PCA Pump vs Epidural: Pain Relief Efficiency
MAR 7, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
PCA Pump and Epidural Pain Management Background and Goals
Pain management represents one of the most critical challenges in modern healthcare, with millions of patients worldwide requiring effective analgesic interventions for various medical conditions. The evolution of pain relief methodologies has progressed significantly from traditional oral medications to sophisticated delivery systems that provide targeted, controlled analgesia. Among these advanced approaches, Patient-Controlled Analgesia (PCA) pumps and epidural anesthesia have emerged as two predominant techniques for managing moderate to severe pain, particularly in perioperative and chronic pain scenarios.
The historical development of pain management technologies began with the recognition that individualized pain control could significantly improve patient outcomes and satisfaction. PCA pump technology, first introduced in the 1970s, revolutionized pain management by allowing patients to self-administer predetermined doses of analgesic medications within safe parameters. This innovation addressed the fundamental challenge of pain perception variability among individuals and the need for timely intervention when pain levels fluctuate.
Epidural anesthesia, with roots dating back to the early 20th century, has evolved into a sophisticated regional anesthesia technique that provides targeted pain relief by delivering medications directly into the epidural space surrounding the spinal cord. This approach has become particularly valuable in obstetrics, major surgical procedures, and chronic pain management, offering the advantage of localized analgesia with reduced systemic side effects.
The contemporary healthcare landscape demands evidence-based approaches to pain management that optimize both efficacy and safety profiles. Current technological objectives focus on developing pain relief systems that minimize opioid consumption while maintaining superior analgesic effectiveness. The integration of smart monitoring systems, programmable delivery mechanisms, and real-time patient feedback has become essential for next-generation pain management solutions.
The primary technical goals driving current research include enhancing the precision of drug delivery, reducing the incidence of breakthrough pain episodes, and minimizing adverse effects associated with systemic medication distribution. Additionally, there is a growing emphasis on developing cost-effective solutions that can be implemented across diverse healthcare settings while maintaining consistent therapeutic outcomes.
Future technological aspirations encompass the development of personalized pain management protocols that leverage patient-specific physiological data, genetic markers, and pain sensitivity profiles. The convergence of artificial intelligence, biomonitoring technologies, and advanced pharmacokinetic modeling promises to create adaptive pain management systems that can automatically adjust treatment parameters based on real-time patient responses and predictive algorithms.
The historical development of pain management technologies began with the recognition that individualized pain control could significantly improve patient outcomes and satisfaction. PCA pump technology, first introduced in the 1970s, revolutionized pain management by allowing patients to self-administer predetermined doses of analgesic medications within safe parameters. This innovation addressed the fundamental challenge of pain perception variability among individuals and the need for timely intervention when pain levels fluctuate.
Epidural anesthesia, with roots dating back to the early 20th century, has evolved into a sophisticated regional anesthesia technique that provides targeted pain relief by delivering medications directly into the epidural space surrounding the spinal cord. This approach has become particularly valuable in obstetrics, major surgical procedures, and chronic pain management, offering the advantage of localized analgesia with reduced systemic side effects.
The contemporary healthcare landscape demands evidence-based approaches to pain management that optimize both efficacy and safety profiles. Current technological objectives focus on developing pain relief systems that minimize opioid consumption while maintaining superior analgesic effectiveness. The integration of smart monitoring systems, programmable delivery mechanisms, and real-time patient feedback has become essential for next-generation pain management solutions.
The primary technical goals driving current research include enhancing the precision of drug delivery, reducing the incidence of breakthrough pain episodes, and minimizing adverse effects associated with systemic medication distribution. Additionally, there is a growing emphasis on developing cost-effective solutions that can be implemented across diverse healthcare settings while maintaining consistent therapeutic outcomes.
Future technological aspirations encompass the development of personalized pain management protocols that leverage patient-specific physiological data, genetic markers, and pain sensitivity profiles. The convergence of artificial intelligence, biomonitoring technologies, and advanced pharmacokinetic modeling promises to create adaptive pain management systems that can automatically adjust treatment parameters based on real-time patient responses and predictive algorithms.
Market Demand for Advanced Pain Relief Solutions
The global pain management market continues to experience substantial growth driven by an aging population, increasing prevalence of chronic pain conditions, and rising awareness of effective pain relief options. Healthcare systems worldwide are seeking more efficient and patient-centered approaches to manage acute and chronic pain, particularly in perioperative and labor settings where traditional methods may fall short of patient expectations and clinical requirements.
Patient-controlled analgesia pumps represent a significant segment within the broader pain management device market, addressing the growing demand for personalized pain relief solutions. These devices cater to patients undergoing major surgical procedures, cancer treatments, and chronic pain management scenarios where conventional oral medications prove insufficient. The market demand stems from healthcare providers' need to improve patient satisfaction scores while reducing nursing workload and medication errors.
