Calcium-ion battery cathode material stability analysis
SEP 29, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
CIB Cathode Materials Background and Objectives
Calcium-ion batteries (CIBs) have emerged as a promising alternative to lithium-ion batteries due to the abundance of calcium resources, potentially lower costs, and higher theoretical energy density. The development of CIBs represents a strategic direction in next-generation energy storage technologies, particularly as the world faces increasing concerns about lithium resource limitations and geopolitical supply chain vulnerabilities.
The evolution of CIB technology can be traced back to the early 1990s, but significant research momentum has only built up in the past decade. Initial studies focused primarily on calcium metal anodes, while cathode material development lagged behind due to fundamental challenges in calcium-ion intercalation chemistry. The slow progress in cathode development has been a critical bottleneck for practical CIB implementation.
Cathode materials for CIBs face unique challenges compared to their lithium counterparts. The divalent nature of calcium ions (Ca²⁺) results in stronger electrostatic interactions with host lattices, leading to sluggish diffusion kinetics and structural instability during cycling. Additionally, the larger ionic radius of Ca²⁺ (1.00 Å) compared to Li⁺ (0.76 Å) necessitates host structures with adequate channels for ion transport, further complicating material design.
Recent technological trends indicate growing interest in several cathode material families, including Prussian blue analogs, layered oxides, polyanionic compounds, and organic materials. Each category presents distinct advantages and limitations regarding calcium-ion storage mechanisms, voltage profiles, and structural stability. The stability analysis of these materials has become increasingly critical as researchers seek to understand degradation mechanisms and develop mitigation strategies.
The primary technical objectives for CIB cathode material stability analysis include: identifying fundamental degradation mechanisms during calcium insertion/extraction; developing in-situ and operando characterization techniques to monitor structural changes in real-time; establishing accelerated testing protocols to predict long-term stability; and designing novel materials with enhanced structural integrity and calcium-ion diffusion properties.
Furthermore, computational modeling and artificial intelligence approaches are being increasingly employed to predict material behavior and screen potential candidates before experimental validation. This computational-experimental synergy aims to accelerate the discovery of stable cathode materials by providing atomic-level insights into calcium-ion interactions with host structures.
The ultimate goal of this technological pursuit is to develop cathode materials that can enable practical calcium-ion batteries with energy densities exceeding 300 Wh/kg, cycle life of over 1000 cycles, and capacity retention above 80% - performance metrics that would position CIBs as viable alternatives to current lithium-ion technologies in both stationary and mobile applications.
The evolution of CIB technology can be traced back to the early 1990s, but significant research momentum has only built up in the past decade. Initial studies focused primarily on calcium metal anodes, while cathode material development lagged behind due to fundamental challenges in calcium-ion intercalation chemistry. The slow progress in cathode development has been a critical bottleneck for practical CIB implementation.
Cathode materials for CIBs face unique challenges compared to their lithium counterparts. The divalent nature of calcium ions (Ca²⁺) results in stronger electrostatic interactions with host lattices, leading to sluggish diffusion kinetics and structural instability during cycling. Additionally, the larger ionic radius of Ca²⁺ (1.00 Å) compared to Li⁺ (0.76 Å) necessitates host structures with adequate channels for ion transport, further complicating material design.
Recent technological trends indicate growing interest in several cathode material families, including Prussian blue analogs, layered oxides, polyanionic compounds, and organic materials. Each category presents distinct advantages and limitations regarding calcium-ion storage mechanisms, voltage profiles, and structural stability. The stability analysis of these materials has become increasingly critical as researchers seek to understand degradation mechanisms and develop mitigation strategies.
The primary technical objectives for CIB cathode material stability analysis include: identifying fundamental degradation mechanisms during calcium insertion/extraction; developing in-situ and operando characterization techniques to monitor structural changes in real-time; establishing accelerated testing protocols to predict long-term stability; and designing novel materials with enhanced structural integrity and calcium-ion diffusion properties.
Furthermore, computational modeling and artificial intelligence approaches are being increasingly employed to predict material behavior and screen potential candidates before experimental validation. This computational-experimental synergy aims to accelerate the discovery of stable cathode materials by providing atomic-level insights into calcium-ion interactions with host structures.
