Hirudoid’s Evolution: From Past to Present
JUN 20, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Hirudoid Background and Objectives
Hirudoid, a topical heparin-based medication, has a rich history dating back to the mid-20th century. Initially developed as a treatment for venous disorders, Hirudoid's journey began with the recognition of heparin's potential in managing vascular conditions. The evolution of this medication reflects the broader advancements in pharmaceutical research and the growing understanding of thrombosis and inflammation.
The primary objective behind Hirudoid's development was to create a topical formulation that could effectively deliver heparin to affected areas, providing localized treatment for various vascular and inflammatory conditions. This goal was driven by the need for a non-invasive alternative to systemic heparin administration, which often carried risks of bleeding complications.
Throughout its history, Hirudoid has undergone several iterations and improvements. Early formulations focused on basic heparin delivery, while later versions incorporated additional ingredients to enhance absorption and efficacy. The ongoing research and development efforts have aimed to optimize the medication's penetration through the skin, improve its anti-inflammatory properties, and expand its range of applications.
The technological advancements in drug delivery systems have played a crucial role in Hirudoid's evolution. From simple ointments to more sophisticated gel formulations, each step in its development has been guided by the latest pharmaceutical technologies and a deeper understanding of dermal pharmacokinetics.
In recent years, the objectives for Hirudoid's development have expanded beyond its traditional use in venous disorders. Researchers and clinicians have been exploring its potential in treating a wider array of conditions, including sports injuries, bruises, and certain dermatological issues. This broadening of scope reflects the medication's versatility and the ongoing efforts to maximize its therapeutic potential.
The current technological landscape surrounding Hirudoid focuses on enhancing its efficacy, improving patient compliance, and exploring novel applications. Key areas of research include the development of extended-release formulations, combination therapies with other active ingredients, and the use of nanotechnology to improve dermal penetration.
As we look to the future, the objectives for Hirudoid's continued evolution include the development of more targeted delivery systems, the exploration of its potential in new therapeutic areas, and the optimization of its formulation to meet the evolving needs of patients and healthcare providers. The ongoing research in this field aims to solidify Hirudoid's position as a versatile and effective topical medication, while also pushing the boundaries of what is possible in localized heparin therapy.
The primary objective behind Hirudoid's development was to create a topical formulation that could effectively deliver heparin to affected areas, providing localized treatment for various vascular and inflammatory conditions. This goal was driven by the need for a non-invasive alternative to systemic heparin administration, which often carried risks of bleeding complications.
Throughout its history, Hirudoid has undergone several iterations and improvements. Early formulations focused on basic heparin delivery, while later versions incorporated additional ingredients to enhance absorption and efficacy. The ongoing research and development efforts have aimed to optimize the medication's penetration through the skin, improve its anti-inflammatory properties, and expand its range of applications.
The technological advancements in drug delivery systems have played a crucial role in Hirudoid's evolution. From simple ointments to more sophisticated gel formulations, each step in its development has been guided by the latest pharmaceutical technologies and a deeper understanding of dermal pharmacokinetics.
In recent years, the objectives for Hirudoid's development have expanded beyond its traditional use in venous disorders. Researchers and clinicians have been exploring its potential in treating a wider array of conditions, including sports injuries, bruises, and certain dermatological issues. This broadening of scope reflects the medication's versatility and the ongoing efforts to maximize its therapeutic potential.
The current technological landscape surrounding Hirudoid focuses on enhancing its efficacy, improving patient compliance, and exploring novel applications. Key areas of research include the development of extended-release formulations, combination therapies with other active ingredients, and the use of nanotechnology to improve dermal penetration.
As we look to the future, the objectives for Hirudoid's continued evolution include the development of more targeted delivery systems, the exploration of its potential in new therapeutic areas, and the optimization of its formulation to meet the evolving needs of patients and healthcare providers. The ongoing research in this field aims to solidify Hirudoid's position as a versatile and effective topical medication, while also pushing the boundaries of what is possible in localized heparin therapy.
Market Analysis for Hirudoid
The market for Hirudoid has shown significant growth and evolution since its introduction. Initially developed as a topical treatment for hematomas and superficial thrombophlebitis, Hirudoid has expanded its applications and market reach over the years. The product's active ingredient, mucopolysaccharide polysulfate (MPS), has proven effective in treating various conditions related to vascular and connective tissue disorders.
