Composite solid electrolytes with enhanced thermal stability
OCT 10, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Composite Solid Electrolytes Background and Objectives
Solid-state batteries have emerged as a promising next-generation energy storage technology, offering potential advantages in safety, energy density, and lifespan compared to conventional lithium-ion batteries with liquid electrolytes. The development of composite solid electrolytes (CSEs) represents a critical advancement in this field, combining different materials to overcome the limitations of single-component solid electrolytes.
The evolution of solid electrolyte technology can be traced back to the 1970s with the discovery of fast ion conductors. However, significant progress has been made in the last decade, driven by increasing demands for safer and higher-energy-density batteries for electric vehicles and portable electronics. Traditional solid electrolytes face challenges including low ionic conductivity at room temperature, poor mechanical properties, and unstable interfaces with electrodes.
Thermal stability has emerged as a particularly crucial factor in solid electrolyte development. Conventional lithium-ion batteries with liquid electrolytes are prone to thermal runaway at elevated temperatures, posing significant safety risks. While solid electrolytes inherently offer improved safety, many promising candidates still suffer from thermal degradation, phase transitions, or interfacial instability at operating or processing temperatures.
Composite solid electrolytes aim to address these challenges by combining complementary materials—typically a primary ion conductor with secondary phases that enhance mechanical properties, interfacial stability, or thermal characteristics. Recent research has focused on polymer-ceramic, ceramic-ceramic, and polymer-ceramic-salt composite systems, each offering unique advantages for specific applications.
The primary technical objectives for enhanced thermally stable CSEs include: achieving ionic conductivity exceeding 10^-4 S/cm across a wide temperature range (-20°C to 100°C); maintaining structural and electrochemical stability at elevated temperatures (>80°C); improving interfacial compatibility with both cathode and anode materials during thermal cycling; and developing scalable, cost-effective manufacturing processes compatible with existing battery production infrastructure.
Current research trends indicate growing interest in hierarchical composite structures, novel interface engineering approaches, and the integration of flame-retardant or thermally conductive additives. Computational modeling and high-throughput screening methods are increasingly being employed to accelerate material discovery and optimization for thermally robust CSEs.
The development of composite solid electrolytes with enhanced thermal stability represents a critical pathway toward commercialization of solid-state batteries, potentially enabling safer, longer-lasting, and higher-energy-density energy storage solutions for transportation, grid storage, and consumer electronics applications.
The evolution of solid electrolyte technology can be traced back to the 1970s with the discovery of fast ion conductors. However, significant progress has been made in the last decade, driven by increasing demands for safer and higher-energy-density batteries for electric vehicles and portable electronics. Traditional solid electrolytes face challenges including low ionic conductivity at room temperature, poor mechanical properties, and unstable interfaces with electrodes.
Thermal stability has emerged as a particularly crucial factor in solid electrolyte development. Conventional lithium-ion batteries with liquid electrolytes are prone to thermal runaway at elevated temperatures, posing significant safety risks. While solid electrolytes inherently offer improved safety, many promising candidates still suffer from thermal degradation, phase transitions, or interfacial instability at operating or processing temperatures.
Composite solid electrolytes aim to address these challenges by combining complementary materials—typically a primary ion conductor with secondary phases that enhance mechanical properties, interfacial stability, or thermal characteristics. Recent research has focused on polymer-ceramic, ceramic-ceramic, and polymer-ceramic-salt composite systems, each offering unique advantages for specific applications.
The primary technical objectives for enhanced thermally stable CSEs include: achieving ionic conductivity exceeding 10^-4 S/cm across a wide temperature range (-20°C to 100°C); maintaining structural and electrochemical stability at elevated temperatures (>80°C); improving interfacial compatibility with both cathode and anode materials during thermal cycling; and developing scalable, cost-effective manufacturing processes compatible with existing battery production infrastructure.
Current research trends indicate growing interest in hierarchical composite structures, novel interface engineering approaches, and the integration of flame-retardant or thermally conductive additives. Computational modeling and high-throughput screening methods are increasingly being employed to accelerate material discovery and optimization for thermally robust CSEs.
The development of composite solid electrolytes with enhanced thermal stability represents a critical pathway toward commercialization of solid-state batteries, potentially enabling safer, longer-lasting, and higher-energy-density energy storage solutions for transportation, grid storage, and consumer electronics applications.
Market Analysis for Thermally Stable Solid Electrolytes
The global market for thermally stable solid electrolytes is experiencing significant growth, driven primarily by the expanding electric vehicle (EV) sector and increasing demand for safer, higher-performance energy storage solutions. Current market valuations indicate that the solid-state battery market, where these electrolytes are a critical component, is projected to reach $8.7 billion by 2027, with a compound annual growth rate of 34.2% from 2022 to 2027.
The demand for composite solid electrolytes with enhanced thermal stability stems from several market factors. Safety concerns with conventional lithium-ion batteries using liquid electrolytes have created a strong market pull for alternatives that can withstand higher temperatures without degradation or safety risks. This is particularly evident in the automotive sector, where thermal runaway prevention has become a priority design consideration for next-generation battery systems.