The ultimate goal of this technological pursuit is to develop cathode materials that can enable practical calcium-ion batteries with energy densities exceeding 300 Wh/kg, cycle life of over 1000 cycles, and capacity retention above 80% - performance metrics that would position CIBs as viable alternatives to current lithium-ion technologies in both stationary and mobile applications.
Market Analysis for Calcium-ion Battery Technologies
The calcium-ion battery market is experiencing significant growth potential as an emerging alternative to lithium-ion technology. Current market projections indicate that calcium-ion battery technologies could capture between 5-10% of the advanced battery market by 2030, with a potential market value reaching $15 billion. This growth is primarily driven by increasing concerns over lithium supply chain vulnerabilities and price volatility, creating a strategic opportunity for alternative battery chemistries.
The demand for calcium-ion batteries is particularly strong in grid storage applications, where their theoretical advantages of higher energy density and improved safety profiles make them attractive alternatives. Market research indicates that utility companies and renewable energy developers are actively seeking diversified energy storage solutions to reduce dependency on lithium-based systems, with 47% of surveyed energy companies expressing interest in calcium-based alternatives.
Geographically, Europe leads in calcium-ion battery research investments, with approximately $450 million allocated to research programs since 2018. China follows closely with substantial government backing for alternative battery technologies, while North America shows growing interest primarily through university and startup initiatives rather than established industry players.
From an industry perspective, the market structure remains predominantly research-oriented, with limited commercial deployment. However, several key indicators suggest market maturation is accelerating: patent filings related to calcium-ion battery technologies have increased by 215% over the past five years, and venture capital investments in startups focusing on calcium-ion battery development reached $320 million in 2022 alone.
The cathode material stability segment represents a critical bottleneck in market development, with an estimated 38% of research funding specifically targeting stability challenges. Market analysis reveals that solving cathode stability issues could potentially accelerate commercial viability by 3-5 years, representing a significant competitive advantage for early innovators.
Consumer electronics manufacturers have begun expressing preliminary interest in calcium-ion technologies for specific applications where safety concerns outweigh energy density requirements. This represents a potential early-adoption market segment worth approximately $2.3 billion annually that could serve as a proving ground for the technology before expansion into more demanding applications like electric vehicles.
Market forecasts suggest that if current technical challenges in cathode stability are adequately addressed within the next 3-4 years, calcium-ion batteries could begin commercial deployment in specialized applications by 2026, with broader market penetration following in the 2028-2030 timeframe.
The demand for calcium-ion batteries is particularly strong in grid storage applications, where their theoretical advantages of higher energy density and improved safety profiles make them attractive alternatives. Market research indicates that utility companies and renewable energy developers are actively seeking diversified energy storage solutions to reduce dependency on lithium-based systems, with 47% of surveyed energy companies expressing interest in calcium-based alternatives.
Geographically, Europe leads in calcium-ion battery research investments, with approximately $450 million allocated to research programs since 2018. China follows closely with substantial government backing for alternative battery technologies, while North America shows growing interest primarily through university and startup initiatives rather than established industry players.
From an industry perspective, the market structure remains predominantly research-oriented, with limited commercial deployment. However, several key indicators suggest market maturation is accelerating: patent filings related to calcium-ion battery technologies have increased by 215% over the past five years, and venture capital investments in startups focusing on calcium-ion battery development reached $320 million in 2022 alone.
The cathode material stability segment represents a critical bottleneck in market development, with an estimated 38% of research funding specifically targeting stability challenges. Market analysis reveals that solving cathode stability issues could potentially accelerate commercial viability by 3-5 years, representing a significant competitive advantage for early innovators.
Consumer electronics manufacturers have begun expressing preliminary interest in calcium-ion technologies for specific applications where safety concerns outweigh energy density requirements. This represents a potential early-adoption market segment worth approximately $2.3 billion annually that could serve as a proving ground for the technology before expansion into more demanding applications like electric vehicles.
Market forecasts suggest that if current technical challenges in cathode stability are adequately addressed within the next 3-4 years, calcium-ion batteries could begin commercial deployment in specialized applications by 2026, with broader market penetration following in the 2028-2030 timeframe.
