Peroxide Variants' Effect on Rubber Vulcanization Temperature
MAR 9, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Peroxide Vulcanization Background and Temperature Objectives
Rubber vulcanization represents one of the most critical chemical processes in polymer science, fundamentally transforming raw rubber from a thermoplastic material into a thermoset elastomer with enhanced mechanical properties, thermal stability, and chemical resistance. This crosslinking process has evolved significantly since Charles Goodyear's accidental discovery of sulfur vulcanization in 1839, with peroxide-based vulcanization emerging as a sophisticated alternative that offers distinct advantages over traditional sulfur systems.
The historical development of peroxide vulcanization began in the 1940s when organic peroxides were first recognized as effective crosslinking agents for rubber compounds. Unlike sulfur vulcanization, which creates polysulfidic crosslinks that can rearrange under stress and temperature, peroxide vulcanization forms stable carbon-carbon bonds between polymer chains. This fundamental difference results in superior heat resistance, better compression set properties, and enhanced chemical stability in the final vulcanized product.
Peroxide vulcanization operates through a free radical mechanism initiated by thermal decomposition of organic peroxides at elevated temperatures. The process involves homolytic cleavage of the peroxide bond, generating highly reactive alkoxy radicals that abstract hydrogen atoms from polymer chains, subsequently forming carbon-centered radicals that combine to create crosslinks. This mechanism is inherently temperature-dependent, with each peroxide variant exhibiting characteristic decomposition kinetics and activation temperatures.
The temperature sensitivity of peroxide vulcanization presents both opportunities and challenges in rubber processing. Different peroxide variants demonstrate varying thermal stability profiles, decomposition rates, and optimal curing temperatures, directly influencing processing windows, cure kinetics, and final product properties. Understanding these temperature relationships is crucial for optimizing manufacturing processes, ensuring consistent product quality, and developing new rubber formulations for demanding applications.
Current technological objectives focus on developing peroxide systems that enable lower-temperature vulcanization while maintaining crosslinking efficiency and final product performance. This pursuit is driven by energy conservation requirements, processing equipment limitations, and the need to incorporate temperature-sensitive additives or fillers. Additionally, there is growing interest in peroxide variants that provide broader processing windows, allowing for more flexible manufacturing conditions and improved process control.
The strategic importance of temperature optimization in peroxide vulcanization extends beyond immediate processing benefits to encompass broader sustainability goals, cost reduction initiatives, and performance enhancement requirements across diverse rubber applications including automotive sealing systems, industrial hoses, and high-performance elastomeric components.
The historical development of peroxide vulcanization began in the 1940s when organic peroxides were first recognized as effective crosslinking agents for rubber compounds. Unlike sulfur vulcanization, which creates polysulfidic crosslinks that can rearrange under stress and temperature, peroxide vulcanization forms stable carbon-carbon bonds between polymer chains. This fundamental difference results in superior heat resistance, better compression set properties, and enhanced chemical stability in the final vulcanized product.
Peroxide vulcanization operates through a free radical mechanism initiated by thermal decomposition of organic peroxides at elevated temperatures. The process involves homolytic cleavage of the peroxide bond, generating highly reactive alkoxy radicals that abstract hydrogen atoms from polymer chains, subsequently forming carbon-centered radicals that combine to create crosslinks. This mechanism is inherently temperature-dependent, with each peroxide variant exhibiting characteristic decomposition kinetics and activation temperatures.
The temperature sensitivity of peroxide vulcanization presents both opportunities and challenges in rubber processing. Different peroxide variants demonstrate varying thermal stability profiles, decomposition rates, and optimal curing temperatures, directly influencing processing windows, cure kinetics, and final product properties. Understanding these temperature relationships is crucial for optimizing manufacturing processes, ensuring consistent product quality, and developing new rubber formulations for demanding applications.
