Comparing 3D Bioprinting and Microfabrication Techniques
MAR 5, 202610 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
3D Bioprinting vs Microfabrication Background and Objectives
The convergence of 3D bioprinting and microfabrication technologies represents a pivotal moment in biomedical engineering, where precision manufacturing meets biological complexity. Both fields have evolved from distinct origins, with 3D bioprinting emerging from additive manufacturing principles adapted for biological materials, while microfabrication stems from semiconductor industry techniques scaled for biological applications. The intersection of these technologies has created unprecedented opportunities for creating functional biological constructs at multiple scales.
3D bioprinting technology has progressed through several evolutionary phases since its inception in the early 2000s. Initial developments focused on adapting conventional 3D printing methods to handle biological materials, progressing from simple cell-laden hydrogel deposition to sophisticated multi-material printing systems. The technology has advanced from proof-of-concept demonstrations to creating complex tissue architectures with integrated vascular networks and multiple cell types.
Microfabrication techniques in biological applications have similarly evolved from traditional photolithography and etching processes used in electronics manufacturing. The adaptation of these precision techniques for biological systems has enabled the creation of microfluidic devices, cell culture platforms, and microscale tissue constructs with features measured in micrometers rather than millimeters.
The primary objective of comparing these technologies lies in understanding their complementary capabilities and identifying optimal application scenarios. 3D bioprinting excels in creating larger-scale tissue constructs with complex geometries and multiple material compositions, while microfabrication provides unmatched precision for creating microscale features and controlled microenvironments essential for cellular behavior studies.
Current technological goals focus on bridging the resolution gap between these approaches, developing hybrid manufacturing strategies that leverage the strengths of both techniques. The integration aims to achieve tissue constructs that possess both macroscale functionality and microscale precision, addressing critical challenges in tissue engineering, drug testing, and regenerative medicine applications.
The strategic importance of this technological comparison extends beyond academic interest, as it directly impacts the development of next-generation biomedical devices, personalized medicine approaches, and scalable tissue manufacturing processes. Understanding the technical boundaries and synergistic potential of these technologies is crucial for directing future research investments and establishing realistic development timelines for clinical applications.
3D bioprinting technology has progressed through several evolutionary phases since its inception in the early 2000s. Initial developments focused on adapting conventional 3D printing methods to handle biological materials, progressing from simple cell-laden hydrogel deposition to sophisticated multi-material printing systems. The technology has advanced from proof-of-concept demonstrations to creating complex tissue architectures with integrated vascular networks and multiple cell types.
Microfabrication techniques in biological applications have similarly evolved from traditional photolithography and etching processes used in electronics manufacturing. The adaptation of these precision techniques for biological systems has enabled the creation of microfluidic devices, cell culture platforms, and microscale tissue constructs with features measured in micrometers rather than millimeters.
The primary objective of comparing these technologies lies in understanding their complementary capabilities and identifying optimal application scenarios. 3D bioprinting excels in creating larger-scale tissue constructs with complex geometries and multiple material compositions, while microfabrication provides unmatched precision for creating microscale features and controlled microenvironments essential for cellular behavior studies.
Current technological goals focus on bridging the resolution gap between these approaches, developing hybrid manufacturing strategies that leverage the strengths of both techniques. The integration aims to achieve tissue constructs that possess both macroscale functionality and microscale precision, addressing critical challenges in tissue engineering, drug testing, and regenerative medicine applications.
The strategic importance of this technological comparison extends beyond academic interest, as it directly impacts the development of next-generation biomedical devices, personalized medicine approaches, and scalable tissue manufacturing processes. Understanding the technical boundaries and synergistic potential of these technologies is crucial for directing future research investments and establishing realistic development timelines for clinical applications.
Market Demand for Advanced Tissue Engineering Solutions
The global tissue engineering market is experiencing unprecedented growth driven by an aging population, increasing prevalence of chronic diseases, and rising demand for organ transplantation alternatives. Traditional treatment approaches for tissue and organ failure face significant limitations, including donor organ shortages, immune rejection risks, and lengthy waiting periods that often result in patient mortality. This critical healthcare gap has created substantial market opportunities for advanced tissue engineering solutions that can provide personalized, biocompatible alternatives.
Healthcare systems worldwide are increasingly recognizing the economic burden of conventional treatments for tissue-related disorders. Chronic wounds, cardiovascular diseases, orthopedic conditions, and organ failures collectively represent billions in annual healthcare expenditures. The potential for tissue engineering technologies to reduce long-term treatment costs while improving patient outcomes has attracted significant attention from healthcare providers, insurance companies, and government health agencies seeking cost-effective solutions.
