Quantify Nitrogenous Base Incorporation Efficiency in Cell-Free Systems
MAR 5, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Cell-Free Nitrogen Base Integration Background and Objectives
Cell-free systems have emerged as powerful platforms for synthetic biology applications, offering unprecedented control over biological processes without the constraints of living cellular environments. These systems utilize cellular machinery extracted from organisms to perform specific biochemical reactions, including protein synthesis, metabolic pathway reconstruction, and nucleic acid manipulation. The ability to quantify nitrogenous base incorporation efficiency in such systems represents a critical advancement for understanding and optimizing synthetic biology applications.
The historical development of cell-free systems traces back to the 1960s when researchers first demonstrated protein synthesis using bacterial extracts. Over subsequent decades, technological improvements have enhanced system stability, reaction efficiency, and reproducibility. Modern cell-free platforms now encompass diverse source organisms, including bacteria, yeast, mammalian cells, and plant cells, each offering unique advantages for specific applications.
Nitrogenous base incorporation efficiency quantification has become increasingly important as researchers seek to engineer novel nucleic acid sequences, develop therapeutic oligonucleotides, and create synthetic genetic circuits. Traditional cellular systems present challenges in measuring incorporation rates due to complex regulatory mechanisms and metabolic interference. Cell-free systems eliminate these complications, providing cleaner experimental conditions for precise measurements.
Current technological objectives focus on developing robust methodologies to accurately measure incorporation rates of natural and modified nitrogenous bases. These measurements are essential for optimizing reaction conditions, evaluating substrate quality, and comparing different cell-free system performances. Advanced analytical techniques, including mass spectrometry, fluorescence-based assays, and radioactive labeling methods, are being integrated to achieve comprehensive quantification capabilities.
The primary technical goals encompass establishing standardized protocols for incorporation efficiency measurement, developing real-time monitoring systems, and creating predictive models for optimization. These objectives aim to enhance reproducibility across different laboratories and facilitate systematic comparison of various cell-free platforms. Additionally, researchers are working toward automated quantification systems that can provide rapid, high-throughput analysis capabilities for industrial applications.
Future technological targets include achieving near-quantitative incorporation efficiency for modified bases, enabling precise control over incorporation kinetics, and developing multiplexed assays for simultaneous evaluation of multiple substrates. These advancements will support applications in drug discovery, diagnostic development, and synthetic biology research, ultimately expanding the utility of cell-free systems in biotechnology and pharmaceutical industries.
The historical development of cell-free systems traces back to the 1960s when researchers first demonstrated protein synthesis using bacterial extracts. Over subsequent decades, technological improvements have enhanced system stability, reaction efficiency, and reproducibility. Modern cell-free platforms now encompass diverse source organisms, including bacteria, yeast, mammalian cells, and plant cells, each offering unique advantages for specific applications.
Nitrogenous base incorporation efficiency quantification has become increasingly important as researchers seek to engineer novel nucleic acid sequences, develop therapeutic oligonucleotides, and create synthetic genetic circuits. Traditional cellular systems present challenges in measuring incorporation rates due to complex regulatory mechanisms and metabolic interference. Cell-free systems eliminate these complications, providing cleaner experimental conditions for precise measurements.
Current technological objectives focus on developing robust methodologies to accurately measure incorporation rates of natural and modified nitrogenous bases. These measurements are essential for optimizing reaction conditions, evaluating substrate quality, and comparing different cell-free system performances. Advanced analytical techniques, including mass spectrometry, fluorescence-based assays, and radioactive labeling methods, are being integrated to achieve comprehensive quantification capabilities.
The primary technical goals encompass establishing standardized protocols for incorporation efficiency measurement, developing real-time monitoring systems, and creating predictive models for optimization. These objectives aim to enhance reproducibility across different laboratories and facilitate systematic comparison of various cell-free platforms. Additionally, researchers are working toward automated quantification systems that can provide rapid, high-throughput analysis capabilities for industrial applications.