Current technological objectives focus on developing peroxide systems that enable lower-temperature vulcanization while maintaining crosslinking efficiency and final product performance. This pursuit is driven by energy conservation requirements, processing equipment limitations, and the need to incorporate temperature-sensitive additives or fillers. Additionally, there is growing interest in peroxide variants that provide broader processing windows, allowing for more flexible manufacturing conditions and improved process control.
The strategic importance of temperature optimization in peroxide vulcanization extends beyond immediate processing benefits to encompass broader sustainability goals, cost reduction initiatives, and performance enhancement requirements across diverse rubber applications including automotive sealing systems, industrial hoses, and high-performance elastomeric components.
Market Demand for Temperature-Optimized Rubber Products
The global rubber industry is experiencing unprecedented demand for temperature-optimized vulcanization processes, driven by stringent performance requirements across multiple sectors. Automotive manufacturers increasingly require rubber components that can withstand extreme temperature variations while maintaining consistent mechanical properties. This demand stems from the growing emphasis on vehicle durability, fuel efficiency, and safety standards that necessitate precise control over vulcanization parameters.
Industrial applications represent another significant market driver, particularly in sectors such as aerospace, oil and gas, and heavy machinery. These industries require rubber seals, gaskets, and components that perform reliably under specific temperature conditions. The ability to fine-tune vulcanization temperatures through peroxide variants enables manufacturers to produce rubber products with tailored thermal resistance and mechanical properties.
The medical device sector has emerged as a high-value market segment demanding temperature-controlled vulcanization processes. Medical-grade rubber products require precise curing temperatures to ensure biocompatibility and sterility while maintaining flexibility and durability. Regulatory compliance in this sector drives the need for consistent and controllable vulcanization processes.
Consumer electronics manufacturing increasingly relies on rubber components with specific thermal properties for device protection and performance. The miniaturization trend in electronics demands rubber materials that can be processed at lower temperatures to prevent damage to sensitive components during manufacturing.
Market research indicates substantial growth potential in emerging economies where industrial expansion and automotive production are accelerating. These regions present opportunities for temperature-optimized rubber processing technologies that can improve product quality while reducing energy consumption.
The sustainability movement has created additional market demand for energy-efficient vulcanization processes. Companies seek peroxide variants that enable lower-temperature curing to reduce carbon footprints and operational costs. This trend aligns with corporate environmental goals and regulatory pressures for greener manufacturing processes.
Specialty rubber applications in renewable energy infrastructure, particularly wind turbine seals and solar panel components, require materials processed under specific temperature conditions to ensure long-term performance in harsh environmental conditions. This emerging market segment represents significant growth potential for temperature-optimized vulcanization technologies.
Industrial applications represent another significant market driver, particularly in sectors such as aerospace, oil and gas, and heavy machinery. These industries require rubber seals, gaskets, and components that perform reliably under specific temperature conditions. The ability to fine-tune vulcanization temperatures through peroxide variants enables manufacturers to produce rubber products with tailored thermal resistance and mechanical properties.
The medical device sector has emerged as a high-value market segment demanding temperature-controlled vulcanization processes. Medical-grade rubber products require precise curing temperatures to ensure biocompatibility and sterility while maintaining flexibility and durability. Regulatory compliance in this sector drives the need for consistent and controllable vulcanization processes.
Consumer electronics manufacturing increasingly relies on rubber components with specific thermal properties for device protection and performance. The miniaturization trend in electronics demands rubber materials that can be processed at lower temperatures to prevent damage to sensitive components during manufacturing.
Market research indicates substantial growth potential in emerging economies where industrial expansion and automotive production are accelerating. These regions present opportunities for temperature-optimized rubber processing technologies that can improve product quality while reducing energy consumption.
The sustainability movement has created additional market demand for energy-efficient vulcanization processes. Companies seek peroxide variants that enable lower-temperature curing to reduce carbon footprints and operational costs. This trend aligns with corporate environmental goals and regulatory pressures for greener manufacturing processes.
