PCA Pump vs On-Demand Systems: User Preference
MAR 7, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
PCA Pump Technology Background and Clinical Goals
Patient-Controlled Analgesia (PCA) pump technology emerged in the 1960s as a revolutionary approach to pain management, fundamentally transforming how healthcare providers deliver analgesic medications. The concept was initially developed to address the limitations of traditional nurse-administered pain relief schedules, which often resulted in inadequate pain control due to fixed dosing intervals and delays in medication delivery.
The foundational principle of PCA technology centers on empowering patients to self-administer predetermined doses of analgesic medication within clinically established safety parameters. This paradigm shift from provider-controlled to patient-controlled analgesia represents a significant advancement in personalized medicine, allowing for more responsive and individualized pain management strategies.
Early PCA systems utilized mechanical infusion pumps with basic safety lockout mechanisms to prevent overdosing. These systems incorporated programmable features including bolus dose amounts, lockout intervals, and maximum dose limits within specified time periods. The technology evolved rapidly through the 1970s and 1980s, incorporating electronic controls, digital displays, and enhanced safety algorithms.
The clinical rationale for PCA pump development stems from pharmacokinetic principles demonstrating significant inter-patient variability in analgesic requirements. Traditional fixed-schedule dosing often results in periods of inadequate analgesia alternating with potential oversedation, creating suboptimal therapeutic outcomes. PCA technology addresses these limitations by enabling real-time dose titration based on individual patient needs and pain perception.
Modern PCA systems have expanded beyond simple bolus delivery to include continuous background infusions, multiple drug compatibility, and sophisticated monitoring capabilities. Contemporary devices integrate wireless connectivity, electronic health record integration, and advanced safety features including biometric authentication and smart pump technology with drug libraries.
The primary clinical goals of PCA pump technology encompass multiple therapeutic objectives. Pain control optimization remains the fundamental goal, aiming to maintain analgesic plasma concentrations within the therapeutic window while minimizing breakthrough pain episodes. Patient satisfaction enhancement represents another critical objective, as PCA systems provide patients with greater autonomy and control over their pain management experience.
Safety optimization constitutes a paramount clinical goal, with modern PCA systems incorporating multiple redundant safety mechanisms to prevent medication errors and adverse events. These include dose limits, temporal restrictions, and integration with clinical decision support systems. Additionally, PCA technology aims to improve clinical efficiency by reducing nursing workload associated with frequent pain assessments and medication administration while maintaining high-quality patient care standards.
The foundational principle of PCA technology centers on empowering patients to self-administer predetermined doses of analgesic medication within clinically established safety parameters. This paradigm shift from provider-controlled to patient-controlled analgesia represents a significant advancement in personalized medicine, allowing for more responsive and individualized pain management strategies.
Early PCA systems utilized mechanical infusion pumps with basic safety lockout mechanisms to prevent overdosing. These systems incorporated programmable features including bolus dose amounts, lockout intervals, and maximum dose limits within specified time periods. The technology evolved rapidly through the 1970s and 1980s, incorporating electronic controls, digital displays, and enhanced safety algorithms.
The clinical rationale for PCA pump development stems from pharmacokinetic principles demonstrating significant inter-patient variability in analgesic requirements. Traditional fixed-schedule dosing often results in periods of inadequate analgesia alternating with potential oversedation, creating suboptimal therapeutic outcomes. PCA technology addresses these limitations by enabling real-time dose titration based on individual patient needs and pain perception.
Modern PCA systems have expanded beyond simple bolus delivery to include continuous background infusions, multiple drug compatibility, and sophisticated monitoring capabilities. Contemporary devices integrate wireless connectivity, electronic health record integration, and advanced safety features including biometric authentication and smart pump technology with drug libraries.
The primary clinical goals of PCA pump technology encompass multiple therapeutic objectives. Pain control optimization remains the fundamental goal, aiming to maintain analgesic plasma concentrations within the therapeutic window while minimizing breakthrough pain episodes. Patient satisfaction enhancement represents another critical objective, as PCA systems provide patients with greater autonomy and control over their pain management experience.
Safety optimization constitutes a paramount clinical goal, with modern PCA systems incorporating multiple redundant safety mechanisms to prevent medication errors and adverse events. These include dose limits, temporal restrictions, and integration with clinical decision support systems. Additionally, PCA technology aims to improve clinical efficiency by reducing nursing workload associated with frequent pain assessments and medication administration while maintaining high-quality patient care standards.
Market Demand for Patient-Controlled Analgesia Systems
The global patient-controlled analgesia systems market demonstrates robust growth driven by increasing surgical procedures, rising prevalence of chronic pain conditions, and growing awareness of personalized pain management approaches. Healthcare institutions worldwide are recognizing the clinical and economic benefits of PCA systems, which enable patients to self-administer predetermined doses of analgesic medications within safe parameters established by healthcare providers.
