Halide solid-state electrolytes for high-voltage cathodes
FEB 14, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Halide Electrolyte Development Background and Objectives
The development of solid-state batteries represents a paradigm shift in energy storage technology, driven by the urgent need for safer, more energy-dense power sources for electric vehicles, portable electronics, and grid-scale applications. Traditional lithium-ion batteries utilizing liquid organic electrolytes face inherent safety risks including flammability and thermal runaway, alongside limitations in energy density due to voltage constraints. These challenges have catalyzed intensive research into solid-state electrolytes as enabling materials for next-generation battery systems.
Halide solid-state electrolytes have emerged as a promising class of materials within the broader solid electrolyte landscape, which also includes oxides, sulfides, and polymers. Metal halides, particularly lithium-based compounds such as Li3YCl6 and Li3InCl6, exhibit exceptional electrochemical stability windows exceeding 5V versus Li/Li+, making them uniquely suited for integration with high-voltage cathode materials. This voltage stability advantage addresses a critical bottleneck in solid-state battery development, as many sulfide electrolytes undergo oxidative decomposition at voltages above 2.5V, severely limiting cathode material selection.
The evolution of halide electrolytes traces back to fundamental studies on lithium halide conductivity in the 1980s, but recent breakthroughs in compositional engineering and structural optimization have dramatically improved their ionic conductivity from 10^-7 S/cm to values approaching 10^-3 S/cm at room temperature. This thousand-fold enhancement has transformed halides from academic curiosities into viable candidates for practical applications, though they still lag behind the best sulfide conductors in absolute conductivity values.
The primary objective of current halide electrolyte research focuses on achieving compatibility with high-voltage cathode materials such as LiCoO2, LiNi0.8Mn0.1Co0.1O2 (NMC811), and lithium-rich layered oxides operating above 4.5V. This compatibility encompasses not only electrochemical stability but also interfacial stability, mechanical integrity during cycling, and scalable manufacturing processes. Secondary objectives include improving room-temperature ionic conductivity, reducing grain boundary resistance, enhancing moisture stability for practical handling, and developing cost-effective synthesis routes suitable for industrial-scale production.
Halide solid-state electrolytes have emerged as a promising class of materials within the broader solid electrolyte landscape, which also includes oxides, sulfides, and polymers. Metal halides, particularly lithium-based compounds such as Li3YCl6 and Li3InCl6, exhibit exceptional electrochemical stability windows exceeding 5V versus Li/Li+, making them uniquely suited for integration with high-voltage cathode materials. This voltage stability advantage addresses a critical bottleneck in solid-state battery development, as many sulfide electrolytes undergo oxidative decomposition at voltages above 2.5V, severely limiting cathode material selection.
The evolution of halide electrolytes traces back to fundamental studies on lithium halide conductivity in the 1980s, but recent breakthroughs in compositional engineering and structural optimization have dramatically improved their ionic conductivity from 10^-7 S/cm to values approaching 10^-3 S/cm at room temperature. This thousand-fold enhancement has transformed halides from academic curiosities into viable candidates for practical applications, though they still lag behind the best sulfide conductors in absolute conductivity values.
The primary objective of current halide electrolyte research focuses on achieving compatibility with high-voltage cathode materials such as LiCoO2, LiNi0.8Mn0.1Co0.1O2 (NMC811), and lithium-rich layered oxides operating above 4.5V. This compatibility encompasses not only electrochemical stability but also interfacial stability, mechanical integrity during cycling, and scalable manufacturing processes. Secondary objectives include improving room-temperature ionic conductivity, reducing grain boundary resistance, enhancing moisture stability for practical handling, and developing cost-effective synthesis routes suitable for industrial-scale production.