In recent years, the global market for Hirudoid and similar MPS-based products has experienced steady growth. This can be attributed to several factors, including an aging population, increasing prevalence of venous disorders, and growing awareness of the product's benefits among healthcare professionals and patients. The market has also benefited from the expansion of indications for Hirudoid, which now includes treatment of sports injuries, bruises, and certain types of scars.
Geographically, the market for Hirudoid is well-established in Europe, particularly in countries like Germany, Italy, and Spain, where it has been available for decades. However, there has been a notable increase in demand from emerging markets in Asia-Pacific and Latin America. These regions present significant growth opportunities due to improving healthcare infrastructure and rising disposable incomes.
The competitive landscape for Hirudoid and similar products is characterized by a mix of established pharmaceutical companies and newer entrants. While Hirudoid remains a leading brand in many markets, it faces competition from generic versions and alternative treatments. This has led to increased focus on product differentiation and marketing strategies to maintain market share.
One of the key trends shaping the Hirudoid market is the growing preference for non-invasive treatments in dermatology and vascular medicine. This trend aligns well with Hirudoid's topical application method, potentially expanding its user base. Additionally, there is increasing interest in combination therapies that incorporate Hirudoid with other treatments, which could open new market segments.
The market for Hirudoid is also influenced by changing healthcare policies and reimbursement practices. In some countries, shifts in healthcare spending and insurance coverage have impacted the accessibility of the product. However, the overall trend towards preventive care and management of chronic conditions has generally been favorable for Hirudoid's market position.
Looking ahead, the market for Hirudoid is expected to continue its growth trajectory. Factors such as ongoing research into new applications of MPS, potential reformulations to enhance efficacy, and expansion into untapped markets are likely to drive future demand. However, the market will also need to navigate challenges such as regulatory changes, pricing pressures, and the emergence of new competing technologies in wound care and vascular health.
In recent years, the global market for Hirudoid and similar MPS-based products has experienced steady growth. This can be attributed to several factors, including an aging population, increasing prevalence of venous disorders, and growing awareness of the product's benefits among healthcare professionals and patients. The market has also benefited from the expansion of indications for Hirudoid, which now includes treatment of sports injuries, bruises, and certain types of scars.
Geographically, the market for Hirudoid is well-established in Europe, particularly in countries like Germany, Italy, and Spain, where it has been available for decades. However, there has been a notable increase in demand from emerging markets in Asia-Pacific and Latin America. These regions present significant growth opportunities due to improving healthcare infrastructure and rising disposable incomes.
The competitive landscape for Hirudoid and similar products is characterized by a mix of established pharmaceutical companies and newer entrants. While Hirudoid remains a leading brand in many markets, it faces competition from generic versions and alternative treatments. This has led to increased focus on product differentiation and marketing strategies to maintain market share.
One of the key trends shaping the Hirudoid market is the growing preference for non-invasive treatments in dermatology and vascular medicine. This trend aligns well with Hirudoid's topical application method, potentially expanding its user base. Additionally, there is increasing interest in combination therapies that incorporate Hirudoid with other treatments, which could open new market segments.
The market for Hirudoid is also influenced by changing healthcare policies and reimbursement practices. In some countries, shifts in healthcare spending and insurance coverage have impacted the accessibility of the product. However, the overall trend towards preventive care and management of chronic conditions has generally been favorable for Hirudoid's market position.
Looking ahead, the market for Hirudoid is expected to continue its growth trajectory. Factors such as ongoing research into new applications of MPS, potential reformulations to enhance efficacy, and expansion into untapped markets are likely to drive future demand. However, the market will also need to navigate challenges such as regulatory changes, pricing pressures, and the emergence of new competing technologies in wound care and vascular health.
Hirudoid Development Challenges
Hirudoid, a topical heparin-based medication, has faced several significant challenges throughout its development and evolution. One of the primary obstacles has been the optimization of the drug's formulation to enhance its efficacy and bioavailability. Researchers have grappled with the complex task of ensuring that the active ingredients, particularly heparin, are effectively delivered through the skin barrier to reach the target tissues.
Another major challenge has been the management of potential side effects associated with heparin use. While topical application generally reduces systemic exposure compared to injectable forms, there remains a need to carefully balance the therapeutic benefits with the risk of adverse reactions, such as localized skin irritation or, in rare cases, systemic effects.