Hospital adoption rates continue to accelerate as healthcare administrators seek solutions that improve patient satisfaction scores while optimizing nursing workflow efficiency. The shift toward value-based healthcare models has intensified focus on patient-reported outcomes, positioning PCA systems as strategic investments that directly impact quality metrics and reimbursement rates. Post-surgical recovery units, oncology departments, and intensive care facilities represent the primary demand drivers for these systems.
Regional market dynamics reveal significant variations in adoption patterns and preferences between PCA pump technologies and on-demand systems. North American healthcare facilities demonstrate strong preference for advanced PCA pump solutions with integrated safety features and electronic health record connectivity. European markets show balanced adoption of both system types, with emphasis on cost-effectiveness and regulatory compliance. Emerging markets in Asia-Pacific regions exhibit growing demand for simplified on-demand systems that require minimal technical infrastructure.
The aging global population creates sustained demand pressure as elderly patients require more frequent surgical interventions and chronic pain management solutions. Cancer treatment centers represent a particularly high-growth segment, where patient comfort during extended treatment protocols directly influences treatment adherence and outcomes. Ambulatory surgical centers are emerging as new demand sources, seeking portable and user-friendly PCA solutions that support same-day discharge protocols.
Healthcare cost containment initiatives influence purchasing decisions, with institutions evaluating total cost of ownership including device acquisition, maintenance, training, and medication waste reduction. The demand for systems that demonstrate measurable improvements in patient satisfaction scores, reduced nursing call frequency, and shorter hospital stays continues to drive market expansion across all healthcare facility types.
Hospital adoption rates continue to accelerate as healthcare administrators seek solutions that improve patient satisfaction scores while optimizing nursing workflow efficiency. The shift toward value-based healthcare models has intensified focus on patient-reported outcomes, positioning PCA systems as strategic investments that directly impact quality metrics and reimbursement rates. Post-surgical recovery units, oncology departments, and intensive care facilities represent the primary demand drivers for these systems.
Regional market dynamics reveal significant variations in adoption patterns and preferences between PCA pump technologies and on-demand systems. North American healthcare facilities demonstrate strong preference for advanced PCA pump solutions with integrated safety features and electronic health record connectivity. European markets show balanced adoption of both system types, with emphasis on cost-effectiveness and regulatory compliance. Emerging markets in Asia-Pacific regions exhibit growing demand for simplified on-demand systems that require minimal technical infrastructure.
The aging global population creates sustained demand pressure as elderly patients require more frequent surgical interventions and chronic pain management solutions. Cancer treatment centers represent a particularly high-growth segment, where patient comfort during extended treatment protocols directly influences treatment adherence and outcomes. Ambulatory surgical centers are emerging as new demand sources, seeking portable and user-friendly PCA solutions that support same-day discharge protocols.
Healthcare cost containment initiatives influence purchasing decisions, with institutions evaluating total cost of ownership including device acquisition, maintenance, training, and medication waste reduction. The demand for systems that demonstrate measurable improvements in patient satisfaction scores, reduced nursing call frequency, and shorter hospital stays continues to drive market expansion across all healthcare facility types.
Current State of PCA vs On-Demand Pain Management
The current landscape of pain management systems presents a complex interplay between Patient-Controlled Analgesia (PCA) pumps and on-demand analgesic delivery systems, each addressing distinct clinical needs and patient populations. PCA pumps have established themselves as the gold standard for post-surgical and chronic pain management in hospital settings, offering continuous baseline infusion with patient-activated bolus doses. These systems typically utilize opioid medications such as morphine, fentanyl, or hydromorphone, delivered through intravenous, subcutaneous, or epidural routes.
On-demand pain management systems encompass a broader category of interventions, ranging from traditional nurse-administered medications to newer smart delivery devices and wearable pain relief technologies. These systems include oral medications, transdermal patches, sublingual tablets, and emerging technologies like transcutaneous electrical nerve stimulation (TENS) units with smartphone connectivity. The fundamental distinction lies in the timing and control mechanism, where on-demand systems require either healthcare provider intervention or patient activation without continuous baseline delivery.
Current clinical practice demonstrates significant variation in system selection based on patient acuity, surgical complexity, and institutional protocols. PCA pumps dominate in intensive care units, post-operative recovery areas, and oncology wards where pain intensity and frequency justify continuous monitoring and precise dosing control. Conversely, on-demand systems find greater application in outpatient settings, emergency departments, and step-down care environments where intermittent pain relief suffices.
The technological infrastructure supporting these systems has evolved considerably, with modern PCA pumps incorporating advanced safety features including dose limits, lockout intervals, and integrated monitoring capabilities. Smart pumps now offer wireless connectivity, enabling real-time data transmission to electronic health records and centralized monitoring stations. On-demand systems have similarly advanced, with digital pill dispensers, mobile applications for pain tracking, and AI-powered dosing recommendations becoming increasingly prevalent.