Regional market analysis reveals that Asia-Pacific currently dominates the solid electrolyte market, with Japan, South Korea, and China leading in both research and commercialization efforts. North America and Europe are rapidly expanding their market presence, with significant investments in research and manufacturing infrastructure. The European market is particularly driven by stringent safety regulations and sustainability goals that favor solid-state battery technologies.
Consumer electronics represents another substantial market segment, with manufacturers seeking higher energy density and safer battery solutions for smartphones, laptops, and wearable devices. This segment values the reduced form factor and enhanced safety that thermally stable solid electrolytes can provide.
Market segmentation by material type shows that composite solid electrolytes combining ceramic and polymer materials are gaining the highest traction due to their balanced properties addressing both ionic conductivity and mechanical flexibility challenges. The market share for oxide-based composites is growing at 38% annually, outpacing other material categories.
Industry forecasts suggest that the initial commercial applications will focus on premium market segments where performance and safety command price premiums, gradually expanding to mass-market applications as manufacturing scales and costs decrease. Current price points for composite solid electrolytes remain 3-5 times higher than conventional liquid electrolytes, presenting a significant barrier to widespread adoption.
Customer surveys indicate that battery manufacturers are willing to pay a premium of up to 40% for solid electrolytes that can demonstrate thermal stability above 150°C while maintaining competitive ionic conductivity. This price sensitivity varies significantly across application sectors, with aerospace and high-end automotive showing the greatest willingness to absorb higher costs for enhanced safety and performance.
The demand for composite solid electrolytes with enhanced thermal stability stems from several market factors. Safety concerns with conventional lithium-ion batteries using liquid electrolytes have created a strong market pull for alternatives that can withstand higher temperatures without degradation or safety risks. This is particularly evident in the automotive sector, where thermal runaway prevention has become a priority design consideration for next-generation battery systems.
Regional market analysis reveals that Asia-Pacific currently dominates the solid electrolyte market, with Japan, South Korea, and China leading in both research and commercialization efforts. North America and Europe are rapidly expanding their market presence, with significant investments in research and manufacturing infrastructure. The European market is particularly driven by stringent safety regulations and sustainability goals that favor solid-state battery technologies.
Consumer electronics represents another substantial market segment, with manufacturers seeking higher energy density and safer battery solutions for smartphones, laptops, and wearable devices. This segment values the reduced form factor and enhanced safety that thermally stable solid electrolytes can provide.
Market segmentation by material type shows that composite solid electrolytes combining ceramic and polymer materials are gaining the highest traction due to their balanced properties addressing both ionic conductivity and mechanical flexibility challenges. The market share for oxide-based composites is growing at 38% annually, outpacing other material categories.
Industry forecasts suggest that the initial commercial applications will focus on premium market segments where performance and safety command price premiums, gradually expanding to mass-market applications as manufacturing scales and costs decrease. Current price points for composite solid electrolytes remain 3-5 times higher than conventional liquid electrolytes, presenting a significant barrier to widespread adoption.
Customer surveys indicate that battery manufacturers are willing to pay a premium of up to 40% for solid electrolytes that can demonstrate thermal stability above 150°C while maintaining competitive ionic conductivity. This price sensitivity varies significantly across application sectors, with aerospace and high-end automotive showing the greatest willingness to absorb higher costs for enhanced safety and performance.
Technical Challenges in Thermal Stability Enhancement
Despite significant advancements in composite solid electrolytes (CSEs), enhancing thermal stability remains one of the most critical technical challenges in this field. Current CSEs often suffer from performance degradation at elevated temperatures, limiting their practical applications in next-generation energy storage systems. The primary challenge lies in maintaining the structural integrity and electrochemical performance of these materials across wide temperature ranges, particularly above 80°C where many polymer-based electrolytes begin to soften or decompose.
Interface stability presents another significant hurdle, as thermal cycling induces mechanical stress at the boundaries between different components within the composite structure. These stresses can lead to delamination, crack formation, and increased interfacial resistance, ultimately compromising the overall performance and safety of the electrolyte system. The challenge is further complicated by the different thermal expansion coefficients of the various components in CSEs, creating additional mechanical strain during temperature fluctuations.
Chemical compatibility issues emerge at elevated temperatures, with accelerated side reactions occurring between the electrolyte components themselves or with electrode materials. These reactions can produce undesirable products that block ion transport pathways, increase impedance, and potentially form dendrites that may cause short circuits. Particularly problematic are the reactions involving polymer matrices and ceramic fillers, which can degrade the polymer chains or alter the surface chemistry of the ceramic particles.
Ion conductivity mechanisms in CSEs are temperature-dependent, with many systems showing non-linear behavior across different temperature ranges. Engineering materials that maintain high ionic conductivity while preserving mechanical strength at elevated temperatures requires sophisticated design approaches that have not yet been fully realized. The trade-off between mechanical properties and ion transport efficiency becomes more pronounced as temperatures increase.