Market Demand for High-Voltage Solid-State Batteries
The global transition toward electrification of transportation and energy storage systems has created unprecedented demand for advanced battery technologies that surpass the performance limitations of conventional lithium-ion batteries. High-voltage solid-state batteries represent a critical evolutionary step in meeting these escalating requirements, driven primarily by the automotive industry's pursuit of electric vehicles with extended driving ranges, faster charging capabilities, and enhanced safety profiles. The integration of halide solid-state electrolytes with high-voltage cathode materials addresses fundamental market needs by enabling energy densities that approach theoretical limits while eliminating flammability risks associated with liquid electrolytes.
Consumer electronics manufacturers are increasingly seeking compact power solutions that can support demanding applications such as augmented reality devices, foldable smartphones, and wearable health monitors. These applications require batteries that maintain stable performance across wide temperature ranges while occupying minimal space, creating substantial market pull for solid-state architectures capable of operating at elevated voltages. The ability of halide electrolytes to remain electrochemically stable against high-voltage cathodes directly responds to this demand for miniaturized yet powerful energy storage solutions.
Grid-scale energy storage represents another significant market driver, as renewable energy integration necessitates large-capacity systems with extended operational lifespans and minimal degradation. High-voltage solid-state batteries utilizing halide electrolytes offer superior cycle stability and reduced maintenance requirements compared to conventional technologies, addressing the economic viability concerns that currently limit widespread deployment of stationary storage infrastructure. The elimination of thermal runaway risks also reduces insurance and safety management costs for utility-scale installations.
Aerospace and defense sectors demonstrate growing interest in high-voltage solid-state battery technologies due to stringent safety requirements and performance demands under extreme conditions. Applications ranging from unmanned aerial vehicles to satellite power systems require energy storage solutions that function reliably across temperature extremes while maintaining consistent voltage output. The chemical stability and mechanical robustness of halide electrolytes position them as viable candidates for these specialized high-value markets, where performance justifies premium pricing structures.
Consumer electronics manufacturers are increasingly seeking compact power solutions that can support demanding applications such as augmented reality devices, foldable smartphones, and wearable health monitors. These applications require batteries that maintain stable performance across wide temperature ranges while occupying minimal space, creating substantial market pull for solid-state architectures capable of operating at elevated voltages. The ability of halide electrolytes to remain electrochemically stable against high-voltage cathodes directly responds to this demand for miniaturized yet powerful energy storage solutions.
Grid-scale energy storage represents another significant market driver, as renewable energy integration necessitates large-capacity systems with extended operational lifespans and minimal degradation. High-voltage solid-state batteries utilizing halide electrolytes offer superior cycle stability and reduced maintenance requirements compared to conventional technologies, addressing the economic viability concerns that currently limit widespread deployment of stationary storage infrastructure. The elimination of thermal runaway risks also reduces insurance and safety management costs for utility-scale installations.
Aerospace and defense sectors demonstrate growing interest in high-voltage solid-state battery technologies due to stringent safety requirements and performance demands under extreme conditions. Applications ranging from unmanned aerial vehicles to satellite power systems require energy storage solutions that function reliably across temperature extremes while maintaining consistent voltage output. The chemical stability and mechanical robustness of halide electrolytes position them as viable candidates for these specialized high-value markets, where performance justifies premium pricing structures.
Current Status and Challenges of Halide Electrolytes
Halide solid-state electrolytes have emerged as promising candidates for next-generation lithium batteries, particularly for pairing with high-voltage cathode materials. These materials, primarily metal halides such as lithium chlorides, bromides, and iodides, exhibit exceptional electrochemical stability windows often exceeding 4.5V versus lithium, making them theoretically compatible with high-voltage cathodes like LiCoO2, LiNi0.8Co0.1Mn0.1O2, and lithium-rich layered oxides. Recent developments have demonstrated ionic conductivities reaching 10^-3 to 10^-2 S/cm at room temperature for optimized compositions, approaching levels suitable for practical applications.