Regulatory frameworks governing both systems continue to evolve, with the FDA implementing stricter guidelines for PCA pump safety features and medication error prevention. The Drug Enforcement Administration maintains rigorous oversight of controlled substance management in both delivery modalities, influencing system design and implementation protocols. Healthcare institutions must navigate complex compliance requirements while balancing patient safety, efficacy, and operational efficiency considerations in their pain management system selection and deployment strategies.
On-demand pain management systems encompass a broader category of interventions, ranging from traditional nurse-administered medications to newer smart delivery devices and wearable pain relief technologies. These systems include oral medications, transdermal patches, sublingual tablets, and emerging technologies like transcutaneous electrical nerve stimulation (TENS) units with smartphone connectivity. The fundamental distinction lies in the timing and control mechanism, where on-demand systems require either healthcare provider intervention or patient activation without continuous baseline delivery.
Current clinical practice demonstrates significant variation in system selection based on patient acuity, surgical complexity, and institutional protocols. PCA pumps dominate in intensive care units, post-operative recovery areas, and oncology wards where pain intensity and frequency justify continuous monitoring and precise dosing control. Conversely, on-demand systems find greater application in outpatient settings, emergency departments, and step-down care environments where intermittent pain relief suffices.
The technological infrastructure supporting these systems has evolved considerably, with modern PCA pumps incorporating advanced safety features including dose limits, lockout intervals, and integrated monitoring capabilities. Smart pumps now offer wireless connectivity, enabling real-time data transmission to electronic health records and centralized monitoring stations. On-demand systems have similarly advanced, with digital pill dispensers, mobile applications for pain tracking, and AI-powered dosing recommendations becoming increasingly prevalent.
Regulatory frameworks governing both systems continue to evolve, with the FDA implementing stricter guidelines for PCA pump safety features and medication error prevention. The Drug Enforcement Administration maintains rigorous oversight of controlled substance management in both delivery modalities, influencing system design and implementation protocols. Healthcare institutions must navigate complex compliance requirements while balancing patient safety, efficacy, and operational efficiency considerations in their pain management system selection and deployment strategies.
Existing PCA and On-Demand System Solutions
01 Patient-controlled analgesia (PCA) pump systems with customizable delivery modes