Specialty rubber applications in renewable energy infrastructure, particularly wind turbine seals and solar panel components, require materials processed under specific temperature conditions to ensure long-term performance in harsh environmental conditions. This emerging market segment represents significant growth potential for temperature-optimized vulcanization technologies.
Current Peroxide Variants and Temperature Control Challenges
The rubber vulcanization industry currently employs several distinct peroxide variants, each presenting unique temperature control characteristics and operational challenges. Dicumyl peroxide (DCP) remains the most widely utilized organic peroxide, typically requiring activation temperatures between 160-180°C. Its decomposition kinetics follow predictable patterns, yet maintaining precise temperature control within the narrow optimal range proves challenging in large-scale production environments.
Benzoyl peroxide and lauroyl peroxide represent alternative variants with lower activation temperatures, operating effectively at 120-140°C and 60-80°C respectively. While these lower-temperature options offer energy efficiency advantages, they introduce complications in processing control due to premature activation risks during mixing and storage phases. The temperature sensitivity of these variants demands sophisticated thermal management systems that many facilities struggle to implement consistently.
Di-tert-butyl peroxide presents another category with high-temperature requirements, necessitating processing temperatures exceeding 200°C. This variant offers superior cross-linking density but creates significant challenges in maintaining uniform temperature distribution across large rubber batches. Temperature variations of even 10-15°C can result in inconsistent vulcanization, leading to mechanical property variations in the final product.
Contemporary temperature control challenges extend beyond simple heating systems to encompass real-time monitoring and feedback mechanisms. Traditional thermocouples often fail to capture rapid temperature fluctuations during exothermic vulcanization reactions, particularly when peroxide decomposition accelerates unexpectedly. This monitoring limitation results in batch-to-batch inconsistencies that compromise product quality standards.
Heat dissipation management represents another critical challenge, especially in thick rubber sections where thermal gradients develop naturally. Peroxide variants with rapid decomposition rates can create localized hot spots, leading to over-vulcanization in some areas while leaving others under-processed. Current cooling systems lack the precision required to address these micro-environmental temperature variations effectively.
The interaction between peroxide concentration and temperature control adds complexity to the vulcanization process. Higher peroxide loadings reduce required processing temperatures but increase the risk of runaway reactions, while lower concentrations demand more precise temperature maintenance over extended periods. This delicate balance requires advanced process control systems that remain economically challenging for many manufacturers to implement.
Benzoyl peroxide and lauroyl peroxide represent alternative variants with lower activation temperatures, operating effectively at 120-140°C and 60-80°C respectively. While these lower-temperature options offer energy efficiency advantages, they introduce complications in processing control due to premature activation risks during mixing and storage phases. The temperature sensitivity of these variants demands sophisticated thermal management systems that many facilities struggle to implement consistently.
Di-tert-butyl peroxide presents another category with high-temperature requirements, necessitating processing temperatures exceeding 200°C. This variant offers superior cross-linking density but creates significant challenges in maintaining uniform temperature distribution across large rubber batches. Temperature variations of even 10-15°C can result in inconsistent vulcanization, leading to mechanical property variations in the final product.
Contemporary temperature control challenges extend beyond simple heating systems to encompass real-time monitoring and feedback mechanisms. Traditional thermocouples often fail to capture rapid temperature fluctuations during exothermic vulcanization reactions, particularly when peroxide decomposition accelerates unexpectedly. This monitoring limitation results in batch-to-batch inconsistencies that compromise product quality standards.
Heat dissipation management represents another critical challenge, especially in thick rubber sections where thermal gradients develop naturally. Peroxide variants with rapid decomposition rates can create localized hot spots, leading to over-vulcanization in some areas while leaving others under-processed. Current cooling systems lack the precision required to address these micro-environmental temperature variations effectively.
The interaction between peroxide concentration and temperature control adds complexity to the vulcanization process. Higher peroxide loadings reduce required processing temperatures but increase the risk of runaway reactions, while lower concentrations demand more precise temperature maintenance over extended periods. This delicate balance requires advanced process control systems that remain economically challenging for many manufacturers to implement.
