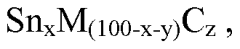Anode material selection for solid-state sodium-ion batteries
FEB 11, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Solid-State Sodium-Ion Anode Development Background and Objectives
Solid-state sodium-ion batteries have emerged as a promising alternative to lithium-ion technology, driven by the abundance and widespread distribution of sodium resources compared to lithium. The development of suitable anode materials represents a critical challenge in advancing this technology from laboratory research to commercial viability. Unlike conventional liquid electrolyte systems, solid-state configurations demand anode materials that can maintain stable interfaces with solid electrolytes while accommodating the volumetric changes associated with sodium insertion and extraction.
The historical evolution of sodium-ion battery technology dates back to the 1980s, when initial research paralleled lithium-ion development. However, the larger ionic radius of sodium compared to lithium presented fundamental challenges in host material design and interfacial stability. The resurgence of interest in sodium-ion systems over the past decade has been catalyzed by concerns over lithium supply chain constraints and the need for cost-effective energy storage solutions for grid-scale applications. The transition to solid-state architectures represents the latest phase in this evolution, aiming to overcome safety limitations and energy density constraints inherent in liquid electrolyte systems.
Current development objectives center on identifying anode materials that satisfy multiple stringent requirements simultaneously. These materials must demonstrate high sodium storage capacity, excellent rate capability, and long-term cycling stability while maintaining chemical and electrochemical compatibility with solid electrolytes. The interfacial resistance between anode and electrolyte remains a primary technical barrier, as does the mechanical stress induced by volume expansion during sodiation cycles.
The strategic importance of this research extends beyond performance metrics to encompass sustainability considerations. Ideal anode materials should utilize earth-abundant elements, enable simplified manufacturing processes, and support the broader goal of developing environmentally responsible energy storage systems. The technology roadmap envisions anode solutions that can operate across wide temperature ranges, withstand high current densities, and achieve cycle lives exceeding ten thousand charge-discharge cycles. These ambitious targets reflect the demanding requirements of both automotive and stationary energy storage applications, where solid-state sodium-ion batteries could provide transformative benefits in safety, cost-effectiveness, and resource sustainability.
The historical evolution of sodium-ion battery technology dates back to the 1980s, when initial research paralleled lithium-ion development. However, the larger ionic radius of sodium compared to lithium presented fundamental challenges in host material design and interfacial stability. The resurgence of interest in sodium-ion systems over the past decade has been catalyzed by concerns over lithium supply chain constraints and the need for cost-effective energy storage solutions for grid-scale applications. The transition to solid-state architectures represents the latest phase in this evolution, aiming to overcome safety limitations and energy density constraints inherent in liquid electrolyte systems.
Current development objectives center on identifying anode materials that satisfy multiple stringent requirements simultaneously. These materials must demonstrate high sodium storage capacity, excellent rate capability, and long-term cycling stability while maintaining chemical and electrochemical compatibility with solid electrolytes. The interfacial resistance between anode and electrolyte remains a primary technical barrier, as does the mechanical stress induced by volume expansion during sodiation cycles.
The strategic importance of this research extends beyond performance metrics to encompass sustainability considerations. Ideal anode materials should utilize earth-abundant elements, enable simplified manufacturing processes, and support the broader goal of developing environmentally responsible energy storage systems. The technology roadmap envisions anode solutions that can operate across wide temperature ranges, withstand high current densities, and achieve cycle lives exceeding ten thousand charge-discharge cycles. These ambitious targets reflect the demanding requirements of both automotive and stationary energy storage applications, where solid-state sodium-ion batteries could provide transformative benefits in safety, cost-effectiveness, and resource sustainability.
Market Demand for Sodium-Ion Battery Applications
The global energy storage market is experiencing a fundamental shift driven by the urgent need for sustainable and cost-effective battery technologies. Sodium-ion batteries have emerged as a compelling alternative to lithium-ion systems, particularly in applications where cost, safety, and resource availability are critical considerations. The market demand for sodium-ion battery applications is expanding across multiple sectors, with stationary energy storage, electric vehicles, and consumer electronics representing the primary growth areas.