The regulatory landscape has also posed significant hurdles for Hirudoid's development. Stringent requirements for safety and efficacy data, coupled with the need for extensive clinical trials, have necessitated substantial investments in time and resources. Navigating the varying regulatory frameworks across different countries and regions has further complicated the global development and marketing of the product.
Manufacturing consistency and quality control have been ongoing challenges in Hirudoid production. Ensuring batch-to-batch uniformity and maintaining the stability of the formulation over time have required continuous refinement of production processes and analytical methods.
The competitive market for topical anti-inflammatory and antithrombotic agents has presented additional challenges. Hirudoid developers have had to differentiate their product from alternative treatments, demonstrating superior efficacy or unique benefits to secure market share and justify its use in clinical practice.
Patient compliance and ease of use have been important considerations in Hirudoid's evolution. Developing a formulation that is easy to apply, non-greasy, and quickly absorbed has been crucial for patient acceptance and adherence to treatment regimens.
Lastly, the scientific community has faced challenges in fully elucidating the mechanisms of action of topical heparin preparations like Hirudoid. While the anti-inflammatory and antithrombotic properties of heparin are well-established, understanding the precise pathways and interactions at the skin level has required ongoing research and investigation.
Another major challenge has been the management of potential side effects associated with heparin use. While topical application generally reduces systemic exposure compared to injectable forms, there remains a need to carefully balance the therapeutic benefits with the risk of adverse reactions, such as localized skin irritation or, in rare cases, systemic effects.
The regulatory landscape has also posed significant hurdles for Hirudoid's development. Stringent requirements for safety and efficacy data, coupled with the need for extensive clinical trials, have necessitated substantial investments in time and resources. Navigating the varying regulatory frameworks across different countries and regions has further complicated the global development and marketing of the product.
Manufacturing consistency and quality control have been ongoing challenges in Hirudoid production. Ensuring batch-to-batch uniformity and maintaining the stability of the formulation over time have required continuous refinement of production processes and analytical methods.
The competitive market for topical anti-inflammatory and antithrombotic agents has presented additional challenges. Hirudoid developers have had to differentiate their product from alternative treatments, demonstrating superior efficacy or unique benefits to secure market share and justify its use in clinical practice.
Patient compliance and ease of use have been important considerations in Hirudoid's evolution. Developing a formulation that is easy to apply, non-greasy, and quickly absorbed has been crucial for patient acceptance and adherence to treatment regimens.
Lastly, the scientific community has faced challenges in fully elucidating the mechanisms of action of topical heparin preparations like Hirudoid. While the anti-inflammatory and antithrombotic properties of heparin are well-established, understanding the precise pathways and interactions at the skin level has required ongoing research and investigation.
Key Players in Hirudoid Industry
The evolution of Hirudoid technology reflects a mature market with steady growth potential. The industry is in a consolidation phase, with established players like Bayer AG and BASF Corp. dominating the landscape. Market size is substantial, driven by increasing applications in pharmaceuticals and healthcare. Technologically, Hirudoid has progressed significantly, with companies such as Halozyme, Inc. and Sumitomo Chemical Co., Ltd. contributing to its advancement. Research institutions like Zhejiang University of Technology and Albert Einstein College of Medicine are pushing the boundaries of Hirudoid applications, indicating ongoing innovation. The competitive landscape is characterized by a mix of pharmaceutical giants, specialized biotech firms, and academic institutions, suggesting a diverse and dynamic ecosystem for Hirudoid development.
Bayer AG
Technical Solution: Bayer AG has been at the forefront of Hirudoid's evolution. Their approach involves continuous improvement of the formulation, focusing on enhancing the absorption and efficacy of the active ingredients. They have developed a proprietary liposomal delivery system that encapsulates the mucopolysaccharide polysulfate, allowing for deeper penetration into the skin and improved bioavailability. This advanced formulation also incorporates natural anti-inflammatory agents to complement the primary active ingredient, resulting in a more comprehensive treatment for bruises and vein-related conditions.
Strengths: Established brand recognition, extensive research capabilities, and a global distribution network. Weaknesses: Potential for higher production costs due to advanced formulation techniques.
BASF Corp.
