Interface stability issues in solid-state sodium-ion batteries and solutions
FEB 11, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Solid-State Sodium-Ion Battery Interface Stability Background and Objectives
Solid-state sodium-ion batteries have emerged as a promising alternative to lithium-ion technology, driven by sodium's natural abundance, lower cost, and more uniform global distribution. Unlike lithium, sodium resources are virtually unlimited and accessible worldwide, making sodium-based energy storage systems particularly attractive for large-scale applications. The transition from liquid to solid electrolytes addresses critical safety concerns associated with flammable organic solvents while potentially enabling higher energy densities and extended operational lifespans.
However, the development of solid-state sodium-ion batteries faces significant challenges at the electrode-electrolyte interfaces. These interfaces represent critical bottlenecks that directly impact battery performance, cycle life, and overall reliability. The larger ionic radius of sodium compared to lithium creates unique interfacial phenomena, including increased interfacial resistance, chemical incompatibility between electrode materials and solid electrolytes, and mechanical stress accumulation during charge-discharge cycles. These issues manifest as capacity degradation, voltage hysteresis, and premature battery failure.
The interface stability problem encompasses multiple dimensions: electrochemical stability windows that determine the voltage range of safe operation, chemical reactivity between contacting materials leading to interphase formation, mechanical contact degradation due to volume changes during sodium insertion and extraction, and space charge layer effects that impede ion transport. Current research indicates that interfacial resistance can account for over 60% of total cell resistance in some solid-state sodium-ion battery configurations, severely limiting practical applications.
The primary objective of this technical investigation is to comprehensively analyze the root causes of interface instability in solid-state sodium-ion batteries and identify viable solutions. This includes understanding the fundamental mechanisms governing interfacial degradation, evaluating existing mitigation strategies, and exploring innovative approaches to enhance interface stability. The ultimate goal is to provide actionable insights that enable the development of solid-state sodium-ion batteries with improved performance metrics, extended cycle life exceeding 3000 cycles, and reduced interfacial resistance below 100 Ω·cm², thereby accelerating their commercialization for grid-scale energy storage and electric vehicle applications.
However, the development of solid-state sodium-ion batteries faces significant challenges at the electrode-electrolyte interfaces. These interfaces represent critical bottlenecks that directly impact battery performance, cycle life, and overall reliability. The larger ionic radius of sodium compared to lithium creates unique interfacial phenomena, including increased interfacial resistance, chemical incompatibility between electrode materials and solid electrolytes, and mechanical stress accumulation during charge-discharge cycles. These issues manifest as capacity degradation, voltage hysteresis, and premature battery failure.
The interface stability problem encompasses multiple dimensions: electrochemical stability windows that determine the voltage range of safe operation, chemical reactivity between contacting materials leading to interphase formation, mechanical contact degradation due to volume changes during sodium insertion and extraction, and space charge layer effects that impede ion transport. Current research indicates that interfacial resistance can account for over 60% of total cell resistance in some solid-state sodium-ion battery configurations, severely limiting practical applications.
The primary objective of this technical investigation is to comprehensively analyze the root causes of interface instability in solid-state sodium-ion batteries and identify viable solutions. This includes understanding the fundamental mechanisms governing interfacial degradation, evaluating existing mitigation strategies, and exploring innovative approaches to enhance interface stability. The ultimate goal is to provide actionable insights that enable the development of solid-state sodium-ion batteries with improved performance metrics, extended cycle life exceeding 3000 cycles, and reduced interfacial resistance below 100 Ω·cm², thereby accelerating their commercialization for grid-scale energy storage and electric vehicle applications.
Market Demand Analysis for Solid-State Sodium-Ion Batteries
The global energy storage market is undergoing a fundamental transformation driven by the urgent need for sustainable and cost-effective alternatives to lithium-ion technology. Solid-state sodium-ion batteries have emerged as a promising candidate to address critical supply chain vulnerabilities and cost constraints associated with lithium-based systems. The abundance of sodium resources and their uniform global distribution present significant advantages for large-scale deployment, particularly in stationary energy storage applications where weight considerations are less critical than in mobile applications.