Manufacturing scalability adds another layer of complexity to thermal stability enhancement. Laboratory-scale techniques that produce thermally stable CSEs often face significant challenges when scaled to industrial production. Ensuring uniform dispersion of fillers, controlling interface quality, and maintaining consistent performance across large-area electrolytes remain difficult to achieve at scale, particularly when optimizing for thermal stability.
Recent research has identified promising approaches including chemically modified interfaces, gradient-structured composites, and thermally resistant polymer blends, yet each solution introduces new challenges related to processing complexity, cost-effectiveness, and long-term reliability. The development of comprehensive testing protocols that can accurately predict long-term thermal stability under real-world operating conditions also remains an ongoing challenge in the field.
Interface stability presents another significant hurdle, as thermal cycling induces mechanical stress at the boundaries between different components within the composite structure. These stresses can lead to delamination, crack formation, and increased interfacial resistance, ultimately compromising the overall performance and safety of the electrolyte system. The challenge is further complicated by the different thermal expansion coefficients of the various components in CSEs, creating additional mechanical strain during temperature fluctuations.
Chemical compatibility issues emerge at elevated temperatures, with accelerated side reactions occurring between the electrolyte components themselves or with electrode materials. These reactions can produce undesirable products that block ion transport pathways, increase impedance, and potentially form dendrites that may cause short circuits. Particularly problematic are the reactions involving polymer matrices and ceramic fillers, which can degrade the polymer chains or alter the surface chemistry of the ceramic particles.
Ion conductivity mechanisms in CSEs are temperature-dependent, with many systems showing non-linear behavior across different temperature ranges. Engineering materials that maintain high ionic conductivity while preserving mechanical strength at elevated temperatures requires sophisticated design approaches that have not yet been fully realized. The trade-off between mechanical properties and ion transport efficiency becomes more pronounced as temperatures increase.
Manufacturing scalability adds another layer of complexity to thermal stability enhancement. Laboratory-scale techniques that produce thermally stable CSEs often face significant challenges when scaled to industrial production. Ensuring uniform dispersion of fillers, controlling interface quality, and maintaining consistent performance across large-area electrolytes remain difficult to achieve at scale, particularly when optimizing for thermal stability.
Recent research has identified promising approaches including chemically modified interfaces, gradient-structured composites, and thermally resistant polymer blends, yet each solution introduces new challenges related to processing complexity, cost-effectiveness, and long-term reliability. The development of comprehensive testing protocols that can accurately predict long-term thermal stability under real-world operating conditions also remains an ongoing challenge in the field.
Current Approaches to Enhance Thermal Stability
01 Polymer-based composite solid electrolytes
Polymer-based composite solid electrolytes incorporate various polymers as matrices with additives to enhance thermal stability. These composites typically combine polymer hosts with ceramic fillers or other stabilizing agents to create electrolytes that maintain structural integrity and ionic conductivity at elevated temperatures. The polymer matrix provides flexibility while additives improve the thermal resistance, making these electrolytes suitable for high-temperature battery applications.- Polymer-based composite solid electrolytes: Polymer-based composite solid electrolytes incorporate polymeric materials as matrices or binders with various additives to enhance thermal stability. These composites typically combine the flexibility and processability of polymers with inorganic components to improve thermal resistance. The polymer matrix provides mechanical support while additives such as ceramic fillers or flame retardants enhance the thermal stability, making these electrolytes suitable for high-temperature applications in batteries and other electrochemical devices.
- Ceramic-based composite solid electrolytes: Ceramic-based composite solid electrolytes utilize ceramic materials as the primary component to achieve superior thermal stability. These electrolytes typically consist of oxide, phosphate, or sulfide-based ceramics that can withstand high temperatures without degradation. The incorporation of different ceramic phases or dopants can further enhance the thermal properties while maintaining ionic conductivity. These materials are particularly valuable in applications requiring operation under extreme temperature conditions.
- Interface engineering for thermal stability: Interface engineering focuses on modifying the boundaries between different components in composite solid electrolytes to enhance thermal stability. This approach involves surface treatments, coatings, or the introduction of interlayers that can reduce interfacial resistance and prevent thermal degradation at high temperatures. By controlling the interface properties, thermal expansion mismatches can be mitigated, and the overall thermal stability of the composite electrolyte system can be significantly improved.
- Additives and dopants for enhanced thermal stability: Various additives and dopants can be incorporated into solid electrolytes to enhance their thermal stability. These include flame retardants, thermal stabilizers, and ionic conductivity enhancers that can withstand high temperatures. The strategic addition of these components can suppress phase transitions, prevent crystallization, and inhibit thermal decomposition at elevated temperatures. This approach allows for the customization of composite solid electrolytes to meet specific thermal stability requirements for different applications.
- Novel composite structures for extreme temperature applications: Advanced composite structures are being developed specifically for extreme temperature applications. These include multi-layered architectures, gradient compositions, and three-dimensional networks that distribute thermal stress and prevent failure under severe conditions. Such novel structures often combine different material classes (ceramics, polymers, and inorganics) in unique configurations to achieve unprecedented thermal stability while maintaining other essential properties like ionic conductivity and mechanical strength.