The pharmaceutical and biotechnology industries are driving substantial demand for advanced tissue models that can replace animal testing and improve drug development processes. Regulatory agencies are increasingly encouraging the adoption of alternative testing methods, creating a robust market for sophisticated in vitro tissue models. These applications require precise control over tissue architecture and cellular organization, making advanced manufacturing techniques essential for meeting industry standards and regulatory requirements.
Regenerative medicine applications represent the fastest-growing segment within tissue engineering markets. The demand spans multiple therapeutic areas including cardiovascular repair, neural tissue regeneration, skin grafts, bone and cartilage reconstruction, and liver tissue engineering. Each application presents unique technical requirements for tissue architecture, mechanical properties, and biological functionality, driving the need for versatile and precise manufacturing approaches.
Research institutions and academic medical centers constitute a significant market segment, requiring advanced tissue engineering platforms for fundamental research and clinical translation studies. The growing emphasis on personalized medicine has intensified demand for technologies capable of producing patient-specific tissue constructs using autologous cells, necessitating flexible and scalable manufacturing processes.
The convergence of digital health initiatives and tissue engineering is creating new market opportunities in precision medicine and therapeutic monitoring. Healthcare providers are seeking integrated solutions that combine advanced tissue manufacturing with digital tracking and quality control systems, reflecting the industry's evolution toward more sophisticated and traceable therapeutic approaches.
Healthcare systems worldwide are increasingly recognizing the economic burden of conventional treatments for tissue-related disorders. Chronic wounds, cardiovascular diseases, orthopedic conditions, and organ failures collectively represent billions in annual healthcare expenditures. The potential for tissue engineering technologies to reduce long-term treatment costs while improving patient outcomes has attracted significant attention from healthcare providers, insurance companies, and government health agencies seeking cost-effective solutions.
The pharmaceutical and biotechnology industries are driving substantial demand for advanced tissue models that can replace animal testing and improve drug development processes. Regulatory agencies are increasingly encouraging the adoption of alternative testing methods, creating a robust market for sophisticated in vitro tissue models. These applications require precise control over tissue architecture and cellular organization, making advanced manufacturing techniques essential for meeting industry standards and regulatory requirements.
Regenerative medicine applications represent the fastest-growing segment within tissue engineering markets. The demand spans multiple therapeutic areas including cardiovascular repair, neural tissue regeneration, skin grafts, bone and cartilage reconstruction, and liver tissue engineering. Each application presents unique technical requirements for tissue architecture, mechanical properties, and biological functionality, driving the need for versatile and precise manufacturing approaches.
Research institutions and academic medical centers constitute a significant market segment, requiring advanced tissue engineering platforms for fundamental research and clinical translation studies. The growing emphasis on personalized medicine has intensified demand for technologies capable of producing patient-specific tissue constructs using autologous cells, necessitating flexible and scalable manufacturing processes.
The convergence of digital health initiatives and tissue engineering is creating new market opportunities in precision medicine and therapeutic monitoring. Healthcare providers are seeking integrated solutions that combine advanced tissue manufacturing with digital tracking and quality control systems, reflecting the industry's evolution toward more sophisticated and traceable therapeutic approaches.
Current State and Challenges in Bioprinting and Microfabrication
The current landscape of 3D bioprinting and microfabrication techniques presents a complex array of technological capabilities alongside significant implementation challenges. Both fields have achieved remarkable progress in recent years, yet face distinct limitations that constrain their widespread adoption in clinical and commercial applications.
3D bioprinting technology has advanced considerably in terms of material compatibility and printing resolution. Current systems can successfully process various bioinks including hydrogels, cell-laden materials, and decellularized extracellular matrix components. Leading bioprinting platforms achieve layer resolutions ranging from 50-200 micrometers, with some specialized systems reaching sub-10 micrometer precision. However, the technology struggles with printing speed limitations, often requiring hours to complete complex tissue constructs, during which cell viability may be compromised.
Microfabrication techniques, particularly photolithography and soft lithography, demonstrate superior precision and reproducibility compared to bioprinting methods. These approaches can consistently produce features at nanometer scales and maintain excellent dimensional accuracy across large substrate areas. Established semiconductor manufacturing processes have been successfully adapted for biological applications, enabling high-throughput production of microfluidic devices and cell culture platforms.
Despite these advances, both technologies face critical challenges in biological compatibility and scalability. Bioprinting systems encounter difficulties in maintaining uniform cell distribution and viability throughout the printing process, particularly when working with multiple cell types or complex geometries. The limited selection of biocompatible printing materials that possess appropriate mechanical properties further constrains design flexibility.
