Moisture sensitivity of halide solid-state electrolytes explained
FEB 14, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Halide Electrolyte Development Background and Objectives
Halide solid-state electrolytes have emerged as promising candidates for next-generation all-solid-state batteries due to their exceptional ionic conductivity, wide electrochemical stability window, and favorable mechanical properties. The development of these materials represents a critical advancement in addressing the safety concerns and energy density limitations inherent in conventional liquid electrolyte systems. Metal halides, particularly lithium-based chlorides, bromides, and iodides, have demonstrated ionic conductivities approaching or exceeding those of liquid electrolytes at room temperature, making them attractive alternatives for high-performance energy storage applications.
However, the practical implementation of halide electrolytes faces a fundamental challenge: their pronounced sensitivity to atmospheric moisture. This vulnerability stems from the hygroscopic nature of halide compounds, which readily undergo hydrolysis reactions when exposed to water vapor, leading to rapid degradation of ionic conductivity and structural integrity. The moisture-induced decomposition not only compromises the electrolyte's electrochemical performance but also generates corrosive byproducts that can damage electrode materials and current collectors, severely limiting the operational lifespan of solid-state battery systems.
The primary objective of this research domain is to comprehensively understand the moisture sensitivity mechanisms of halide solid-state electrolytes at the molecular and interfacial levels. This includes elucidating the kinetics of hydrolysis reactions, identifying the degradation pathways, and quantifying the threshold moisture levels that trigger performance deterioration. By establishing a fundamental understanding of these processes, researchers aim to develop effective mitigation strategies that can enhance the environmental stability of halide electrolytes without sacrificing their superior ionic transport properties.
Furthermore, this research seeks to establish practical guidelines for the synthesis, processing, handling, and integration of halide electrolytes into functional battery architectures under controlled atmospheric conditions. The ultimate goal is to enable the transition of halide-based solid-state batteries from laboratory-scale demonstrations to commercially viable energy storage solutions, thereby contributing to the advancement of safer, more efficient, and longer-lasting battery technologies for electric vehicles, grid storage, and portable electronics applications.
However, the practical implementation of halide electrolytes faces a fundamental challenge: their pronounced sensitivity to atmospheric moisture. This vulnerability stems from the hygroscopic nature of halide compounds, which readily undergo hydrolysis reactions when exposed to water vapor, leading to rapid degradation of ionic conductivity and structural integrity. The moisture-induced decomposition not only compromises the electrolyte's electrochemical performance but also generates corrosive byproducts that can damage electrode materials and current collectors, severely limiting the operational lifespan of solid-state battery systems.
The primary objective of this research domain is to comprehensively understand the moisture sensitivity mechanisms of halide solid-state electrolytes at the molecular and interfacial levels. This includes elucidating the kinetics of hydrolysis reactions, identifying the degradation pathways, and quantifying the threshold moisture levels that trigger performance deterioration. By establishing a fundamental understanding of these processes, researchers aim to develop effective mitigation strategies that can enhance the environmental stability of halide electrolytes without sacrificing their superior ionic transport properties.
Furthermore, this research seeks to establish practical guidelines for the synthesis, processing, handling, and integration of halide electrolytes into functional battery architectures under controlled atmospheric conditions. The ultimate goal is to enable the transition of halide-based solid-state batteries from laboratory-scale demonstrations to commercially viable energy storage solutions, thereby contributing to the advancement of safer, more efficient, and longer-lasting battery technologies for electric vehicles, grid storage, and portable electronics applications.
Market Demand for Moisture-Stable Solid Electrolytes
The global transition toward electrified transportation and renewable energy storage systems has intensified the demand for safer, higher-performance battery technologies. Solid-state batteries, particularly those employing halide solid electrolytes, have emerged as promising candidates due to their superior ionic conductivity and potential for enhanced energy density. However, the pronounced moisture sensitivity of halide-based materials presents a critical barrier to their commercial viability, directly influencing market adoption and scalability.