Epidural anesthesia and analgesia services constitute another substantial market segment, particularly within obstetric and surgical specialties. The demand for epidural procedures has grown consistently as more patients seek effective pain relief during childbirth and major surgeries. Healthcare facilities are investing in advanced epidural delivery systems and training programs to meet this increasing demand while ensuring safety and efficacy standards.
The comparative efficiency between PCA pumps and epidural techniques has become a critical consideration for healthcare administrators and clinicians making procurement and protocol decisions. Hospitals and surgical centers are evaluating these technologies based on patient outcomes, cost-effectiveness, staff training requirements, and overall operational efficiency. This evaluation process drives demand for evidence-based solutions that demonstrate superior pain relief effectiveness.
Market drivers include regulatory emphasis on patient satisfaction metrics, value-based healthcare initiatives, and the need to reduce opioid consumption through more targeted delivery methods. Healthcare providers are increasingly seeking pain management solutions that offer measurable outcomes, reduced side effects, and improved patient mobility during recovery periods.
The demand landscape also reflects regional variations in healthcare infrastructure, reimbursement policies, and clinical practice patterns. Developed markets show preference for technologically advanced systems with integrated monitoring capabilities, while emerging markets focus on cost-effective solutions that provide reliable pain relief without extensive infrastructure requirements.
Patient-controlled analgesia pumps represent a significant segment within the broader pain management device market, addressing the growing demand for personalized pain relief solutions. These devices cater to patients undergoing major surgical procedures, cancer treatments, and chronic pain management scenarios where conventional oral medications prove insufficient. The market demand stems from healthcare providers' need to improve patient satisfaction scores while reducing nursing workload and medication errors.
Epidural anesthesia and analgesia services constitute another substantial market segment, particularly within obstetric and surgical specialties. The demand for epidural procedures has grown consistently as more patients seek effective pain relief during childbirth and major surgeries. Healthcare facilities are investing in advanced epidural delivery systems and training programs to meet this increasing demand while ensuring safety and efficacy standards.
The comparative efficiency between PCA pumps and epidural techniques has become a critical consideration for healthcare administrators and clinicians making procurement and protocol decisions. Hospitals and surgical centers are evaluating these technologies based on patient outcomes, cost-effectiveness, staff training requirements, and overall operational efficiency. This evaluation process drives demand for evidence-based solutions that demonstrate superior pain relief effectiveness.
Market drivers include regulatory emphasis on patient satisfaction metrics, value-based healthcare initiatives, and the need to reduce opioid consumption through more targeted delivery methods. Healthcare providers are increasingly seeking pain management solutions that offer measurable outcomes, reduced side effects, and improved patient mobility during recovery periods.
The demand landscape also reflects regional variations in healthcare infrastructure, reimbursement policies, and clinical practice patterns. Developed markets show preference for technologically advanced systems with integrated monitoring capabilities, while emerging markets focus on cost-effective solutions that provide reliable pain relief without extensive infrastructure requirements.
Current State and Challenges in Pain Management Technologies
Pain management technologies have evolved significantly over the past decades, with Patient-Controlled Analgesia (PCA) pumps and epidural anesthesia emerging as two dominant approaches for managing moderate to severe pain in clinical settings. Both technologies represent sophisticated solutions that have transformed pain management practices, yet each faces distinct technical and operational challenges that impact their effectiveness and adoption rates.