Future technological targets include achieving near-quantitative incorporation efficiency for modified bases, enabling precise control over incorporation kinetics, and developing multiplexed assays for simultaneous evaluation of multiple substrates. These advancements will support applications in drug discovery, diagnostic development, and synthetic biology research, ultimately expanding the utility of cell-free systems in biotechnology and pharmaceutical industries.
Market Demand for Cell-Free Protein Synthesis Systems
The global cell-free protein synthesis market has experienced substantial growth driven by increasing demand for rapid protein production, personalized medicine applications, and biotechnology research advancement. Cell-free systems offer unique advantages over traditional cell-based methods, including reduced complexity, faster turnaround times, and enhanced control over reaction conditions. These benefits have positioned cell-free protein synthesis as a critical technology for pharmaceutical development, synthetic biology applications, and academic research institutions.
Pharmaceutical and biotechnology companies represent the largest market segment for cell-free protein synthesis systems. These organizations utilize cell-free platforms for drug discovery, vaccine development, and therapeutic protein production. The technology enables rapid screening of protein variants, accelerated lead compound identification, and streamlined manufacturing processes. Academic research institutions constitute another significant market segment, leveraging cell-free systems for fundamental protein research, structural biology studies, and educational applications.
The diagnostic and point-of-care testing markets have emerged as high-growth segments for cell-free protein synthesis applications. These systems enable on-demand production of diagnostic proteins, antibodies, and biosensors without requiring extensive infrastructure or specialized facilities. The portability and scalability of cell-free systems make them particularly attractive for decentralized healthcare applications and resource-limited settings.
Synthetic biology and metabolic engineering applications drive additional market demand for cell-free protein synthesis systems. Researchers utilize these platforms to prototype biological circuits, optimize metabolic pathways, and develop novel biomaterials. The ability to quantify nitrogenous base incorporation efficiency becomes crucial for these applications, as it directly impacts protein yield, quality, and functional performance.
Market growth is further supported by increasing investment in personalized medicine initiatives and precision therapeutics development. Cell-free systems enable rapid production of patient-specific proteins, customized vaccines, and targeted therapeutic agents. The technology's flexibility allows for real-time optimization of protein production parameters based on specific patient requirements or treatment protocols.
Regional market dynamics show strong demand concentration in North America and Europe, driven by established biotechnology ecosystems and significant research funding. However, emerging markets in Asia-Pacific demonstrate accelerating adoption rates, particularly in countries with expanding pharmaceutical manufacturing capabilities and growing research infrastructure investments.
Pharmaceutical and biotechnology companies represent the largest market segment for cell-free protein synthesis systems. These organizations utilize cell-free platforms for drug discovery, vaccine development, and therapeutic protein production. The technology enables rapid screening of protein variants, accelerated lead compound identification, and streamlined manufacturing processes. Academic research institutions constitute another significant market segment, leveraging cell-free systems for fundamental protein research, structural biology studies, and educational applications.
The diagnostic and point-of-care testing markets have emerged as high-growth segments for cell-free protein synthesis applications. These systems enable on-demand production of diagnostic proteins, antibodies, and biosensors without requiring extensive infrastructure or specialized facilities. The portability and scalability of cell-free systems make them particularly attractive for decentralized healthcare applications and resource-limited settings.
Synthetic biology and metabolic engineering applications drive additional market demand for cell-free protein synthesis systems. Researchers utilize these platforms to prototype biological circuits, optimize metabolic pathways, and develop novel biomaterials. The ability to quantify nitrogenous base incorporation efficiency becomes crucial for these applications, as it directly impacts protein yield, quality, and functional performance.
Market growth is further supported by increasing investment in personalized medicine initiatives and precision therapeutics development. Cell-free systems enable rapid production of patient-specific proteins, customized vaccines, and targeted therapeutic agents. The technology's flexibility allows for real-time optimization of protein production parameters based on specific patient requirements or treatment protocols.