Current lithium-ion battery technologies face inherent safety risks associated with flammable liquid electrolytes, driving manufacturers and research institutions to seek solid-state alternatives. The automotive sector, which represents a substantial portion of battery demand, requires electrolytes that can withstand diverse environmental conditions during manufacturing, assembly, and operation. Moisture-stable solid electrolytes would significantly reduce production costs by eliminating the need for stringent dry-room facilities and enabling compatibility with existing manufacturing infrastructure.
Consumer electronics manufacturers are similarly constrained by the hygroscopic nature of halide electrolytes, which complicates device assembly and limits product lifespan. The market increasingly demands battery solutions that maintain performance integrity across varying humidity levels without requiring hermetic sealing or continuous desiccant systems. This need extends to grid-scale energy storage applications, where long-term environmental exposure and maintenance accessibility are paramount considerations.
The aerospace and defense sectors present additional market segments requiring moisture-resistant solid electrolytes. These applications demand materials capable of reliable operation across extreme temperature and humidity ranges while maintaining structural and electrochemical stability. The inability of current halide electrolytes to meet these environmental resilience standards restricts their deployment in mission-critical systems.
Emerging markets in portable medical devices and Internet of Things applications further amplify demand for environmentally robust solid electrolytes. These sectors prioritize miniaturization and operational reliability in uncontrolled ambient conditions, characteristics that moisture-sensitive materials cannot currently provide. The development of moisture-stable halide electrolytes would unlock these high-growth market segments and accelerate the broader adoption of solid-state battery technology across multiple industries.
Current lithium-ion battery technologies face inherent safety risks associated with flammable liquid electrolytes, driving manufacturers and research institutions to seek solid-state alternatives. The automotive sector, which represents a substantial portion of battery demand, requires electrolytes that can withstand diverse environmental conditions during manufacturing, assembly, and operation. Moisture-stable solid electrolytes would significantly reduce production costs by eliminating the need for stringent dry-room facilities and enabling compatibility with existing manufacturing infrastructure.
Consumer electronics manufacturers are similarly constrained by the hygroscopic nature of halide electrolytes, which complicates device assembly and limits product lifespan. The market increasingly demands battery solutions that maintain performance integrity across varying humidity levels without requiring hermetic sealing or continuous desiccant systems. This need extends to grid-scale energy storage applications, where long-term environmental exposure and maintenance accessibility are paramount considerations.
The aerospace and defense sectors present additional market segments requiring moisture-resistant solid electrolytes. These applications demand materials capable of reliable operation across extreme temperature and humidity ranges while maintaining structural and electrochemical stability. The inability of current halide electrolytes to meet these environmental resilience standards restricts their deployment in mission-critical systems.
Emerging markets in portable medical devices and Internet of Things applications further amplify demand for environmentally robust solid electrolytes. These sectors prioritize miniaturization and operational reliability in uncontrolled ambient conditions, characteristics that moisture-sensitive materials cannot currently provide. The development of moisture-stable halide electrolytes would unlock these high-growth market segments and accelerate the broader adoption of solid-state battery technology across multiple industries.
Moisture Sensitivity Challenges in Halide Electrolytes
Halide solid-state electrolytes, particularly metal halides such as lithium chloride, lithium bromide, and their derivatives, have emerged as promising candidates for next-generation all-solid-state batteries due to their high ionic conductivity and favorable electrochemical stability windows. However, their practical implementation faces a critical obstacle: extreme sensitivity to atmospheric moisture. This vulnerability stems from the inherently hygroscopic nature of halide compounds, which readily absorb water molecules from ambient air, leading to rapid degradation of their electrochemical properties and structural integrity.