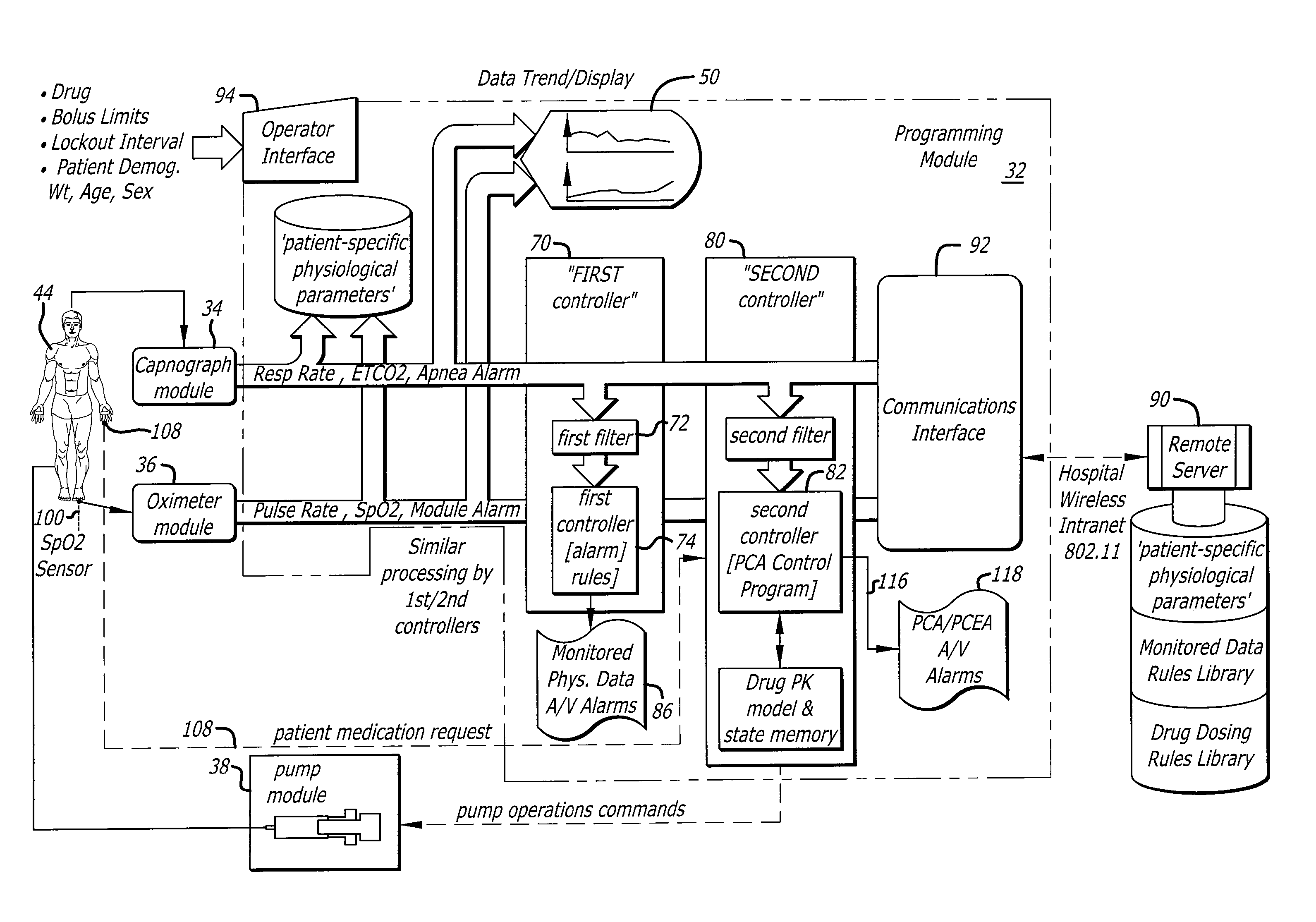
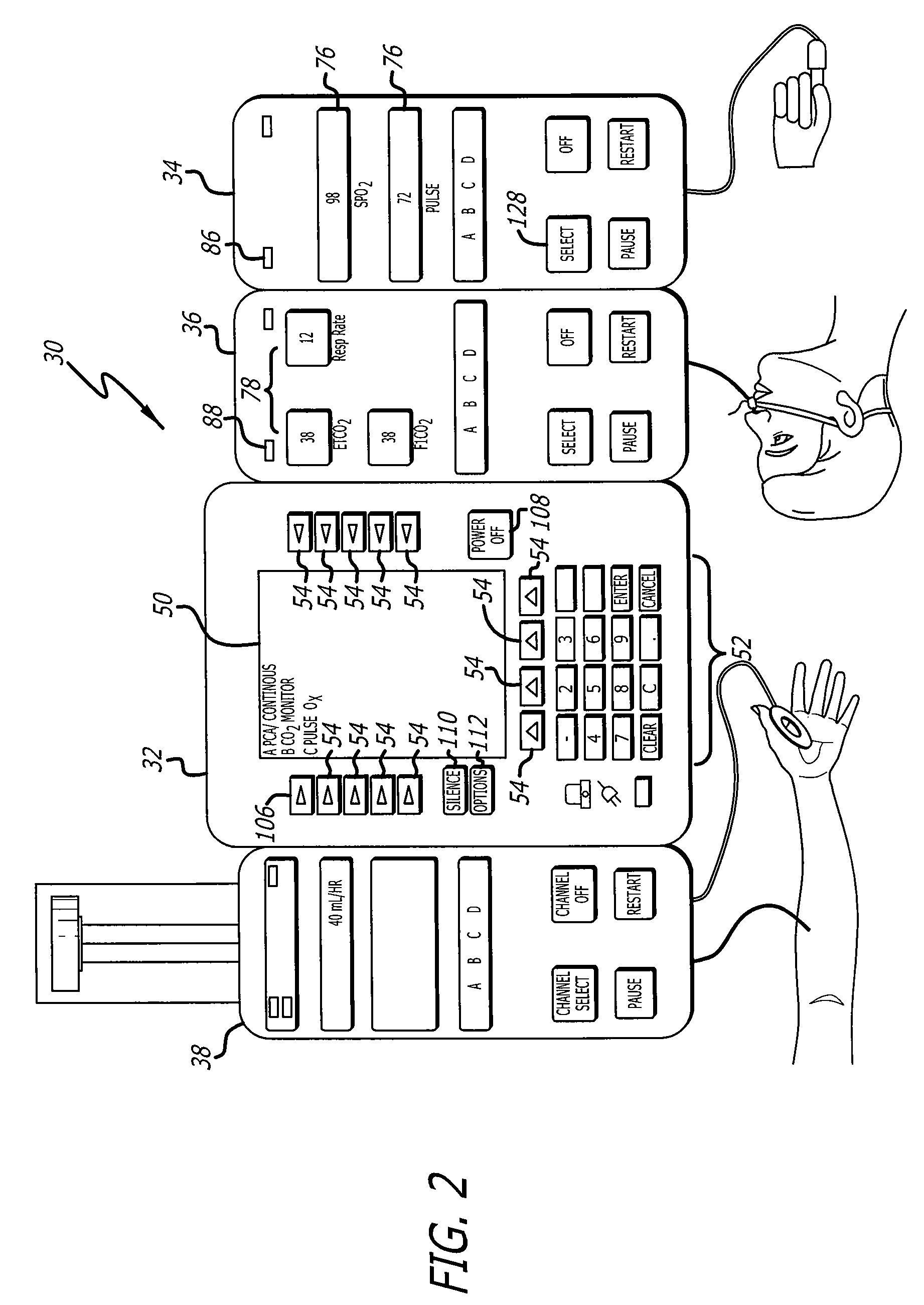
PCA pump technology currently operates through programmable infusion systems that allow patients to self-administer predetermined doses of analgesic medications within clinician-defined safety parameters. Modern PCA systems incorporate advanced microprocessor controls, wireless connectivity, and integrated safety mechanisms including lockout intervals and maximum dose limits. However, these systems face significant challenges including medication calculation errors, device programming complexities, and potential for patient misuse or overreliance on opioid-based medications.
Epidural pain management represents a more invasive but highly effective approach, utilizing catheter-based delivery systems to administer anesthetic agents directly into the epidural space. Current epidural technologies feature improved catheter designs, enhanced imaging guidance systems, and sophisticated drug delivery mechanisms. The primary challenges include technical difficulties in catheter placement, risk of complications such as dural puncture or infection, and the requirement for specialized clinical expertise for administration and monitoring.
Both technologies struggle with standardization issues across different healthcare systems and geographic regions. PCA pumps face interoperability challenges between different manufacturer systems and electronic health record integration difficulties. Epidural procedures encounter variability in technique protocols, inconsistent training standards among practitioners, and limited availability of specialized equipment in certain healthcare facilities.
Patient safety remains a critical challenge for both approaches. PCA systems must address concerns regarding opioid dependency, respiratory depression risks, and the need for continuous patient monitoring. Epidural techniques face challenges related to procedural complications, contraindications in certain patient populations, and the requirement for immediate access to emergency interventions when complications arise.