Stationary energy storage systems constitute the most immediate and substantial market opportunity for sodium-ion batteries. Grid-scale energy storage facilities require massive battery deployments to support renewable energy integration and load balancing. The abundance of sodium resources and lower material costs make sodium-ion technology particularly attractive for these large-scale installations where energy density requirements are less stringent than mobile applications. Utility companies and renewable energy operators are increasingly evaluating sodium-ion solutions as economically viable alternatives for peak shaving, frequency regulation, and backup power systems.
The electric vehicle sector presents a differentiated market opportunity, particularly in the entry-level and urban mobility segments. While lithium-ion batteries dominate high-performance electric vehicles, sodium-ion batteries are gaining traction in low-speed electric vehicles, electric bicycles, and short-range urban transportation. Manufacturers in emerging markets are especially interested in sodium-ion technology due to its potential to reduce battery costs significantly, making electric mobility more accessible to broader consumer segments. The technology's superior low-temperature performance and enhanced safety characteristics further strengthen its appeal for specific vehicle categories.
Consumer electronics and portable power applications represent an emerging demand segment. Although energy density limitations currently restrict sodium-ion batteries from competing directly with lithium-ion in smartphones and laptops, opportunities exist in power tools, backup power supplies, and less weight-sensitive devices. The growing emphasis on supply chain diversification and reduced dependence on lithium resources is prompting electronics manufacturers to explore sodium-ion alternatives for specific product lines.
Industrial applications including telecommunications base stations, uninterruptible power supplies, and material handling equipment are demonstrating increasing interest in sodium-ion technology. These applications prioritize reliability, safety, and total cost of ownership over maximum energy density, creating favorable conditions for sodium-ion adoption. The technology's extended cycle life and robust performance across varying environmental conditions align well with industrial operational requirements.
Stationary energy storage systems constitute the most immediate and substantial market opportunity for sodium-ion batteries. Grid-scale energy storage facilities require massive battery deployments to support renewable energy integration and load balancing. The abundance of sodium resources and lower material costs make sodium-ion technology particularly attractive for these large-scale installations where energy density requirements are less stringent than mobile applications. Utility companies and renewable energy operators are increasingly evaluating sodium-ion solutions as economically viable alternatives for peak shaving, frequency regulation, and backup power systems.
The electric vehicle sector presents a differentiated market opportunity, particularly in the entry-level and urban mobility segments. While lithium-ion batteries dominate high-performance electric vehicles, sodium-ion batteries are gaining traction in low-speed electric vehicles, electric bicycles, and short-range urban transportation. Manufacturers in emerging markets are especially interested in sodium-ion technology due to its potential to reduce battery costs significantly, making electric mobility more accessible to broader consumer segments. The technology's superior low-temperature performance and enhanced safety characteristics further strengthen its appeal for specific vehicle categories.
Consumer electronics and portable power applications represent an emerging demand segment. Although energy density limitations currently restrict sodium-ion batteries from competing directly with lithium-ion in smartphones and laptops, opportunities exist in power tools, backup power supplies, and less weight-sensitive devices. The growing emphasis on supply chain diversification and reduced dependence on lithium resources is prompting electronics manufacturers to explore sodium-ion alternatives for specific product lines.
Industrial applications including telecommunications base stations, uninterruptible power supplies, and material handling equipment are demonstrating increasing interest in sodium-ion technology. These applications prioritize reliability, safety, and total cost of ownership over maximum energy density, creating favorable conditions for sodium-ion adoption. The technology's extended cycle life and robust performance across varying environmental conditions align well with industrial operational requirements.