Despite these advantages, halide electrolytes face several critical challenges that impede their commercial deployment. The primary obstacle lies in their mechanical properties, as most halide compounds exhibit brittleness and poor ductility compared to sulfide-based electrolytes. This mechanical fragility complicates electrode-electrolyte interface formation and increases susceptibility to crack propagation during battery cycling, leading to increased interfacial resistance and potential failure pathways.
Interfacial compatibility represents another significant challenge. While halide electrolytes demonstrate wide electrochemical windows in isolation, practical cathode-electrolyte interfaces often suffer from space charge layer formation, chemical side reactions, and mutual diffusion phenomena. These interfacial issues manifest as increased impedance and capacity fade during cycling, particularly at elevated voltages where oxidative decomposition becomes thermodynamically favorable. The formation of resistive interphases containing mixed halide-oxide phases further degrades performance.
Manufacturing scalability poses additional constraints. Current synthesis methods for halide electrolytes typically involve high-temperature solid-state reactions or mechanochemical ball milling, both presenting challenges for large-scale production. Moisture sensitivity of many halide compositions necessitates stringent atmospheric control during processing and assembly, significantly increasing production costs. Furthermore, achieving uniform particle size distribution and consistent ionic conductivity across large batches remains technically demanding.
The geographical distribution of halide electrolyte research shows concentration in East Asian countries, particularly Japan, South Korea, and China, alongside significant contributions from North American and European research institutions. This distribution reflects both fundamental research capabilities and strategic interests in next-generation battery technologies for electric vehicles and grid storage applications.
Despite these advantages, halide electrolytes face several critical challenges that impede their commercial deployment. The primary obstacle lies in their mechanical properties, as most halide compounds exhibit brittleness and poor ductility compared to sulfide-based electrolytes. This mechanical fragility complicates electrode-electrolyte interface formation and increases susceptibility to crack propagation during battery cycling, leading to increased interfacial resistance and potential failure pathways.
Interfacial compatibility represents another significant challenge. While halide electrolytes demonstrate wide electrochemical windows in isolation, practical cathode-electrolyte interfaces often suffer from space charge layer formation, chemical side reactions, and mutual diffusion phenomena. These interfacial issues manifest as increased impedance and capacity fade during cycling, particularly at elevated voltages where oxidative decomposition becomes thermodynamically favorable. The formation of resistive interphases containing mixed halide-oxide phases further degrades performance.
Manufacturing scalability poses additional constraints. Current synthesis methods for halide electrolytes typically involve high-temperature solid-state reactions or mechanochemical ball milling, both presenting challenges for large-scale production. Moisture sensitivity of many halide compositions necessitates stringent atmospheric control during processing and assembly, significantly increasing production costs. Furthermore, achieving uniform particle size distribution and consistent ionic conductivity across large batches remains technically demanding.
The geographical distribution of halide electrolyte research shows concentration in East Asian countries, particularly Japan, South Korea, and China, alongside significant contributions from North American and European research institutions. This distribution reflects both fundamental research capabilities and strategic interests in next-generation battery technologies for electric vehicles and grid storage applications.
Existing Halide Electrolyte Solutions
01 Halide-based solid electrolyte composition and ionic conductivity
Halide solid-state electrolytes utilize specific halide compositions to achieve high ionic conductivity at room temperature. These electrolytes typically comprise metal halides combined with lithium or sodium halides to form crystalline or amorphous structures that facilitate ion transport. The composition and stoichiometry of halide components directly influence the electrochemical window and voltage stability of the electrolyte system.- Halide-based solid electrolyte composition and ionic conductivity: Halide solid-state electrolytes utilize specific halide compositions to achieve high ionic conductivity at room temperature. These electrolytes typically comprise metal halides combined with lithium or sodium halides to form crystalline or amorphous structures that facilitate ion transport. The composition can be optimized by adjusting the ratio of different halide components and incorporating dopants to enhance electrochemical performance and stability across a wide voltage window.