The integration of digital health technologies presents both opportunities and challenges. While smart PCA pumps with connectivity features offer improved monitoring capabilities, they also introduce cybersecurity vulnerabilities and data privacy concerns. Similarly, epidural procedures benefit from advanced imaging technologies but require significant capital investments and specialized training programs that many healthcare facilities struggle to implement effectively.
PCA pump technology currently operates through programmable infusion systems that allow patients to self-administer predetermined doses of analgesic medications within clinician-defined safety parameters. Modern PCA systems incorporate advanced microprocessor controls, wireless connectivity, and integrated safety mechanisms including lockout intervals and maximum dose limits. However, these systems face significant challenges including medication calculation errors, device programming complexities, and potential for patient misuse or overreliance on opioid-based medications.
Epidural pain management represents a more invasive but highly effective approach, utilizing catheter-based delivery systems to administer anesthetic agents directly into the epidural space. Current epidural technologies feature improved catheter designs, enhanced imaging guidance systems, and sophisticated drug delivery mechanisms. The primary challenges include technical difficulties in catheter placement, risk of complications such as dural puncture or infection, and the requirement for specialized clinical expertise for administration and monitoring.
Both technologies struggle with standardization issues across different healthcare systems and geographic regions. PCA pumps face interoperability challenges between different manufacturer systems and electronic health record integration difficulties. Epidural procedures encounter variability in technique protocols, inconsistent training standards among practitioners, and limited availability of specialized equipment in certain healthcare facilities.