Current Anode Material Challenges in Solid-State Sodium Batteries
Solid-state sodium-ion batteries represent a promising alternative to lithium-based systems, yet the selection and implementation of suitable anode materials face significant technical obstacles that currently limit their commercial viability. The primary challenge stems from the inherently larger ionic radius of sodium ions compared to lithium ions, which creates substantial difficulties in achieving efficient ion insertion and extraction processes within anode structures.
Volume expansion during sodiation cycles poses one of the most critical challenges for anode materials in solid-state configurations. Hard carbon and other carbonaceous materials, while demonstrating reasonable sodium storage capacity, experience considerable structural strain during repeated charge-discharge cycles. This mechanical stress becomes particularly problematic at the solid-solid interface between the anode and solid electrolyte, where maintaining intimate contact is essential for sustained electrochemical performance. The volume changes can lead to interfacial delamination and increased resistance over time.
Interface compatibility between anode materials and solid electrolytes presents another fundamental constraint. Many promising anode candidates exhibit chemical or electrochemical instability when in direct contact with solid electrolytes, particularly sulfide-based systems. Side reactions at these interfaces result in the formation of resistive interphases that impede sodium-ion transport and accelerate capacity degradation. The challenge intensifies with metallic sodium anodes, which theoretically offer the highest energy density but suffer from dendrite formation and severe interfacial reactions.
The limited electronic and ionic conductivity of many anode materials further restricts performance capabilities. Materials such as titanium-based oxides and certain alloy-type anodes demonstrate inadequate rate performance due to sluggish sodium-ion diffusion kinetics within their crystal structures. This limitation becomes more pronounced in solid-state configurations where the absence of liquid electrolytes eliminates the buffering effect that typically facilitates ion transport in conventional battery systems.
Manufacturing and scalability challenges compound these technical issues. Achieving uniform contact between rigid solid electrolytes and anode materials requires high-pressure processing or elevated temperatures, which increase production costs and complexity. The brittleness of many solid electrolytes makes it difficult to accommodate the mechanical stresses generated by anode volume changes without compromising structural integrity. These factors collectively represent the key technical barriers that must be addressed to advance solid-state sodium-ion battery technology toward practical applications.
Volume expansion during sodiation cycles poses one of the most critical challenges for anode materials in solid-state configurations. Hard carbon and other carbonaceous materials, while demonstrating reasonable sodium storage capacity, experience considerable structural strain during repeated charge-discharge cycles. This mechanical stress becomes particularly problematic at the solid-solid interface between the anode and solid electrolyte, where maintaining intimate contact is essential for sustained electrochemical performance. The volume changes can lead to interfacial delamination and increased resistance over time.
Interface compatibility between anode materials and solid electrolytes presents another fundamental constraint. Many promising anode candidates exhibit chemical or electrochemical instability when in direct contact with solid electrolytes, particularly sulfide-based systems. Side reactions at these interfaces result in the formation of resistive interphases that impede sodium-ion transport and accelerate capacity degradation. The challenge intensifies with metallic sodium anodes, which theoretically offer the highest energy density but suffer from dendrite formation and severe interfacial reactions.
The limited electronic and ionic conductivity of many anode materials further restricts performance capabilities. Materials such as titanium-based oxides and certain alloy-type anodes demonstrate inadequate rate performance due to sluggish sodium-ion diffusion kinetics within their crystal structures. This limitation becomes more pronounced in solid-state configurations where the absence of liquid electrolytes eliminates the buffering effect that typically facilitates ion transport in conventional battery systems.
Manufacturing and scalability challenges compound these technical issues. Achieving uniform contact between rigid solid electrolytes and anode materials requires high-pressure processing or elevated temperatures, which increase production costs and complexity. The brittleness of many solid electrolytes makes it difficult to accommodate the mechanical stresses generated by anode volume changes without compromising structural integrity. These factors collectively represent the key technical barriers that must be addressed to advance solid-state sodium-ion battery technology toward practical applications.