Grid-scale energy storage represents the most substantial market opportunity for solid-state sodium-ion batteries. The rapid expansion of renewable energy infrastructure, particularly solar and wind installations, has created unprecedented demand for reliable storage solutions that can balance intermittent power generation. Current market dynamics favor technologies that can deliver long cycle life, enhanced safety profiles, and reduced material costs, all areas where solid-state sodium-ion systems demonstrate competitive advantages over conventional liquid electrolyte batteries.
The electric vehicle sector, while traditionally dominated by lithium-ion technology, is increasingly exploring sodium-ion alternatives for entry-level and urban mobility segments. Manufacturers in emerging markets are particularly interested in sodium-based solutions that can reduce dependency on imported lithium while maintaining acceptable performance metrics. However, interface stability challenges currently limit the energy density and cycle life required for widespread automotive adoption, creating a clear technology gap that must be addressed to unlock this market segment.
Industrial and commercial energy storage applications represent another significant demand driver. Data centers, telecommunications infrastructure, and industrial facilities require backup power systems with high reliability and safety standards. Solid-state configurations offer inherent safety advantages by eliminating flammable liquid electrolytes, making them attractive for applications where thermal management and fire risk mitigation are paramount concerns.
The residential energy storage market is gradually recognizing the potential of sodium-ion technology, particularly in regions with abundant solar resources but limited access to affordable lithium-ion systems. Consumer acceptance will largely depend on resolving interface stability issues that currently compromise long-term performance and warranty viability. Market penetration in this segment requires demonstrated reliability over extended operational periods, typically spanning ten to fifteen years.
Grid-scale energy storage represents the most substantial market opportunity for solid-state sodium-ion batteries. The rapid expansion of renewable energy infrastructure, particularly solar and wind installations, has created unprecedented demand for reliable storage solutions that can balance intermittent power generation. Current market dynamics favor technologies that can deliver long cycle life, enhanced safety profiles, and reduced material costs, all areas where solid-state sodium-ion systems demonstrate competitive advantages over conventional liquid electrolyte batteries.
The electric vehicle sector, while traditionally dominated by lithium-ion technology, is increasingly exploring sodium-ion alternatives for entry-level and urban mobility segments. Manufacturers in emerging markets are particularly interested in sodium-based solutions that can reduce dependency on imported lithium while maintaining acceptable performance metrics. However, interface stability challenges currently limit the energy density and cycle life required for widespread automotive adoption, creating a clear technology gap that must be addressed to unlock this market segment.
Industrial and commercial energy storage applications represent another significant demand driver. Data centers, telecommunications infrastructure, and industrial facilities require backup power systems with high reliability and safety standards. Solid-state configurations offer inherent safety advantages by eliminating flammable liquid electrolytes, making them attractive for applications where thermal management and fire risk mitigation are paramount concerns.
The residential energy storage market is gradually recognizing the potential of sodium-ion technology, particularly in regions with abundant solar resources but limited access to affordable lithium-ion systems. Consumer acceptance will largely depend on resolving interface stability issues that currently compromise long-term performance and warranty viability. Market penetration in this segment requires demonstrated reliability over extended operational periods, typically spanning ten to fifteen years.
Current Interface Stability Challenges in Solid-State Sodium Batteries
Solid-state sodium-ion batteries represent a promising alternative to lithium-based systems, yet their commercial viability remains hindered by critical interface stability challenges. The primary technical obstacles stem from the complex interactions between solid electrolytes and electrode materials, which fundamentally differ from conventional liquid electrolyte systems. These challenges manifest across multiple dimensions, creating significant barriers to achieving reliable long-term performance.