Patient safety remains a critical challenge for both approaches. PCA systems must address concerns regarding opioid dependency, respiratory depression risks, and the need for continuous patient monitoring. Epidural techniques face challenges related to procedural complications, contraindications in certain patient populations, and the requirement for immediate access to emergency interventions when complications arise.
The integration of digital health technologies presents both opportunities and challenges. While smart PCA pumps with connectivity features offer improved monitoring capabilities, they also introduce cybersecurity vulnerabilities and data privacy concerns. Similarly, epidural procedures benefit from advanced imaging technologies but require significant capital investments and specialized training programs that many healthcare facilities struggle to implement effectively.
Existing PCA and Epidural Technology Solutions
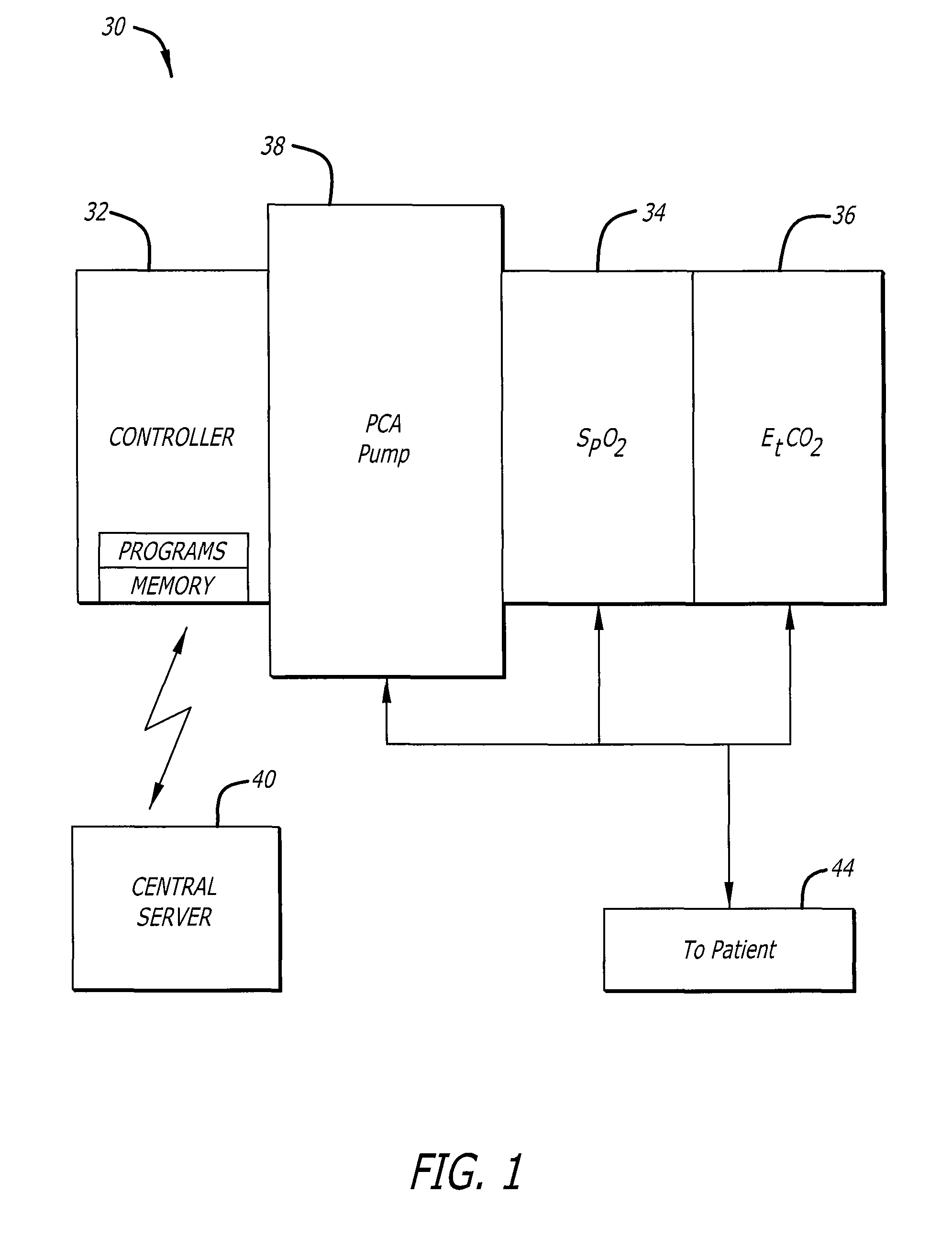
01 Patient-controlled analgesia (PCA) pump systems and devices
PCA pump systems allow patients to self-administer pain medication at controlled intervals, providing on-demand pain relief. These systems typically include programmable pumps with safety features such as lockout intervals, dose limits, and monitoring capabilities. The technology enables patients to manage their pain more effectively while maintaining safety through preset parameters that prevent overdosing.- Patient-controlled analgesia (PCA) pump systems and devices: PCA pump systems allow patients to self-administer pain medication at controlled intervals, providing on-demand pain relief. These systems typically include programmable pumps with safety lockout features to prevent overdosing. The technology enables patients to manage their pain levels actively while maintaining safety parameters set by healthcare providers. PCA pumps can deliver various analgesic medications intravenously or subcutaneously, offering flexibility in pain management approaches.