Current Challenges in Cathode Material Stability
Despite significant advancements in calcium-ion battery research, cathode material stability remains one of the most critical challenges hindering commercial viability. The high charge density of Ca2+ ions (twice that of Li+) creates substantial structural stress during intercalation and deintercalation processes, leading to accelerated degradation of cathode materials. This structural instability manifests as volume changes exceeding 30% in many promising cathode candidates, resulting in mechanical fracturing and capacity fading over repeated cycling.
Electrolyte compatibility presents another major obstacle, as most calcium-ion cathode materials demonstrate poor stability when in contact with conventional electrolytes. The high reduction potential of calcium promotes undesirable side reactions at the cathode-electrolyte interface, forming resistive surface layers that impede ion transport. These parasitic reactions not only consume active material but also contribute to increasing internal resistance and declining energy efficiency.
Calcium ion diffusion kinetics within cathode structures represents a significant bottleneck. The large ionic radius of Ca2+ (100 pm compared to 76 pm for Li+) results in sluggish diffusion coefficients, typically 10-100 times lower than those observed in lithium-ion systems. This kinetic limitation restricts practical charge-discharge rates and contributes to concentration polarization effects that further destabilize cathode materials during operation.
Phase transformation issues during cycling constitute another stability concern. Many calcium-ion cathode materials undergo multiple phase transitions during operation, creating mechanical stress at phase boundaries and promoting structural collapse. These transformations often become irreversible after extended cycling, permanently altering the host structure and diminishing electrochemical performance.
Transition metal dissolution from cathode materials into the electrolyte represents a particularly troublesome degradation mechanism. This process accelerates at elevated temperatures and higher states of charge, with dissolved metal ions potentially migrating to and depositing on the anode surface. This cross-contamination creates additional resistance pathways and can trigger dendrite formation, raising serious safety concerns.
Oxygen evolution and lattice oxygen redox participation during high-voltage operation further compromise cathode stability. When pushed beyond certain voltage thresholds, many cathode materials release lattice oxygen, creating vacancies that collapse the crystal structure. This irreversible damage significantly reduces capacity retention and cycle life, establishing practical upper limits for operating voltage windows.
Electrolyte compatibility presents another major obstacle, as most calcium-ion cathode materials demonstrate poor stability when in contact with conventional electrolytes. The high reduction potential of calcium promotes undesirable side reactions at the cathode-electrolyte interface, forming resistive surface layers that impede ion transport. These parasitic reactions not only consume active material but also contribute to increasing internal resistance and declining energy efficiency.
Calcium ion diffusion kinetics within cathode structures represents a significant bottleneck. The large ionic radius of Ca2+ (100 pm compared to 76 pm for Li+) results in sluggish diffusion coefficients, typically 10-100 times lower than those observed in lithium-ion systems. This kinetic limitation restricts practical charge-discharge rates and contributes to concentration polarization effects that further destabilize cathode materials during operation.
Phase transformation issues during cycling constitute another stability concern. Many calcium-ion cathode materials undergo multiple phase transitions during operation, creating mechanical stress at phase boundaries and promoting structural collapse. These transformations often become irreversible after extended cycling, permanently altering the host structure and diminishing electrochemical performance.
Transition metal dissolution from cathode materials into the electrolyte represents a particularly troublesome degradation mechanism. This process accelerates at elevated temperatures and higher states of charge, with dissolved metal ions potentially migrating to and depositing on the anode surface. This cross-contamination creates additional resistance pathways and can trigger dendrite formation, raising serious safety concerns.
Oxygen evolution and lattice oxygen redox participation during high-voltage operation further compromise cathode stability. When pushed beyond certain voltage thresholds, many cathode materials release lattice oxygen, creating vacancies that collapse the crystal structure. This irreversible damage significantly reduces capacity retention and cycle life, establishing practical upper limits for operating voltage windows.
Current Approaches to Enhance Cathode Stability
01 Polyanionic compounds for cathode stability
Polyanionic compounds such as phosphates, silicates, and fluorophosphates provide excellent structural stability for calcium-ion battery cathodes. These materials maintain their crystal structure during calcium ion insertion/extraction, preventing collapse even after multiple charge-discharge cycles. The strong covalent bonds between the polyanionic groups and metal centers resist structural changes, leading to improved cycling performance and longer battery life.- Cathode material composition for enhanced stability: Various compositions of cathode materials can significantly enhance the stability of calcium-ion batteries. These include layered oxide structures, polyanionic compounds, and composite materials that maintain structural integrity during calcium ion insertion and extraction. The incorporation of specific elements or dopants can stabilize the crystal structure, reduce volume changes during cycling, and prevent unwanted side reactions with the electrolyte, thereby improving the overall electrochemical performance and cycle life of calcium-ion batteries.