The most pressing issue involves the formation of high interfacial resistance at both cathode and anode interfaces. Solid electrolytes, particularly oxide-based materials like NASICON and sulfide-based compounds, exhibit poor physical contact with electrode particles due to rigid solid-solid interfaces. This contact limitation restricts ionic transport pathways and increases overall cell resistance, directly impacting power density and rate capability. The problem intensifies during cycling as volume changes in electrode materials further deteriorate interfacial contact.
Chemical incompatibility between solid electrolytes and electrode materials constitutes another major challenge. Many solid electrolytes undergo undesirable side reactions with cathode materials at elevated voltages, forming resistive interphase layers that impede sodium-ion conduction. Sulfide electrolytes are particularly susceptible to oxidation at high potentials, while oxide electrolytes may react with certain cathode compositions, leading to capacity fade and impedance growth over extended cycling.
The sodium metal anode interface presents unique difficulties due to sodium's high reactivity and low melting point. Dendrite formation remains a critical concern, as inhomogeneous sodium deposition can penetrate grain boundaries in polycrystalline electrolytes, causing short circuits. Additionally, the continuous consumption of sodium through side reactions with electrolyte materials results in irreversible capacity loss and unstable solid electrolyte interphase formation.
Mechanical stress accumulation at interfaces during charge-discharge cycles further exacerbates stability issues. The volumetric expansion and contraction of electrode materials generate interfacial gaps and microcracks, progressively degrading ionic pathways. This mechanical degradation is particularly severe in all-solid-state configurations where external pressure application proves insufficient to maintain consistent interfacial contact throughout battery lifetime. Temperature variations during operation compound these mechanical challenges, as thermal expansion mismatches between components create additional interfacial strain.
The most pressing issue involves the formation of high interfacial resistance at both cathode and anode interfaces. Solid electrolytes, particularly oxide-based materials like NASICON and sulfide-based compounds, exhibit poor physical contact with electrode particles due to rigid solid-solid interfaces. This contact limitation restricts ionic transport pathways and increases overall cell resistance, directly impacting power density and rate capability. The problem intensifies during cycling as volume changes in electrode materials further deteriorate interfacial contact.
Chemical incompatibility between solid electrolytes and electrode materials constitutes another major challenge. Many solid electrolytes undergo undesirable side reactions with cathode materials at elevated voltages, forming resistive interphase layers that impede sodium-ion conduction. Sulfide electrolytes are particularly susceptible to oxidation at high potentials, while oxide electrolytes may react with certain cathode compositions, leading to capacity fade and impedance growth over extended cycling.
The sodium metal anode interface presents unique difficulties due to sodium's high reactivity and low melting point. Dendrite formation remains a critical concern, as inhomogeneous sodium deposition can penetrate grain boundaries in polycrystalline electrolytes, causing short circuits. Additionally, the continuous consumption of sodium through side reactions with electrolyte materials results in irreversible capacity loss and unstable solid electrolyte interphase formation.
Mechanical stress accumulation at interfaces during charge-discharge cycles further exacerbates stability issues. The volumetric expansion and contraction of electrode materials generate interfacial gaps and microcracks, progressively degrading ionic pathways. This mechanical degradation is particularly severe in all-solid-state configurations where external pressure application proves insufficient to maintain consistent interfacial contact throughout battery lifetime. Temperature variations during operation compound these mechanical challenges, as thermal expansion mismatches between components create additional interfacial strain.
Current Interface Engineering Solutions for Sodium Batteries
01 Solid electrolyte composition and interface modification
Solid-state sodium-ion batteries utilize specific solid electrolyte compositions to enhance interface stability. These compositions may include sulfide-based, oxide-based, or polymer-based electrolytes with tailored ionic conductivity. Interface modification techniques involve the introduction of buffer layers or coating materials between the electrolyte and electrodes to reduce interfacial resistance and prevent unwanted chemical reactions. The optimization of electrolyte composition and interface engineering are critical for achieving stable electrochemical performance and long cycle life.- Solid electrolyte composition and interface modification: Solid-state sodium-ion batteries utilize specific solid electrolyte compositions to enhance interface stability. These compositions may include sulfide-based, oxide-based, or polymer-based electrolytes with tailored ionic conductivity. Interface modification techniques involve coating layers or buffer layers between the electrolyte and electrodes to reduce interfacial resistance and prevent unwanted chemical reactions. The selection of appropriate solid electrolyte materials and interface engineering strategies is crucial for achieving stable ion transport and minimizing degradation at the electrode-electrolyte interface.