PCA pump systems can be designed with multiple delivery modes that allow patients to select their preferred method of pain medication administration. These systems typically offer both continuous infusion and bolus dose options, enabling patients to choose between steady medication delivery or on-demand doses based on their pain management needs. The flexibility in delivery modes enhances patient satisfaction and allows for personalized pain control strategies that can be adjusted according to individual comfort levels and medical requirements.- Patient-controlled analgesia (PCA) pump systems with programmable delivery modes: PCA pump systems can be designed with programmable delivery modes that allow patients to self-administer pain medication within preset safety limits. These systems typically include features for bolus dosing, continuous infusion, and lockout intervals to prevent overdosing. The programmable nature allows healthcare providers to customize delivery parameters based on individual patient needs while maintaining safety protocols. Advanced systems may include memory functions to track usage patterns and adjust settings accordingly.
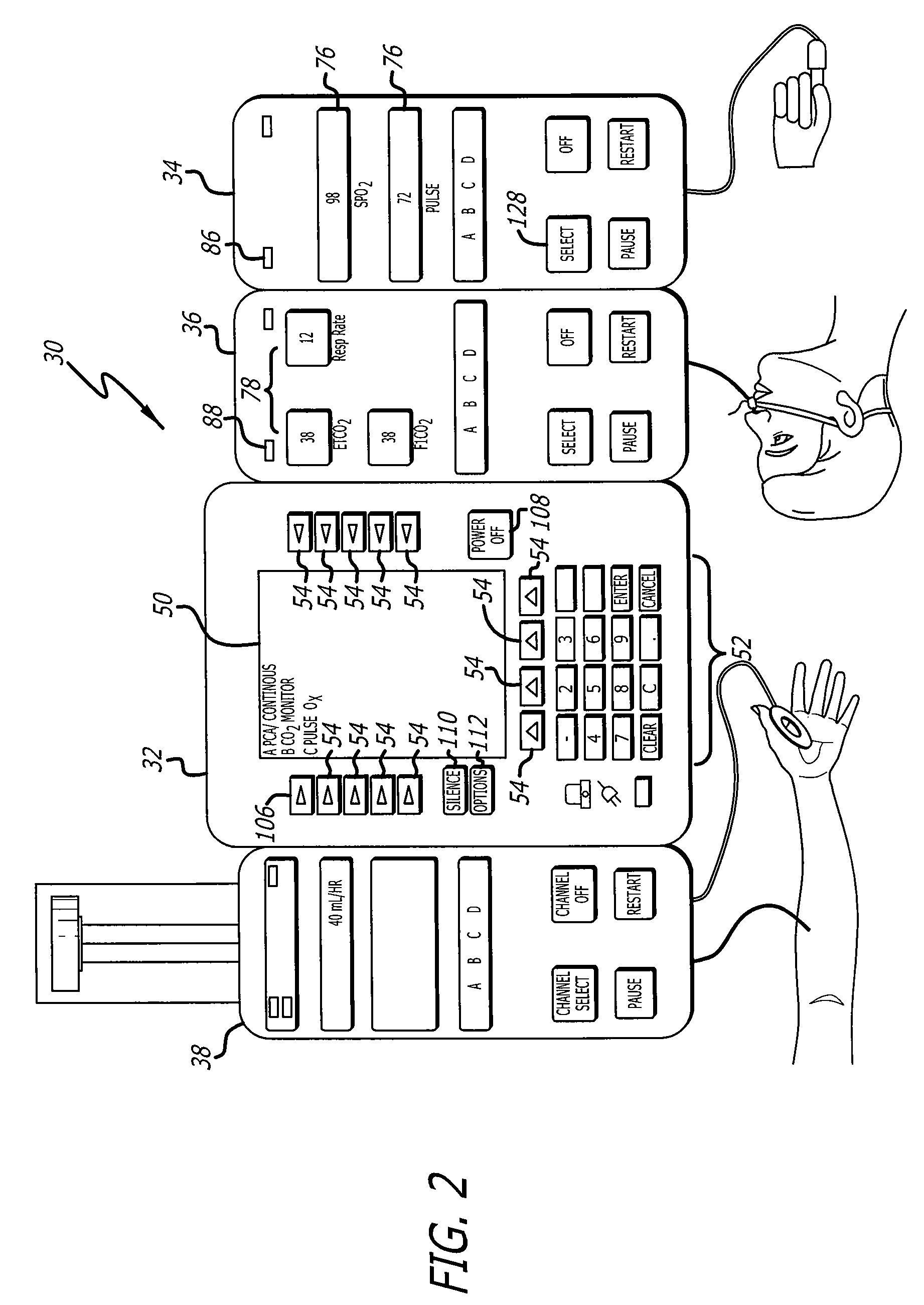
- User interface design and control mechanisms for enhanced patient interaction: Modern infusion systems incorporate intuitive user interfaces with simplified control mechanisms to improve patient interaction and satisfaction. These interfaces may include touch screens, button controls, or wireless remote devices that allow patients to easily request medication doses. The design focuses on reducing complexity while providing clear feedback about system status, remaining medication, and lockout periods. Enhanced visual and auditory indicators help patients understand when doses are available and successfully delivered.
- On-demand bolus delivery with safety lockout features: On-demand systems incorporate safety mechanisms that allow patients to request additional medication doses while preventing excessive administration through lockout intervals. These features include time-based lockout periods between doses, maximum dose limits per time period, and cumulative dose tracking. The systems are designed to balance patient autonomy with clinical safety requirements, ensuring that patients can manage breakthrough pain while staying within therapeutic ranges established by healthcare providers.
- Comparative studies and preference analysis between PCA and on-demand delivery modes: Research and clinical evaluations have been conducted to assess patient preferences between continuous PCA delivery and on-demand bolus systems. These studies examine factors such as pain control effectiveness, patient satisfaction, ease of use, and overall comfort with different delivery modalities. Findings help guide the development of hybrid systems that combine features of both approaches, allowing customization based on patient preferences, medical conditions, and treatment protocols.
- Smart monitoring and feedback systems for optimizing user experience: Advanced pump systems integrate monitoring capabilities that track patient usage patterns, medication consumption, and pain management effectiveness. These systems can provide real-time feedback to both patients and healthcare providers, enabling data-driven adjustments to therapy protocols. Features may include wireless connectivity for remote monitoring, alert systems for unusual usage patterns, and analytics tools that help identify optimal delivery parameters for individual patients. The integration of smart technology enhances both safety and patient satisfaction.
