of drug candidates with nucleic acids.
Academic research institutions constitute another significant market driver, with growing emphasis on fundamental studies of gene regulation, epigenetics, and molecular recognition mechanisms. Universities and research centers require sophisticated analytical tools to investigate how small molecules modulate DNA structure, influence transcription factor binding, and affect chromatin organization. This demand is amplified by increased funding for genomics research and personalized medicine initiatives.
The biotechnology sector shows substantial growth in demand for these analytical capabilities, particularly companies developing DNA-based therapeutics, gene editing tools, and diagnostic platforms. Biotech firms working on antisense oligonucleotides, aptamers, and CRISPR technologies need precise measurement tools to optimize their molecular designs and validate therapeutic mechanisms.
Diagnostic and clinical testing markets are emerging as important demand drivers, especially for applications in pharmacogenomics and companion diagnostics. Healthcare providers increasingly require tools to predict patient responses to medications based on drug-DNA interactions, driving demand for standardized, high-throughput analytical platforms suitable for clinical environments.
The agrochemical industry represents a growing market segment, with companies developing herbicides, pesticides, and plant growth regulators that target specific genetic pathways in crops or pests. These applications require specialized analytical approaches to ensure selectivity and minimize environmental impact.
Contract research organizations are experiencing increased demand for outsourced small molecule-DNA interaction studies, as pharmaceutical and biotech companies seek specialized expertise and cost-effective solutions. This trend is creating opportunities for service providers offering comprehensive analytical packages combining multiple measurement techniques.
Regulatory requirements are also shaping market demand, as agencies increasingly expect detailed molecular interaction data for drug approvals, particularly for novel therapeutic modalities targeting nucleic acids or requiring genetic biomarkers for patient selection.
Academic research institutions constitute another significant market driver, with growing emphasis on fundamental studies of gene regulation, epigenetics, and molecular recognition mechanisms. Universities and research centers require sophisticated analytical tools to investigate how small molecules modulate DNA structure, influence transcription factor binding, and affect chromatin organization. This demand is amplified by increased funding for genomics research and personalized medicine initiatives.
The biotechnology sector shows substantial growth in demand for these analytical capabilities, particularly companies developing DNA-based therapeutics, gene editing tools, and diagnostic platforms. Biotech firms working on antisense oligonucleotides, aptamers, and CRISPR technologies need precise measurement tools to optimize their molecular designs and validate therapeutic mechanisms.
Diagnostic and clinical testing markets are emerging as important demand drivers, especially for applications in pharmacogenomics and companion diagnostics. Healthcare providers increasingly require tools to predict patient responses to medications based on drug-DNA interactions, driving demand for standardized, high-throughput analytical platforms suitable for clinical environments.
The agrochemical industry represents a growing market segment, with companies developing herbicides, pesticides, and plant growth regulators that target specific genetic pathways in crops or pests. These applications require specialized analytical approaches to ensure selectivity and minimize environmental impact.
Contract research organizations are experiencing increased demand for outsourced small molecule-DNA interaction studies, as pharmaceutical and biotech companies seek specialized expertise and cost-effective solutions. This trend is creating opportunities for service providers offering comprehensive analytical packages combining multiple measurement techniques.
Regulatory requirements are also shaping market demand, as agencies increasingly expect detailed molecular interaction data for drug approvals, particularly for novel therapeutic modalities targeting nucleic acids or requiring genetic biomarkers for patient selection.
Current State and Challenges in Base-Molecule Interaction Measurement
The measurement of interactions between nitrogenous bases and small molecules represents a critical frontier in molecular biology and drug discovery, yet current methodologies face significant limitations that constrain research progress. Contemporary approaches primarily rely on spectroscopic techniques, surface plasmon resonance, isothermal titration calorimetry, and nuclear magnetic resonance spectroscopy, each presenting distinct advantages and inherent challenges.
Spectroscopic methods, including UV-Vis and fluorescence spectroscopy, offer real-time monitoring capabilities but suffer from limited sensitivity when detecting weak interactions. The signal-to-noise ratio becomes particularly problematic when studying low-affinity binding events, which are common in base-small molecule interactions. Additionally, spectral overlap and interference from buffer components frequently compromise data quality and interpretation accuracy.
