How Nanotechnology Enhances Lithium Sulfur Battery Functionality
OCT 24, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Nanotechnology in Li-S Batteries: Background and Objectives
Lithium-sulfur (Li-S) batteries have emerged as promising candidates to succeed conventional lithium-ion batteries due to their theoretical energy density of 2600 Wh/kg, which is approximately five times higher than current commercial lithium-ion technologies. The development of Li-S batteries can be traced back to the 1960s when the first conceptual designs were proposed. However, significant progress in this field only began to accelerate in the early 2000s with the advent of advanced nanotechnology techniques and materials science innovations.
The evolution of Li-S battery technology has been characterized by persistent efforts to overcome three fundamental challenges: the insulating nature of sulfur, the shuttle effect caused by soluble polysulfide intermediates, and significant volume expansion during cycling. These challenges have historically limited the practical application of Li-S batteries despite their theoretical advantages. The technological trajectory has shifted from basic sulfur-carbon composites to sophisticated nanostructured materials designed to address these specific limitations.
Current global research trends indicate a growing interest in nanotechnology-based solutions for Li-S batteries, with annual publications in this field increasing by approximately 25% year-over-year since 2015. This surge reflects the scientific community's recognition of nanotechnology's potential to revolutionize energy storage systems through precise material engineering at the nanoscale.
The primary technical objectives for nanotechnology in Li-S batteries include enhancing sulfur utilization through conductive nanostructures, mitigating polysulfide shuttling via physical confinement and chemical binding strategies, and accommodating volume changes through flexible nanoarchitectures. Additionally, researchers aim to improve the ionic and electronic conductivity of cathode materials, enhance the stability of the solid-electrolyte interphase, and develop novel electrolyte systems compatible with sulfur electrochemistry.
Recent breakthroughs in carbon nanomaterials, metal-organic frameworks, and two-dimensional materials have provided new platforms for sulfur immobilization and polysulfide trapping. Concurrently, advances in atomic layer deposition and solution-based synthesis methods have enabled precise control over nanostructure formation, facilitating the development of multifunctional materials tailored for Li-S battery applications.
The anticipated trajectory for nanotechnology in Li-S batteries points toward integrated approaches that simultaneously address multiple failure mechanisms through hierarchical nanostructures and multifunctional interfaces. The ultimate goal is to develop commercially viable Li-S batteries with energy densities exceeding 500 Wh/kg, cycle life greater than 1000 cycles, and cost-effective manufacturing processes that can be scaled for mass production.
The evolution of Li-S battery technology has been characterized by persistent efforts to overcome three fundamental challenges: the insulating nature of sulfur, the shuttle effect caused by soluble polysulfide intermediates, and significant volume expansion during cycling. These challenges have historically limited the practical application of Li-S batteries despite their theoretical advantages. The technological trajectory has shifted from basic sulfur-carbon composites to sophisticated nanostructured materials designed to address these specific limitations.
Current global research trends indicate a growing interest in nanotechnology-based solutions for Li-S batteries, with annual publications in this field increasing by approximately 25% year-over-year since 2015. This surge reflects the scientific community's recognition of nanotechnology's potential to revolutionize energy storage systems through precise material engineering at the nanoscale.
The primary technical objectives for nanotechnology in Li-S batteries include enhancing sulfur utilization through conductive nanostructures, mitigating polysulfide shuttling via physical confinement and chemical binding strategies, and accommodating volume changes through flexible nanoarchitectures. Additionally, researchers aim to improve the ionic and electronic conductivity of cathode materials, enhance the stability of the solid-electrolyte interphase, and develop novel electrolyte systems compatible with sulfur electrochemistry.
Recent breakthroughs in carbon nanomaterials, metal-organic frameworks, and two-dimensional materials have provided new platforms for sulfur immobilization and polysulfide trapping. Concurrently, advances in atomic layer deposition and solution-based synthesis methods have enabled precise control over nanostructure formation, facilitating the development of multifunctional materials tailored for Li-S battery applications.
The anticipated trajectory for nanotechnology in Li-S batteries points toward integrated approaches that simultaneously address multiple failure mechanisms through hierarchical nanostructures and multifunctional interfaces. The ultimate goal is to develop commercially viable Li-S batteries with energy densities exceeding 500 Wh/kg, cycle life greater than 1000 cycles, and cost-effective manufacturing processes that can be scaled for mass production.
Market Analysis for Next-Generation Energy Storage Solutions
The global energy storage market is experiencing unprecedented growth, driven by the increasing adoption of renewable energy sources and the electrification of transportation. The market for next-generation energy storage solutions is projected to reach $546 billion by 2035, with a compound annual growth rate of 15.2% between 2023 and 2035. Within this expanding landscape, lithium-sulfur (Li-S) batteries enhanced by nanotechnology represent one of the most promising technologies poised to disrupt conventional lithium-ion dominance.
Current market analysis indicates that the demand for high-energy-density batteries is accelerating across multiple sectors. The electric vehicle segment, which currently constitutes approximately 38% of advanced battery demand, requires solutions that extend range while reducing weight and cost. Li-S batteries offer theoretical energy densities up to 2,600 Wh/kg, significantly outperforming traditional lithium-ion batteries that typically max out at 260 Wh/kg.