02 User interface design and control mechanisms for enhanced patient interaction
Modern infusion systems incorporate intuitive user interfaces that allow patients to easily request medication doses through simple button presses or touch controls. These interfaces are designed with patient safety in mind, featuring lockout intervals to prevent overdosing and clear visual or auditory feedback to confirm dose delivery. The control mechanisms are engineered to be accessible for patients with varying levels of mobility and cognitive function, ensuring that all users can effectively manage their pain relief while maintaining safety protocols.Expand Specific Solutions03 Programmable dosing parameters and safety features
Advanced pump systems allow healthcare providers to program specific dosing parameters including bolus dose amounts, lockout intervals, and maximum dose limits based on individual patient needs and preferences. These programmable features ensure that patient preferences can be accommodated while maintaining strict safety boundaries. The systems include fail-safe mechanisms and alarm functions that alert both patients and medical staff to any irregularities in medication delivery, providing an additional layer of security in patient-controlled administration.Expand Specific Solutions04 Hybrid systems combining continuous and on-demand delivery
Hybrid infusion systems integrate both basal continuous infusion and patient-controlled bolus capabilities, allowing patients to maintain a baseline level of medication while having the option to self-administer additional doses as needed. This dual-mode approach caters to patient preferences by providing consistent pain management with the flexibility to address breakthrough pain episodes. The systems can be configured to emphasize either continuous or on-demand delivery based on patient feedback and clinical assessment of pain patterns.Expand Specific Solutions05 Monitoring and feedback systems for patient satisfaction assessment
Contemporary infusion devices incorporate monitoring capabilities that track patient usage patterns, including frequency of on-demand requests and timing of dose administrations. This data collection enables healthcare providers to assess patient satisfaction with their current pain management regimen and make informed adjustments to better align with patient preferences. Some systems include patient feedback mechanisms that allow users to rate their pain levels and satisfaction, creating a closed-loop system that continuously optimizes the balance between automated and patient-controlled delivery based on real-world usage and patient-reported outcomes.Expand Specific Solutions
Key Players in PCA Pump and Pain Management Industry
The PCA pump versus on-demand systems market represents a mature healthcare technology sector experiencing steady growth driven by aging demographics and chronic pain management needs. The industry has reached technological maturity with established players like Baxter International and Nipro Corp. leading innovation in patient-controlled analgesia systems, while companies such as Avent (part of Avanos Medical) focus on specialized delivery mechanisms. Market dynamics show increasing preference for PCA systems due to improved patient satisfaction and clinical outcomes, though on-demand alternatives maintain relevance in specific care settings. Technology integration capabilities from firms like IBM and NEC Corp. are enhancing system connectivity and data analytics, while the competitive landscape remains consolidated among major medical device manufacturers who continue investing in user-centric design improvements and smart pump technologies.