Microfabrication techniques, while offering superior precision, are limited by their predominantly two-dimensional nature and reliance on potentially cytotoxic processing chemicals. Creating truly three-dimensional structures requires multiple fabrication steps and complex assembly processes, increasing production costs and potential failure points.
Manufacturing scalability represents another significant challenge for both approaches. Bioprinting systems typically operate as single-unit production platforms with limited throughput capacity, making them unsuitable for large-scale manufacturing applications. Microfabrication, though inherently scalable through established semiconductor processes, requires substantial capital investment in cleanroom facilities and specialized equipment.
Integration challenges also persist in combining these technologies effectively. While hybrid approaches show promise for leveraging the strengths of both methods, current integration strategies often result in increased complexity and reduced overall system reliability, limiting their practical implementation in production environments.
3D bioprinting technology has advanced considerably in terms of material compatibility and printing resolution. Current systems can successfully process various bioinks including hydrogels, cell-laden materials, and decellularized extracellular matrix components. Leading bioprinting platforms achieve layer resolutions ranging from 50-200 micrometers, with some specialized systems reaching sub-10 micrometer precision. However, the technology struggles with printing speed limitations, often requiring hours to complete complex tissue constructs, during which cell viability may be compromised.
Microfabrication techniques, particularly photolithography and soft lithography, demonstrate superior precision and reproducibility compared to bioprinting methods. These approaches can consistently produce features at nanometer scales and maintain excellent dimensional accuracy across large substrate areas. Established semiconductor manufacturing processes have been successfully adapted for biological applications, enabling high-throughput production of microfluidic devices and cell culture platforms.
Despite these advances, both technologies face critical challenges in biological compatibility and scalability. Bioprinting systems encounter difficulties in maintaining uniform cell distribution and viability throughout the printing process, particularly when working with multiple cell types or complex geometries. The limited selection of biocompatible printing materials that possess appropriate mechanical properties further constrains design flexibility.
Microfabrication techniques, while offering superior precision, are limited by their predominantly two-dimensional nature and reliance on potentially cytotoxic processing chemicals. Creating truly three-dimensional structures requires multiple fabrication steps and complex assembly processes, increasing production costs and potential failure points.
Manufacturing scalability represents another significant challenge for both approaches. Bioprinting systems typically operate as single-unit production platforms with limited throughput capacity, making them unsuitable for large-scale manufacturing applications. Microfabrication, though inherently scalable through established semiconductor processes, requires substantial capital investment in cleanroom facilities and specialized equipment.
Integration challenges also persist in combining these technologies effectively. While hybrid approaches show promise for leveraging the strengths of both methods, current integration strategies often result in increased complexity and reduced overall system reliability, limiting their practical implementation in production environments.
Current Technical Solutions in Bioprinting vs Microfabrication
01 Bioink formulations and cell-laden hydrogels for 3D bioprinting
Development of specialized bioink compositions that incorporate living cells within hydrogel matrices to enable precise 3D bioprinting of tissue constructs. These formulations focus on optimizing rheological properties, cell viability, and printability while maintaining biocompatibility. The bioinks are designed to support cell proliferation and differentiation during and after the printing process, enabling the creation of complex tissue architectures with controlled spatial distribution of multiple cell types.- Bioink formulations and material compositions for 3D bioprinting: Development of specialized bioink materials that incorporate living cells, biomaterials, and growth factors to create printable compositions suitable for tissue engineering. These formulations focus on achieving optimal viscosity, cell viability, and structural integrity during and after the printing process. The bioinks are designed to mimic natural extracellular matrix properties while maintaining printability and supporting cell proliferation and differentiation.
- Multi-material and multi-nozzle bioprinting systems: Advanced bioprinting apparatus featuring multiple print heads or nozzles capable of simultaneously depositing different cell types, biomaterials, or bioinks. These systems enable the creation of complex heterogeneous tissue structures with precise spatial control over material placement. The technology allows for gradient formations and intricate architectural designs that better replicate native tissue organization.
- Microfabrication techniques for scaffold and microstructure production: Methods for creating microscale features and scaffolds using techniques such as photolithography, soft lithography, and micromolding. These approaches enable precise control over pore size, channel dimensions, and surface topography at the micrometer scale. The fabricated structures provide mechanical support and guide cellular organization in tissue engineering applications.
- Layer-by-layer assembly and crosslinking strategies: Techniques for sequential deposition and stabilization of bioprinted layers through various crosslinking mechanisms including photocrosslinking, thermal gelation, and chemical crosslinking. These methods ensure structural stability of printed constructs while maintaining cell viability. The strategies focus on optimizing the timing and conditions of crosslinking to achieve desired mechanical properties and biological functionality.