- Epidural drug delivery systems and catheter technologies: Epidural delivery systems involve the administration of analgesic medications directly into the epidural space surrounding the spinal cord. These systems utilize specialized catheters and infusion devices designed for continuous or intermittent delivery of pain relief medications. The technology focuses on precise placement and maintenance of epidural catheters to ensure effective pain control while minimizing complications. Advanced epidural systems may include features for controlled flow rates and pressure monitoring.
- Comparative pain management protocols and monitoring systems: Systems and methods for comparing different pain relief modalities involve monitoring patient outcomes, pain scores, and medication consumption. These technologies enable healthcare providers to assess the relative effectiveness of different analgesic approaches through data collection and analysis. Monitoring systems may track vital signs, pain levels, and adverse events to determine optimal pain management strategies. The comparison protocols help establish evidence-based guidelines for selecting appropriate pain relief methods for specific patient populations.
- Combination and hybrid pain management delivery systems: Hybrid systems integrate features of both patient-controlled and continuous infusion methods to optimize pain relief. These devices may allow switching between different delivery modes or combining multiple analgesic approaches. The technology enables customization of pain management protocols based on individual patient needs and clinical situations. Advanced systems may incorporate feedback mechanisms to adjust delivery parameters automatically based on patient response.
- Safety features and adverse event prevention in analgesic delivery: Safety mechanisms in pain management systems include dose limiting features, alarm systems, and fail-safe protocols to prevent medication errors and overdoses. These technologies incorporate monitoring capabilities to detect potential complications such as respiratory depression or catheter displacement. Advanced safety features may include biometric authentication, automated dose calculation, and real-time patient monitoring integration. The systems are designed to balance effective pain relief with minimization of risks associated with analgesic administration.
02 Epidural drug delivery systems and catheter technologies
Epidural delivery systems involve the administration of analgesic medications through catheters placed in the epidural space. These systems provide continuous or intermittent pain relief through specialized catheter designs, infusion pumps, and delivery mechanisms. The technology focuses on precise placement, secure fixation, and controlled medication delivery to the epidural space for effective pain management.Expand Specific Solutions03 Comparative pain management protocols and monitoring systems
Systems and methods for comparing different pain relief modalities through monitoring patient outcomes, pain scores, and medication consumption. These technologies include data collection devices, assessment tools, and analytical systems that evaluate the effectiveness of different pain management approaches. The systems may incorporate sensors, feedback mechanisms, and reporting capabilities to track pain relief efficiency.Expand Specific Solutions04 Combined or hybrid pain management delivery systems
Integrated systems that combine features of both patient-controlled and continuous delivery methods, allowing for flexible pain management strategies. These hybrid approaches may include devices capable of switching between different delivery modes, combining epidural and systemic delivery, or integrating multiple pain relief technologies into a single platform for optimized patient care.Expand Specific Solutions05 Safety features and drug administration control mechanisms
Advanced safety systems designed to prevent medication errors, overdosing, and complications in pain management. These include lockout mechanisms, dose verification systems, tamper-resistant features, and automated monitoring capabilities. The technologies focus on ensuring patient safety while maintaining effective pain control through intelligent control algorithms and fail-safe mechanisms.Expand Specific Solutions
Key Players in Pain Management Device Industry
The PCA pump versus epidural pain relief efficiency landscape represents a mature market segment within the broader pain management industry, currently valued at several billion dollars globally and experiencing steady growth driven by aging populations and increased surgical procedures. The technology maturity varies significantly across key players, with established medical device manufacturers like Baxter International, Koninklijke Philips NV, and Nipro Corp. leading in traditional infusion pump technologies, while specialized companies such as Jiangsu Apon Medical Technology, Medcaptain Medical Technology, and Curlin Medical focus on advanced PCA systems with wireless capabilities and smart monitoring features. Chinese manufacturers including Jiangsu Renxian Medical Technology are emerging as competitive forces, particularly in IoT-enabled analgesic management systems. The competitive landscape shows consolidation among major players while innovation centers on digitalization, remote monitoring, and personalized pain management protocols, indicating a transition from hardware-focused to software-integrated solutions.
Baxter International, Inc.