Surface plasmon resonance technology provides label-free detection and kinetic analysis capabilities, yet faces substantial challenges in studying small molecule interactions due to mass transport limitations and low molecular weight analytes producing minimal refractive index changes. The requirement for surface immobilization can also alter the natural binding characteristics of nitrogenous bases, potentially leading to artifacts in binding affinity measurements.
Isothermal titration calorimetry remains the gold standard for thermodynamic characterization but demands relatively high concentrations of both binding partners, making it unsuitable for studying rare or expensive compounds. The technique also requires significant sample volumes and extended measurement times, limiting throughput for screening applications.
Nuclear magnetic resonance spectroscopy offers detailed structural information about binding interactions but faces sensitivity constraints and requires specialized expertise for data interpretation. The technique struggles with larger molecular complexes due to line broadening effects and demands isotopic labeling for comprehensive analysis.
Current computational approaches, while advancing rapidly, still lack sufficient accuracy in predicting binding affinities and interaction modes for novel base-small molecule pairs. Force field limitations and inadequate sampling of conformational space continue to challenge molecular dynamics simulations.
The field urgently requires more sensitive, high-throughput methodologies capable of detecting weak interactions while providing detailed mechanistic insights. Integration of multiple complementary techniques and development of novel detection principles represent critical needs for advancing our understanding of these fundamental molecular interactions.
Spectroscopic methods, including UV-Vis and fluorescence spectroscopy, offer real-time monitoring capabilities but suffer from limited sensitivity when detecting weak interactions. The signal-to-noise ratio becomes particularly problematic when studying low-affinity binding events, which are common in base-small molecule interactions. Additionally, spectral overlap and interference from buffer components frequently compromise data quality and interpretation accuracy.
Surface plasmon resonance technology provides label-free detection and kinetic analysis capabilities, yet faces substantial challenges in studying small molecule interactions due to mass transport limitations and low molecular weight analytes producing minimal refractive index changes. The requirement for surface immobilization can also alter the natural binding characteristics of nitrogenous bases, potentially leading to artifacts in binding affinity measurements.
Isothermal titration calorimetry remains the gold standard for thermodynamic characterization but demands relatively high concentrations of both binding partners, making it unsuitable for studying rare or expensive compounds. The technique also requires significant sample volumes and extended measurement times, limiting throughput for screening applications.
Nuclear magnetic resonance spectroscopy offers detailed structural information about binding interactions but faces sensitivity constraints and requires specialized expertise for data interpretation. The technique struggles with larger molecular complexes due to line broadening effects and demands isotopic labeling for comprehensive analysis.
Current computational approaches, while advancing rapidly, still lack sufficient accuracy in predicting binding affinities and interaction modes for novel base-small molecule pairs. Force field limitations and inadequate sampling of conformational space continue to challenge molecular dynamics simulations.
The field urgently requires more sensitive, high-throughput methodologies capable of detecting weak interactions while providing detailed mechanistic insights. Integration of multiple complementary techniques and development of novel detection principles represent critical needs for advancing our understanding of these fundamental molecular interactions.
Existing Methods for Base-Small Molecule Interaction Detection
01 Nucleotide analogs and derivatives for therapeutic applications
Modified nitrogenous bases and nucleotide analogs can be designed to interact with biological targets for therapeutic purposes. These modifications enhance binding affinity, specificity, and stability of the molecules. Such compounds can be used in antiviral, anticancer, and other pharmaceutical applications by interfering with nucleic acid metabolism or enzyme function.- Nucleotide and nucleoside derivatives for therapeutic applications: Modified nitrogenous bases and their derivatives, including nucleotides and nucleosides, can interact with small molecules to form therapeutic compounds. These modifications enhance binding affinity and specificity for target molecules, improving drug efficacy. The interactions involve hydrogen bonding, pi-stacking, and electrostatic forces between the nitrogenous bases and small molecular entities, leading to enhanced biological activity and stability.
- Base pairing interactions in molecular recognition: Nitrogenous bases exhibit specific pairing interactions with complementary small molecules through Watson-Crick or Hoogsteen base pairing mechanisms. These interactions are fundamental for molecular recognition processes and can be exploited in drug design and biosensor development. The specificity of base pairing allows for selective binding and detection of target molecules in complex biological systems.
- Small molecule intercalation with nucleic acid bases: Small molecules can intercalate between stacked nitrogenous bases, disrupting or modifying the normal base-base interactions. This intercalation mechanism is important for developing anticancer agents and antimicrobial compounds. The planar aromatic structures of certain small molecules allow them to insert between base pairs, affecting DNA/RNA structure and function.