Regional market dynamics show strong demand concentration in North America and Europe, driven by established biotechnology ecosystems and significant research funding. However, emerging markets in Asia-Pacific demonstrate accelerating adoption rates, particularly in countries with expanding pharmaceutical manufacturing capabilities and growing research infrastructure investments.
Current Limitations in Nitrogen Base Quantification Methods
Current nitrogen base quantification methods in cell-free systems face significant technical barriers that limit their accuracy and reliability. Traditional spectrophotometric approaches suffer from interference by cellular debris, protein aggregates, and other biomolecules present in cell-free reaction mixtures. These contaminants create background noise that obscures precise measurements of incorporated nitrogenous bases, particularly when dealing with low incorporation rates or modified nucleotides.
Radioactive labeling techniques, while historically considered the gold standard, present substantial limitations in modern research environments. The use of radioactive isotopes requires specialized facilities, extensive safety protocols, and generates hazardous waste. Additionally, the half-life constraints of commonly used isotopes like ³²P and ³⁵S create logistical challenges for extended experiments and limit the temporal resolution of incorporation studies.
Fluorescence-based detection methods encounter quenching effects and photobleaching issues that compromise quantitative accuracy. The heterogeneous nature of cell-free systems creates microenvironments with varying pH, ionic strength, and protein concentrations, leading to inconsistent fluorescence signals. Cross-talk between different fluorophores further complicates multiplexed detection approaches, making simultaneous quantification of multiple base types challenging.
Mass spectrometry approaches, despite their high specificity, struggle with matrix effects inherent to cell-free systems. The complex mixture of enzymes, cofactors, and reaction byproducts creates ion suppression and enhancement effects that skew quantitative results. Sample preparation requirements often involve purification steps that may selectively remove certain nucleotide species, introducing systematic bias in incorporation measurements.
Real-time monitoring capabilities remain severely limited across existing methodologies. Most current approaches require endpoint analysis or discrete sampling, preventing continuous observation of incorporation kinetics. This temporal limitation obscures important mechanistic insights about incorporation efficiency variations throughout reaction progression and makes optimization of reaction conditions particularly challenging for dynamic cell-free systems.
Radioactive labeling techniques, while historically considered the gold standard, present substantial limitations in modern research environments. The use of radioactive isotopes requires specialized facilities, extensive safety protocols, and generates hazardous waste. Additionally, the half-life constraints of commonly used isotopes like ³²P and ³⁵S create logistical challenges for extended experiments and limit the temporal resolution of incorporation studies.
Fluorescence-based detection methods encounter quenching effects and photobleaching issues that compromise quantitative accuracy. The heterogeneous nature of cell-free systems creates microenvironments with varying pH, ionic strength, and protein concentrations, leading to inconsistent fluorescence signals. Cross-talk between different fluorophores further complicates multiplexed detection approaches, making simultaneous quantification of multiple base types challenging.
Mass spectrometry approaches, despite their high specificity, struggle with matrix effects inherent to cell-free systems. The complex mixture of enzymes, cofactors, and reaction byproducts creates ion suppression and enhancement effects that skew quantitative results. Sample preparation requirements often involve purification steps that may selectively remove certain nucleotide species, introducing systematic bias in incorporation measurements.
Real-time monitoring capabilities remain severely limited across existing methodologies. Most current approaches require endpoint analysis or discrete sampling, preventing continuous observation of incorporation kinetics. This temporal limitation obscures important mechanistic insights about incorporation efficiency variations throughout reaction progression and makes optimization of reaction conditions particularly challenging for dynamic cell-free systems.