Technical Solution: Baxter has developed advanced PCA pump systems with sophisticated drug delivery algorithms and safety features. Their PCA pumps incorporate multi-modal pain management protocols, allowing for precise opioid titration while minimizing side effects. The company's technology includes smart pump features with drug libraries, dose error reduction systems, and wireless connectivity for remote monitoring. Their pumps support various administration routes and can be integrated with hospital information systems for comprehensive pain management tracking. Baxter's approach emphasizes patient-controlled analgesia optimization through real-time monitoring and automated safety checks, providing healthcare providers with detailed analytics on pain relief effectiveness and medication consumption patterns.
Strengths: Established market leader with comprehensive safety features and hospital system integration. Weaknesses: Higher cost compared to basic PCA systems and requires extensive staff training.
CareFusion 303, Inc.
Technical Solution: CareFusion has developed intelligent PCA pump technology focusing on medication safety and clinical workflow optimization. Their systems feature advanced drug recognition technology, barcode scanning capabilities, and integrated clinical decision support tools. The pumps include sophisticated alarm management systems and provide detailed reporting on pain management outcomes. CareFusion's technology emphasizes reducing medication errors through automated verification processes and real-time monitoring of patient responses. Their PCA systems support multiple pain management protocols and can adapt dosing based on patient feedback and physiological parameters, enabling personalized pain relief strategies.
Strengths: Strong focus on medication safety and error reduction with user-friendly interfaces. Weaknesses: Limited market presence compared to larger competitors and higher maintenance requirements.
Core Innovations in Pain Relief Efficiency Technologies
System and method for optimizing control of PCA and PCEA system
PatentActiveUS7871394B2
Innovation
- A system and method that utilize a second controller to process physiological signals and request signals differently from the first controller, filtering data with techniques like moving averages and adaptive filters, and incorporating pharmacokinetic modeling to optimize PCA device operation, allowing for automatic inhibition of medication delivery during potential respiratory depression while minimizing false alarms.
Secure patient-controlled analgesia
PatentWO2021236679A1
Innovation
- A system that includes a drug delivery device, a drug control device, and a control unit capable of capturing biometric information, such as fingerprints, to authenticate the patient before administering medication, ensuring only authorized patients can self-administer doses based on their biometric data and real-time physiological signals.
Clinical Safety Standards for Pain Management Devices
Clinical safety standards for pain management devices represent a critical framework governing the deployment and operation of both Patient-Controlled Analgesia (PCA) pumps and epidural systems in healthcare environments. These standards encompass comprehensive protocols designed to minimize patient risk while maximizing therapeutic efficacy across different pain management modalities.
The regulatory landscape for pain management devices is primarily governed by FDA guidelines in the United States, CE marking requirements in Europe, and corresponding national health authorities worldwide. PCA pumps must comply with IEC 60601-2-24 standards, which specifically address the safety and essential performance requirements for infusion pumps and controllers. These devices require rigorous validation of dosing accuracy, occlusion detection capabilities, and fail-safe mechanisms to prevent overdose scenarios.
Epidural pain management systems operate under distinct safety protocols due to their invasive nature and proximity to critical neurological structures. Clinical standards mandate specific training certifications for healthcare providers, sterile insertion techniques, and continuous monitoring protocols. The American Society of Anesthesiologists has established comprehensive guidelines addressing patient selection criteria, contraindication screening, and post-procedural monitoring requirements.
Device interoperability and data security represent emerging safety considerations as pain management systems increasingly integrate with electronic health records and hospital information systems. Standards such as HL7 FHIR and IHE profiles ensure secure data transmission while maintaining patient privacy compliance under HIPAA regulations.
Quality assurance protocols require regular calibration schedules for PCA pumps, typically every six months, with documented accuracy verification within ±5% tolerance levels. Epidural equipment undergoes stringent sterilization validation following CDC guidelines, with biological indicators confirming sterility assurance levels of 10^-6.
Risk management frameworks incorporate failure mode and effects analysis (FMEA) methodologies to identify potential device malfunctions and their clinical consequences. These analyses inform preventive maintenance schedules, staff training protocols, and emergency response procedures. Clinical governance structures mandate incident reporting systems that track device-related adverse events, enabling continuous improvement of safety protocols and informing future standard revisions.
The regulatory landscape for pain management devices is primarily governed by FDA guidelines in the United States, CE marking requirements in Europe, and corresponding national health authorities worldwide. PCA pumps must comply with IEC 60601-2-24 standards, which specifically address the safety and essential performance requirements for infusion pumps and controllers. These devices require rigorous validation of dosing accuracy, occlusion detection capabilities, and fail-safe mechanisms to prevent overdose scenarios.