- Chemical modification of nitrogenous bases for enhanced binding: Chemical modifications at specific positions of nitrogenous bases can enhance their interaction with small molecules. These modifications include alkylation, acylation, and addition of functional groups that increase binding affinity and selectivity. The modified bases demonstrate improved stability and resistance to enzymatic degradation while maintaining their ability to interact with target small molecules.
- Coordination complexes of nitrogenous bases with metal-containing small molecules: Nitrogenous bases can form coordination complexes with metal-containing small molecules through their nitrogen atoms. These metal-base interactions create stable complexes with unique properties useful in catalysis, sensing, and therapeutic applications. The coordination geometry and binding strength depend on the metal ion type and the specific nitrogenous base involved in the interaction.
02 Small molecule inhibitors targeting nucleic acid binding proteins
Small molecules can be designed to interact with proteins that bind to nitrogenous bases, thereby modulating gene expression or replication processes. These inhibitors work by competing with natural substrates or inducing conformational changes in target proteins. This approach is valuable for developing drugs against diseases involving aberrant nucleic acid-protein interactions.Expand Specific Solutions03 Base pairing modifications and synthetic nucleic acids
Synthetic modifications to nitrogenous bases can create novel base pairing systems with altered hydrogen bonding patterns. These modifications enable the development of expanded genetic alphabets and improved oligonucleotide therapeutics. Applications include enhanced antisense oligonucleotides, aptamers, and diagnostic probes with increased stability and specificity.Expand Specific Solutions04 Metal ion coordination with nitrogenous bases
Nitrogenous bases can form coordination complexes with metal ions, creating compounds with unique properties. These metal-base interactions can be exploited for catalysis, sensing applications, and as structural scaffolds in supramolecular chemistry. The coordination affects the electronic properties and reactivity of both the metal center and the base.Expand Specific Solutions05 Intercalating agents and DNA groove binders
Small molecules can interact with nitrogenous bases through intercalation between base pairs or binding in DNA grooves. These interactions can affect DNA structure, stability, and biological function. Such compounds are important in developing anticancer agents, antimicrobial drugs, and molecular probes for studying nucleic acid structure and dynamics.Expand Specific Solutions
Key Players in Molecular Interaction and Biosensor Industry
The field of measuring interactions between nitrogenous bases and small molecules represents an emerging biotechnology sector with significant growth potential, driven by applications in drug discovery, diagnostics, and personalized medicine. The market is experiencing rapid expansion as pharmaceutical companies increasingly recognize the importance of understanding molecular interactions for therapeutic development. Technology maturity varies considerably across different approaches, with established players like Hitachi Ltd. and Robert Bosch GmbH leveraging advanced instrumentation capabilities, while specialized biotechnology companies such as Base4 Biotechnology, Depixus SAS, and Ontera Inc. are pioneering novel single-molecule detection platforms. Academic institutions including Harvard College, Tsinghua University, and Osaka University are contributing fundamental research breakthroughs that drive innovation. The competitive landscape shows a convergence of traditional analytical instrument manufacturers, emerging biotech startups, and research institutions, indicating a technology transition phase where next-generation molecular interaction measurement platforms are being developed to meet growing pharmaceutical and diagnostic market demands.
President & Fellows of Harvard College
Technical Solution: Harvard has developed advanced computational methods and experimental techniques for measuring nitrogenous base-small molecule interactions. Their approach combines molecular dynamics simulations with high-resolution NMR spectroscopy and isothermal titration calorimetry to quantify binding affinities and kinetic parameters. The university's research focuses on developing novel fluorescence-based assays and surface plasmon resonance techniques to study DNA-drug interactions in real-time. Their interdisciplinary teams have created innovative microfluidic platforms that enable single-molecule detection of base-pairing interactions with small therapeutic compounds, providing unprecedented insights into binding mechanisms and selectivity patterns.
Strengths: Leading research institution with cutting-edge facilities and interdisciplinary expertise. Weaknesses: Academic focus may limit commercial scalability and industrial application speed.