- Vascularization and perfusion systems in bioprinted constructs: Integration of vascular networks and perfusion channels within bioprinted tissues to enable nutrient delivery and waste removal. These systems incorporate methods for creating hollow channels, embedding endothelial cells, and establishing functional blood vessel-like structures. The technology addresses the critical challenge of maintaining cell viability in thick tissue constructs by ensuring adequate mass transport throughout the printed structure.
02 Multi-material and multi-nozzle bioprinting systems
Advanced bioprinting apparatus featuring multiple print heads or nozzles capable of simultaneously depositing different biomaterials and cell types. These systems enable the fabrication of heterogeneous tissue structures with spatially defined compositions and properties. The technology allows for precise control over material deposition patterns, layer-by-layer assembly, and the creation of complex geometries that mimic native tissue organization and functionality.Expand Specific Solutions03 Microfluidic and microfabrication platforms for tissue engineering
Microfabrication techniques utilizing microfluidic devices and micro-scale manufacturing processes to create precise tissue constructs and organ-on-chip systems. These platforms enable controlled manipulation of cells and biomaterials at the microscale, facilitating the generation of tissue models with defined microarchitecture. The technology supports high-throughput fabrication, real-time monitoring, and the creation of vascularized tissue structures through integrated channel networks.Expand Specific Solutions04 Scaffold-based bioprinting and support structure design
Methods for creating biodegradable scaffolds and support structures that provide mechanical stability and guide tissue formation in bioprinted constructs. These approaches involve the design and fabrication of porous architectures with controlled pore size, interconnectivity, and degradation rates. The scaffolds serve as temporary frameworks that support cell attachment, migration, and tissue maturation while gradually degrading as native extracellular matrix is deposited by cells.Expand Specific Solutions05 Post-printing processing and tissue maturation techniques