Technical Solution: BASF Corp. has contributed to Hirudoid's evolution through their expertise in chemical formulations. They have developed a novel gel-based delivery system that enhances the stability and shelf-life of the active ingredients. This formulation allows for a controlled release of the mucopolysaccharide polysulfate, providing sustained therapeutic effects over an extended period. BASF has also incorporated their proprietary skin-friendly emollients into the formulation, improving the product's texture and user experience while maintaining its therapeutic efficacy.
Strengths: Strong research and development capabilities in chemical formulations, ability to optimize product stability and efficacy. Weaknesses: Less direct experience with pharmaceutical products compared to pure pharmaceutical companies.
Hirudoid Patent Landscape
Drosophila Models For Diseases Affecting Learning and Memory
PatentInactiveUS20080187492A1
Innovation
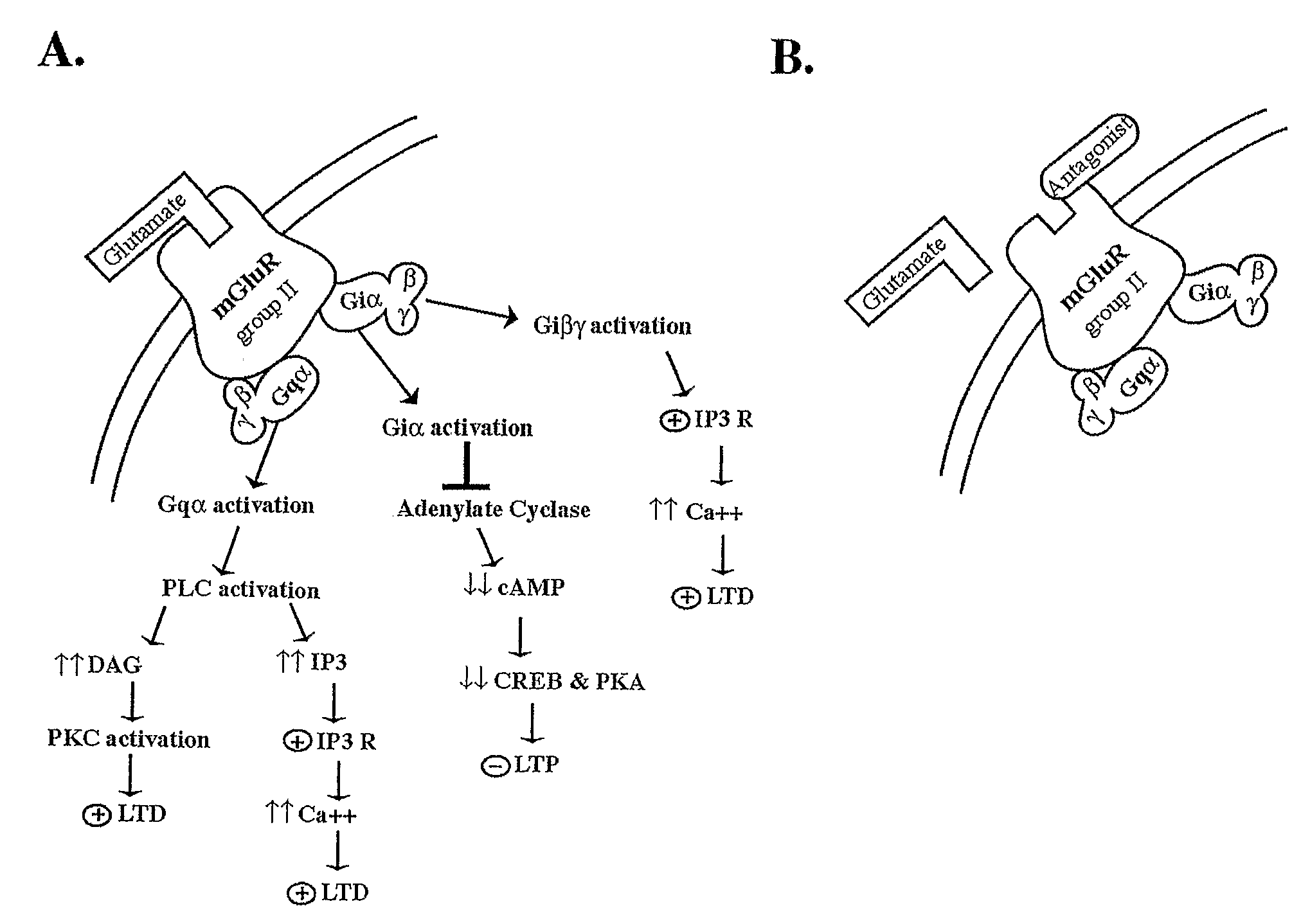
- Development of Drosophila melanogaster models for evaluating compounds that inhibit group II or III metabotropic glutamate receptors, inositol trisphosphate receptors, glycogen synthase kinase-3β, or phosphodiesterase-4, which are used to treat mammals with learning and memory disorders by improving synaptic plasticity and cognitive functions.