Mainstream Anode Material Solutions and Strategies
01 Carbon-based anode materials for solid-state sodium-ion batteries
Carbon-based materials, including hard carbon, soft carbon, and graphite derivatives, are widely used as anode materials in solid-state sodium-ion batteries. These materials offer good electrical conductivity, structural stability, and the ability to accommodate sodium ion insertion and extraction. The carbon structure can be modified through various treatments to optimize porosity, surface area, and interlayer spacing, which enhances sodium storage capacity and cycling performance. These materials provide a cost-effective solution with reasonable electrochemical performance for sodium-ion battery applications.- Carbon-based anode materials for solid-state sodium-ion batteries: Carbon-based materials, including hard carbon, soft carbon, and graphite derivatives, are widely used as anode materials in solid-state sodium-ion batteries. These materials offer good electrical conductivity, structural stability, and the ability to intercalate sodium ions. The carbon structure can be modified through various treatments to optimize sodium storage capacity and cycling performance. Surface modifications and doping strategies are employed to enhance the electrochemical properties and reduce volume expansion during charge-discharge cycles.
- Metal oxide and metal compound-based anode materials: Metal oxides and metal compounds serve as promising anode materials for solid-state sodium-ion batteries due to their high theoretical capacity. These materials undergo conversion or alloying reactions with sodium ions during the electrochemical process. Various transition metal oxides and their composites demonstrate excellent sodium storage capabilities. The materials can be engineered in different morphologies and structures to improve ion diffusion pathways and electronic conductivity, thereby enhancing overall battery performance.
- Composite and hybrid anode materials: Composite anode materials combine multiple components to leverage the advantages of different materials while mitigating their individual limitations. These hybrid structures typically integrate carbon materials with metal compounds or other active materials to achieve improved conductivity, capacity, and structural stability. The synergistic effects between components enhance sodium ion storage and transport properties. Such composite designs help address issues like volume expansion and poor cycling stability commonly encountered in single-component anodes.
- Nanostructured and porous anode materials: Nanostructured anode materials with controlled morphologies, such as nanoparticles, nanowires, nanotubes, and porous structures, offer enhanced electrochemical performance for solid-state sodium-ion batteries. The nanoscale dimensions provide shortened ion diffusion paths and increased electrode-electrolyte contact area. Porous architectures facilitate electrolyte penetration and accommodate volume changes during cycling. These structural features contribute to improved rate capability, higher capacity retention, and better long-term cycling stability.
- Surface modification and coating strategies for anode materials: Surface modification techniques and protective coatings are applied to anode materials to enhance their compatibility with solid-state electrolytes and improve overall battery performance. These strategies include the application of conductive layers, protective films, and interface engineering to reduce interfacial resistance and prevent unwanted side reactions. Surface treatments help stabilize the solid-electrolyte interface, suppress dendrite formation, and improve sodium ion transport kinetics. Such modifications are crucial for achieving stable long-term cycling and high coulombic efficiency in solid-state sodium-ion batteries.