- Voltage stability and electrochemical window enhancement: Improving the electrochemical stability window of halide solid-state electrolytes is critical for high-voltage battery applications. This can be achieved through material design strategies including surface modification, interface engineering, and the incorporation of stabilizing additives. These approaches help prevent decomposition reactions at high voltages and maintain stable ion conduction, enabling compatibility with high-voltage cathode materials.
- Interface engineering between halide electrolytes and electrodes: The interface between halide solid electrolytes and electrode materials significantly affects voltage performance and cycling stability. Interface engineering techniques include the application of protective coatings, buffer layers, or interfacial modification agents to reduce interfacial resistance and prevent unwanted side reactions. These methods improve charge transfer kinetics and extend the operational voltage range of solid-state batteries.
- Doping and substitution strategies for voltage performance: Doping halide solid electrolytes with various elements or partial substitution of halide ions can significantly enhance voltage stability and ionic conductivity. These modifications alter the crystal structure and electronic properties, leading to improved electrochemical windows and reduced interfacial reactions at high voltages. The selection of appropriate dopants depends on their compatibility with the host structure and their ability to stabilize the electrolyte under operating conditions.
- Composite halide electrolytes for enhanced voltage characteristics: Composite halide electrolytes combine halide materials with polymers, oxides, or other solid electrolytes to create hybrid systems with improved voltage stability and mechanical properties. These composites leverage the advantages of different materials to achieve better interfacial contact, enhanced ionic conductivity, and extended electrochemical stability windows. The composite approach also helps mitigate individual material limitations and enables operation at higher voltages.
02 Voltage stability and electrochemical window enhancement
The electrochemical stability window of halide solid-state electrolytes determines the maximum operating voltage of batteries. Strategies to enhance voltage stability include doping with specific elements, forming protective interfacial layers, and optimizing the crystal structure. These modifications prevent decomposition reactions at high voltages and extend the operational voltage range of solid-state batteries.Expand Specific Solutions03 Interface engineering between halide electrolytes and electrodes
The interface between halide solid electrolytes and electrode materials significantly affects voltage performance and cycling stability. Interface engineering techniques include applying buffer layers, surface coating modifications, and compositional gradients to reduce interfacial resistance and prevent side reactions. These approaches maintain stable voltage profiles during charge-discharge cycles and improve overall battery performance.Expand Specific Solutions04 Halide electrolyte synthesis and processing methods
Manufacturing methods for halide solid-state electrolytes impact their voltage characteristics and electrochemical properties. Synthesis techniques such as mechanical ball milling, solid-state reactions, and solution-based processing affect particle size, crystallinity, and defect concentration. Optimized processing conditions result in electrolytes with improved voltage stability and reduced polarization during operation.Expand Specific Solutions05 Composite halide electrolyte systems for voltage optimization
Composite halide electrolyte systems combine multiple halide phases or incorporate polymer matrices to optimize voltage performance. These hybrid structures leverage the advantages of different components to achieve enhanced ionic conductivity, wider electrochemical windows, and better mechanical properties. The synergistic effects in composite systems enable stable operation at higher voltages compared to single-phase halide electrolytes.Expand Specific Solutions
Key Players in Halide Electrolyte Research
The halide solid-state electrolyte research for high-voltage cathodes represents an emerging yet rapidly advancing field within next-generation battery technology. The industry is transitioning from laboratory-scale development to early commercialization, with significant investments from automotive manufacturers like Guangzhou Automobile Group and Zhejiang Geely Holding Group, alongside specialized battery developers such as QuantumScape Corp., QingTao Energy Development, and Honeycomb Battery Co. The market demonstrates substantial growth potential driven by electric vehicle demands and energy storage requirements. Technology maturity varies considerably across players, with leading research institutions including Centre National de la Recherche Scientifique, Beijing Institute of Technology, and Georgia Tech Research Corp. advancing fundamental science, while companies like Factorial Inc., BASF Corp., and ZEON Corp. focus on materials commercialization. Academic-industry collaborations involving University of Waterloo, University of Michigan, and Kyushu University accelerate innovation, though challenges in scalability, interfacial stability, and manufacturing processes remain critical barriers to widespread adoption.