Protocols and systems for post-bioprinting treatment of tissue constructs to enhance structural integrity, promote cell maturation, and improve functional properties. These techniques include crosslinking methods, bioreactor cultivation, mechanical stimulation, and controlled environmental conditions that facilitate tissue development. The processes aim to transform freshly printed constructs into mature, functional tissues with appropriate mechanical properties and biological characteristics suitable for implantation or in vitro applications.Expand Specific Solutions
Core Patents in 3D Bioprinting and Microfabrication
Method for producing a three-dimensional biological structure and said structure thus obtained
PatentInactiveUS20200263138A1
Innovation
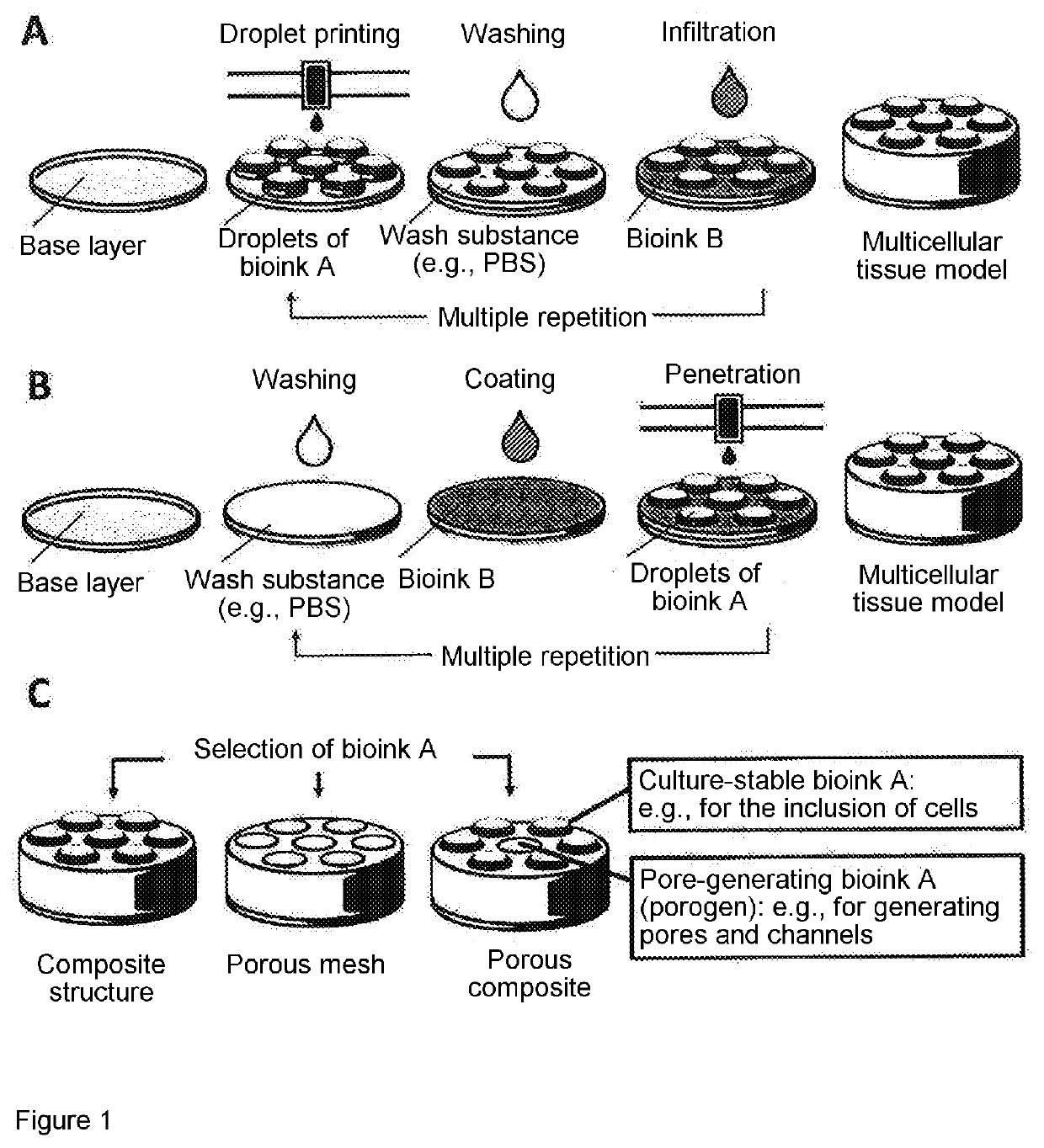
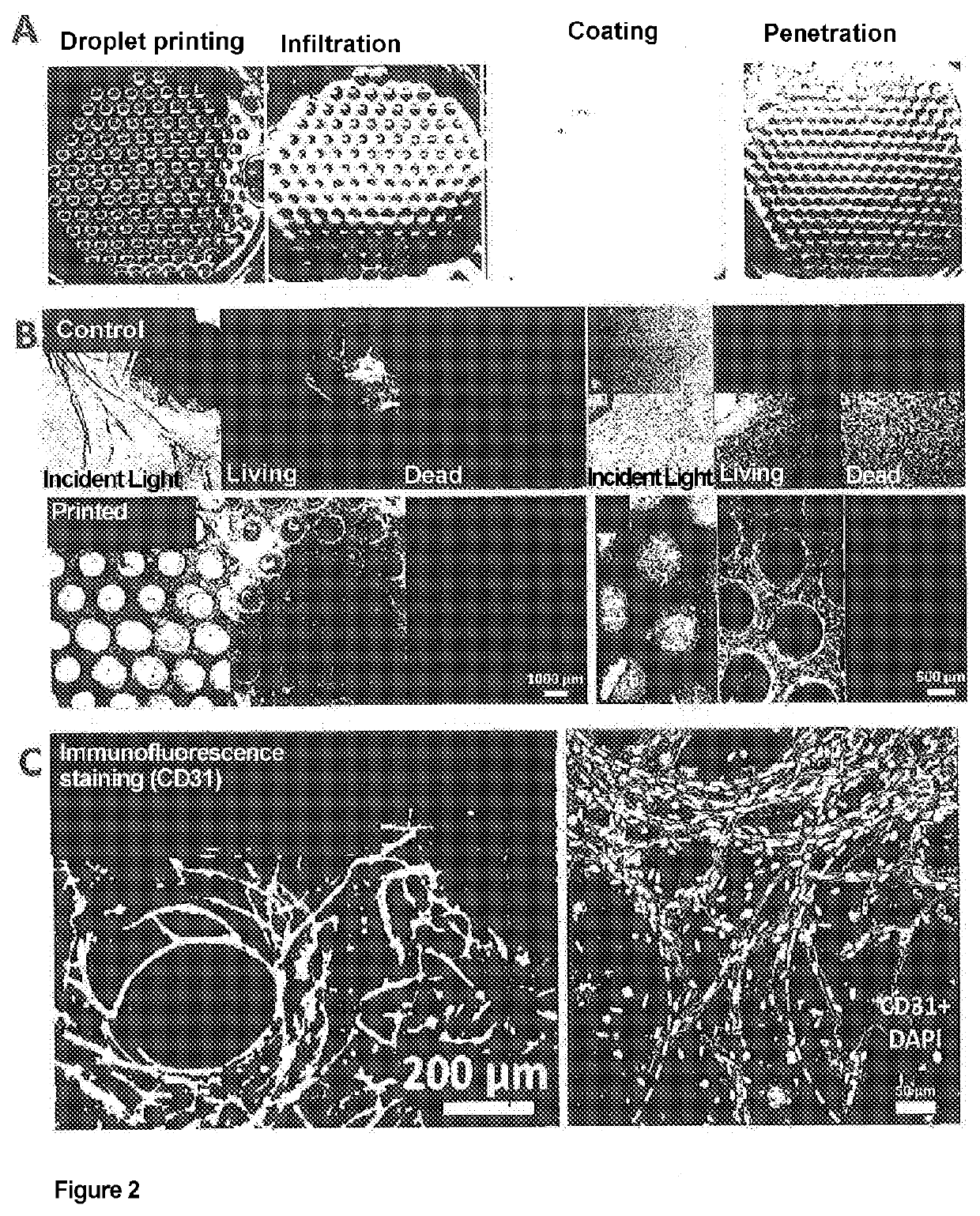
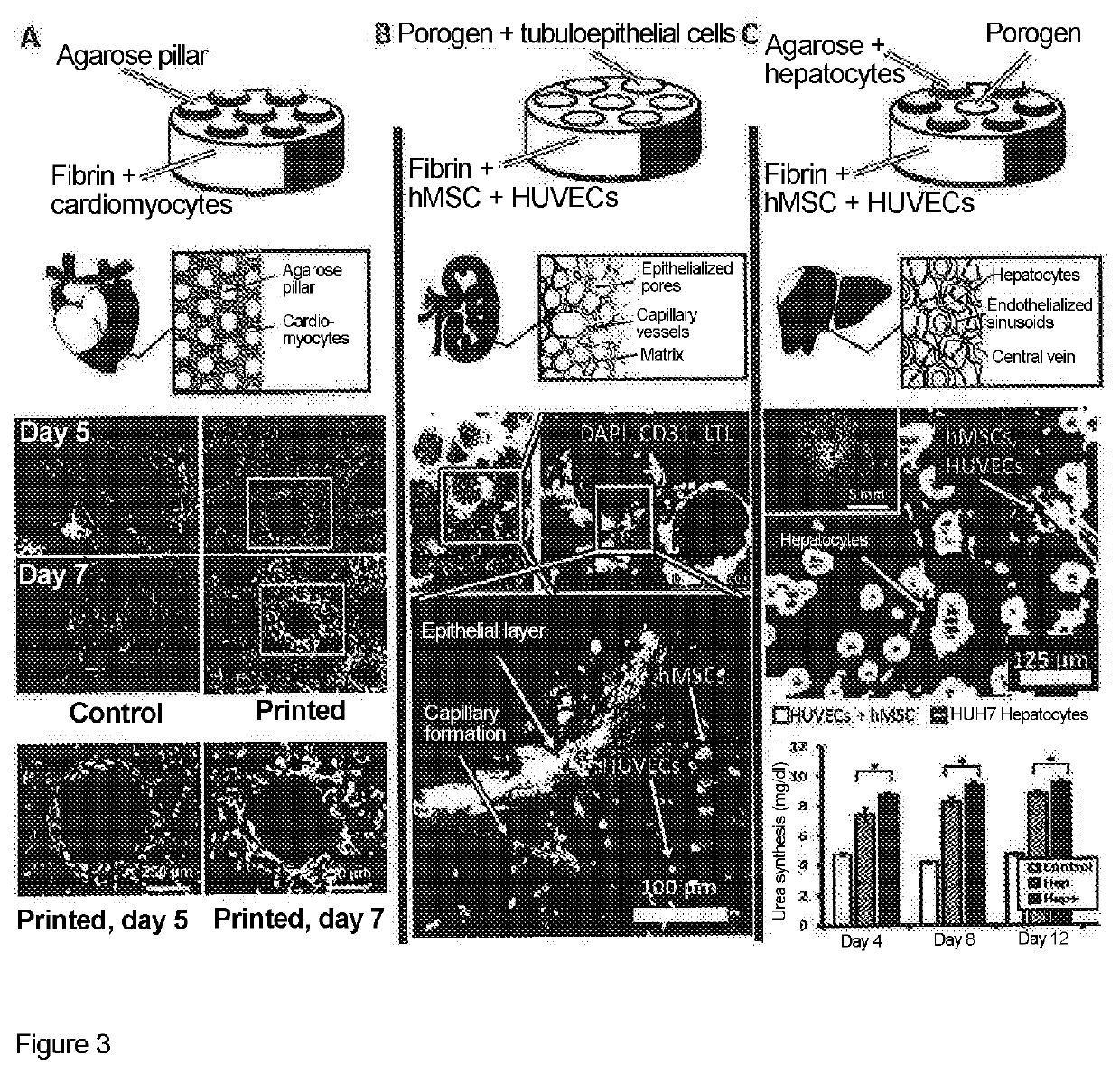
- A drop-on-demand bioprinting process that uses at least two different materials to form subregions, with one material applied by droplet printing, allowing for the creation of complex tissue structures by focusing on biological function rather than anatomy, and enabling the formation of various tissue types through infiltration or penetration methods.