The moisture sensitivity challenge manifests through multiple degradation pathways. Upon exposure to humid environments, halide electrolytes undergo hydrolysis reactions that form hydrated phases, significantly reducing ionic conductivity by orders of magnitude. The absorbed moisture disrupts the crystalline structure essential for efficient ion transport, creating insulating barriers at grain boundaries and interfaces. Additionally, water molecules can react with halide ions to generate acidic byproducts, which further corrode electrode materials and compromise cell performance. This degradation process is often irreversible, making moisture protection a paramount concern throughout the entire lifecycle from synthesis to device operation.
Current manufacturing and handling protocols require stringent environmental controls, including ultra-low humidity glove boxes with dew points below -60°C and hermetically sealed packaging systems. These requirements substantially increase production costs and complicate scalability for commercial applications. The challenge extends beyond laboratory settings, as real-world battery applications demand long-term stability under varying atmospheric conditions. Even trace amounts of moisture penetration through packaging materials or during assembly processes can trigger progressive degradation, limiting the practical shelf life and operational reliability of halide-based solid-state batteries.
The fundamental trade-off between achieving high ionic conductivity and maintaining moisture stability represents a core technical dilemma. Strategies to enhance one property often compromise the other, necessitating innovative approaches that address both requirements simultaneously. Understanding the molecular-level mechanisms of moisture interaction and developing effective mitigation strategies remain critical priorities for advancing halide electrolytes toward commercial viability in solid-state energy storage systems.
The moisture sensitivity challenge manifests through multiple degradation pathways. Upon exposure to humid environments, halide electrolytes undergo hydrolysis reactions that form hydrated phases, significantly reducing ionic conductivity by orders of magnitude. The absorbed moisture disrupts the crystalline structure essential for efficient ion transport, creating insulating barriers at grain boundaries and interfaces. Additionally, water molecules can react with halide ions to generate acidic byproducts, which further corrode electrode materials and compromise cell performance. This degradation process is often irreversible, making moisture protection a paramount concern throughout the entire lifecycle from synthesis to device operation.
Current manufacturing and handling protocols require stringent environmental controls, including ultra-low humidity glove boxes with dew points below -60°C and hermetically sealed packaging systems. These requirements substantially increase production costs and complicate scalability for commercial applications. The challenge extends beyond laboratory settings, as real-world battery applications demand long-term stability under varying atmospheric conditions. Even trace amounts of moisture penetration through packaging materials or during assembly processes can trigger progressive degradation, limiting the practical shelf life and operational reliability of halide-based solid-state batteries.
The fundamental trade-off between achieving high ionic conductivity and maintaining moisture stability represents a core technical dilemma. Strategies to enhance one property often compromise the other, necessitating innovative approaches that address both requirements simultaneously. Understanding the molecular-level mechanisms of moisture interaction and developing effective mitigation strategies remain critical priorities for advancing halide electrolytes toward commercial viability in solid-state energy storage systems.
Current Moisture Protection Solutions
01 Surface coating and modification strategies for moisture protection
Halide solid-state electrolytes can be protected from moisture through surface coating and modification techniques. These methods involve applying protective layers or chemically modifying the surface to create a barrier against water vapor. Surface treatments can include the application of hydrophobic materials, polymer coatings, or inorganic protective layers that prevent direct contact between the halide electrolyte and atmospheric moisture. These strategies help maintain the structural integrity and ionic conductivity of the electrolyte while reducing degradation caused by moisture exposure.- Surface coating and modification strategies for moisture protection: Halide solid-state electrolytes can be protected from moisture through surface coating and modification techniques. These methods involve applying protective layers or chemically modifying the surface to create a barrier against water vapor. Surface treatments can include organic coatings, inorganic oxide layers, or hybrid materials that prevent direct contact between the halide electrolyte and atmospheric moisture. These strategies help maintain the structural integrity and ionic conductivity of the electrolyte while reducing degradation caused by hydrolysis reactions.