02 Ceramic-reinforced solid electrolytes
Ceramic-reinforced solid electrolytes utilize thermally stable ceramic materials to enhance the overall thermal stability of the electrolyte system. These composites incorporate ceramic particles or layers that can withstand high temperatures without degradation. The ceramic components act as thermal barriers and structural reinforcements, preventing thermal runaway and maintaining electrochemical performance under extreme temperature conditions.Expand Specific Solutions03 Interface engineering for thermal stability
Interface engineering focuses on modifying the boundaries between different components in composite solid electrolytes to enhance thermal stability. This approach involves surface treatments, interlayer additions, or gradient structures that minimize thermal stress at interfaces. By controlling the interfacial chemistry and structure, these techniques reduce thermal expansion mismatches and prevent degradation pathways that typically initiate at component boundaries under thermal stress.Expand Specific Solutions04 Inorganic-organic hybrid electrolytes
Inorganic-organic hybrid electrolytes combine the advantages of both material classes to achieve superior thermal stability. These systems integrate inorganic components known for high thermal resistance with organic materials that provide flexibility and processability. The synergistic combination results in composite electrolytes that maintain structural integrity and ionic conductivity across wide temperature ranges, making them suitable for applications requiring thermal resilience.Expand Specific Solutions05 Additives and dopants for enhanced thermal stability
Various additives and dopants can be incorporated into solid electrolytes to significantly improve their thermal stability. These include flame retardants, thermal stabilizers, and specialized dopants that increase the decomposition temperature of the electrolyte system. Such additives can modify the crystalline structure, suppress phase transitions, or create protective layers that activate under thermal stress, thereby extending the operational temperature range of composite solid electrolytes.Expand Specific Solutions
Key Industry Players and Research Institutions
The composite solid electrolyte market for enhanced thermal stability is currently in a growth phase, with increasing demand driven by electric vehicle and energy storage applications. The market size is expanding rapidly, projected to reach significant value by 2030 as battery safety concerns become paramount. Technologically, the field shows moderate maturity with ongoing innovation. Leading players represent diverse geographical and industrial backgrounds: LG Energy Solution and LG Chem dominate commercial applications; Honda Motor and GS Yuasa bring automotive expertise; while academic institutions like Central South University and Kyushu University contribute fundamental research. JSR Corp, BASF, and Toyobo lead in materials development, while Chinese institutions (Tianmu Lake Institute, Yangtze River Delta Physics Research Center) are emerging as significant contributors, indicating a competitive landscape with both established corporations and research-focused entities.
BASF Corp.
Technical Solution: BASF has developed a comprehensive composite solid electrolyte platform utilizing their expertise in advanced materials chemistry. Their approach centers on a hierarchical composite structure combining nano-sized ceramic particles (primarily LLZO and LATP) with a thermally resistant fluoropolymer matrix. BASF employs a proprietary core-shell particle engineering technique where ceramic particles are encapsulated with a thin layer of thermally stable polymer that serves as a buffer zone between the ceramic and bulk polymer matrix. This design significantly reduces thermal expansion mismatch issues that typically lead to cracking at elevated temperatures. Their composite electrolytes incorporate specially designed thermal stabilizers and antioxidants that prevent polymer degradation even at temperatures exceeding 150°C. BASF has pioneered an innovative processing technique using supercritical CO2 to create a microporous structure within the composite, enhancing ionic transport while maintaining mechanical integrity. Their materials demonstrate exceptional dimensional stability with less than 2% change in thickness after exposure to temperature cycling between -20°C and 150°C. Recent developments include the incorporation of self-healing polymers that can repair microcracks formed during thermal cycling, significantly extending operational lifetime[9][10].
Strengths: Hierarchical composite structure optimizes both thermal and mechanical properties; proprietary core-shell particle technology minimizes interfacial thermal stress; exceptional dimensional stability during temperature cycling. Weaknesses: Higher material costs due to complex particle engineering; potential for increased manufacturing complexity; challenges in achieving uniform particle distribution in large-format applications.
Chinese Academy of Sciences Institute of Physics
Technical Solution: The Chinese Academy of Sciences Institute of Physics has developed an advanced composite solid electrolyte system based on a three-dimensional interconnected network structure. Their innovative approach combines nano-sized ceramic fillers (primarily Li1.3Al0.3Ti1.7(PO4)3 and Li6.4La3Zr1.4Ta0.6O12) with a cross-linked polymer matrix using a sol-gel synthesis method. This creates a continuous ceramic framework that provides both mechanical support and high-temperature stability. The institute has pioneered a novel surface functionalization technique for ceramic particles using silane coupling agents that form strong covalent bonds with the polymer matrix, significantly reducing interfacial thermal stress. Their composite electrolyte incorporates thermally stable flame-retardant additives derived from phosphazene compounds, which release non-flammable gases upon heating to actively suppress thermal runaway. Extensive testing has demonstrated that their composite electrolytes maintain structural integrity and over 80% of their room temperature ionic conductivity even after exposure to temperatures of 180°C for extended periods. The institute has also developed computational models to predict thermal behavior and optimize composition ratios for specific application requirements[6][8].