- Electrode material optimization for interface compatibility: The stability of interfaces in solid-state sodium-ion batteries can be improved through careful selection and modification of electrode materials. This includes the use of cathode materials with compatible crystal structures and anode materials that minimize volume expansion during cycling. Surface treatments and coatings on electrode materials help to establish stable solid-solid contacts with the electrolyte. Material engineering approaches focus on reducing interfacial impedance and preventing side reactions that could compromise battery performance and longevity.
- Interlayer design and buffer layer implementation: Implementation of interlayers or buffer layers between the solid electrolyte and electrodes represents an effective strategy for enhancing interface stability. These intermediate layers can accommodate volume changes during charge-discharge cycles, improve ionic conductivity at interfaces, and prevent direct contact between incompatible materials. The buffer layers may consist of composite materials, conductive polymers, or specially designed compounds that facilitate smooth sodium-ion transport while maintaining mechanical and chemical stability at the interface.
- Interface characterization and stability assessment methods: Advanced characterization techniques are employed to evaluate and monitor interface stability in solid-state sodium-ion batteries. These methods include electrochemical impedance spectroscopy, in-situ microscopy, and surface analysis techniques to understand interfacial phenomena. Stability assessment involves studying the evolution of interfacial resistance, chemical composition changes, and mechanical integrity under various operating conditions. Such analytical approaches enable the identification of degradation mechanisms and guide the development of strategies to enhance long-term interface stability.
- Manufacturing processes for stable interface formation: The manufacturing process plays a critical role in establishing stable interfaces in solid-state sodium-ion batteries. Techniques such as controlled sintering, hot-pressing, and thin-film deposition are utilized to create intimate contact between electrolyte and electrode materials. Process parameters including temperature, pressure, and atmosphere are optimized to minimize interfacial defects and ensure uniform contact. Advanced fabrication methods focus on achieving dense, void-free interfaces with low resistance and high mechanical strength to support stable battery operation over extended cycling.
02 Electrode material design for interface compatibility
The selection and design of electrode materials play a crucial role in maintaining interface stability in solid-state sodium-ion batteries. Cathode and anode materials are engineered to have compatible chemical and mechanical properties with the solid electrolyte. Surface treatments, doping strategies, and composite structures are employed to minimize volume changes during cycling and reduce interfacial degradation. These approaches help maintain intimate contact between electrodes and electrolyte, ensuring efficient sodium-ion transport across interfaces.Expand Specific Solutions03 Interfacial layer formation and control
The formation of stable interfacial layers is essential for preventing side reactions and maintaining long-term stability in solid-state sodium-ion batteries. Controlled interfacial layer formation can be achieved through in-situ or ex-situ methods, including the application of protective coatings or the use of additives that promote the formation of stable sodium-conducting interphases. These interfacial layers act as barriers against electrolyte decomposition and electrode dissolution while facilitating sodium-ion conduction.Expand Specific Solutions04 Mechanical stress management at interfaces
Managing mechanical stress at the electrode-electrolyte interface is critical for maintaining interface stability during battery operation. Volume changes in electrode materials during sodium insertion and extraction can cause mechanical degradation and loss of contact with the solid electrolyte. Strategies to address this include the use of flexible or compliant interface materials, optimization of particle size and morphology, and the application of external pressure to maintain interfacial contact. These approaches help prevent crack formation and delamination at interfaces.Expand Specific Solutions05 Interface characterization and stability evaluation methods