Baxter International, Inc.
Technical Solution: Baxter has developed comprehensive PCA pump systems with advanced safety features including dose limits, lockout intervals, and continuous monitoring capabilities. Their PCA pumps feature user-friendly interfaces with clear displays and simple button operations for patient-controlled analgesia. The systems incorporate smart pump technology with drug libraries and dose error reduction systems to prevent medication errors. Baxter's PCA solutions also include wireless connectivity for remote monitoring and data collection, enabling healthcare providers to track patient usage patterns and pain management effectiveness in real-time.
Strengths: Market leader with proven safety record and comprehensive drug libraries. Weaknesses: Higher cost compared to basic on-demand systems and requires specialized training for optimal use.
Avent, Inc.
Technical Solution: Avent specializes in innovative drug delivery systems including patient-controlled analgesia devices that emphasize ease of use and patient comfort. Their PCA systems feature ergonomic design with intuitive controls that allow patients to self-administer pain medication within prescribed safety parameters. The company focuses on developing lightweight, portable PCA devices with extended battery life for improved patient mobility. Avent's systems incorporate advanced flow control mechanisms and anti-tampering features to ensure medication security while maintaining patient autonomy in pain management.
Strengths: Focus on patient comfort and mobility with lightweight designs. Weaknesses: Smaller market presence compared to major competitors and limited global distribution network.
Core Innovations in User-Centric Pain Control
System and method for optimizing control of PCA and PCEA system
PatentActiveUS7871394B2
Innovation
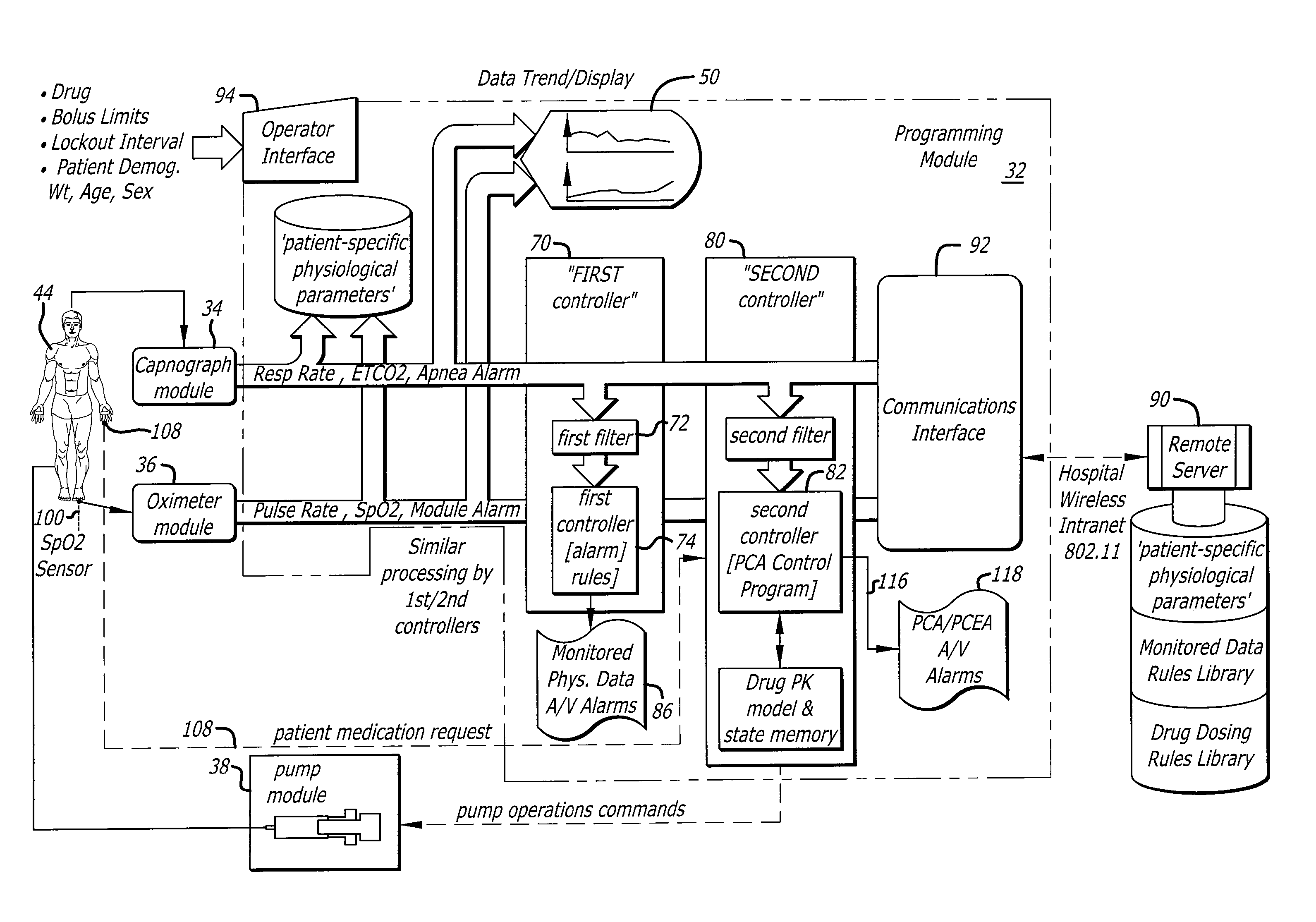
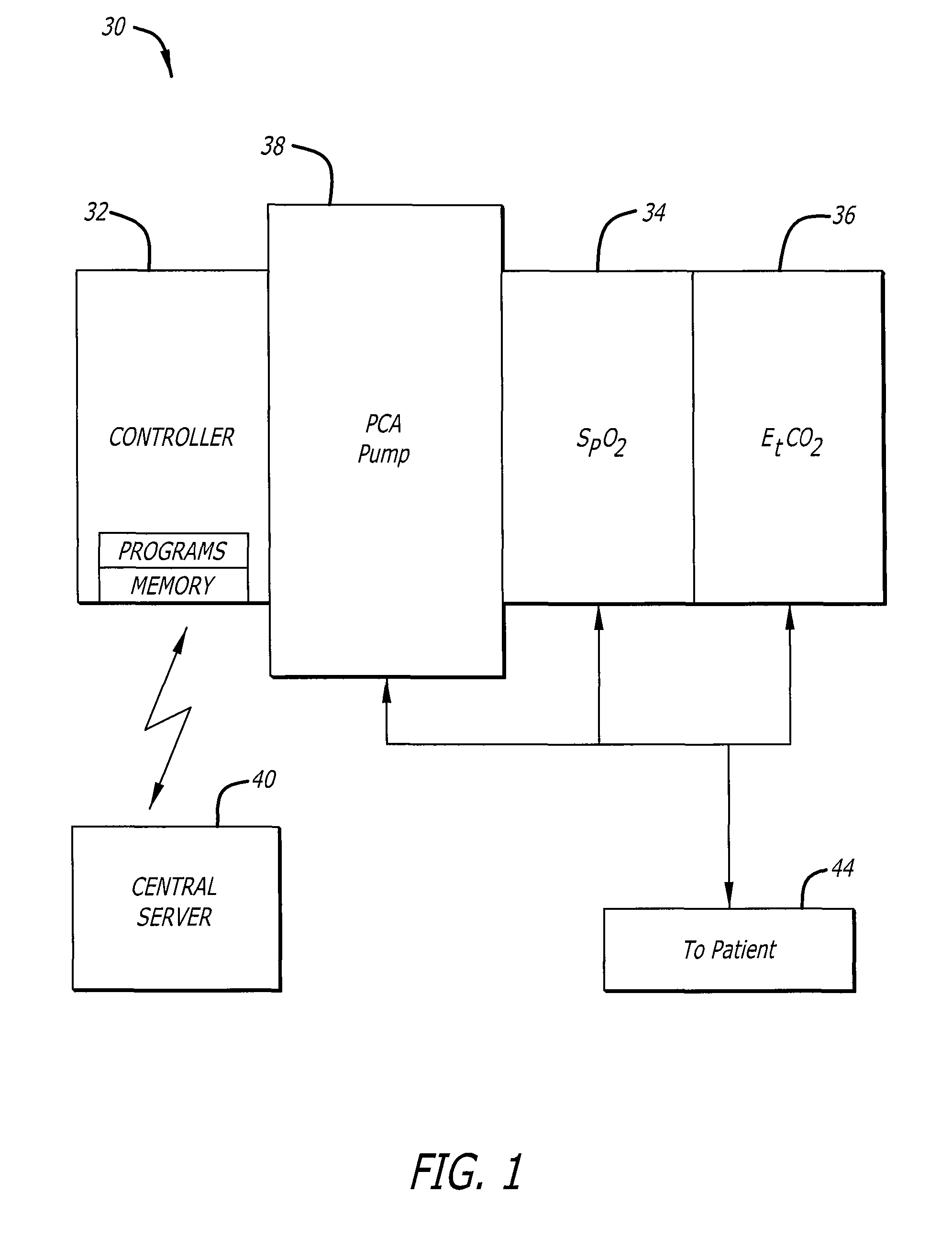
- A system and method that utilize a second controller to process physiological signals and request signals differently from the first controller, filtering data with techniques like moving averages and adaptive filters, and incorporating pharmacokinetic modeling to optimize PCA device operation, allowing for automatic inhibition of medication delivery during potential respiratory depression while minimizing false alarms.
Secure patient-controlled analgesia
PatentWO2021236679A1
Innovation
- A system that includes a drug delivery device, a drug control device, and a control unit capable of capturing biometric information, such as fingerprints, to authenticate the patient before administering medication, ensuring only authorized patients can self-administer doses based on their biometric data and real-time physiological signals.
Regulatory Framework for PCA Medical Devices