Strengths: Three-dimensional interconnected structure provides superior mechanical and thermal stability; novel surface functionalization techniques significantly improve interfacial properties; active flame-retardant mechanisms provide additional safety features. Weaknesses: Complex synthesis process may present challenges for large-scale production; higher content of ceramic materials may reduce overall flexibility; potential for increased brittleness compared to polymer-dominant composites.
Critical Patents and Scientific Breakthroughs
Advanced solid acid electrolyte composites
PatentInactiveUS20070128491A1
Innovation
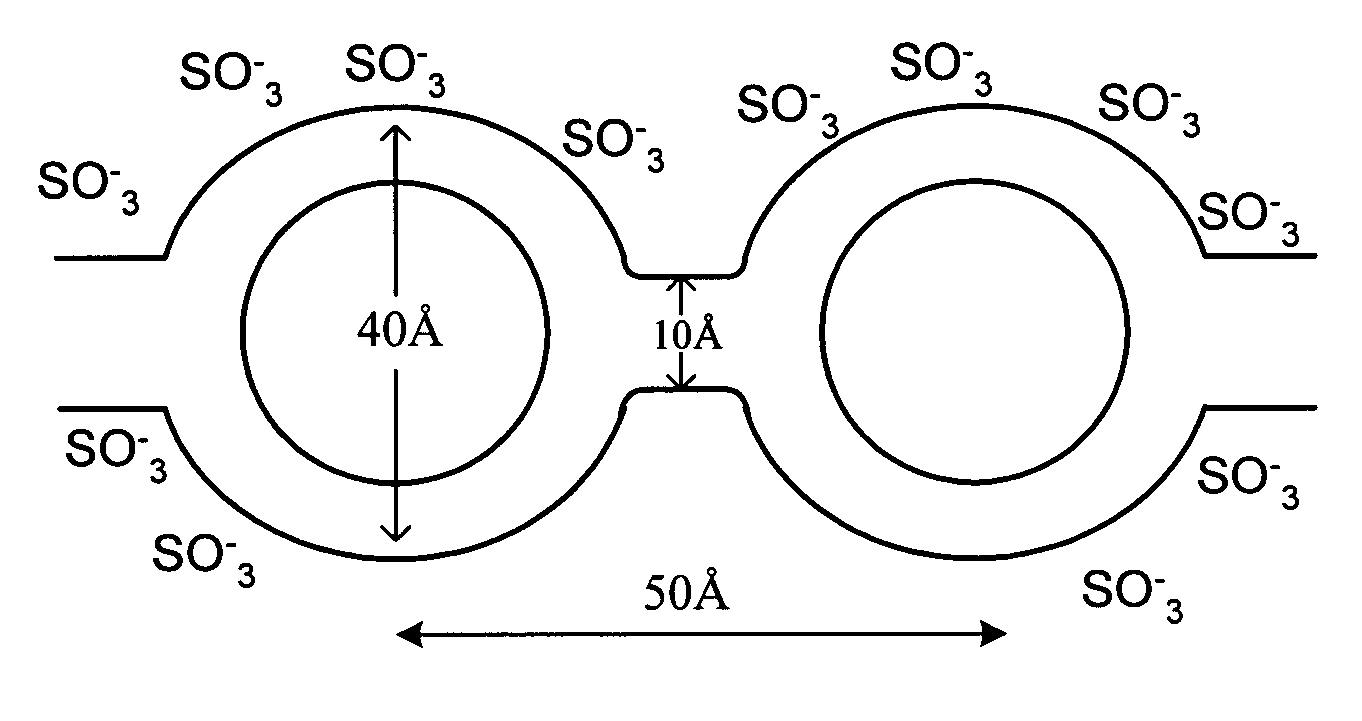
- A proton conducting membrane comprising a stable electrolyte composite material with a solid acid component and a surface-hydrogen-containing secondary component, forming interfaces through hydrogen bonding interactions, which enhances mechanical stability and allows operation at higher temperatures.