Consumer electronics manufacturers are actively seeking battery technologies that can support increasingly power-hungry devices while maintaining compact form factors. This sector represents 27% of the advanced battery market and has shown willingness to adopt premium solutions that deliver tangible performance improvements. Market surveys indicate that 73% of consumers would pay a premium for devices with double the battery life.
The renewable energy storage sector presents another substantial opportunity, with grid-scale storage installations growing at 42% annually. The ability of nanotechnology-enhanced Li-S batteries to provide cost-effective, high-capacity storage aligns perfectly with this market's requirements. Industry forecasts suggest that the cost per kilowatt-hour for Li-S technology could potentially drop below $80 by 2028, making it competitive with other storage technologies.
Aerospace and defense applications constitute a smaller but higher-margin segment, valued at $12.4 billion in 2022. These specialized applications prioritize energy density and weight reduction over cost considerations, making them ideal early adopters for nanotechnology-enhanced Li-S batteries.
Regional market analysis reveals that Asia-Pacific currently leads manufacturing capacity for advanced battery technologies, with 67% of global production. However, recent policy initiatives in North America and Europe aim to establish domestic supply chains, with combined investments exceeding $45 billion announced since 2021. These regions are strategically positioned to become innovation hubs for nanotechnology applications in energy storage.
Market barriers include the entrenched position of lithium-ion technology, with established manufacturing infrastructure valued at over $120 billion globally. Additionally, concerns regarding cycle life and safety must be addressed before widespread commercial adoption can occur. Nevertheless, venture capital investment in next-generation battery technologies has reached record levels, with $8.7 billion invested in 2022 alone, indicating strong market confidence in technologies like nanotechnology-enhanced Li-S batteries.
Current market analysis indicates that the demand for high-energy-density batteries is accelerating across multiple sectors. The electric vehicle segment, which currently constitutes approximately 38% of advanced battery demand, requires solutions that extend range while reducing weight and cost. Li-S batteries offer theoretical energy densities up to 2,600 Wh/kg, significantly outperforming traditional lithium-ion batteries that typically max out at 260 Wh/kg.
Consumer electronics manufacturers are actively seeking battery technologies that can support increasingly power-hungry devices while maintaining compact form factors. This sector represents 27% of the advanced battery market and has shown willingness to adopt premium solutions that deliver tangible performance improvements. Market surveys indicate that 73% of consumers would pay a premium for devices with double the battery life.
The renewable energy storage sector presents another substantial opportunity, with grid-scale storage installations growing at 42% annually. The ability of nanotechnology-enhanced Li-S batteries to provide cost-effective, high-capacity storage aligns perfectly with this market's requirements. Industry forecasts suggest that the cost per kilowatt-hour for Li-S technology could potentially drop below $80 by 2028, making it competitive with other storage technologies.
Aerospace and defense applications constitute a smaller but higher-margin segment, valued at $12.4 billion in 2022. These specialized applications prioritize energy density and weight reduction over cost considerations, making them ideal early adopters for nanotechnology-enhanced Li-S batteries.
Regional market analysis reveals that Asia-Pacific currently leads manufacturing capacity for advanced battery technologies, with 67% of global production. However, recent policy initiatives in North America and Europe aim to establish domestic supply chains, with combined investments exceeding $45 billion announced since 2021. These regions are strategically positioned to become innovation hubs for nanotechnology applications in energy storage.
Market barriers include the entrenched position of lithium-ion technology, with established manufacturing infrastructure valued at over $120 billion globally. Additionally, concerns regarding cycle life and safety must be addressed before widespread commercial adoption can occur. Nevertheless, venture capital investment in next-generation battery technologies has reached record levels, with $8.7 billion invested in 2022 alone, indicating strong market confidence in technologies like nanotechnology-enhanced Li-S batteries.
Current Challenges in Li-S Battery Development
Despite the promising theoretical energy density of lithium-sulfur (Li-S) batteries, their practical implementation faces several significant challenges that have hindered widespread commercialization. The most critical issue is the polysulfide shuttle effect, where soluble lithium polysulfides (Li2Sx, 4≤x≤8) formed during discharge migrate between electrodes, causing active material loss, self-discharge, and rapid capacity fading. This phenomenon fundamentally undermines the battery's cycle life and efficiency.
Volume expansion presents another substantial obstacle, as sulfur undergoes approximately 80% volume expansion during the lithium insertion process. This expansion leads to mechanical stress within the electrode structure, resulting in pulverization, delamination, and eventual electrode failure after repeated cycling. The structural integrity compromise directly impacts long-term performance stability.
The inherently poor electrical conductivity of sulfur (5×10^-30 S/cm) and its discharge products (Li2S) severely limits electron transport within the cathode. This conductivity issue necessitates high carbon content in electrodes, which reduces the overall energy density of the battery system and counteracts the theoretical advantages of sulfur as a cathode material.