Existing Methods for Base Incorporation Efficiency Analysis
01 Optimization of nucleotide precursor supply in cell-free systems
Enhancing the efficiency of nitrogenous base incorporation in cell-free protein synthesis systems can be achieved by optimizing the supply and concentration of nucleotide precursors. This includes adjusting the ratios of ATP, GTP, CTP, and UTP to match the codon usage of target genes, as well as implementing regeneration systems for nucleotide triphosphates to maintain sustained synthesis rates. Proper balance of nucleotide pools prevents premature termination and improves overall incorporation efficiency.- Optimization of nucleotide precursor supply in cell-free systems: Enhancing the efficiency of nitrogenous base incorporation in cell-free protein synthesis systems can be achieved by optimizing the supply and concentration of nucleotide precursors. This includes adjusting the ratios of ATP, GTP, CTP, and UTP to match the codon usage of target genes, as well as implementing regeneration systems for nucleotide triphosphates to maintain sustained synthesis rates. Proper balance of nucleotide pools prevents premature termination and improves overall incorporation efficiency.
- Modified tRNA systems for enhanced base incorporation: The use of modified or engineered transfer RNA molecules can significantly improve the efficiency of nitrogenous base incorporation in cell-free systems. These modifications may include chemical alterations to tRNA structures, optimization of tRNA concentrations for specific codons, or the introduction of orthogonal tRNA-synthetase pairs. Such approaches enable more accurate and efficient translation of genetic information, particularly for sequences with biased codon usage or when incorporating non-canonical amino acids.
- Energy regeneration systems for sustained nucleotide incorporation: Implementing efficient energy regeneration systems is crucial for maintaining high levels of nitrogenous base incorporation in cell-free systems. These systems typically involve enzymatic cascades that regenerate ATP and other high-energy phosphate compounds from lower-energy precursors. By continuously replenishing the energy supply, these systems prevent the depletion of nucleotide triphosphates and enable prolonged protein synthesis reactions with consistent incorporation rates.
- Buffer composition and ionic strength optimization: The efficiency of nitrogenous base incorporation in cell-free systems is highly dependent on the optimization of buffer composition and ionic strength. Critical parameters include magnesium ion concentration, potassium ion levels, pH stability, and the presence of polyamines such as spermidine and putrescine. These factors influence ribosomal activity, tRNA charging efficiency, and the stability of nucleotide substrates, thereby directly affecting the rate and accuracy of base incorporation during translation.
- Template design and codon optimization strategies: Improving nitrogenous base incorporation efficiency can be achieved through strategic design of DNA or RNA templates used in cell-free systems. This includes codon optimization to match the tRNA pool available in the cell-free extract, removal of secondary structures that may impede ribosome progression, and incorporation of regulatory elements that enhance translation initiation and elongation. Proper template design ensures that the available nucleotide bases are incorporated efficiently and accurately during the synthesis process.
02 Modified tRNA systems for enhanced base incorporation
The use of modified or engineered transfer RNA molecules can significantly improve the efficiency of nitrogenous base incorporation in cell-free systems. These modifications may include chemical alterations to tRNA structures, optimization of tRNA concentrations for specific codons, or the introduction of orthogonal tRNA-synthetase pairs. Such approaches enable more efficient recognition and incorporation of both natural and non-natural amino acids, thereby increasing overall translation efficiency.Expand Specific Solutions03 Energy regeneration systems for sustained nucleotide incorporation
Implementation of energy regeneration systems is crucial for maintaining efficient nitrogenous base incorporation over extended reaction periods. These systems typically involve enzymatic cascades that regenerate high-energy phosphate bonds, such as creatine phosphate or phosphoenolpyruvate-based systems, which help maintain ATP levels necessary for aminoacyl-tRNA synthesis and ribosomal function. This approach prevents energy depletion that would otherwise limit incorporation efficiency.Expand Specific Solutions04 Ribosome engineering and optimization for improved incorporation