The regulatory framework governing Patient-Controlled Analgesia (PCA) medical devices represents a complex landscape that significantly influences user preferences between PCA pumps and on-demand systems. Regulatory bodies worldwide, including the FDA in the United States, the European Medicines Agency (EMA), and Health Canada, have established comprehensive guidelines that directly impact device design, functionality, and ultimately user acceptance.
Medical device classification systems categorize PCA devices as Class II or Class III products, depending on their complexity and risk profile. This classification determines the regulatory pathway, with 510(k) clearance required for most PCA pumps in the US market, while novel on-demand systems may require more stringent Pre-Market Approval (PMA) processes. These regulatory requirements create barriers to innovation that can influence the availability and features of different system types.
Safety standards mandated by regulatory frameworks significantly shape user preferences through their impact on device usability. IEC 60601-2-24 standards for infusion pumps establish requirements for alarm systems, drug libraries, and dose error reduction systems. These safety features, while essential for patient protection, can affect user workflow and preference between traditional PCA pumps and newer on-demand alternatives.
Post-market surveillance requirements create ongoing obligations for manufacturers that influence product development strategies. Adverse event reporting, mandatory device registrations, and periodic safety updates affect how companies prioritize features and improvements. Users often prefer systems with established regulatory track records, creating market advantages for traditional PCA pump designs over innovative on-demand systems.
International regulatory harmonization efforts, such as the Medical Device Single Audit Program (MDSAP), are gradually standardizing requirements across markets. However, regional differences in regulatory approaches continue to influence which PCA technologies are available in specific markets, thereby affecting local user preferences and adoption patterns.
The regulatory framework's emphasis on clinical evidence requirements also impacts user choice by determining which comparative effectiveness data is available. Rigorous clinical trial requirements for new on-demand systems versus established predicate devices for traditional PCA pumps create information asymmetries that influence healthcare provider decision-making and ultimately shape user preferences in clinical settings.
Medical device classification systems categorize PCA devices as Class II or Class III products, depending on their complexity and risk profile. This classification determines the regulatory pathway, with 510(k) clearance required for most PCA pumps in the US market, while novel on-demand systems may require more stringent Pre-Market Approval (PMA) processes. These regulatory requirements create barriers to innovation that can influence the availability and features of different system types.
Safety standards mandated by regulatory frameworks significantly shape user preferences through their impact on device usability. IEC 60601-2-24 standards for infusion pumps establish requirements for alarm systems, drug libraries, and dose error reduction systems. These safety features, while essential for patient protection, can affect user workflow and preference between traditional PCA pumps and newer on-demand alternatives.