Advanced characterization techniques are employed to evaluate and monitor interface stability in solid-state sodium-ion batteries. These methods include electrochemical impedance spectroscopy, scanning electron microscopy, transmission electron microscopy, and X-ray photoelectron spectroscopy to analyze interfacial resistance, morphology, and chemical composition. In-situ and operando techniques provide real-time insights into interfacial changes during battery cycling. Understanding interface evolution through these characterization methods enables the development of strategies to enhance long-term stability and performance.Expand Specific Solutions
Major Players in Solid-State Sodium-Ion Battery Development
The solid-state sodium-ion battery sector is experiencing intensified competition as the technology transitions from early research to commercialization stages. The market remains relatively nascent compared to lithium-ion alternatives, with moderate growth potential driven by cost advantages and abundant sodium resources. Interface stability challenges represent a critical technical bottleneck requiring breakthrough solutions. Leading players include Contemporary Amperex Technology Co., Ltd. and Murata Manufacturing Co. Ltd., who are advancing solid electrolyte interface engineering, while research institutions like Massachusetts Institute of Technology, Beijing Institute of Technology, and Lawrence Berkeley National Laboratory are pioneering novel coating technologies and composite electrolyte designs. Automotive manufacturers such as Toyota Motor Corp., Fisker Inc., and Rivian Holdings LLC are actively investing in next-generation battery solutions. Material suppliers including Shenzhen Capchem Technology Co., Ltd., Resonac Holdings Corp., and TDK Corp. are developing advanced electrolyte formulations to enhance interfacial compatibility and cycling stability, positioning the technology for broader commercial adoption.
The Regents of the University of California
Technical Solution: UC researchers have pioneered fundamental solutions to interface stability in solid-state sodium-ion batteries through innovative electrolyte design and interfacial chemistry control. Their research focuses on developing sodium superionic conductor (NASICON)-type solid electrolytes with modified surface chemistry to reduce interfacial resistance with both cathode and anode materials. The university has developed artificial solid-electrolyte interphase (SEI) layers using atomic layer deposition techniques that provide stable sodium-ion transport while blocking electronic conduction and preventing electrolyte decomposition. UC's approach includes computational modeling to predict interfacial reactions and guide material selection for compatible electrode-electrolyte combinations. They have demonstrated mixed ionic-electronic conducting interlayers that accommodate mechanical stress and maintain electrochemical stability across wide temperature ranges, addressing thermal expansion mismatch issues at interfaces.
Strengths: Cutting-edge fundamental research with deep understanding of interfacial phenomena and access to advanced characterization tools. Weaknesses: Academic focus means technology requires industrial partnership for scale-up and commercialization, longer path to market implementation.
Contemporary Amperex Technology Co., Ltd.
Technical Solution: CATL has developed advanced solid-state sodium-ion battery technology focusing on interface stability through multi-layered interface engineering approaches. Their solution incorporates composite solid electrolytes combining sulfide and oxide materials to create stable interfacial contact with sodium metal anodes. The company employs in-situ formed buffer layers at the electrode-electrolyte interface to accommodate volume changes during cycling and reduce interfacial resistance. CATL's technology utilizes surface coating modifications on cathode materials with sodium-conducting polymers to enhance interfacial compatibility and prevent side reactions. They have implemented advanced manufacturing processes including controlled atmosphere processing to minimize interfacial degradation from moisture and oxygen exposure during cell assembly.
Strengths: Industry-leading manufacturing capabilities and extensive R&D resources enable rapid commercialization. Weaknesses: Technology still in development phase with limited public performance data on long-term interface stability.
Core Patents on Interface Stabilization Techniques
Solid-state electrolyte, secondary battery, battery module, battery pack, and electrical device
PatentPendingEP4528874A1
Innovation
- A solid electrolyte with a contact angle of molten sodium less than 82° is developed, utilizing inorganic or polymer electrolytes with controlled residual alkali content and surface treatments like nanosecond laser, magnetron sputtering bias cleaning, or plasma cleaning to enhance wettability and reduce interface resistance.