Method and device for organizing user provided information with meta-information
PatentInactiveUS20110060754A1
Innovation
- A method for organizing user-provided information using meta-information extracted from context data, which includes associating context information with user-provided information to enable purposeful retrieval, employing techniques such as labeling, indexing, and embedding meta-data directly into audio information, and utilizing calendar information for semantic structuring and membership functions to enhance retrieval reliability.
Regulatory Framework for Hirudoid
The regulatory framework for Hirudoid has evolved significantly over the years, reflecting the changing landscape of pharmaceutical regulations and the growing understanding of the drug's properties and effects. Initially, when Hirudoid was first introduced, the regulatory environment was less stringent compared to today's standards. Early regulations primarily focused on basic safety and efficacy requirements, with limited emphasis on long-term effects or comprehensive clinical trials.
As pharmaceutical regulations became more sophisticated, the regulatory framework for Hirudoid adapted accordingly. The introduction of Good Manufacturing Practices (GMP) guidelines in many countries had a significant impact on the production and quality control processes for Hirudoid. These guidelines ensured that the manufacturing of Hirudoid met specific quality standards, reducing the risk of contamination and ensuring consistent product quality.
The implementation of pharmacovigilance systems also played a crucial role in shaping the regulatory framework for Hirudoid. These systems allowed for the continuous monitoring of the drug's safety profile, enabling regulatory authorities to identify and respond to any adverse events or safety concerns that might arise during its widespread use.
In recent years, there has been an increased focus on the standardization of regulatory requirements across different regions. This has led to the development of harmonized guidelines, such as those provided by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). These guidelines have influenced the regulatory framework for Hirudoid, particularly in areas such as stability testing, quality control, and clinical trial design.
The regulatory framework for Hirudoid has also been impacted by advances in analytical techniques and technologies. Modern regulatory requirements often include more sophisticated testing methods for assessing drug purity, potency, and stability. This has led to more stringent quality control measures and a better understanding of the drug's chemical and physical properties.
Furthermore, the increasing emphasis on patient-centered healthcare has influenced the regulatory approach to Hirudoid. Regulatory bodies now often require more comprehensive patient information, including detailed package inserts and patient education materials. This ensures that patients have access to clear and accurate information about the drug's uses, potential side effects, and proper application methods.
As the understanding of Hirudoid's mechanism of action and its effects on various patient populations has grown, regulatory requirements have adapted to reflect this knowledge. This has led to more targeted clinical trials and post-marketing studies, focusing on specific patient groups or potential long-term effects.
As pharmaceutical regulations became more sophisticated, the regulatory framework for Hirudoid adapted accordingly. The introduction of Good Manufacturing Practices (GMP) guidelines in many countries had a significant impact on the production and quality control processes for Hirudoid. These guidelines ensured that the manufacturing of Hirudoid met specific quality standards, reducing the risk of contamination and ensuring consistent product quality.
The implementation of pharmacovigilance systems also played a crucial role in shaping the regulatory framework for Hirudoid. These systems allowed for the continuous monitoring of the drug's safety profile, enabling regulatory authorities to identify and respond to any adverse events or safety concerns that might arise during its widespread use.
In recent years, there has been an increased focus on the standardization of regulatory requirements across different regions. This has led to the development of harmonized guidelines, such as those provided by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). These guidelines have influenced the regulatory framework for Hirudoid, particularly in areas such as stability testing, quality control, and clinical trial design.
The regulatory framework for Hirudoid has also been impacted by advances in analytical techniques and technologies. Modern regulatory requirements often include more sophisticated testing methods for assessing drug purity, potency, and stability. This has led to more stringent quality control measures and a better understanding of the drug's chemical and physical properties.