Existing Peroxide Solutions for Temperature Management
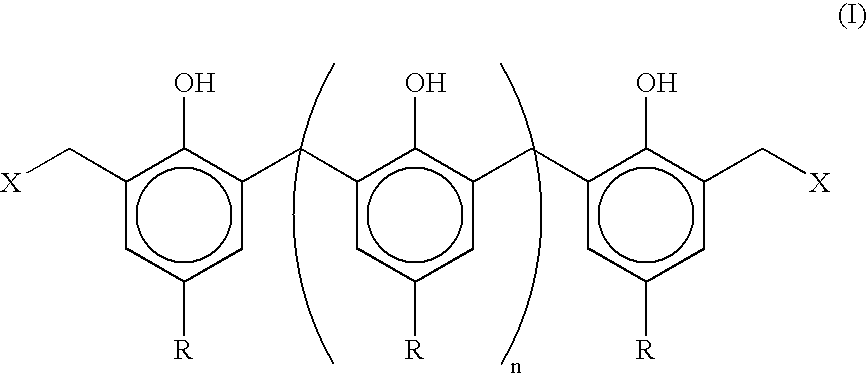
01 Use of organic peroxides with different decomposition temperatures for vulcanization
Different organic peroxides exhibit varying decomposition temperatures, which directly affect the vulcanization temperature of rubber compounds. By selecting peroxides with specific decomposition characteristics, the vulcanization process can be optimized for different temperature ranges. This allows manufacturers to tailor the curing process to specific production requirements and equipment capabilities.- Use of organic peroxides with different decomposition temperatures for vulcanization: Different organic peroxides exhibit varying decomposition temperatures, which directly affect the vulcanization temperature of rubber compounds. By selecting peroxides with specific decomposition characteristics, the vulcanization process can be optimized for different temperature ranges. This allows manufacturers to tailor the curing process to specific production requirements and equipment capabilities.
- Combination of multiple peroxide types to control vulcanization temperature range: Using combinations of different peroxide variants enables precise control over the vulcanization temperature profile. By blending peroxides with high and low decomposition temperatures, a broader or more specific curing window can be achieved. This approach provides flexibility in processing conditions and can improve the uniformity of vulcanization across different product geometries.
- Addition of co-agents and activators to modify peroxide vulcanization temperature: Co-agents and activators can be incorporated into peroxide vulcanization systems to adjust the effective curing temperature. These additives interact with peroxide radicals to enhance crosslinking efficiency and can shift the optimal vulcanization temperature. The use of such modifiers allows for lower temperature processing or faster cure rates at given temperatures.
- Development of low-temperature peroxide vulcanization systems: Specialized peroxide formulations have been developed to enable vulcanization at reduced temperatures. These systems are particularly useful for heat-sensitive materials or energy-efficient manufacturing processes. Low-temperature peroxide variants maintain adequate curing efficiency while operating below conventional vulcanization temperatures.
- High-temperature stable peroxide variants for demanding applications: Certain peroxide variants are designed to remain stable at elevated temperatures and provide vulcanization at higher temperature ranges. These formulations are suitable for applications requiring enhanced thermal resistance or rapid curing cycles. High-temperature peroxide systems offer improved scorch safety during processing while maintaining effective crosslinking at elevated cure temperatures.