Hybrid solid electrolyte for secondary battery and manufacturing method therefor
PatentWO2018056491A1
Innovation
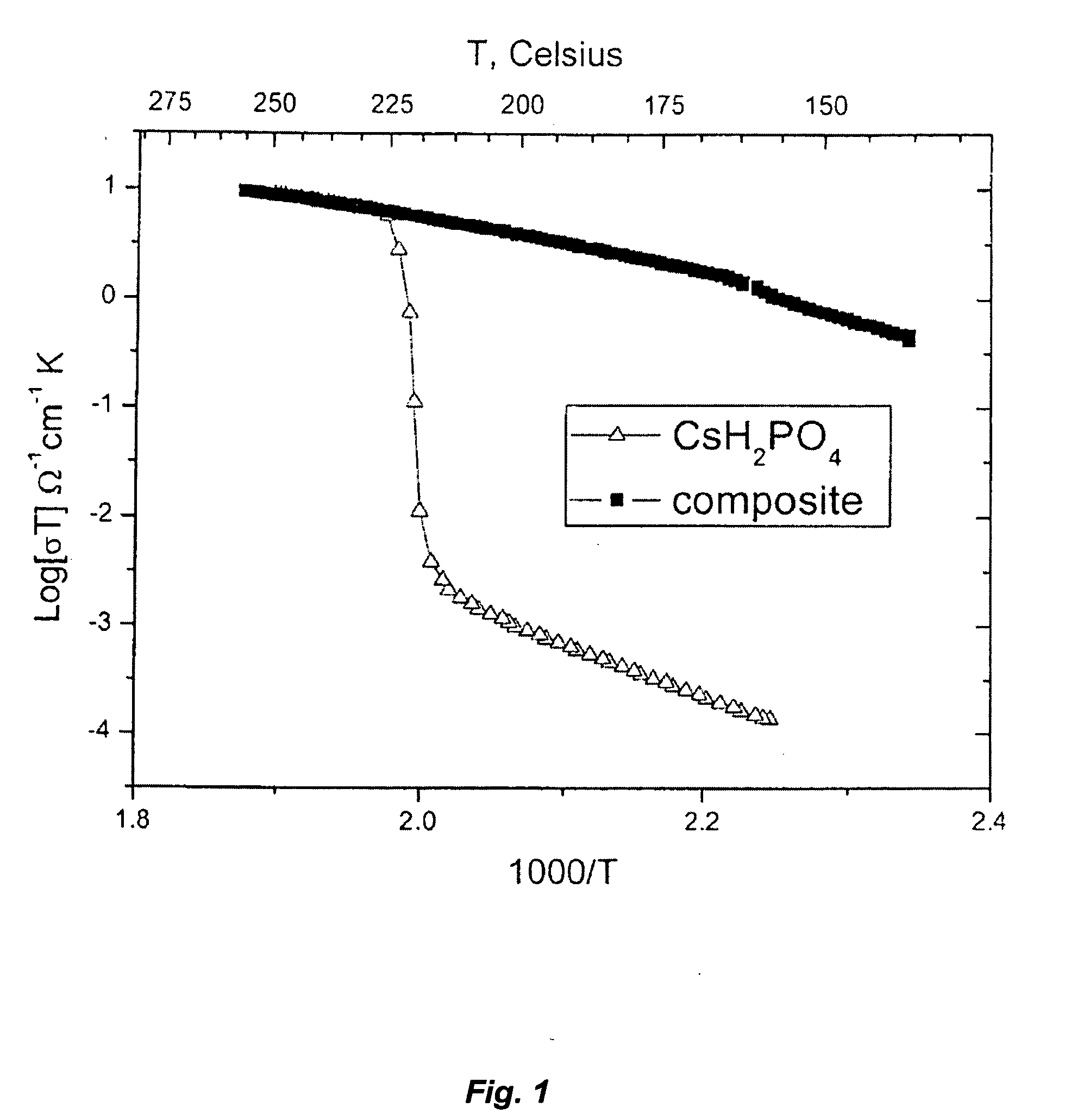
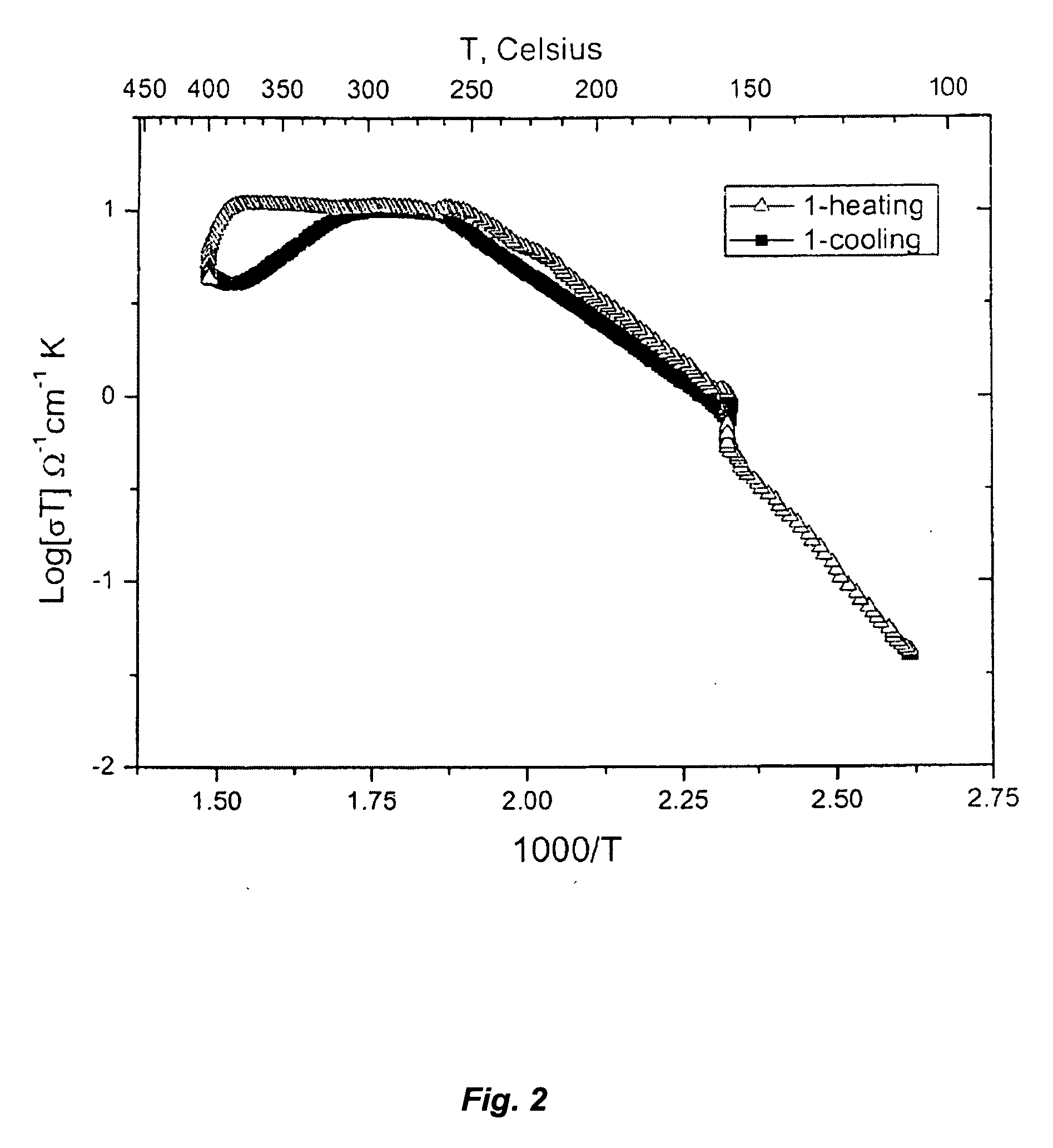
- A composite solid electrolyte is developed by combining ion conductive ceramic, polymer, and liquid electrolyte, with a specific weight ratio and solvent composition, including ionic liquids, to enhance stability and electrochemical properties, such as thermal stability above 400°C and reduced interfacial resistance.
Safety Standards and Testing Protocols
The development of composite solid electrolytes with enhanced thermal stability necessitates rigorous safety standards and testing protocols to ensure their reliable implementation in energy storage systems. Current international standards, including IEC 62660 and ISO 12405, provide foundational frameworks for evaluating solid electrolytes, though they require adaptation to address the unique properties of composite materials.
Thermal stability testing represents a critical component of safety evaluation, typically involving differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA) to determine phase transition temperatures and decomposition points. These tests must be conducted under standardized conditions, with heating rates typically set at 5-10°C/min and temperature ranges extending from ambient to 500°C to fully characterize thermal behavior.
Mechanical integrity testing protocols have evolved significantly, now incorporating compression, tensile, and shear tests to evaluate the structural stability of composite electrolytes under various stress conditions. The industry standard requires materials to maintain integrity under pressures of 10-100 MPa, simulating real-world battery operation conditions.
Electrochemical stability assessment protocols measure the voltage window within which the electrolyte remains stable, typically using cyclic voltammetry and impedance spectroscopy. For advanced energy storage applications, composite solid electrolytes must demonstrate stability within a 0-5V range against lithium metal reference electrodes.