02 Metal oxide and composite anode materials
Metal oxides and their composites serve as promising anode materials for solid-state sodium-ion batteries due to their high theoretical capacity and diverse structural configurations. These materials include transition metal oxides and their combinations with carbon or other conductive matrices. The composite structure helps to improve electrical conductivity, buffer volume expansion during cycling, and enhance overall electrochemical stability. Surface modifications and nanostructuring techniques are employed to optimize the performance of these oxide-based anodes, resulting in improved capacity retention and rate capability.Expand Specific Solutions03 Alloy-based anode materials for sodium storage
Alloy-based materials that can form compounds with sodium through alloying reactions are utilized as high-capacity anode materials. These materials typically include elements that can reversibly react with sodium ions, offering significantly higher theoretical capacities compared to intercalation-type materials. However, they often experience substantial volume changes during sodiation and desodiation processes. To address this challenge, various strategies such as nanostructuring, composite formation with conductive matrices, and structural design are implemented to accommodate volume expansion and maintain structural integrity during cycling.Expand Specific Solutions04 Layered and intercalation-type anode materials
Layered structures and intercalation-type materials provide stable frameworks for reversible sodium ion insertion and extraction in solid-state batteries. These materials feature well-defined crystal structures with suitable interlayer spacing that facilitates sodium ion diffusion. The layered architecture allows for structural stability during repeated charge-discharge cycles while maintaining good ionic conductivity. Various modifications to the layered structure, including doping and surface treatment, are employed to enhance sodium ion mobility and improve the overall electrochemical performance of the anode.Expand Specific Solutions05 Nanostructured and surface-modified anode materials
Nanostructuring and surface modification techniques are critical approaches to enhance the performance of anode materials in solid-state sodium-ion batteries. These methods include creating nanoparticles, nanowires, nanotubes, or porous structures that provide increased surface area, shortened ion diffusion pathways, and improved contact with solid electrolytes. Surface treatments such as coating with conductive layers, functionalization, or interface engineering help to stabilize the electrode-electrolyte interface, reduce interfacial resistance, and prevent unwanted side reactions. These modifications result in improved rate capability, cycling stability, and overall battery performance.Expand Specific Solutions
Key Players in Solid-State Sodium Battery Development
The solid-state sodium-ion battery anode material sector represents an emerging technology landscape at the early commercialization stage, with market development driven by cost advantages over lithium alternatives and growing energy storage demands. The competitive arena encompasses diverse players spanning automotive giants like Toyota Motor Corp., BMW, and Ford Global Technologies LLC exploring next-generation battery integration, established electronics manufacturers including Samsung Electronics and Sharp Corp. advancing material innovations, and specialized battery material developers such as Honeycomb Battery Co., Beijing Zhongke Haina Technology, and Shenzhen Jiana Energy Technology focusing on sodium-ion specific solutions. Academic institutions like Central South University, Shandong University, and Indian Institute of Technology Bombay contribute fundamental research, while recycling specialists Guangdong Bangpu and Hunan Bangpu address sustainability. Technology maturity varies significantly across participants, with most entities in pilot-scale development phases as they optimize anode compositions, cycling stability, and manufacturing scalability for commercial viability.
Bayerische Motoren Werke AG
Technical Solution: BMW has developed solid-state sodium-ion battery technology focusing on hard carbon and sodium metal composite anodes for automotive applications. Their approach integrates pre-sodiation techniques to enhance initial coulombic efficiency and cycle stability. The anode material strategy emphasizes cost-effective hard carbon derived from biomass sources, combined with surface modification layers to improve interfacial compatibility with solid electrolytes. BMW's research prioritizes scalability and manufacturing feasibility, targeting energy densities of 150-200 Wh/kg for next-generation electric vehicle platforms. Their anode design incorporates protective coatings to mitigate dendrite formation and volume expansion issues inherent in sodium metal systems.
Strengths: Strong automotive integration expertise, focus on manufacturing scalability, robust safety protocols. Weaknesses: Lower energy density compared to lithium systems, early-stage commercialization, limited public technical disclosure.
Toyota Motor Corp.
Technical Solution: Toyota's solid-state sodium-ion battery anode strategy centers on sulfide-based solid electrolyte compatibility with layered hard carbon anodes. Their proprietary technology employs gradient-structured carbon materials with controlled porosity to optimize sodium-ion insertion kinetics and minimize irreversible capacity loss. Toyota has developed advanced binder systems and conductive additives specifically designed for solid-state configurations, achieving cycle life exceeding 1000 cycles in prototype cells. The company's anode research emphasizes thermal stability and abuse tolerance, critical for automotive safety standards. Their material selection process prioritizes domestic resource availability and supply chain independence from lithium-based systems.
Strengths: Extensive solid-state battery R&D experience, superior manufacturing capabilities, strong patent portfolio. Weaknesses: Conservative commercialization timeline, higher material processing costs, performance gap versus lithium solid-state batteries.