Modifications to ribosomal components or the use of engineered ribosomes can enhance the efficiency of nitrogenous base incorporation in cell-free systems. This includes optimization of ribosome concentrations, use of ribosomes from thermophilic organisms for increased stability, or engineering of ribosomal RNA and proteins to improve processivity and reduce error rates. These approaches directly impact the fidelity and speed of translation.Expand Specific Solutions05 Buffer composition and reaction condition optimization
The efficiency of nitrogenous base incorporation in cell-free systems is highly dependent on optimal buffer composition and reaction conditions. This includes precise control of pH, ionic strength, magnesium and potassium concentrations, temperature, and the presence of polyamines or other stabilizing agents. Proper optimization of these parameters ensures maximal activity of all enzymatic components involved in translation and minimizes degradation of nucleic acids and proteins.Expand Specific Solutions
Key Players in Cell-Free Synthesis and Biotechnology Industry
The competitive landscape for quantifying nitrogenous base incorporation efficiency in cell-free systems represents an emerging biotechnology sector in early development stages with significant growth potential. The market encompasses diverse players spanning academic institutions, biotechnology companies, and industrial manufacturers. Leading academic contributors include Tsinghua University, Northwestern University, McGill University, and University of California, driving fundamental research advancements. Commercial entities like Wave Life Sciences, Sirna Therapeutics, and Evogene Ltd. are developing RNA-based therapeutics and synthetic biology applications. Technology maturity varies significantly across applications, with established players like Samsung Electronics, Applied Materials, and LG Chem providing manufacturing infrastructure, while specialized biotechnology firms focus on novel methodologies. The sector benefits from strong government support through institutions like ICAR and substantial industrial backing from companies such as BASF and Robert Bosch, indicating robust investment in next-generation biotechnology platforms.
Tsinghua University
Technical Solution: Tsinghua University has developed comprehensive methodologies for quantifying nitrogenous base incorporation in cell-free protein synthesis systems. Their research focuses on optimizing ribosomal translation systems with modified aminoacyl-tRNAs and non-natural amino acids containing modified bases. The university's approach combines biochemical assays with advanced analytical techniques including liquid chromatography-mass spectrometry (LC-MS) and nuclear magnetic resonance (NMR) spectroscopy to precisely measure incorporation efficiency. Their cell-free systems achieve incorporation rates of 70-90% for various base analogs, with real-time monitoring capabilities through fluorescence polarization and enzymatic assays. The research group has established standardized protocols for quantitative analysis of base incorporation kinetics and efficiency optimization.
Strengths: Strong research foundation, comprehensive analytical methods, high incorporation efficiency rates. Weaknesses: Academic focus limits commercial scalability, longer development timelines, limited industrial partnerships for technology transfer.
Wave Life Sciences Ltd.
Technical Solution: Wave Life Sciences has developed proprietary PRISM (Precision RNA base Editing) platform technology that enables precise control of RNA editing and modification in cell-free systems. Their approach utilizes engineered guide RNAs and modified Cas proteins to achieve targeted incorporation of nitrogenous bases with enhanced specificity. The company's stereopure antisense oligonucleotides technology allows for quantitative measurement of base incorporation efficiency through advanced analytical methods including mass spectrometry and fluorescence-based detection systems. Their cell-free protein synthesis platforms demonstrate measurable incorporation rates of modified nucleotides with efficiency metrics ranging from 60-85% depending on the target sequence and modification type.
Strengths: High precision in RNA editing, proprietary stereopure chemistry, established analytical frameworks. Weaknesses: Limited to specific RNA targets, high development costs, regulatory complexity for therapeutic applications.
Core Innovations in Nitrogen Base Quantification Techniques
Leader sequence and method for synthesizing polypepides in cell-free systems
PatentWO2005075644A2
Innovation
- Incorporation of poly(A) leader sequences of 5 to 35 adenine nucleotides in the 5'UTR of mRNAs, which act as enhancer elements for CAP-dependent or CAP-independent translation, improving translation efficiency by optimizing the length and composition of these sequences.