02 Combination of multiple peroxide types to control vulcanization temperature range
Using combinations of different peroxide variants enables precise control over the vulcanization temperature profile. By blending peroxides with high and low decomposition temperatures, a broader or more specific curing window can be achieved. This approach provides flexibility in processing conditions and can improve the efficiency of the vulcanization process while maintaining product quality.Expand Specific Solutions03 Low-temperature vulcanization systems using specialized peroxide formulations
Specialized peroxide formulations have been developed to enable vulcanization at reduced temperatures, which can save energy and prevent thermal degradation of heat-sensitive materials. These systems typically incorporate peroxides with lower activation energies or are combined with activators and accelerators that facilitate crosslinking at milder conditions. Low-temperature vulcanization is particularly beneficial for certain polymer types and composite materials.Expand Specific Solutions04 High-temperature resistant peroxide systems for demanding applications
For applications requiring elevated processing temperatures or enhanced thermal stability, high-temperature resistant peroxide systems are employed. These formulations utilize peroxides with higher decomposition temperatures and improved thermal stability, allowing vulcanization to occur at elevated temperatures without premature decomposition. Such systems are essential for producing rubber products that must withstand extreme operating conditions.Expand Specific Solutions05 Temperature-adjustable vulcanization through peroxide concentration and additives
The vulcanization temperature can be effectively adjusted by varying the concentration of peroxide and incorporating specific additives or co-agents. Higher peroxide concentrations generally increase the rate of crosslinking, while certain additives can modify the decomposition kinetics and effective curing temperature. This approach provides a practical method for fine-tuning the vulcanization process to match specific production requirements and achieve desired material properties.Expand Specific Solutions
Key Players in Peroxide and Rubber Vulcanization Industry
The peroxide variants' effect on rubber vulcanization temperature represents a mature technology sector within the broader rubber and chemical industries, currently experiencing steady growth driven by automotive and industrial applications. The global rubber chemicals market, valued at approximately $5 billion, demonstrates moderate expansion with established players dominating through extensive R&D capabilities. Technology maturity is evidenced by major tire manufacturers like Goodyear, Bridgestone, and Sumitomo Rubber Industries implementing advanced vulcanization processes, while chemical giants including Arkema, Bayer AG, and LANXESS provide specialized peroxide formulations. Japanese companies such as JSR Corp and Mitsui Chemicals lead innovation in synthetic rubber technologies, complemented by emerging Chinese players like Anhui Zhongding and PetroChina expanding manufacturing capabilities. The competitive landscape shows consolidation among established firms while newer entrants focus on cost-effective solutions and regional market penetration.
Goodyear Tire & Rubber Co.
Technical Solution: Goodyear has developed advanced peroxide vulcanization systems that enable lower temperature processing for specialty rubber compounds. Their technology focuses on dicumyl peroxide and bis(tert-butylperoxyisopropyl)benzene variants that activate at temperatures 15-20°C lower than conventional sulfur systems. The company's proprietary formulations incorporate peroxide activators and co-agents that enhance crosslinking efficiency while reducing vulcanization temperature from typical 160-180°C range to 140-160°C. This approach is particularly effective for EPDM and silicone rubber applications where heat-sensitive additives are present.
Strengths: Extensive tire industry experience, proven commercial applications, strong R&D capabilities. Weaknesses: Limited to specific rubber types, higher material costs compared to sulfur systems.
Mitsui Chemicals, Inc.
Technical Solution: Mitsui Chemicals has pioneered low-temperature peroxide vulcanization through their LUCANT series of peroxide initiators. These specialized variants include temperature-modified organic peroxides that decompose at 120-140°C, significantly lower than standard peroxide systems. Their technology incorporates synergistic combinations of different peroxide types with metallic activators that enhance decomposition efficiency. The company's approach focuses on molecular design of peroxide structures to achieve optimal balance between storage stability and low-temperature reactivity, enabling vulcanization temperatures reduced by up to 40°C compared to conventional systems.
Strengths: Chemical expertise in peroxide synthesis, innovative molecular design capabilities, broad industrial applications. Weaknesses: Requires specialized handling procedures, limited compatibility with certain rubber formulations.
Core Innovations in Peroxide Chemistry for Vulcanization
Thermoplastic elastomer composition and process for producing the same
PatentInactiveUS20050038186A1
Innovation
- A thermoplastic elastomer composition is developed by dynamically heat-treating a polymer composition including a rubber and an olefinic resin in the presence of a crosslinking agent, with specific parameters such as a gel fraction of 80% or higher and rubber particles with a number-averaged particle size of 3 μm or less, along with the use of additives like ethylene•α-olefin copolymer rubber and hydrogenated diene polymer, to enhance mechanical properties and adhesion.