Interface compatibility testing has emerged as a crucial protocol area, evaluating the chemical and electrochemical interactions between composite electrolytes and electrode materials. Standardized procedures now include extended contact tests at elevated temperatures (60-80°C) for periods exceeding 1000 hours to simulate long-term usage scenarios.
Accelerated aging protocols subject materials to elevated temperatures (typically 60-85°C) for extended periods (500-2000 hours) to predict long-term stability. These tests are complemented by thermal cycling procedures that evaluate material performance through repeated temperature fluctuations between -40°C and 85°C.
Safety certification requirements vary by region, with UL 1642 in North America, IEC 62133 in Europe, and GB/T 31485 in China representing the primary standards for battery components. Manufacturers must navigate these regulatory frameworks to ensure market access, with certification processes typically requiring 6-12 months of comprehensive testing.
Recent advancements in testing methodologies include in-situ characterization techniques that monitor structural and chemical changes during thermal events, providing deeper insights into failure mechanisms and enabling more precise safety assessments for next-generation composite solid electrolytes.
Thermal stability testing represents a critical component of safety evaluation, typically involving differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA) to determine phase transition temperatures and decomposition points. These tests must be conducted under standardized conditions, with heating rates typically set at 5-10°C/min and temperature ranges extending from ambient to 500°C to fully characterize thermal behavior.
Mechanical integrity testing protocols have evolved significantly, now incorporating compression, tensile, and shear tests to evaluate the structural stability of composite electrolytes under various stress conditions. The industry standard requires materials to maintain integrity under pressures of 10-100 MPa, simulating real-world battery operation conditions.
Electrochemical stability assessment protocols measure the voltage window within which the electrolyte remains stable, typically using cyclic voltammetry and impedance spectroscopy. For advanced energy storage applications, composite solid electrolytes must demonstrate stability within a 0-5V range against lithium metal reference electrodes.