Tsinghua University
Technical Solution: Tsinghua University has established comprehensive research programs for studying nitrogenous base-small molecule interactions using both computational and experimental approaches. Their methodology integrates quantum mechanical calculations, molecular dynamics simulations, and experimental validation through various biophysical techniques including circular dichroism spectroscopy, UV-Vis absorption, and fluorescence quenching studies. The university has developed novel synthetic biology approaches to create engineered nucleic acid structures that can serve as highly specific sensors for small molecule detection. Their research extends to developing new analytical methods for studying epigenetic modifications and their interactions with small molecule modulators.
Strengths: Strong computational capabilities and comprehensive research infrastructure with government support. Weaknesses: Academic institution with potential limitations in technology transfer and commercial development timelines.
Core Technologies in Molecular Recognition and Binding Analysis
Small molecule binding pockets in nucleic acids
PatentActiveUS20170032078A1
Innovation
- A method involving isotopic labeling of nucleotides in polynucleotides to determine 2-D or 3-D atomic resolution structures using NMR spectroscopy, allowing for the identification of key small molecule-nucleic acid interactions and the optimization of binding pockets, enabling efficient hit identification and rational design of new compounds.
Solid phase methods for thermodynamic and kinetic quantification of interactions between nucleic acids and small molecules
PatentInactiveUS20120295805A1
Innovation
- A solid-phase based method where a single BA in solution is exposed to many NA sequences immobilized on an array, allowing interactions to take place and being quantified by comparing denaturation profiles before and after BA interaction, without labeling, in situ, and capable of high-throughput analysis of both reversible and adduct-forming compounds.
Regulatory Framework for Molecular Diagnostic Technologies
The regulatory landscape for molecular diagnostic technologies measuring interactions between nitrogenous bases and small molecules encompasses multiple jurisdictional frameworks that govern the development, validation, and commercialization of these sophisticated analytical systems. In the United States, the Food and Drug Administration (FDA) oversees these technologies through its Center for Devices and Radiological Health (CDRH), classifying most base-molecule interaction measurement devices as Class II medical devices requiring 510(k) premarket notification or Class III devices necessitating premarket approval (PMA) depending on their intended clinical applications.
The European Union operates under the In Vitro Diagnostic Medical Devices Regulation (IVDR 2017/746), which became fully applicable in May 2022, establishing stringent requirements for diagnostic technologies that analyze molecular interactions. These regulations mandate comprehensive clinical evidence, quality management systems compliant with ISO 13485, and conformity assessment procedures conducted by notified bodies for higher-risk classifications.
Analytical validation requirements focus on demonstrating the accuracy, precision, and reproducibility of base-molecule interaction measurements across different sample types and concentrations. Regulatory bodies require extensive validation studies encompassing limit of detection, limit of quantification, analytical specificity, and interference testing to ensure reliable detection of molecular binding events.
Clinical validation presents additional complexity, as manufacturers must demonstrate clinical utility and analytical validity through well-controlled studies that correlate interaction measurements with clinically relevant outcomes. This includes establishing reference ranges, clinical sensitivity, and specificity for specific patient populations and disease states.
Quality assurance frameworks mandate implementation of robust quality control measures, including regular calibration protocols, proficiency testing participation, and comprehensive documentation systems. Manufacturers must establish risk management processes following ISO 14971 standards, addressing potential failure modes in molecular interaction detection systems.
International harmonization efforts through organizations like the International Council for Harmonisation (ICH) and the Global Harmonization Task Force (GHTF) aim to streamline regulatory requirements across different markets, though significant regional variations persist in approval timelines and specific technical requirements for these advanced molecular diagnostic platforms.
The European Union operates under the In Vitro Diagnostic Medical Devices Regulation (IVDR 2017/746), which became fully applicable in May 2022, establishing stringent requirements for diagnostic technologies that analyze molecular interactions. These regulations mandate comprehensive clinical evidence, quality management systems compliant with ISO 13485, and conformity assessment procedures conducted by notified bodies for higher-risk classifications.
Analytical validation requirements focus on demonstrating the accuracy, precision, and reproducibility of base-molecule interaction measurements across different sample types and concentrations. Regulatory bodies require extensive validation studies encompassing limit of detection, limit of quantification, analytical specificity, and interference testing to ensure reliable detection of molecular binding events.
Clinical validation presents additional complexity, as manufacturers must demonstrate clinical utility and analytical validity through well-controlled studies that correlate interaction measurements with clinically relevant outcomes. This includes establishing reference ranges, clinical sensitivity, and specificity for specific patient populations and disease states.