Centre National de la Recherche Scientifique
Technical Solution: CNRS has conducted fundamental research on halide solid electrolytes, particularly focusing on lithium metal halide families (Li3MX6 where M=Y, Er, In and X=Cl, Br) for high-voltage solid-state battery applications. Their research demonstrates that chloride-based electrolytes such as Li3YCl6 and Li3InCl6 exhibit wide electrochemical stability windows ranging from 0.4V to 5.5V vs Li/Li+, making them inherently compatible with high-voltage cathode materials including LiCoO2 and LNMO spinel structures. The materials achieve room-temperature ionic conductivities between 10^-3 and 10^-4 S/cm with relatively low grain boundary resistances. CNRS research groups have investigated doping strategies using mixed halide compositions (Cl/Br combinations) to optimize both ionic transport and interfacial properties. Laboratory-scale cells using these halide electrolytes with high-voltage cathodes have demonstrated initial capacities exceeding 140 mAh/g with stable cycling performance over 200 cycles.
Strengths: Deep fundamental understanding of halide electrolyte chemistry and structure-property relationships; extensive characterization capabilities; strong publication record advancing scientific knowledge. Weaknesses: Research-focused organization with limited direct commercialization pathways; laboratory-scale demonstrations require significant development for industrial application; technology transfer mechanisms needed for market impact.
Factorial, Inc.
Technical Solution: Factorial has developed a proprietary solid-state battery technology utilizing halide-based electrolytes specifically designed for high-voltage cathode compatibility. Their FEST (Factorial Electrolyte System Technology) platform incorporates metal halide compounds that demonstrate exceptional electrochemical stability windows exceeding 4.5V vs Li/Li+, enabling compatibility with high-voltage cathode materials such as NMC811 and LNMO. The halide electrolyte formulation exhibits ionic conductivity in the range of 10^-3 to 10^-4 S/cm at room temperature while maintaining interfacial stability with lithium metal anodes. The company has successfully demonstrated full-cell prototypes achieving energy densities above 350 Wh/kg with extended cycle life performance over 500 cycles at C/3 rate.
Strengths: High voltage stability window enabling next-generation cathode materials; strong industry partnerships with major automotive OEMs for commercialization. Weaknesses: Relatively lower ionic conductivity compared to sulfide electrolytes; scalable manufacturing processes still under development for mass production.
Core Patents in Halide-Cathode Interface Engineering
Design principle of solid electrolyte for high-voltage all-solid-state battery and utilization thereof
PatentPendingUS20250372699A1
Innovation
- A lithium halide-based solid electrolyte is developed by partially substituting chloride with fluoride and selecting appropriate metal elements, optimized through first-principle calculations to enhance structural stability and ionic conductivity.