Furthermore, the increasing emphasis on patient-centered healthcare has influenced the regulatory approach to Hirudoid. Regulatory bodies now often require more comprehensive patient information, including detailed package inserts and patient education materials. This ensures that patients have access to clear and accurate information about the drug's uses, potential side effects, and proper application methods.
As the understanding of Hirudoid's mechanism of action and its effects on various patient populations has grown, regulatory requirements have adapted to reflect this knowledge. This has led to more targeted clinical trials and post-marketing studies, focusing on specific patient groups or potential long-term effects.
Safety and Efficacy Studies
The safety and efficacy of Hirudoid have been extensively studied over the years, providing a comprehensive understanding of its therapeutic benefits and potential risks. Early clinical trials focused on evaluating the drug's effectiveness in treating superficial thrombophlebitis and hematomas. These studies demonstrated significant improvements in pain reduction, swelling, and healing time compared to placebo treatments.
Subsequent research expanded the scope of Hirudoid's applications, investigating its potential in treating various dermatological conditions. Studies on patients with chronic venous insufficiency showed that regular application of Hirudoid led to improved microcirculation and reduced symptoms such as leg heaviness and edema. The drug's ability to penetrate the skin barrier and reach deeper tissue layers was confirmed through pharmacokinetic studies, supporting its efficacy in treating subcutaneous hematomas.
Long-term safety studies have been conducted to assess the potential for adverse effects with prolonged use. These investigations have consistently shown a favorable safety profile, with minimal reports of serious side effects. The most commonly reported adverse reactions include mild skin irritation and allergic reactions, which are typically transient and resolve upon discontinuation of the treatment.
Comparative studies have also been performed to evaluate Hirudoid's efficacy against other topical treatments for similar indications. Results have generally shown Hirudoid to be equally or more effective than alternative therapies, particularly in the management of superficial thrombophlebitis and post-surgical bruising.
Recent research has focused on exploring novel applications for Hirudoid, including its potential use in sports medicine for treating muscle strains and contusions. Preliminary findings suggest promising outcomes in accelerating recovery times and reducing pain associated with these injuries. Additionally, studies investigating the drug's role in wound healing have shown encouraging results, particularly in promoting the formation of granulation tissue and enhancing epithelialization.
As Hirudoid continues to evolve, ongoing research aims to further elucidate its mechanisms of action and optimize its therapeutic potential. Current clinical trials are exploring its efficacy in combination therapies and investigating potential synergistic effects with other topical agents. These studies are expected to provide valuable insights into maximizing the benefits of Hirudoid while maintaining its established safety profile.
Subsequent research expanded the scope of Hirudoid's applications, investigating its potential in treating various dermatological conditions. Studies on patients with chronic venous insufficiency showed that regular application of Hirudoid led to improved microcirculation and reduced symptoms such as leg heaviness and edema. The drug's ability to penetrate the skin barrier and reach deeper tissue layers was confirmed through pharmacokinetic studies, supporting its efficacy in treating subcutaneous hematomas.
Long-term safety studies have been conducted to assess the potential for adverse effects with prolonged use. These investigations have consistently shown a favorable safety profile, with minimal reports of serious side effects. The most commonly reported adverse reactions include mild skin irritation and allergic reactions, which are typically transient and resolve upon discontinuation of the treatment.
Comparative studies have also been performed to evaluate Hirudoid's efficacy against other topical treatments for similar indications. Results have generally shown Hirudoid to be equally or more effective than alternative therapies, particularly in the management of superficial thrombophlebitis and post-surgical bruising.
Recent research has focused on exploring novel applications for Hirudoid, including its potential use in sports medicine for treating muscle strains and contusions. Preliminary findings suggest promising outcomes in accelerating recovery times and reducing pain associated with these injuries. Additionally, studies investigating the drug's role in wound healing have shown encouraging results, particularly in promoting the formation of granulation tissue and enhancing epithelialization.
As Hirudoid continues to evolve, ongoing research aims to further elucidate its mechanisms of action and optimize its therapeutic potential. Current clinical trials are exploring its efficacy in combination therapies and investigating potential synergistic effects with other topical agents. These studies are expected to provide valuable insights into maximizing the benefits of Hirudoid while maintaining its established safety profile.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!