Three dimensional microtissue bioprinter
PatentPendingUS20220323649A1
Innovation
- A computer-controlled, programmable 3D bioprinter with dispensing units featuring a syringe, actuator, and nozzle system that allows for precise XYZ motion and nanoliter volumetric dispensing of bioink, including stem cells, cartilage cells, and extracellular matrix, enabling the creation of microtissues with morphology and function similar to native tissues.
Regulatory Framework for Bioprinted Medical Products
The regulatory landscape for bioprinted medical products represents one of the most complex and evolving areas in biotechnology governance. Current frameworks primarily rely on existing medical device regulations, with agencies like the FDA, EMA, and other national authorities adapting traditional approval pathways to accommodate these novel manufacturing technologies. The challenge lies in the fundamental difference between conventional medical devices and bioprinted products, which often incorporate living cells, biomaterials, and patient-specific customization.
Regulatory classification varies significantly depending on the intended use and complexity of bioprinted products. Simple bioprinted scaffolds without living cells typically fall under medical device regulations, following Class II or III pathways based on risk assessment. However, products incorporating viable cells face additional scrutiny under biologics regulations, requiring extensive safety and efficacy data. The FDA has established specific guidance documents addressing tissue-engineered medical products, while the EMA has developed advanced therapy medicinal product regulations that encompass certain bioprinted applications.
Quality control and manufacturing standards present unique challenges for bioprinted medical products. Traditional Good Manufacturing Practice guidelines require adaptation to address the variability inherent in biological materials and the complexity of bioprinting processes. Regulatory bodies are developing new frameworks that emphasize process validation, material traceability, and real-time quality monitoring throughout the bioprinting workflow.
International harmonization efforts are underway to establish consistent regulatory approaches across different jurisdictions. Organizations like the International Council for Harmonisation are working to develop unified standards for bioprinted medical products, though significant variations remain between regions. The regulatory pathway often requires extensive preclinical testing, including biocompatibility studies, sterility validation, and long-term stability assessments.
Emerging regulatory considerations include patient-specific customization protocols, data integrity requirements for digital manufacturing files, and post-market surveillance strategies tailored to bioprinted products. As the technology matures, regulatory frameworks continue evolving to balance innovation acceleration with patient safety assurance.
Regulatory classification varies significantly depending on the intended use and complexity of bioprinted products. Simple bioprinted scaffolds without living cells typically fall under medical device regulations, following Class II or III pathways based on risk assessment. However, products incorporating viable cells face additional scrutiny under biologics regulations, requiring extensive safety and efficacy data. The FDA has established specific guidance documents addressing tissue-engineered medical products, while the EMA has developed advanced therapy medicinal product regulations that encompass certain bioprinted applications.
Quality control and manufacturing standards present unique challenges for bioprinted medical products. Traditional Good Manufacturing Practice guidelines require adaptation to address the variability inherent in biological materials and the complexity of bioprinting processes. Regulatory bodies are developing new frameworks that emphasize process validation, material traceability, and real-time quality monitoring throughout the bioprinting workflow.
International harmonization efforts are underway to establish consistent regulatory approaches across different jurisdictions. Organizations like the International Council for Harmonisation are working to develop unified standards for bioprinted medical products, though significant variations remain between regions. The regulatory pathway often requires extensive preclinical testing, including biocompatibility studies, sterility validation, and long-term stability assessments.
Emerging regulatory considerations include patient-specific customization protocols, data integrity requirements for digital manufacturing files, and post-market surveillance strategies tailored to bioprinted products. As the technology matures, regulatory frameworks continue evolving to balance innovation acceleration with patient safety assurance.
Bioethical Considerations in Tissue Engineering Manufacturing
The advancement of 3D bioprinting and microfabrication techniques in tissue engineering manufacturing has introduced unprecedented capabilities for creating complex biological structures, yet these innovations simultaneously raise profound ethical questions that demand careful consideration. The ability to fabricate living tissues and potentially entire organs challenges traditional boundaries between natural and artificial biological systems, necessitating a comprehensive ethical framework to guide responsible development and implementation.