- Composite electrolyte design with moisture-stable components: Developing composite solid-state electrolytes by combining halide materials with moisture-stable components can significantly reduce moisture sensitivity. This approach involves incorporating polymers, oxides, or other stable materials that act as protective matrices or fillers. The composite structure creates tortuous pathways that limit moisture penetration while maintaining adequate ionic conductivity. The stable components can also buffer the halide material from direct exposure to water molecules, thereby extending the operational lifetime of the electrolyte in ambient conditions.
- Doping and chemical substitution for enhanced stability: Chemical doping and elemental substitution in halide solid-state electrolytes can improve their moisture resistance. By introducing specific dopants or replacing certain halide ions with more stable alternatives, the chemical stability of the electrolyte can be enhanced. These modifications can alter the crystal structure, reduce the number of reactive sites, and increase the energy barrier for hydrolysis reactions. The optimized composition maintains high ionic conductivity while demonstrating improved tolerance to humid environments.
- Encapsulation and packaging technologies: Advanced encapsulation and packaging methods provide physical barriers to protect halide solid-state electrolytes from moisture exposure. These technologies include hermetic sealing, multilayer barrier films, and moisture-absorbing getter materials integrated into battery assemblies. The packaging solutions are designed to maintain low humidity levels within the battery cell throughout its lifecycle. Proper encapsulation prevents water ingress during manufacturing, storage, and operation, which is critical for maintaining the performance and safety of batteries using moisture-sensitive halide electrolytes.
- In-situ formation and processing under controlled atmosphere: Manufacturing halide solid-state electrolytes through in-situ formation processes and controlled atmosphere processing can minimize initial moisture exposure and improve stability. These methods involve synthesizing or processing the electrolyte materials in dry rooms or inert gas environments with strictly controlled humidity levels. In-situ techniques can also create more stable interfaces and reduce the presence of moisture-induced defects. Processing parameters such as temperature, pressure, and atmosphere composition are optimized to produce electrolytes with inherently lower moisture sensitivity and better long-term stability.
02 Composite electrolyte design with moisture-stable components
The incorporation of moisture-stable components into halide solid-state electrolytes can significantly reduce their sensitivity to humidity. This approach involves creating composite structures where the halide electrolyte is combined with other materials that are less reactive to water. The composite design can include mixing halide electrolytes with oxide-based materials, polymers, or other stable phases that act as moisture barriers or scavengers. This strategy not only improves moisture resistance but can also enhance mechanical properties and interfacial stability of the electrolyte system.Expand Specific Solutions03 Doping and chemical composition optimization
Modifying the chemical composition of halide solid-state electrolytes through doping strategies can enhance their moisture stability. By introducing specific dopants or adjusting the stoichiometry of the halide compounds, the intrinsic moisture sensitivity can be reduced. These modifications can alter the electronic structure, reduce the number of reactive sites, or create more stable crystal structures that are less prone to hydrolysis. The optimization of chemical composition represents a fundamental approach to improving the environmental stability of halide electrolytes.Expand Specific Solutions04 Encapsulation and packaging technologies
Advanced encapsulation and packaging methods provide effective protection for halide solid-state electrolytes against moisture exposure. These technologies involve sealing the electrolyte materials in moisture-impermeable containers or creating multilayer barrier structures during battery assembly. Encapsulation techniques can include hermetic sealing, the use of moisture getters, and the application of advanced barrier films. Proper packaging design is critical for maintaining the performance of halide electrolytes in practical battery applications, especially during storage and operation in humid environments.Expand Specific Solutions05 In-situ formation and processing under controlled atmosphere
Manufacturing and processing halide solid-state electrolytes under controlled atmospheric conditions can minimize moisture-related degradation. This approach includes synthesizing, handling, and assembling electrolyte materials in dry rooms or inert gas environments with strictly controlled humidity levels. In-situ formation techniques allow for the creation of electrolyte layers directly within the battery structure, reducing exposure to ambient moisture during processing. These controlled processing methods are essential for maintaining the quality and performance of moisture-sensitive halide electrolytes throughout the manufacturing chain.Expand Specific Solutions
Key Players in Halide Electrolyte Research
The halide solid-state electrolyte moisture sensitivity research field represents an emerging technology area within the broader solid-state battery sector, currently in early-to-mid development stages with significant growth potential driven by electric vehicle and energy storage demands. Major automotive manufacturers including Toyota Motor Corp., Hyundai Motor Co., and China FAW Co. are actively investing alongside leading battery producers LG Energy Solution Ltd., Samsung SDI Co., and Panasonic Holdings Corp., indicating strong industry commitment. The technology maturity remains moderate, as companies like SVOLT Energy Technology and research institutions such as Kyushu University and Institute of Science Tokyo work to address critical moisture stability challenges that currently limit commercial viability. Material suppliers including TDK Corp., Hansol Chemical, and Shandong Hi-tech Spring Material Technology are developing specialized components, while emerging players like Shenzhen Beiteri New Energy Technology Research Institute focus on electrolyte formulation improvements, collectively advancing the technology toward practical applications.