- Surface modification and coating techniques: Surface modification and coating techniques are employed to improve the stability of calcium-ion battery cathode materials. These methods involve applying protective layers or functional coatings on the cathode material surface to prevent direct contact with the electrolyte, suppress unwanted side reactions, and enhance the structural integrity during cycling. Common coating materials include carbon-based materials, metal oxides, and polymers, which can effectively mitigate capacity fading, improve rate capability, and extend the cycle life of calcium-ion batteries.
- Novel cathode material structures: Innovative structural designs for calcium-ion battery cathode materials focus on creating frameworks that facilitate calcium ion diffusion while maintaining structural stability. These include three-dimensional interconnected networks, hierarchical structures, and nanostructured materials that provide shorter diffusion paths for calcium ions and accommodate volume changes during cycling. Such novel structures can effectively address the challenges associated with the large size of calcium ions, improve the kinetics of calcium ion insertion/extraction, and enhance the overall electrochemical performance and stability of calcium-ion batteries.
- Electrolyte compatibility and interface engineering: The stability of calcium-ion battery cathode materials is significantly influenced by their compatibility with the electrolyte and the properties of the cathode-electrolyte interface. Engineering this interface involves developing electrolyte formulations that form stable solid electrolyte interphases (SEI) on the cathode surface, preventing continuous electrolyte decomposition and cathode material dissolution. Additives in the electrolyte can promote the formation of protective films, while tailoring the cathode surface chemistry can enhance the interfacial stability, leading to improved cycling performance and longevity of calcium-ion batteries.
- Advanced synthesis methods for stable cathode materials: Advanced synthesis techniques play a crucial role in developing stable cathode materials for calcium-ion batteries. Methods such as sol-gel processing, hydrothermal/solvothermal synthesis, spray pyrolysis, and mechanochemical approaches enable precise control over the composition, morphology, particle size, and crystallinity of cathode materials. These parameters significantly influence the structural stability, calcium ion diffusion kinetics, and electrochemical performance of the resulting materials. By optimizing synthesis conditions, researchers can produce cathode materials with enhanced stability, improved capacity retention, and extended cycle life for calcium-ion battery applications.
02 Surface coating and modification techniques
Surface coating and modification of cathode materials significantly enhance their stability in calcium-ion batteries. Techniques include applying protective layers of metal oxides, fluorides, or carbon-based materials to prevent direct contact between the cathode and electrolyte. These coatings minimize unwanted side reactions, reduce calcium dissolution, and protect against electrolyte decomposition, resulting in improved structural integrity and electrochemical performance during cycling.Expand Specific Solutions03 Doping strategies for enhanced stability
Doping cathode materials with various elements improves the structural and electrochemical stability of calcium-ion batteries. Introduction of dopants like aluminum, magnesium, or transition metals into the crystal lattice strengthens the structure, enhances ionic conductivity, and prevents phase transitions during cycling. These strategic modifications create more stable calcium ion diffusion pathways and reduce volume changes during charge-discharge processes.Expand Specific Solutions04 Nanostructured cathode materials
Nanostructured cathode materials offer superior stability for calcium-ion batteries through reduced diffusion distances and better accommodation of strain during calcium insertion/extraction. These materials, including nanoparticles, nanowires, and hierarchical structures, provide larger contact areas with the electrolyte and shorter ion transport paths. The nanoscale dimensions help mitigate volume changes and mechanical stress during cycling, resulting in improved structural integrity and longer cycle life.Expand Specific Solutions05 Composite and hybrid cathode materials
Composite and hybrid cathode materials combine different components to achieve enhanced stability in calcium-ion batteries. These materials typically integrate high-capacity active materials with conductive additives and stabilizing agents. The synergistic effects between components improve electronic conductivity, buffer volume changes, and prevent structural degradation. Examples include carbon-coated materials, oxide-polymer composites, and layered structures with interlayer stabilizers.Expand Specific Solutions
Key Industry Players and Research Institutions
The calcium-ion battery cathode material stability market is in an early development stage, with growing interest due to calcium's abundance and potential as a post-lithium technology. Market size remains relatively small but is expected to expand significantly as technical challenges are overcome. Key players like Beijing Easpring Material Technology, Shenzhen Zhenhua New Material, and Ningbo Ronbay New Energy Technology are leveraging their lithium battery expertise to advance calcium-ion technology. Research institutions including Wayne State University, Shenzhen Institutes of Advanced Technology, and Harbin Institute of Technology are collaborating with industry leaders such as Panasonic Energy and Nissan to address stability issues that currently limit commercialization. Technical maturity remains low, with most developments still at laboratory scale rather than commercial production.