Unconjugated cyclopolyene copolymer, rubber composition, and use
PatentInactiveEP1217015B1
Innovation
- A random copolymer based on non-conjugated cyclic polyene, specifically formulated with structural units from α-olefins, non-conjugated cyclic polyenes, and non-conjugated linear polyenes, is developed, offering improved mechanical strength, workability, and a balanced composition that enhances braking performance and reduces rolling resistance.
Environmental Regulations for Peroxide Usage in Rubber
The regulatory landscape governing peroxide usage in rubber manufacturing has evolved significantly over the past two decades, driven by increasing environmental awareness and worker safety concerns. Current environmental regulations primarily focus on volatile organic compound emissions, workplace exposure limits, and waste disposal protocols for peroxide-based vulcanization systems.
In the United States, the Environmental Protection Agency regulates peroxide compounds under the Toxic Substances Control Act, with specific attention to dicumyl peroxide and benzoyl peroxide emissions during rubber processing. The Occupational Safety and Health Administration has established permissible exposure limits for organic peroxides, typically ranging from 0.1 to 5 ppm depending on the specific compound's toxicity profile.
European Union regulations under REACH require comprehensive registration and evaluation of peroxide variants used in rubber vulcanization. The Classification, Labelling and Packaging Regulation categorizes most organic peroxides as oxidizing substances, mandating specific handling and storage requirements. Additionally, the Industrial Emissions Directive sets strict limits on volatile organic compound emissions from rubber processing facilities.
Asian markets, particularly China and Japan, have implemented increasingly stringent environmental standards. China's National Standard GB 27632 specifically addresses rubber industry emissions, while Japan's Air Pollution Control Law regulates peroxide-related volatile organic compounds. These regulations often require advanced emission control systems and regular monitoring protocols.
Emerging regulatory trends indicate a shift toward lifecycle assessment approaches, considering the environmental impact from peroxide production through disposal. Future regulations are expected to emphasize sustainable alternatives and closed-loop processing systems. Companies must increasingly demonstrate compliance through detailed emission monitoring, worker exposure assessments, and environmental impact documentation.
The regulatory complexity varies significantly by peroxide type, with newer variants often facing more stringent approval processes. This regulatory environment directly influences the selection of peroxide systems for rubber vulcanization, as manufacturers must balance performance requirements with compliance costs and environmental responsibilities.
In the United States, the Environmental Protection Agency regulates peroxide compounds under the Toxic Substances Control Act, with specific attention to dicumyl peroxide and benzoyl peroxide emissions during rubber processing. The Occupational Safety and Health Administration has established permissible exposure limits for organic peroxides, typically ranging from 0.1 to 5 ppm depending on the specific compound's toxicity profile.
European Union regulations under REACH require comprehensive registration and evaluation of peroxide variants used in rubber vulcanization. The Classification, Labelling and Packaging Regulation categorizes most organic peroxides as oxidizing substances, mandating specific handling and storage requirements. Additionally, the Industrial Emissions Directive sets strict limits on volatile organic compound emissions from rubber processing facilities.
Asian markets, particularly China and Japan, have implemented increasingly stringent environmental standards. China's National Standard GB 27632 specifically addresses rubber industry emissions, while Japan's Air Pollution Control Law regulates peroxide-related volatile organic compounds. These regulations often require advanced emission control systems and regular monitoring protocols.
Emerging regulatory trends indicate a shift toward lifecycle assessment approaches, considering the environmental impact from peroxide production through disposal. Future regulations are expected to emphasize sustainable alternatives and closed-loop processing systems. Companies must increasingly demonstrate compliance through detailed emission monitoring, worker exposure assessments, and environmental impact documentation.
The regulatory complexity varies significantly by peroxide type, with newer variants often facing more stringent approval processes. This regulatory environment directly influences the selection of peroxide systems for rubber vulcanization, as manufacturers must balance performance requirements with compliance costs and environmental responsibilities.