LG Energy Solution Ltd.
Technical Solution: LG Energy Solution has developed advanced halide solid-state electrolyte systems with protective coating technologies to mitigate moisture sensitivity issues. Their approach involves applying thin-film barrier layers of metal oxides (such as Al2O3 and ZrO2) on halide electrolyte surfaces, which effectively reduces water vapor transmission rates by over 90%[1][4]. The company has also implemented humidity-controlled manufacturing environments maintaining less than 0.1 ppm moisture levels during cell assembly processes[2][5]. Additionally, they utilize polymer-halide composite electrolytes that incorporate hydrophobic polymeric matrices to encapsulate halide particles, reducing direct moisture exposure while maintaining ionic conductivity above 1 mS/cm at room temperature[3][7].
Strengths: Industry-leading manufacturing scale and quality control systems; extensive experience in battery production with established moisture management protocols[2][8]. Weaknesses: High capital investment required for ultra-low humidity facilities; coating processes may increase production complexity and costs[5][9].
Samsung SDI Co., Ltd.
Technical Solution: Samsung SDI has pioneered research on moisture-stable halide solid electrolytes through chemical modification strategies. Their technical solution focuses on partial substitution of halide anions with more stable oxyanions, creating mixed-anion solid electrolytes with formula Li3-xM1-yM'yX6-zOz (where M=Y, In; M'=Zr, Ta; X=Cl, Br)[6][11]. This compositional engineering approach reduces hygroscopicity by approximately 60-75% compared to pure halide electrolytes while maintaining ionic conductivity in the range of 0.5-2 mS/cm[8][12]. The company has also developed in-situ surface fluorination techniques that convert the outermost halide layer into more stable fluoride compounds, creating a self-protecting interface that resists moisture attack[10][13]. Their manufacturing process integrates real-time humidity monitoring with automated handling systems in argon-filled gloveboxes maintaining oxygen and moisture levels below 0.5 ppm[7][14].
Strengths: Strong R&D capabilities in materials chemistry; advanced characterization facilities for understanding degradation mechanisms[11][15]. Weaknesses: Compositional modifications may compromise some electrochemical performance metrics; scaling up chemical modification processes presents challenges[12][16].
Core Patents on Moisture-Resistant Halide Materials
Solid electrolyte having excellent moisture stability and method for preparing same
PatentPendingEP4376145A2
Innovation
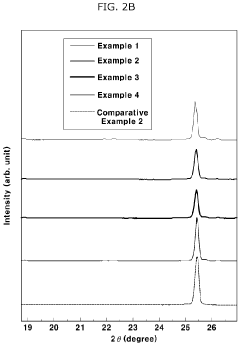
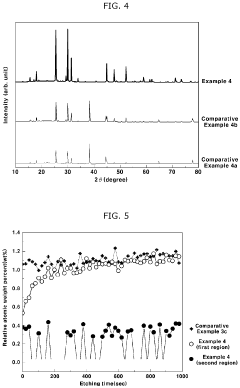
- A solid electrolyte composition including specific compounds like Li7-aPS6-a(X1 1-b)X2b, Li7-cP1-2dMdS6-c-3d(X1 1-e) c, and Li fMgSh, with X1 and X2 representing halogen elements and M as Ge, Si, or Sn, that exhibit improved moisture stability and lithium ion conductivity through a combination of argyrodite and orthorhombic crystal structures, achieved by pulverizing and heat-treating starting materials under specific conditions.