Solid-state electrolyte, cathode electrode, and methods of making same for sulfide-based all-solid-state-batteries
PatentPendingUS20230055896A1
Innovation
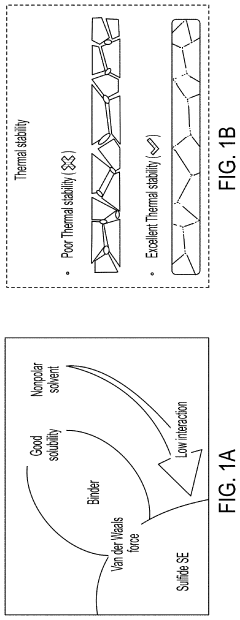
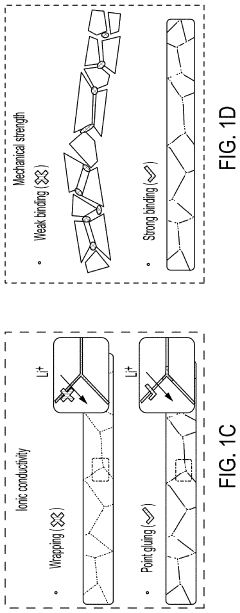
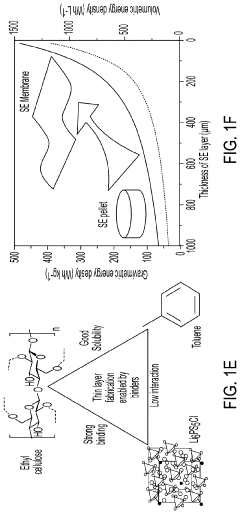
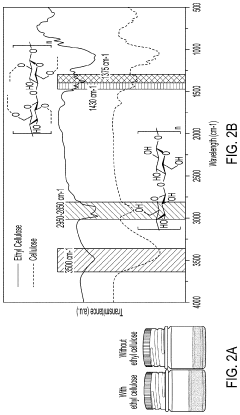
- A method involving the use of ethyl cellulose as a binder and solvent in the vacuum filtration process to create a thin, robust, and highly ion-conductive sulfide SE membrane with Li6PS5Cl, coupled with a water-mediated synthesis of Li3InCl6 for the cathode layer, to achieve a stable and efficient battery configuration.
Safety Standards for Solid-State Battery Systems
The development of halide solid-state electrolytes for high-voltage cathode applications necessitates comprehensive safety standards to ensure reliable commercialization and widespread adoption. Currently, the regulatory framework for solid-state battery systems remains fragmented, with various international organizations working to establish unified protocols. The International Electrotechnical Commission (IEC) and Underwriters Laboratories (UL) have initiated preliminary guidelines, yet specific standards addressing halide-based electrolytes and their unique safety considerations are still evolving. These standards must address material stability, thermal runaway prevention, and interface integrity under extreme operating conditions.
Safety certification for solid-state batteries incorporating halide electrolytes requires rigorous testing protocols that differ substantially from conventional lithium-ion systems. Key evaluation criteria include electrochemical stability windows, moisture sensitivity, and chemical compatibility with high-voltage cathode materials. Testing methodologies must encompass mechanical abuse scenarios, thermal cycling, and long-term degradation assessments. Particular attention is directed toward halide electrolyte decomposition products and their potential toxicity, necessitating specialized containment and handling procedures during manufacturing and end-of-life processing.
Regulatory bodies are increasingly focusing on establishing threshold values for critical safety parameters specific to halide solid-state systems. These include maximum allowable interfacial resistance growth rates, minimum ionic conductivity retention after thermal exposure, and acceptable levels of gas generation during operation. The standards must also define qualification procedures for high-voltage operation beyond 4.5V, where halide electrolytes demonstrate promising stability but require validated safety margins.
International harmonization efforts are underway to create unified safety standards that facilitate global market access while maintaining stringent protection requirements. Industry consortia and research institutions are collaborating to generate comprehensive safety databases, incorporating failure mode analysis and risk assessment methodologies tailored to halide electrolyte chemistry. These collaborative initiatives aim to accelerate standard development while ensuring that emerging technologies can be safely integrated into various applications, from consumer electronics to electric vehicles and grid storage systems.
Safety certification for solid-state batteries incorporating halide electrolytes requires rigorous testing protocols that differ substantially from conventional lithium-ion systems. Key evaluation criteria include electrochemical stability windows, moisture sensitivity, and chemical compatibility with high-voltage cathode materials. Testing methodologies must encompass mechanical abuse scenarios, thermal cycling, and long-term degradation assessments. Particular attention is directed toward halide electrolyte decomposition products and their potential toxicity, necessitating specialized containment and handling procedures during manufacturing and end-of-life processing.
Regulatory bodies are increasingly focusing on establishing threshold values for critical safety parameters specific to halide solid-state systems. These include maximum allowable interfacial resistance growth rates, minimum ionic conductivity retention after thermal exposure, and acceptable levels of gas generation during operation. The standards must also define qualification procedures for high-voltage operation beyond 4.5V, where halide electrolytes demonstrate promising stability but require validated safety margins.