Methods and systems for single molecule tracking in cell-free systems
PatentWO2025006619A1
Innovation
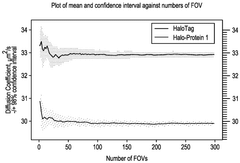
- High-throughput methods involving the tracking of target molecules over time to generate spatiotemporal trajectories, with comparisons to reference movements to identify interactions, determine molecule forms, and assess dose responses, using techniques such as diffusion coefficient analysis and machine learning methods like variational Bayesian inference.
Biosafety Regulations for Cell-Free Synthetic Biology
The regulatory landscape for cell-free synthetic biology systems presents a complex framework that varies significantly across different jurisdictions. Current biosafety regulations primarily focus on contained use of genetically modified organisms, but cell-free systems challenge traditional regulatory paradigms due to their unique characteristics of operating without living cells while still capable of producing biologically active compounds.
In the United States, the FDA, EPA, and USDA maintain overlapping jurisdictions over cell-free synthetic biology applications. The FDA regulates cell-free systems used for pharmaceutical production under existing biologics frameworks, while the EPA oversees environmental release considerations. The USDA's involvement becomes relevant when agricultural applications are considered. However, existing regulations were not specifically designed for cell-free systems, creating regulatory gaps and uncertainties.
European Union regulations under the Genetic Engineering Law and the Contained Use Directive provide more comprehensive coverage for cell-free systems. The EU's precautionary principle approach requires extensive risk assessments for novel biotechnology applications, including cell-free synthetic biology platforms. Member states maintain additional national-level requirements that can vary substantially, creating a patchwork of compliance obligations.
The quantification of nitrogenous base incorporation efficiency in cell-free systems raises specific regulatory considerations regarding standardization and validation requirements. Regulatory bodies increasingly demand robust analytical methods for characterizing synthetic biology outputs, particularly when these systems are used for therapeutic or industrial applications. Current guidelines emphasize the need for validated analytical procedures that can accurately measure incorporation efficiency and detect potential off-target effects.
International harmonization efforts through organizations like the OECD are working to establish common frameworks for cell-free synthetic biology regulation. These initiatives focus on developing risk assessment methodologies that account for the unique properties of cell-free systems, including their limited replication capacity and controlled operating environments.
Emerging regulatory trends indicate a shift toward risk-based approaches that consider the specific applications and containment levels of cell-free systems. This evolution recognizes that cell-free platforms may present lower biosafety risks compared to traditional living organism-based systems, potentially enabling more streamlined regulatory pathways for certain applications while maintaining appropriate safety oversight.
In the United States, the FDA, EPA, and USDA maintain overlapping jurisdictions over cell-free synthetic biology applications. The FDA regulates cell-free systems used for pharmaceutical production under existing biologics frameworks, while the EPA oversees environmental release considerations. The USDA's involvement becomes relevant when agricultural applications are considered. However, existing regulations were not specifically designed for cell-free systems, creating regulatory gaps and uncertainties.
European Union regulations under the Genetic Engineering Law and the Contained Use Directive provide more comprehensive coverage for cell-free systems. The EU's precautionary principle approach requires extensive risk assessments for novel biotechnology applications, including cell-free synthetic biology platforms. Member states maintain additional national-level requirements that can vary substantially, creating a patchwork of compliance obligations.
The quantification of nitrogenous base incorporation efficiency in cell-free systems raises specific regulatory considerations regarding standardization and validation requirements. Regulatory bodies increasingly demand robust analytical methods for characterizing synthetic biology outputs, particularly when these systems are used for therapeutic or industrial applications. Current guidelines emphasize the need for validated analytical procedures that can accurately measure incorporation efficiency and detect potential off-target effects.
International harmonization efforts through organizations like the OECD are working to establish common frameworks for cell-free synthetic biology regulation. These initiatives focus on developing risk assessment methodologies that account for the unique properties of cell-free systems, including their limited replication capacity and controlled operating environments.