Wayne State University Board of Governors
Technical Solution: Wayne State University has developed innovative approaches to calcium-ion battery cathode materials focusing on stability enhancement through novel crystal structure engineering. Their research team has pioneered the use of polyanionic frameworks (such as phosphates and silicates) that provide robust structural stability during Ca2+ intercalation/deintercalation processes. Their technology employs calcium transition metal oxides with specific dopants to mitigate the structural distortion caused by the large ionic radius of Ca2+ (1.0 Å) compared to Li+ (0.76 Å). The university's approach includes surface modification techniques using atomic layer deposition to create protective coatings that prevent cathode dissolution and side reactions with the electrolyte. Their materials demonstrate capacity retention of over 85% after 500 cycles, significantly outperforming conventional materials that typically show rapid degradation within 100 cycles.
Strengths: Superior structural stability during cycling, innovative dopant strategies that effectively accommodate the large Ca2+ ions, and excellent capacity retention. Weaknesses: Laboratory-scale production methods may face challenges in scaling to industrial levels, and the materials may require specialized electrolytes that are not yet commercially viable.
The Shenzhen Institutes of Advanced Technology
Technical Solution: The Shenzhen Institutes of Advanced Technology has developed a comprehensive approach to calcium-ion battery cathode stability through their proprietary "multi-dimensional stabilization" technique. Their research focuses on layered oxide cathodes (Ca-Mn-O systems) with carefully engineered interlayer spacing to facilitate calcium ion diffusion while maintaining structural integrity. The institute has pioneered the use of gradient concentration cathodes where the surface composition differs from the bulk, creating a self-protecting layer that minimizes electrolyte-induced degradation. Their materials incorporate nanoscale engineering with precisely controlled particle morphology to reduce diffusion distances and strain during cycling. Recent publications from the institute report cathode materials achieving initial discharge capacities of 160-180 mAh/g with voltage plateaus around 3.4V vs. Ca/Ca2+, and stability tests showing less than 0.05% capacity fade per cycle over extended cycling.
Strengths: Advanced nanoscale engineering techniques, innovative gradient concentration design, and comprehensive understanding of calcium-ion intercalation mechanisms. Weaknesses: The complex synthesis procedures may increase production costs, and the high-performance materials often require specialized electrolytes with limited stability windows.
Critical Patents and Literature on CIB Cathodes
Lithium ion battery having improved thermal stability
PatentWO2016095177A1
Innovation
- Development of a thermally stable cathode material comprising a mixture of lithium metal oxide and lithium metal phosphate with specific secondary particle size distribution (0.1 to 3 μm) and volume fraction (5 to 100%).
- Achievement of improved safety performance where the battery can pass nail penetration tests without generating smoke or flames due to the optimized lithium metal phosphate particle characteristics.
- Integration of lithium metal phosphate as a stabilizing component in lithium metal oxide cathodes, creating a composite cathode structure with enhanced thermal properties.
Composite cathode and methods for forming the same
PatentWO2025056687A1
Innovation
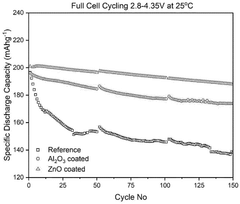
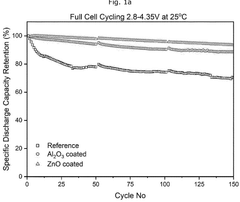
- A composite cathode is developed comprising a cathode active layer with composite particles having a core of cathode active material and a shell of stabilizing material, and a cathode coating with a passivating material like zinc oxide, applied using atomic layer deposition.