Interface protection for sodium all-solid-state batteries
PatentWO2021188976A1
Innovation
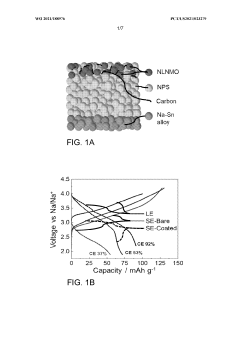
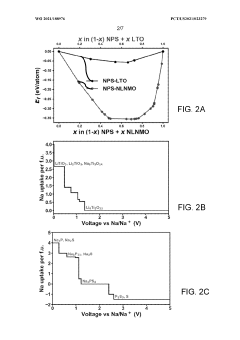
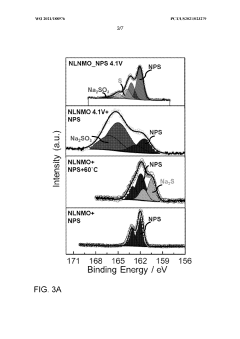
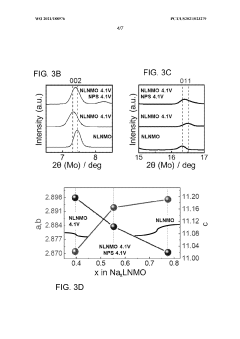
- Applying a Lithium Titanate Oxide (LTO) coating to the cathode of sodium all-solid-state batteries, specifically to the NLNMO or Nao.8[Lio.12Nio.22Mno.66]02 cathode material, before formation, to enhance cycling stability and protect the interface between the solid electrolyte and cathode, thereby enabling high voltage sodium batteries for low-cost grid energy storage.
Material Compatibility and Interfacial Reaction Mechanisms
Material compatibility at solid-solid interfaces represents a fundamental challenge in solid-state sodium-ion batteries, where chemical and electrochemical stability between electrode materials and solid electrolytes directly determines device performance and longevity. The interfacial region serves as the critical pathway for sodium-ion transport, yet it simultaneously becomes a reactive zone where thermodynamic and kinetic instabilities manifest. Understanding the underlying reaction mechanisms at these interfaces is essential for developing effective mitigation strategies.
The chemical incompatibility between cathode materials and solid electrolytes often stems from redox reactions driven by the electrochemical potential difference during battery operation. Oxide-based cathodes such as layered sodium transition metal oxides can trigger oxidative decomposition of sulfide electrolytes, forming insulating interphases that impede ion conduction. Similarly, thiophosphate electrolytes may react with oxide cathodes, producing secondary phases like sodium sulfate or metal sulfides that increase interfacial resistance. These reactions are particularly pronounced at elevated temperatures or high voltage conditions, accelerating capacity fade and power degradation.
At the anode-electrolyte interface, metallic sodium exhibits strong reducing properties that can destabilize most solid electrolytes through chemical reduction reactions. This leads to the formation of mixed ionic-electronic conducting interphases or electronically insulating layers, both detrimental to battery cycling stability. The situation becomes more complex with alloy-type anodes, where volume expansion during sodiation introduces mechanical stress that exacerbates interfacial contact loss and promotes side reactions.
Interfacial reaction kinetics are governed by multiple factors including material composition, crystal structure, surface chemistry, and operating conditions. Grain boundaries and surface defects in polycrystalline electrolytes often serve as preferential sites for reaction initiation due to their higher chemical activity. The formation of space charge layers at interfaces further complicates ion transport by creating additional energy barriers. Computational studies using density functional theory have revealed that electronic band alignment and work function differences between contacting materials significantly influence interfacial stability, providing theoretical guidance for material selection and interface engineering approaches.
The chemical incompatibility between cathode materials and solid electrolytes often stems from redox reactions driven by the electrochemical potential difference during battery operation. Oxide-based cathodes such as layered sodium transition metal oxides can trigger oxidative decomposition of sulfide electrolytes, forming insulating interphases that impede ion conduction. Similarly, thiophosphate electrolytes may react with oxide cathodes, producing secondary phases like sodium sulfate or metal sulfides that increase interfacial resistance. These reactions are particularly pronounced at elevated temperatures or high voltage conditions, accelerating capacity fade and power degradation.