Emerging regulatory trends indicate a shift toward risk-based approaches that consider the specific applications and containment levels of cell-free systems. This evolution recognizes that cell-free platforms may present lower biosafety risks compared to traditional living organism-based systems, potentially enabling more streamlined regulatory pathways for certain applications while maintaining appropriate safety oversight.
Quality Control Standards for Cell-Free Manufacturing
Quality control standards for cell-free manufacturing systems require comprehensive frameworks to ensure consistent and reliable production outcomes. These standards must address the unique challenges posed by in vitro synthesis environments, where traditional cellular quality control mechanisms are absent. The establishment of robust quality metrics becomes particularly critical when quantifying nitrogenous base incorporation efficiency, as this parameter directly impacts the fidelity and functionality of synthesized nucleic acids.
Current quality control protocols in cell-free systems typically encompass multiple analytical checkpoints throughout the manufacturing process. Pre-reaction quality assessments focus on raw material purity, enzyme activity verification, and buffer composition validation. Real-time monitoring standards include temperature control, pH stability, and reaction kinetics tracking. Post-synthesis quality measures evaluate product yield, sequence accuracy, and structural integrity through various analytical techniques.
Standardization of nitrogenous base incorporation measurements requires precise analytical methodologies with defined acceptance criteria. High-performance liquid chromatography (HPLC) and mass spectrometry protocols must maintain coefficient of variation below 5% for quantitative measurements. Spectrophotometric assays should demonstrate linear response ranges spanning at least three orders of magnitude. Quality control samples with known incorporation efficiencies must be processed alongside test samples to ensure measurement accuracy and reproducibility.
Regulatory compliance frameworks for cell-free manufacturing increasingly emphasize process validation and documentation requirements. Good Manufacturing Practice (GMP) guidelines mandate comprehensive batch records, equipment qualification protocols, and personnel training documentation. Quality control laboratories must maintain calibrated instrumentation, validated analytical methods, and appropriate environmental controls. Deviation investigation procedures and corrective action protocols ensure continuous improvement of manufacturing processes.
Future quality control standards will likely incorporate advanced process analytical technologies and real-time release testing capabilities. Automated monitoring systems with integrated data analytics will enable continuous quality assessment throughout production cycles. These evolving standards will facilitate more efficient manufacturing processes while maintaining stringent quality requirements for therapeutic and research applications.
Current quality control protocols in cell-free systems typically encompass multiple analytical checkpoints throughout the manufacturing process. Pre-reaction quality assessments focus on raw material purity, enzyme activity verification, and buffer composition validation. Real-time monitoring standards include temperature control, pH stability, and reaction kinetics tracking. Post-synthesis quality measures evaluate product yield, sequence accuracy, and structural integrity through various analytical techniques.
Standardization of nitrogenous base incorporation measurements requires precise analytical methodologies with defined acceptance criteria. High-performance liquid chromatography (HPLC) and mass spectrometry protocols must maintain coefficient of variation below 5% for quantitative measurements. Spectrophotometric assays should demonstrate linear response ranges spanning at least three orders of magnitude. Quality control samples with known incorporation efficiencies must be processed alongside test samples to ensure measurement accuracy and reproducibility.
Regulatory compliance frameworks for cell-free manufacturing increasingly emphasize process validation and documentation requirements. Good Manufacturing Practice (GMP) guidelines mandate comprehensive batch records, equipment qualification protocols, and personnel training documentation. Quality control laboratories must maintain calibrated instrumentation, validated analytical methods, and appropriate environmental controls. Deviation investigation procedures and corrective action protocols ensure continuous improvement of manufacturing processes.
Future quality control standards will likely incorporate advanced process analytical technologies and real-time release testing capabilities. Automated monitoring systems with integrated data analytics will enable continuous quality assessment throughout production cycles. These evolving standards will facilitate more efficient manufacturing processes while maintaining stringent quality requirements for therapeutic and research applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!