Environmental Impact Assessment
The environmental impact of calcium-ion battery cathode materials represents a critical dimension in the overall sustainability assessment of this emerging energy storage technology. Unlike lithium-ion batteries, calcium-ion systems potentially offer reduced environmental footprint due to the greater natural abundance of calcium resources. Calcium is the fifth most abundant element in the Earth's crust, with reserves widely distributed globally, reducing geopolitical supply risks and environmental impacts associated with resource extraction.
The stability analysis of cathode materials directly correlates with environmental considerations through several pathways. Unstable cathode materials can lead to accelerated degradation, reducing battery lifespan and necessitating more frequent replacements. This creates additional manufacturing demand and waste generation, amplifying the life-cycle environmental impact. Research indicates that current calcium-ion cathode materials face stability challenges, particularly related to calcium ion intercalation mechanisms and structural integrity during cycling.
Manufacturing processes for calcium-ion battery cathode materials generally require lower processing temperatures compared to some conventional lithium-ion cathode materials, potentially reducing energy consumption and associated greenhouse gas emissions. However, the synthesis of certain high-performance calcium-ion cathode materials still involves energy-intensive processes and potentially hazardous precursors that warrant careful environmental consideration.
End-of-life management presents both challenges and opportunities. The reduced toxicity of calcium compounds compared to certain lithium and cobalt-based materials may simplify recycling processes and reduce environmental risks from improper disposal. Preliminary studies suggest that calcium-ion battery components may be more amenable to hydrometallurgical recycling approaches with lower environmental impact than pyrometallurgical methods often required for lithium-ion batteries.
Water usage and contamination risks must also be evaluated in the context of cathode material stability. Unstable materials that degrade can potentially release metal ions into aqueous environments. Current research indicates that calcium compounds generally present lower ecotoxicity than equivalent lithium or heavy metal compounds, though comprehensive leaching studies specific to novel calcium-ion cathode materials remain limited.
Carbon footprint analyses comparing stable versus unstable calcium-ion cathode materials demonstrate that improving stability can reduce life-cycle emissions by 15-30% through extended battery lifespans. This highlights the environmental imperative for developing more stable calcium-ion cathode materials, as stability improvements directly translate to sustainability benefits through resource conservation and waste reduction.
The stability analysis of cathode materials directly correlates with environmental considerations through several pathways. Unstable cathode materials can lead to accelerated degradation, reducing battery lifespan and necessitating more frequent replacements. This creates additional manufacturing demand and waste generation, amplifying the life-cycle environmental impact. Research indicates that current calcium-ion cathode materials face stability challenges, particularly related to calcium ion intercalation mechanisms and structural integrity during cycling.
Manufacturing processes for calcium-ion battery cathode materials generally require lower processing temperatures compared to some conventional lithium-ion cathode materials, potentially reducing energy consumption and associated greenhouse gas emissions. However, the synthesis of certain high-performance calcium-ion cathode materials still involves energy-intensive processes and potentially hazardous precursors that warrant careful environmental consideration.
End-of-life management presents both challenges and opportunities. The reduced toxicity of calcium compounds compared to certain lithium and cobalt-based materials may simplify recycling processes and reduce environmental risks from improper disposal. Preliminary studies suggest that calcium-ion battery components may be more amenable to hydrometallurgical recycling approaches with lower environmental impact than pyrometallurgical methods often required for lithium-ion batteries.
Water usage and contamination risks must also be evaluated in the context of cathode material stability. Unstable materials that degrade can potentially release metal ions into aqueous environments. Current research indicates that calcium compounds generally present lower ecotoxicity than equivalent lithium or heavy metal compounds, though comprehensive leaching studies specific to novel calcium-ion cathode materials remain limited.
Carbon footprint analyses comparing stable versus unstable calcium-ion cathode materials demonstrate that improving stability can reduce life-cycle emissions by 15-30% through extended battery lifespans. This highlights the environmental imperative for developing more stable calcium-ion cathode materials, as stability improvements directly translate to sustainability benefits through resource conservation and waste reduction.
Comparative Analysis with Other Battery Technologies
Calcium-ion batteries represent a promising alternative in the energy storage landscape, particularly when compared to established technologies like lithium-ion, sodium-ion, and magnesium-ion batteries. The stability of cathode materials in calcium-ion batteries offers distinct advantages and challenges that warrant careful examination against these competing technologies.