At the anode-electrolyte interface, metallic sodium exhibits strong reducing properties that can destabilize most solid electrolytes through chemical reduction reactions. This leads to the formation of mixed ionic-electronic conducting interphases or electronically insulating layers, both detrimental to battery cycling stability. The situation becomes more complex with alloy-type anodes, where volume expansion during sodiation introduces mechanical stress that exacerbates interfacial contact loss and promotes side reactions.
Interfacial reaction kinetics are governed by multiple factors including material composition, crystal structure, surface chemistry, and operating conditions. Grain boundaries and surface defects in polycrystalline electrolytes often serve as preferential sites for reaction initiation due to their higher chemical activity. The formation of space charge layers at interfaces further complicates ion transport by creating additional energy barriers. Computational studies using density functional theory have revealed that electronic band alignment and work function differences between contacting materials significantly influence interfacial stability, providing theoretical guidance for material selection and interface engineering approaches.
Manufacturing Scalability of Interface Modification Methods
The transition from laboratory-scale interface modification techniques to industrial-scale manufacturing represents a critical bottleneck in commercializing solid-state sodium-ion batteries. While numerous interface engineering strategies have demonstrated promising results at small scales, their practical implementation faces significant challenges related to cost-effectiveness, production throughput, and quality consistency. Current manufacturing scalability concerns primarily revolve around three aspects: process complexity, equipment requirements, and material utilization efficiency.
Coating-based interface modification methods, such as atomic layer deposition and chemical vapor deposition, offer precise control over interface composition but suffer from inherently slow deposition rates and high capital investment requirements. These techniques typically process substrates sequentially or in small batches, making them economically unfeasible for large-volume battery production. Alternative approaches like solution-based coating and spray deposition present better scalability potential due to their compatibility with roll-to-roll processing, though they face challenges in achieving uniform coating thickness and composition control across large substrate areas.
In-situ interface formation strategies demonstrate superior scalability advantages as they integrate interface modification directly into the battery assembly process, eliminating separate processing steps. However, these methods require careful optimization of processing parameters to ensure reproducible interface quality across different production batches. The scalability of buffer layer incorporation depends heavily on material availability and processing compatibility with existing manufacturing infrastructure.
Economic viability analysis reveals that manufacturing costs for interface-modified solid-state sodium-ion batteries must decrease by approximately 60-70% to compete with conventional lithium-ion technologies. This necessitates development of high-throughput processing techniques that maintain interface quality while significantly reducing production time per unit. Emerging approaches combining multiple interface modification strategies in single processing steps show promise for achieving both performance targets and manufacturing scalability requirements.
Coating-based interface modification methods, such as atomic layer deposition and chemical vapor deposition, offer precise control over interface composition but suffer from inherently slow deposition rates and high capital investment requirements. These techniques typically process substrates sequentially or in small batches, making them economically unfeasible for large-volume battery production. Alternative approaches like solution-based coating and spray deposition present better scalability potential due to their compatibility with roll-to-roll processing, though they face challenges in achieving uniform coating thickness and composition control across large substrate areas.
In-situ interface formation strategies demonstrate superior scalability advantages as they integrate interface modification directly into the battery assembly process, eliminating separate processing steps. However, these methods require careful optimization of processing parameters to ensure reproducible interface quality across different production batches. The scalability of buffer layer incorporation depends heavily on material availability and processing compatibility with existing manufacturing infrastructure.
Economic viability analysis reveals that manufacturing costs for interface-modified solid-state sodium-ion batteries must decrease by approximately 60-70% to compete with conventional lithium-ion technologies. This necessitates development of high-throughput processing techniques that maintain interface quality while significantly reducing production time per unit. Emerging approaches combining multiple interface modification strategies in single processing steps show promise for achieving both performance targets and manufacturing scalability requirements.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!