Patient autonomy and informed consent represent fundamental ethical pillars in tissue engineering manufacturing. When utilizing these advanced fabrication techniques, patients must be thoroughly informed about the nature of bioprinted or microfabricated tissues, including their synthetic origins, potential limitations, and long-term implications. The complexity of these technologies requires enhanced consent processes that ensure patients truly understand the experimental nature of such treatments and their participation in advancing medical science.
The question of biological ownership emerges as a critical concern when comparing these manufacturing approaches. Both 3D bioprinting and microfabrication may utilize patient-derived cells or genetic material, raising questions about intellectual property rights over the resulting tissues. The commercialization potential of these technologies creates tension between patient rights and corporate interests, particularly when considering who owns the engineered tissues and any subsequent medical breakthroughs derived from them.
Equity and accessibility concerns become amplified when examining the resource-intensive nature of both manufacturing techniques. The sophisticated equipment, specialized materials, and technical expertise required for 3D bioprinting and microfabrication may create disparities in healthcare access. These technologies risk exacerbating existing healthcare inequalities if they remain available only to affluent populations or developed nations, raising questions about distributive justice in medical innovation.
The enhancement versus treatment distinction presents another ethical dimension unique to these manufacturing capabilities. While therapeutic applications for replacing damaged tissues align with traditional medical goals, the potential for enhancement applications challenges conventional bioethical boundaries. Both techniques could theoretically create tissues with enhanced properties beyond normal human capabilities, requiring careful consideration of what constitutes appropriate medical intervention.
Safety and precautionary principles demand rigorous evaluation given the experimental nature of both manufacturing approaches. The long-term effects of implanting bioprinted or microfabricated tissues remain largely unknown, necessitating robust risk assessment protocols and ongoing monitoring systems. The irreversible nature of many tissue engineering interventions amplifies the importance of comprehensive safety evaluation before clinical implementation.
Human dignity considerations encompass concerns about commodification of human biology through industrial manufacturing processes. The transformation of tissue creation from natural biological processes to controlled manufacturing environments raises philosophical questions about the sanctity of human biological systems and the appropriate limits of technological intervention in human biology.
Regulatory frameworks must evolve to address the unique ethical challenges posed by these manufacturing techniques while fostering continued innovation. International cooperation becomes essential to establish consistent ethical standards that prevent regulatory arbitrage while ensuring responsible development of these transformative technologies across global research communities.
Patient autonomy and informed consent represent fundamental ethical pillars in tissue engineering manufacturing. When utilizing these advanced fabrication techniques, patients must be thoroughly informed about the nature of bioprinted or microfabricated tissues, including their synthetic origins, potential limitations, and long-term implications. The complexity of these technologies requires enhanced consent processes that ensure patients truly understand the experimental nature of such treatments and their participation in advancing medical science.
The question of biological ownership emerges as a critical concern when comparing these manufacturing approaches. Both 3D bioprinting and microfabrication may utilize patient-derived cells or genetic material, raising questions about intellectual property rights over the resulting tissues. The commercialization potential of these technologies creates tension between patient rights and corporate interests, particularly when considering who owns the engineered tissues and any subsequent medical breakthroughs derived from them.
Equity and accessibility concerns become amplified when examining the resource-intensive nature of both manufacturing techniques. The sophisticated equipment, specialized materials, and technical expertise required for 3D bioprinting and microfabrication may create disparities in healthcare access. These technologies risk exacerbating existing healthcare inequalities if they remain available only to affluent populations or developed nations, raising questions about distributive justice in medical innovation.
The enhancement versus treatment distinction presents another ethical dimension unique to these manufacturing capabilities. While therapeutic applications for replacing damaged tissues align with traditional medical goals, the potential for enhancement applications challenges conventional bioethical boundaries. Both techniques could theoretically create tissues with enhanced properties beyond normal human capabilities, requiring careful consideration of what constitutes appropriate medical intervention.
Safety and precautionary principles demand rigorous evaluation given the experimental nature of both manufacturing approaches. The long-term effects of implanting bioprinted or microfabricated tissues remain largely unknown, necessitating robust risk assessment protocols and ongoing monitoring systems. The irreversible nature of many tissue engineering interventions amplifies the importance of comprehensive safety evaluation before clinical implementation.
Human dignity considerations encompass concerns about commodification of human biology through industrial manufacturing processes. The transformation of tissue creation from natural biological processes to controlled manufacturing environments raises philosophical questions about the sanctity of human biological systems and the appropriate limits of technological intervention in human biology.
Regulatory frameworks must evolve to address the unique ethical challenges posed by these manufacturing techniques while fostering continued innovation. International cooperation becomes essential to establish consistent ethical standards that prevent regulatory arbitrage while ensuring responsible development of these transformative technologies across global research communities.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!