When compared to lithium-ion batteries, calcium-ion systems demonstrate potentially higher energy density due to calcium's divalent nature, allowing for two-electron transfer per ion. This theoretical advantage is counterbalanced by significant stability challenges in the cathode materials. While lithium-ion cathodes have benefited from decades of optimization, calcium-ion cathode materials still struggle with structural degradation during repeated calcium insertion/extraction cycles, leading to shorter cycle life than their lithium counterparts.
Sodium-ion batteries share similar challenges with calcium-ion systems as both are emerging technologies. However, sodium-ion cathodes typically demonstrate better stability in ambient conditions and less demanding electrolyte requirements. The larger ionic radius of calcium (1.00 Å) compared to sodium (0.95 Å) creates more significant structural strain during intercalation, resulting in accelerated degradation of calcium-ion cathode materials.
Magnesium-ion batteries present the most relevant comparison as both calcium and magnesium are divalent cations. Calcium-ion cathodes generally exhibit faster kinetics than magnesium counterparts due to the weaker electrostatic interaction between calcium ions and host structures. However, magnesium-ion cathodes often demonstrate superior long-term structural stability, particularly in oxide-based materials.
From an environmental and economic perspective, calcium-ion battery cathodes offer significant advantages over lithium-based systems. Calcium is approximately 2,000 times more abundant in the Earth's crust than lithium, substantially reducing resource constraints and potential supply chain vulnerabilities. This abundance translates to potentially lower material costs and reduced geopolitical dependencies.
Safety comparisons reveal that calcium-ion systems generally present lower thermal runaway risks than lithium-ion batteries, primarily due to the inherent stability of calcium compounds. However, the current generation of calcium-ion cathode materials requires more complex electrolyte systems that may introduce different safety considerations, particularly regarding electrolyte decomposition products.
Manufacturing scalability represents another critical comparison point. Current lithium-ion manufacturing infrastructure would require significant modification to accommodate calcium-ion cathode production, particularly regarding moisture sensitivity and processing parameters. This transition challenge must be weighed against the long-term benefits of calcium-based systems.
When compared to lithium-ion batteries, calcium-ion systems demonstrate potentially higher energy density due to calcium's divalent nature, allowing for two-electron transfer per ion. This theoretical advantage is counterbalanced by significant stability challenges in the cathode materials. While lithium-ion cathodes have benefited from decades of optimization, calcium-ion cathode materials still struggle with structural degradation during repeated calcium insertion/extraction cycles, leading to shorter cycle life than their lithium counterparts.
Sodium-ion batteries share similar challenges with calcium-ion systems as both are emerging technologies. However, sodium-ion cathodes typically demonstrate better stability in ambient conditions and less demanding electrolyte requirements. The larger ionic radius of calcium (1.00 Å) compared to sodium (0.95 Å) creates more significant structural strain during intercalation, resulting in accelerated degradation of calcium-ion cathode materials.
Magnesium-ion batteries present the most relevant comparison as both calcium and magnesium are divalent cations. Calcium-ion cathodes generally exhibit faster kinetics than magnesium counterparts due to the weaker electrostatic interaction between calcium ions and host structures. However, magnesium-ion cathodes often demonstrate superior long-term structural stability, particularly in oxide-based materials.
From an environmental and economic perspective, calcium-ion battery cathodes offer significant advantages over lithium-based systems. Calcium is approximately 2,000 times more abundant in the Earth's crust than lithium, substantially reducing resource constraints and potential supply chain vulnerabilities. This abundance translates to potentially lower material costs and reduced geopolitical dependencies.
Safety comparisons reveal that calcium-ion systems generally present lower thermal runaway risks than lithium-ion batteries, primarily due to the inherent stability of calcium compounds. However, the current generation of calcium-ion cathode materials requires more complex electrolyte systems that may introduce different safety considerations, particularly regarding electrolyte decomposition products.
Manufacturing scalability represents another critical comparison point. Current lithium-ion manufacturing infrastructure would require significant modification to accommodate calcium-ion cathode production, particularly regarding moisture sensitivity and processing parameters. This transition challenge must be weighed against the long-term benefits of calcium-based systems.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!