Epidural pain management systems operate under distinct safety protocols due to their invasive nature and proximity to critical neurological structures. Clinical standards mandate specific training certifications for healthcare providers, sterile insertion techniques, and continuous monitoring protocols. The American Society of Anesthesiologists has established comprehensive guidelines addressing patient selection criteria, contraindication screening, and post-procedural monitoring requirements.
Device interoperability and data security represent emerging safety considerations as pain management systems increasingly integrate with electronic health records and hospital information systems. Standards such as HL7 FHIR and IHE profiles ensure secure data transmission while maintaining patient privacy compliance under HIPAA regulations.
Quality assurance protocols require regular calibration schedules for PCA pumps, typically every six months, with documented accuracy verification within ±5% tolerance levels. Epidural equipment undergoes stringent sterilization validation following CDC guidelines, with biological indicators confirming sterility assurance levels of 10^-6.
Risk management frameworks incorporate failure mode and effects analysis (FMEA) methodologies to identify potential device malfunctions and their clinical consequences. These analyses inform preventive maintenance schedules, staff training protocols, and emergency response procedures. Clinical governance structures mandate incident reporting systems that track device-related adverse events, enabling continuous improvement of safety protocols and informing future standard revisions.
Patient Safety and Opioid Crisis Considerations
Patient safety considerations represent a critical dimension in evaluating PCA pumps versus epidural analgesia, particularly given the heightened awareness of opioid-related risks in healthcare settings. Both modalities carry distinct safety profiles that must be carefully weighed against their analgesic benefits, with particular attention to medication errors, respiratory depression, and long-term dependency risks.
PCA pump systems present unique safety challenges related to programming errors, device malfunctions, and patient misuse. Studies indicate that medication errors occur in approximately 1-5% of PCA administrations, with programming mistakes being the most common cause. Modern PCA devices incorporate multiple safety features including dose limits, lockout intervals, and smart pump technology with drug libraries to minimize these risks. However, the potential for respiratory depression remains a concern, particularly in opioid-naive patients or those with comorbidities affecting respiratory function.
Epidural analgesia demonstrates a different safety profile, with risks primarily associated with catheter placement and maintenance. Serious complications such as epidural hematoma or abscess occur in less than 0.1% of cases, while temporary neurological deficits affect approximately 0.02% of patients. The technique's advantage lies in its ability to provide effective analgesia with significantly lower systemic opioid exposure, thereby reducing respiratory depression risk and opioid-related side effects.
The opioid crisis has fundamentally altered the risk-benefit analysis of pain management strategies. Healthcare institutions increasingly prioritize multimodal approaches that minimize opioid consumption while maintaining adequate analgesia. Epidural techniques align well with these objectives, as they can reduce total opioid requirements by 30-50% compared to systemic administration. This reduction translates to decreased risk of postoperative opioid dependence, which affects approximately 6-10% of surgical patients receiving traditional opioid regimens.
Regulatory responses to the opioid crisis have introduced additional safety requirements for both modalities. Enhanced monitoring protocols, mandatory education programs, and stricter documentation requirements now govern opioid administration regardless of delivery method. These measures, while improving safety, have increased implementation complexity and associated costs for healthcare providers seeking to optimize pain management protocols.
PCA pump systems present unique safety challenges related to programming errors, device malfunctions, and patient misuse. Studies indicate that medication errors occur in approximately 1-5% of PCA administrations, with programming mistakes being the most common cause. Modern PCA devices incorporate multiple safety features including dose limits, lockout intervals, and smart pump technology with drug libraries to minimize these risks. However, the potential for respiratory depression remains a concern, particularly in opioid-naive patients or those with comorbidities affecting respiratory function.
Epidural analgesia demonstrates a different safety profile, with risks primarily associated with catheter placement and maintenance. Serious complications such as epidural hematoma or abscess occur in less than 0.1% of cases, while temporary neurological deficits affect approximately 0.02% of patients. The technique's advantage lies in its ability to provide effective analgesia with significantly lower systemic opioid exposure, thereby reducing respiratory depression risk and opioid-related side effects.
The opioid crisis has fundamentally altered the risk-benefit analysis of pain management strategies. Healthcare institutions increasingly prioritize multimodal approaches that minimize opioid consumption while maintaining adequate analgesia. Epidural techniques align well with these objectives, as they can reduce total opioid requirements by 30-50% compared to systemic administration. This reduction translates to decreased risk of postoperative opioid dependence, which affects approximately 6-10% of surgical patients receiving traditional opioid regimens.
Regulatory responses to the opioid crisis have introduced additional safety requirements for both modalities. Enhanced monitoring protocols, mandatory education programs, and stricter documentation requirements now govern opioid administration regardless of delivery method. These measures, while improving safety, have increased implementation complexity and associated costs for healthcare providers seeking to optimize pain management protocols.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!