Post-market surveillance requirements create ongoing obligations for manufacturers that influence product development strategies. Adverse event reporting, mandatory device registrations, and periodic safety updates affect how companies prioritize features and improvements. Users often prefer systems with established regulatory track records, creating market advantages for traditional PCA pump designs over innovative on-demand systems.
International regulatory harmonization efforts, such as the Medical Device Single Audit Program (MDSAP), are gradually standardizing requirements across markets. However, regional differences in regulatory approaches continue to influence which PCA technologies are available in specific markets, thereby affecting local user preferences and adoption patterns.
The regulatory framework's emphasis on clinical evidence requirements also impacts user choice by determining which comparative effectiveness data is available. Rigorous clinical trial requirements for new on-demand systems versus established predicate devices for traditional PCA pumps create information asymmetries that influence healthcare provider decision-making and ultimately shape user preferences in clinical settings.
Patient Safety and Addiction Prevention Protocols
Patient safety protocols in PCA pump and on-demand pain management systems represent critical frameworks designed to minimize adverse events while preventing substance abuse. These protocols encompass multi-layered safety mechanisms that address both immediate clinical risks and long-term addiction concerns through systematic monitoring and intervention strategies.
Contemporary safety protocols integrate real-time physiological monitoring with advanced alarm systems to detect respiratory depression, oversedation, and other opioid-related complications. PCA systems typically employ lockout intervals, dose limits, and continuous pulse oximetry monitoring to prevent overdose scenarios. On-demand systems incorporate similar safeguards but rely more heavily on healthcare provider assessment and intervention protocols.
Addiction prevention protocols focus on comprehensive patient screening, risk stratification, and behavioral monitoring throughout treatment duration. Pre-treatment assessments utilize validated screening tools such as the Opioid Risk Tool (ORT) and Screener and Opioid Assessment for Patients with Pain (SOAPP) to identify high-risk patients requiring enhanced monitoring protocols.
Implementation strategies vary significantly between system types, with PCA pumps offering automated compliance tracking and usage pattern analysis, while on-demand systems depend on manual documentation and clinical observation. Both approaches incorporate standardized weaning protocols and transition planning to minimize withdrawal symptoms and reduce long-term dependency risks.
Emerging protocols integrate artificial intelligence algorithms to analyze usage patterns and predict potential abuse behaviors. These systems can automatically adjust dosing parameters, trigger clinical alerts, and recommend intervention strategies based on individual patient risk profiles and historical usage data.
Regulatory compliance frameworks mandate specific documentation requirements, staff training protocols, and quality assurance measures. Healthcare institutions must maintain detailed records of medication administration, patient responses, and adverse events while ensuring staff competency in recognizing early signs of addiction or misuse.
The effectiveness of these protocols depends heavily on interdisciplinary collaboration between pain management specialists, nursing staff, pharmacists, and addiction counselors. Regular protocol updates incorporate latest clinical evidence and regulatory guidelines to maintain optimal patient safety standards while addressing evolving addiction prevention strategies.
Contemporary safety protocols integrate real-time physiological monitoring with advanced alarm systems to detect respiratory depression, oversedation, and other opioid-related complications. PCA systems typically employ lockout intervals, dose limits, and continuous pulse oximetry monitoring to prevent overdose scenarios. On-demand systems incorporate similar safeguards but rely more heavily on healthcare provider assessment and intervention protocols.
Addiction prevention protocols focus on comprehensive patient screening, risk stratification, and behavioral monitoring throughout treatment duration. Pre-treatment assessments utilize validated screening tools such as the Opioid Risk Tool (ORT) and Screener and Opioid Assessment for Patients with Pain (SOAPP) to identify high-risk patients requiring enhanced monitoring protocols.
Implementation strategies vary significantly between system types, with PCA pumps offering automated compliance tracking and usage pattern analysis, while on-demand systems depend on manual documentation and clinical observation. Both approaches incorporate standardized weaning protocols and transition planning to minimize withdrawal symptoms and reduce long-term dependency risks.
Emerging protocols integrate artificial intelligence algorithms to analyze usage patterns and predict potential abuse behaviors. These systems can automatically adjust dosing parameters, trigger clinical alerts, and recommend intervention strategies based on individual patient risk profiles and historical usage data.
Regulatory compliance frameworks mandate specific documentation requirements, staff training protocols, and quality assurance measures. Healthcare institutions must maintain detailed records of medication administration, patient responses, and adverse events while ensuring staff competency in recognizing early signs of addiction or misuse.
The effectiveness of these protocols depends heavily on interdisciplinary collaboration between pain management specialists, nursing staff, pharmacists, and addiction counselors. Regular protocol updates incorporate latest clinical evidence and regulatory guidelines to maintain optimal patient safety standards while addressing evolving addiction prevention strategies.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!