Core Patents in Sodium Anode Interface Engineering
Anode compositions for sodium-ion batteries and methods of making same
PatentWO2014081786A1
Innovation
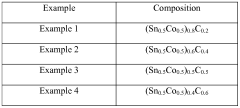
- Development of an electrochemically active anode material comprising alloy particles with an electrochemically inactive phase and nanocrystalline or amorphous structure, free of crystalline grains greater than 40 nm, to limit volume expansion and enhance cycle life.
Anode material for a solid-state battery and solid-state battery
PatentPendingDE102021121349A1
Innovation
- An anode material comprising secondary particles with a porous matrix and primary particles that serve as a growth surface for lithium, surrounded by an ion-conducting protective layer, allowing lithium ions to penetrate while preventing decomposition and mechanical stress.
Resource Availability and Sustainability of Sodium Anodes
Sodium's exceptional abundance in the Earth's crust, estimated at approximately 2.6% by weight, positions it as one of the most accessible metallic elements for large-scale battery applications. Unlike lithium, which is predominantly concentrated in specific geographical regions such as South America's lithium triangle and certain areas of Australia, sodium resources are widely distributed across all continents. This geographical diversity significantly reduces supply chain vulnerabilities and geopolitical dependencies that currently challenge lithium-ion battery production. Sodium can be extracted from various sources including seawater, salt lakes, and mineral deposits, with extraction processes generally requiring less energy and generating lower environmental impact compared to lithium mining operations.
The sustainability profile of sodium anodes extends beyond mere resource availability to encompass environmental and economic considerations. Sodium extraction from seawater and brine sources presents minimal ecological disruption, as these processes do not require extensive land excavation or habitat destruction. The abundance of sodium translates directly into cost advantages, with sodium compounds typically priced at a fraction of their lithium equivalents. This economic accessibility makes solid-state sodium-ion batteries particularly attractive for grid-scale energy storage applications where cost per kilowatt-hour remains a critical factor.
From a circular economy perspective, sodium-based battery systems demonstrate promising recyclability characteristics. The recovery and reprocessing of sodium materials require less complex separation techniques compared to lithium systems, potentially enabling more efficient closed-loop recycling processes. The lower toxicity of sodium compounds relative to certain lithium-based materials further enhances the environmental credentials of sodium anode systems. Additionally, the manufacturing infrastructure for sodium-based materials can leverage existing industrial processes from the chlor-alkali industry, reducing the need for entirely new production facilities and associated capital investments.
The long-term sustainability of sodium anodes is further reinforced by their compatibility with abundant supporting materials. Electrolytes, current collectors, and other battery components for sodium systems can utilize more readily available and less expensive materials, contributing to an overall reduction in resource pressure across the entire battery value chain.
The sustainability profile of sodium anodes extends beyond mere resource availability to encompass environmental and economic considerations. Sodium extraction from seawater and brine sources presents minimal ecological disruption, as these processes do not require extensive land excavation or habitat destruction. The abundance of sodium translates directly into cost advantages, with sodium compounds typically priced at a fraction of their lithium equivalents. This economic accessibility makes solid-state sodium-ion batteries particularly attractive for grid-scale energy storage applications where cost per kilowatt-hour remains a critical factor.
From a circular economy perspective, sodium-based battery systems demonstrate promising recyclability characteristics. The recovery and reprocessing of sodium materials require less complex separation techniques compared to lithium systems, potentially enabling more efficient closed-loop recycling processes. The lower toxicity of sodium compounds relative to certain lithium-based materials further enhances the environmental credentials of sodium anode systems. Additionally, the manufacturing infrastructure for sodium-based materials can leverage existing industrial processes from the chlor-alkali industry, reducing the need for entirely new production facilities and associated capital investments.