Energy Efficiency Considerations in Vulcanization Processes
Energy efficiency in rubber vulcanization processes has become increasingly critical as manufacturers face rising energy costs and environmental regulations. The selection of peroxide variants significantly impacts the thermal requirements and overall energy consumption during vulcanization, making it essential to understand the relationship between catalyst choice and process efficiency.
Traditional sulfur-based vulcanization systems typically require temperatures ranging from 140-180°C, while peroxide-initiated systems often demand higher temperatures of 160-200°C. However, the energy efficiency equation extends beyond simple temperature requirements. Peroxide variants that enable lower vulcanization temperatures can substantially reduce energy consumption, particularly in continuous processes where maintaining elevated temperatures represents a significant operational cost.
The thermal efficiency of peroxide vulcanization is influenced by the decomposition kinetics of different peroxide types. Fast-acting peroxides like dicumyl peroxide require precise temperature control but can achieve complete crosslinking in shorter cure cycles, reducing overall energy input per unit of product. Conversely, slower-acting peroxides may operate at lower peak temperatures but require extended heating periods, potentially offsetting initial energy savings.
Heat transfer optimization becomes crucial when implementing peroxide vulcanization systems. The uniform distribution of thermal energy throughout rubber compounds affects both cure quality and energy utilization. Peroxide variants with lower activation temperatures can improve heat transfer efficiency by reducing the temperature differential between heating elements and the rubber matrix, minimizing thermal losses and improving overall process efficiency.
Process integration strategies can further enhance energy efficiency in peroxide vulcanization. Multi-stage heating profiles, where initial heating activates peroxide decomposition followed by controlled temperature maintenance, can optimize energy consumption while ensuring complete crosslinking. Additionally, heat recovery systems that capture and reuse thermal energy from cooling phases can significantly improve overall process efficiency.
The economic implications of energy-efficient vulcanization extend beyond direct energy costs. Reduced thermal stress on equipment, lower maintenance requirements, and improved product consistency contribute to overall operational efficiency. Peroxide selection strategies that prioritize energy efficiency while maintaining product quality represent a key competitive advantage in modern rubber manufacturing operations.
Traditional sulfur-based vulcanization systems typically require temperatures ranging from 140-180°C, while peroxide-initiated systems often demand higher temperatures of 160-200°C. However, the energy efficiency equation extends beyond simple temperature requirements. Peroxide variants that enable lower vulcanization temperatures can substantially reduce energy consumption, particularly in continuous processes where maintaining elevated temperatures represents a significant operational cost.
The thermal efficiency of peroxide vulcanization is influenced by the decomposition kinetics of different peroxide types. Fast-acting peroxides like dicumyl peroxide require precise temperature control but can achieve complete crosslinking in shorter cure cycles, reducing overall energy input per unit of product. Conversely, slower-acting peroxides may operate at lower peak temperatures but require extended heating periods, potentially offsetting initial energy savings.
Heat transfer optimization becomes crucial when implementing peroxide vulcanization systems. The uniform distribution of thermal energy throughout rubber compounds affects both cure quality and energy utilization. Peroxide variants with lower activation temperatures can improve heat transfer efficiency by reducing the temperature differential between heating elements and the rubber matrix, minimizing thermal losses and improving overall process efficiency.
Process integration strategies can further enhance energy efficiency in peroxide vulcanization. Multi-stage heating profiles, where initial heating activates peroxide decomposition followed by controlled temperature maintenance, can optimize energy consumption while ensuring complete crosslinking. Additionally, heat recovery systems that capture and reuse thermal energy from cooling phases can significantly improve overall process efficiency.
The economic implications of energy-efficient vulcanization extend beyond direct energy costs. Reduced thermal stress on equipment, lower maintenance requirements, and improved product consistency contribute to overall operational efficiency. Peroxide selection strategies that prioritize energy efficiency while maintaining product quality represent a key competitive advantage in modern rubber manufacturing operations.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!