International harmonization efforts are underway to create unified safety standards that facilitate global market access while maintaining stringent protection requirements. Industry consortia and research institutions are collaborating to generate comprehensive safety databases, incorporating failure mode analysis and risk assessment methodologies tailored to halide electrolyte chemistry. These collaborative initiatives aim to accelerate standard development while ensuring that emerging technologies can be safely integrated into various applications, from consumer electronics to electric vehicles and grid storage systems.
Scalable Manufacturing of Halide Electrolytes
The transition from laboratory-scale synthesis to industrial-scale production represents a critical bottleneck in commercializing halide solid-state electrolytes for high-voltage cathode applications. Current manufacturing approaches predominantly rely on batch processing methods such as ball milling and solid-state sintering, which are inherently limited in throughput and reproducibility. These conventional techniques face significant challenges in maintaining compositional uniformity and controlling particle size distribution when scaled beyond kilogram quantities, directly impacting the electrochemical performance consistency required for battery applications.
Recent advancements have introduced continuous manufacturing processes that show promise for large-scale production. Mechanochemical synthesis using continuous flow reactors enables precise control over reaction parameters while achieving production rates exceeding several kilograms per hour. This approach minimizes batch-to-batch variations and reduces processing time from days to hours compared to traditional methods. Additionally, spray drying and flame spray pyrolysis techniques have emerged as viable alternatives, offering rapid production rates and excellent compositional control, though they require substantial capital investment in specialized equipment.
The economic viability of halide electrolyte manufacturing hinges on raw material costs and energy consumption. Lithium and rare-earth halides constitute the primary cost drivers, with material expenses accounting for approximately sixty to seventy percent of total production costs. Energy-intensive processes such as high-temperature annealing and extended milling operations further constrain cost competitiveness. Strategies to address these challenges include developing lower-temperature synthesis routes, implementing solvent-based processing methods, and establishing closed-loop recycling systems for precursor materials.
Quality control and standardization present additional manufacturing complexities. Halide electrolytes exhibit extreme sensitivity to moisture and oxygen, necessitating stringent environmental controls throughout production and handling. Establishing industry-wide specifications for purity levels, particle morphology, and ionic conductivity remains essential for enabling supply chain integration. Furthermore, developing in-line monitoring techniques using spectroscopic and electrochemical methods will be crucial for ensuring consistent product quality at industrial scales while minimizing waste and production downtime.
Recent advancements have introduced continuous manufacturing processes that show promise for large-scale production. Mechanochemical synthesis using continuous flow reactors enables precise control over reaction parameters while achieving production rates exceeding several kilograms per hour. This approach minimizes batch-to-batch variations and reduces processing time from days to hours compared to traditional methods. Additionally, spray drying and flame spray pyrolysis techniques have emerged as viable alternatives, offering rapid production rates and excellent compositional control, though they require substantial capital investment in specialized equipment.
The economic viability of halide electrolyte manufacturing hinges on raw material costs and energy consumption. Lithium and rare-earth halides constitute the primary cost drivers, with material expenses accounting for approximately sixty to seventy percent of total production costs. Energy-intensive processes such as high-temperature annealing and extended milling operations further constrain cost competitiveness. Strategies to address these challenges include developing lower-temperature synthesis routes, implementing solvent-based processing methods, and establishing closed-loop recycling systems for precursor materials.
Quality control and standardization present additional manufacturing complexities. Halide electrolytes exhibit extreme sensitivity to moisture and oxygen, necessitating stringent environmental controls throughout production and handling. Establishing industry-wide specifications for purity levels, particle morphology, and ionic conductivity remains essential for enabling supply chain integration. Furthermore, developing in-line monitoring techniques using spectroscopic and electrochemical methods will be crucial for ensuring consistent product quality at industrial scales while minimizing waste and production downtime.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!