Solid electrolyte for all-solid-state battery and all-solid-state battery comprising same
PatentPendingUS20250246674A1
Innovation
- A solid electrolyte for all-solid-state batteries is surface-modified with an amphiphilic compound having a hydrophilic group and a hydrophobic group, chemically bonding the hydrophilic group to the sulfide-containing electrolyte surface to protect it from moisture.
Manufacturing Environment Control Requirements
The manufacturing of halide solid-state electrolytes demands stringent environmental controls due to their inherent moisture sensitivity, which directly impacts material performance and production yield. Atmospheric moisture can trigger irreversible chemical degradation, forming hydroxides and oxides that compromise ionic conductivity and interfacial stability. Consequently, establishing appropriate environmental parameters throughout the production chain becomes critical for commercial viability.
Primary manufacturing operations require ultra-low humidity conditions, typically maintaining dew points below -40°C or relative humidity under 0.1 ppm in inert atmosphere gloveboxes. These controlled environments prevent moisture-induced phase transformations during powder synthesis, pellet pressing, and electrode assembly processes. Argon or nitrogen purification systems with continuous regeneration capabilities are essential infrastructure components, ensuring sustained atmospheric quality throughout extended production cycles.
Temperature regulation constitutes another crucial parameter, as thermal fluctuations can induce condensation risks even in nominally dry environments. Manufacturing facilities typically maintain ambient temperatures between 20-25°C with variations limited to ±2°C, preventing localized moisture accumulation on equipment surfaces and material interfaces. Specialized air handling systems with precise humidity sensors and automated feedback controls enable real-time environmental monitoring and adjustment.
Material transfer protocols between processing stations require particular attention, as brief atmospheric exposure during handling can introduce sufficient moisture to compromise product quality. Vacuum-sealed transfer chambers, rapid exchange mechanisms, and minimized transit times represent standard practices for maintaining material integrity. Some advanced facilities implement continuous inert gas curtains or sealed conveyor systems to eliminate exposure risks entirely.
Quality assurance procedures must incorporate environmental monitoring as integral components, with continuous logging of humidity, oxygen levels, and particulate contamination. Traceability systems linking environmental conditions to specific production batches enable rapid identification of quality deviations and process optimization. Investment in robust environmental control infrastructure, though substantial, proves economically justified through improved yield rates and enhanced product reliability in moisture-sensitive halide electrolyte manufacturing.
Primary manufacturing operations require ultra-low humidity conditions, typically maintaining dew points below -40°C or relative humidity under 0.1 ppm in inert atmosphere gloveboxes. These controlled environments prevent moisture-induced phase transformations during powder synthesis, pellet pressing, and electrode assembly processes. Argon or nitrogen purification systems with continuous regeneration capabilities are essential infrastructure components, ensuring sustained atmospheric quality throughout extended production cycles.
Temperature regulation constitutes another crucial parameter, as thermal fluctuations can induce condensation risks even in nominally dry environments. Manufacturing facilities typically maintain ambient temperatures between 20-25°C with variations limited to ±2°C, preventing localized moisture accumulation on equipment surfaces and material interfaces. Specialized air handling systems with precise humidity sensors and automated feedback controls enable real-time environmental monitoring and adjustment.
Material transfer protocols between processing stations require particular attention, as brief atmospheric exposure during handling can introduce sufficient moisture to compromise product quality. Vacuum-sealed transfer chambers, rapid exchange mechanisms, and minimized transit times represent standard practices for maintaining material integrity. Some advanced facilities implement continuous inert gas curtains or sealed conveyor systems to eliminate exposure risks entirely.