Interface compatibility testing has emerged as a crucial protocol area, evaluating the chemical and electrochemical interactions between composite electrolytes and electrode materials. Standardized procedures now include extended contact tests at elevated temperatures (60-80°C) for periods exceeding 1000 hours to simulate long-term usage scenarios.
Accelerated aging protocols subject materials to elevated temperatures (typically 60-85°C) for extended periods (500-2000 hours) to predict long-term stability. These tests are complemented by thermal cycling procedures that evaluate material performance through repeated temperature fluctuations between -40°C and 85°C.
Safety certification requirements vary by region, with UL 1642 in North America, IEC 62133 in Europe, and GB/T 31485 in China representing the primary standards for battery components. Manufacturers must navigate these regulatory frameworks to ensure market access, with certification processes typically requiring 6-12 months of comprehensive testing.
Recent advancements in testing methodologies include in-situ characterization techniques that monitor structural and chemical changes during thermal events, providing deeper insights into failure mechanisms and enabling more precise safety assessments for next-generation composite solid electrolytes.
Manufacturing Scalability Assessment
The scalability of manufacturing processes for composite solid electrolytes (CSEs) with enhanced thermal stability presents significant challenges that must be addressed for commercial viability. Current laboratory-scale production methods typically involve complex multi-step processes including powder preparation, mixing, pressing, and sintering, which are difficult to translate directly to industrial scales. The transition from gram-scale to kilogram or ton-scale production requires substantial process engineering and optimization.
Material consistency represents a primary challenge in scaling up CSE manufacturing. Ensuring uniform distribution of ceramic fillers within polymer matrices across large batches demands precise control over mixing parameters and environmental conditions. Variations in particle size distribution, agglomeration tendencies, and interfacial interactions can significantly impact the final electrolyte performance, particularly thermal stability properties that are critical for safety.
Equipment adaptation constitutes another major consideration. While laboratory production often utilizes small batch processing equipment, industrial manufacturing requires continuous or semi-continuous production lines. Specialized equipment for high-throughput mixing, casting, and thermal treatment must be developed with careful attention to maintaining material properties throughout scaled processes.
Cost efficiency becomes increasingly important at larger scales. Raw material sourcing strategies must balance performance requirements with economic considerations. For instance, some high-performance ceramic additives that enhance thermal stability may be prohibitively expensive for mass production, necessitating alternative formulations or novel synthesis routes for cost reduction.
Quality control methodologies must evolve alongside production scaling. In-line monitoring techniques capable of rapidly assessing critical parameters such as ionic conductivity, mechanical properties, and thermal stability characteristics are essential for maintaining consistent product quality. Non-destructive testing methods are particularly valuable for continuous production environments.
Several promising approaches are emerging to address these challenges. Roll-to-roll processing techniques adapted from polymer film industries show potential for continuous production of thin-film composite electrolytes. Solution-based processing methods offer advantages in achieving homogeneous dispersion of ceramic components while reducing energy requirements compared to traditional solid-state routes.
Collaborative efforts between academic institutions and industrial partners are accelerating the development of scalable manufacturing protocols. Pilot-scale demonstration projects have successfully produced CSEs with enhanced thermal stability at scales of 5-10 kg per batch, representing an important intermediate step toward full commercialization.
Material consistency represents a primary challenge in scaling up CSE manufacturing. Ensuring uniform distribution of ceramic fillers within polymer matrices across large batches demands precise control over mixing parameters and environmental conditions. Variations in particle size distribution, agglomeration tendencies, and interfacial interactions can significantly impact the final electrolyte performance, particularly thermal stability properties that are critical for safety.
Equipment adaptation constitutes another major consideration. While laboratory production often utilizes small batch processing equipment, industrial manufacturing requires continuous or semi-continuous production lines. Specialized equipment for high-throughput mixing, casting, and thermal treatment must be developed with careful attention to maintaining material properties throughout scaled processes.
Cost efficiency becomes increasingly important at larger scales. Raw material sourcing strategies must balance performance requirements with economic considerations. For instance, some high-performance ceramic additives that enhance thermal stability may be prohibitively expensive for mass production, necessitating alternative formulations or novel synthesis routes for cost reduction.
Quality control methodologies must evolve alongside production scaling. In-line monitoring techniques capable of rapidly assessing critical parameters such as ionic conductivity, mechanical properties, and thermal stability characteristics are essential for maintaining consistent product quality. Non-destructive testing methods are particularly valuable for continuous production environments.
Several promising approaches are emerging to address these challenges. Roll-to-roll processing techniques adapted from polymer film industries show potential for continuous production of thin-film composite electrolytes. Solution-based processing methods offer advantages in achieving homogeneous dispersion of ceramic components while reducing energy requirements compared to traditional solid-state routes.
Collaborative efforts between academic institutions and industrial partners are accelerating the development of scalable manufacturing protocols. Pilot-scale demonstration projects have successfully produced CSEs with enhanced thermal stability at scales of 5-10 kg per batch, representing an important intermediate step toward full commercialization.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!