The long-term sustainability of sodium anodes is further reinforced by their compatibility with abundant supporting materials. Electrolytes, current collectors, and other battery components for sodium systems can utilize more readily available and less expensive materials, contributing to an overall reduction in resource pressure across the entire battery value chain.
Manufacturing Scalability of Solid-State Sodium Anodes
Manufacturing scalability represents a critical bottleneck in transitioning solid-state sodium anode technologies from laboratory demonstrations to commercial production. The fabrication of solid-state sodium-ion batteries with metallic sodium or sodium alloy anodes involves processes fundamentally different from conventional lithium-ion battery manufacturing, requiring substantial modifications to existing production infrastructure. Current pilot-scale production faces challenges in achieving uniform electrode-electrolyte interfaces, maintaining moisture-free environments during assembly, and ensuring consistent material quality across large-format cells.
The primary manufacturing challenge stems from sodium metal's high reactivity and low melting point, necessitating stringent atmospheric control throughout the production chain. Roll-to-roll processing techniques, widely adopted in conventional battery manufacturing, require adaptation to accommodate the mechanical properties of solid electrolytes and the chemical sensitivity of sodium anodes. Dry room specifications must be significantly more rigorous than those for lithium-ion production, with dew points below -60°C to prevent sodium degradation and interfacial reactions.
Electrode fabrication methods currently under development include vapor deposition, mechanical pressing, and in-situ formation approaches. Each method presents distinct scalability considerations regarding throughput, material utilization efficiency, and capital equipment requirements. Vapor deposition offers excellent interface quality but suffers from low deposition rates and high equipment costs. Mechanical pressing methods provide better throughput but face challenges in achieving uniform pressure distribution across large electrode areas while maintaining interface integrity.
Quality control and process monitoring present additional complexities in scaled manufacturing. Non-destructive testing methods capable of detecting interfacial defects, electrolyte cracks, and sodium distribution inhomogeneities must be integrated into production lines without compromising throughput. The development of automated inspection systems compatible with inert atmosphere requirements remains an active area of industrial research.
Cost modeling studies indicate that achieving price parity with conventional sodium-ion batteries requires production volumes exceeding several hundred megawatt-hours annually, necessitating significant upfront capital investment in specialized equipment and facility infrastructure. Supply chain establishment for high-purity solid electrolyte materials and sodium metal represents another critical factor influencing manufacturing scalability and overall production economics.
The primary manufacturing challenge stems from sodium metal's high reactivity and low melting point, necessitating stringent atmospheric control throughout the production chain. Roll-to-roll processing techniques, widely adopted in conventional battery manufacturing, require adaptation to accommodate the mechanical properties of solid electrolytes and the chemical sensitivity of sodium anodes. Dry room specifications must be significantly more rigorous than those for lithium-ion production, with dew points below -60°C to prevent sodium degradation and interfacial reactions.
Electrode fabrication methods currently under development include vapor deposition, mechanical pressing, and in-situ formation approaches. Each method presents distinct scalability considerations regarding throughput, material utilization efficiency, and capital equipment requirements. Vapor deposition offers excellent interface quality but suffers from low deposition rates and high equipment costs. Mechanical pressing methods provide better throughput but face challenges in achieving uniform pressure distribution across large electrode areas while maintaining interface integrity.
Quality control and process monitoring present additional complexities in scaled manufacturing. Non-destructive testing methods capable of detecting interfacial defects, electrolyte cracks, and sodium distribution inhomogeneities must be integrated into production lines without compromising throughput. The development of automated inspection systems compatible with inert atmosphere requirements remains an active area of industrial research.
Cost modeling studies indicate that achieving price parity with conventional sodium-ion batteries requires production volumes exceeding several hundred megawatt-hours annually, necessitating significant upfront capital investment in specialized equipment and facility infrastructure. Supply chain establishment for high-purity solid electrolyte materials and sodium metal represents another critical factor influencing manufacturing scalability and overall production economics.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!