Quality assurance procedures must incorporate environmental monitoring as integral components, with continuous logging of humidity, oxygen levels, and particulate contamination. Traceability systems linking environmental conditions to specific production batches enable rapid identification of quality deviations and process optimization. Investment in robust environmental control infrastructure, though substantial, proves economically justified through improved yield rates and enhanced product reliability in moisture-sensitive halide electrolyte manufacturing.
Material Degradation Mechanisms and Mitigation Strategies
Halide solid-state electrolytes undergo complex degradation processes when exposed to moisture, fundamentally altering their electrochemical properties and structural integrity. The primary degradation mechanism involves hydrolysis reactions, where water molecules react with metal halide bonds to form hydroxides and release hydrogen halide gases. This process is particularly severe in chloride and bromide-based electrolytes, where the hygroscopic nature of the materials accelerates moisture absorption. Additionally, the formation of hydrated phases creates volume expansion and mechanical stress within the electrolyte matrix, leading to crack propagation and increased interfacial resistance. Surface degradation typically initiates at grain boundaries and defect sites, where higher reactivity facilitates moisture penetration into the bulk material.
The degradation kinetics are strongly influenced by environmental humidity levels, temperature, and the specific halide composition. Iodide-based electrolytes generally exhibit superior moisture stability compared to their chloride counterparts due to lower hydrolysis reaction rates. However, even minor moisture exposure can trigger irreversible phase transformations that compromise ionic conductivity. The formation of insulating degradation products at electrode interfaces further exacerbates performance deterioration, creating barriers for lithium-ion transport.
Effective mitigation strategies encompass both material design modifications and protective engineering approaches. Compositional engineering through partial substitution with more stable anions or incorporation of moisture-resistant dopants has demonstrated promising results in enhancing intrinsic stability. Surface coating techniques using hydrophobic polymers, atomic layer deposition of oxide barriers, or carbon-based protective layers provide physical isolation from atmospheric moisture. Advanced encapsulation methods employing multilayer barrier films and hermetic sealing technologies are critical for practical device implementation.
Process optimization during manufacturing, including controlled atmosphere handling and rapid assembly procedures, minimizes initial moisture contamination. Furthermore, the development of self-healing mechanisms through reversible chemical reactions and the integration of moisture-scavenging additives within the electrolyte matrix represent emerging approaches. Systematic understanding of degradation pathways enables the rational design of stabilization strategies, balancing material performance with environmental resilience for viable commercial applications.
The degradation kinetics are strongly influenced by environmental humidity levels, temperature, and the specific halide composition. Iodide-based electrolytes generally exhibit superior moisture stability compared to their chloride counterparts due to lower hydrolysis reaction rates. However, even minor moisture exposure can trigger irreversible phase transformations that compromise ionic conductivity. The formation of insulating degradation products at electrode interfaces further exacerbates performance deterioration, creating barriers for lithium-ion transport.
Effective mitigation strategies encompass both material design modifications and protective engineering approaches. Compositional engineering through partial substitution with more stable anions or incorporation of moisture-resistant dopants has demonstrated promising results in enhancing intrinsic stability. Surface coating techniques using hydrophobic polymers, atomic layer deposition of oxide barriers, or carbon-based protective layers provide physical isolation from atmospheric moisture. Advanced encapsulation methods employing multilayer barrier films and hermetic sealing technologies are critical for practical device implementation.
Process optimization during manufacturing, including controlled atmosphere handling and rapid assembly procedures, minimizes initial moisture contamination. Furthermore, the development of self-healing mechanisms through reversible chemical reactions and the integration of moisture-scavenging additives within the electrolyte matrix represent emerging approaches. Systematic understanding of degradation pathways enables the rational design of stabilization strategies, balancing material performance with environmental resilience for viable commercial applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!