Quality assurance frameworks mandate implementation of robust quality control measures, including regular calibration protocols, proficiency testing participation, and comprehensive documentation systems. Manufacturers must establish risk management processes following ISO 14971 standards, addressing potential failure modes in molecular interaction detection systems.
International harmonization efforts through organizations like the International Council for Harmonisation (ICH) and the Global Harmonization Task Force (GHTF) aim to streamline regulatory requirements across different markets, though significant regional variations persist in approval timelines and specific technical requirements for these advanced molecular diagnostic platforms.
Standardization Challenges in Molecular Interaction Assays
The standardization of molecular interaction assays for measuring nitrogenous base-small molecule interactions faces significant methodological and technical challenges that impede reproducibility and cross-laboratory comparability. Current assay protocols vary substantially across research institutions, with different buffer systems, pH conditions, ionic strengths, and temperature controls being employed for similar experimental objectives. This variability creates substantial barriers to data integration and meta-analysis efforts.
Protocol harmonization represents one of the most pressing standardization challenges. Different laboratories often utilize distinct experimental conditions that can dramatically influence binding affinity measurements and kinetic parameters. For instance, variations in buffer composition can alter the protonation states of nitrogenous bases, while temperature fluctuations affect both binding thermodynamics and molecular conformational dynamics. The absence of universally accepted reference standards compounds these issues, making it difficult to establish baseline measurements for quality control.
Instrumentation diversity poses another significant standardization hurdle. Surface plasmon resonance, isothermal titration calorimetry, fluorescence polarization, and nuclear magnetic resonance spectroscopy each provide different perspectives on molecular interactions, but their outputs are not easily comparable. Each technique has distinct sensitivity ranges, detection limits, and optimal experimental conditions, making direct comparison of results challenging without appropriate conversion factors or standardized calibration procedures.
Data reporting and analysis methodologies lack consistency across the field. Different research groups employ varying curve-fitting algorithms, statistical approaches, and error analysis methods when processing interaction data. The absence of standardized data formats and reporting requirements makes it difficult to compare results from different studies or integrate findings into comprehensive databases.
Quality control measures remain inadequately standardized, with limited consensus on appropriate reference compounds, control experiments, and validation procedures. Many laboratories lack standardized protocols for instrument calibration, sample preparation validation, and systematic error detection. This situation is further complicated by the diverse chemical properties of small molecules used in these assays, which require different handling and storage conditions.
Regulatory and metrological frameworks for molecular interaction measurements are still evolving, with limited guidance from international standards organizations. The development of certified reference materials and standardized measurement protocols remains in early stages, hindering the establishment of traceability chains necessary for reliable quantitative measurements in pharmaceutical and biotechnology applications.
Protocol harmonization represents one of the most pressing standardization challenges. Different laboratories often utilize distinct experimental conditions that can dramatically influence binding affinity measurements and kinetic parameters. For instance, variations in buffer composition can alter the protonation states of nitrogenous bases, while temperature fluctuations affect both binding thermodynamics and molecular conformational dynamics. The absence of universally accepted reference standards compounds these issues, making it difficult to establish baseline measurements for quality control.
Instrumentation diversity poses another significant standardization hurdle. Surface plasmon resonance, isothermal titration calorimetry, fluorescence polarization, and nuclear magnetic resonance spectroscopy each provide different perspectives on molecular interactions, but their outputs are not easily comparable. Each technique has distinct sensitivity ranges, detection limits, and optimal experimental conditions, making direct comparison of results challenging without appropriate conversion factors or standardized calibration procedures.
Data reporting and analysis methodologies lack consistency across the field. Different research groups employ varying curve-fitting algorithms, statistical approaches, and error analysis methods when processing interaction data. The absence of standardized data formats and reporting requirements makes it difficult to compare results from different studies or integrate findings into comprehensive databases.
Quality control measures remain inadequately standardized, with limited consensus on appropriate reference compounds, control experiments, and validation procedures. Many laboratories lack standardized protocols for instrument calibration, sample preparation validation, and systematic error detection. This situation is further complicated by the diverse chemical properties of small molecules used in these assays, which require different handling and storage conditions.
Regulatory and metrological frameworks for molecular interaction measurements are still evolving, with limited guidance from international standards organizations. The development of certified reference materials and standardized measurement protocols remains in early stages, hindering the establishment of traceability chains necessary for reliable quantitative measurements in pharmaceutical and biotechnology applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!