Lithium metal anodes used in Li-S batteries suffer from dendrite formation during cycling, creating serious safety concerns due to potential short circuits. Additionally, the high reactivity between lithium metal and polysulfides accelerates anode degradation, further compromising battery performance and safety.
The slow reaction kinetics of sulfur conversion reactions, particularly the sluggish transformation between Li2S2 and Li2S during discharge, creates significant overpotential and reduces energy efficiency. This kinetic limitation becomes especially pronounced at high discharge rates, severely restricting power capability.
Manufacturing scalability remains challenging due to the complex electrode structures required to address the aforementioned issues. Current laboratory-scale solutions often involve intricate nanostructured materials that are difficult to produce at industrial scales with consistent quality and reasonable cost.
The electrolyte systems for Li-S batteries face a fundamental dilemma: they must dissolve enough polysulfides to enable the electrochemical reactions while simultaneously limiting their diffusion to prevent the shuttle effect. This contradictory requirement has made electrolyte optimization particularly challenging, with most current formulations representing compromises rather than ideal solutions.
Volume expansion presents another substantial obstacle, as sulfur undergoes approximately 80% volume expansion during the lithium insertion process. This expansion leads to mechanical stress within the electrode structure, resulting in pulverization, delamination, and eventual electrode failure after repeated cycling. The structural integrity compromise directly impacts long-term performance stability.
The inherently poor electrical conductivity of sulfur (5×10^-30 S/cm) and its discharge products (Li2S) severely limits electron transport within the cathode. This conductivity issue necessitates high carbon content in electrodes, which reduces the overall energy density of the battery system and counteracts the theoretical advantages of sulfur as a cathode material.
Lithium metal anodes used in Li-S batteries suffer from dendrite formation during cycling, creating serious safety concerns due to potential short circuits. Additionally, the high reactivity between lithium metal and polysulfides accelerates anode degradation, further compromising battery performance and safety.
The slow reaction kinetics of sulfur conversion reactions, particularly the sluggish transformation between Li2S2 and Li2S during discharge, creates significant overpotential and reduces energy efficiency. This kinetic limitation becomes especially pronounced at high discharge rates, severely restricting power capability.
Manufacturing scalability remains challenging due to the complex electrode structures required to address the aforementioned issues. Current laboratory-scale solutions often involve intricate nanostructured materials that are difficult to produce at industrial scales with consistent quality and reasonable cost.
The electrolyte systems for Li-S batteries face a fundamental dilemma: they must dissolve enough polysulfides to enable the electrochemical reactions while simultaneously limiting their diffusion to prevent the shuttle effect. This contradictory requirement has made electrolyte optimization particularly challenging, with most current formulations representing compromises rather than ideal solutions.
Nanomaterial Solutions for Polysulfide Shuttling Effect
01 Cathode materials and structures for lithium-sulfur batteries
Cathode materials and structures play a crucial role in lithium-sulfur battery functionality. Advanced cathode designs incorporate sulfur with conductive materials to improve electron transport and mitigate the shuttle effect. Various carbon-based materials, such as graphene, carbon nanotubes, and porous carbon, are used as hosts for sulfur to enhance conductivity and contain polysulfides. Structural modifications of cathodes, including hierarchical porous structures and core-shell configurations, help to accommodate volume changes during cycling and improve overall battery performance.- Cathode materials and structures for lithium-sulfur batteries: Advanced cathode materials and structures are crucial for improving lithium-sulfur battery functionality. These include sulfur-carbon composites, conductive polymers, and nanostructured materials that can effectively contain sulfur and prevent polysulfide dissolution. Innovative cathode designs with hierarchical pore structures help accommodate volume changes during cycling and enhance the utilization of active materials, leading to improved capacity and cycle life of lithium-sulfur batteries.
- Electrolyte modifications for enhanced performance: Specialized electrolyte formulations play a vital role in lithium-sulfur battery functionality. Additives and solvents that suppress the shuttle effect of polysulfides can significantly improve battery efficiency. Electrolyte modifications include the use of ionic liquids, solid-state electrolytes, and functional additives that form protective interfaces on electrodes. These modifications help control the dissolution of polysulfides, enhance ionic conductivity, and improve the overall electrochemical stability of the battery system.
- Anode protection strategies: Protecting the lithium metal anode is essential for lithium-sulfur battery functionality. Various approaches include the use of protective layers, artificial solid electrolyte interphases, and structured lithium hosts. These strategies prevent direct contact between lithium and polysulfides, mitigate dendrite formation, and enhance the stability of the anode-electrolyte interface. Improved anode protection leads to reduced side reactions, enhanced coulombic efficiency, and extended cycle life of lithium-sulfur batteries.
- Interlayers and separators for polysulfide retention: Functional interlayers and modified separators are designed to physically and chemically trap polysulfides within the cathode region. These components act as barriers that allow lithium ion transport while blocking polysulfide migration. Materials used include carbon-based films, polymer membranes with functional groups, and ceramic-polymer composites. By effectively containing polysulfides, these components reduce capacity fading, improve cycling stability, and enhance the overall energy efficiency of lithium-sulfur batteries.
- Novel cell designs and system integration: Innovative cell architectures and system-level designs address the unique challenges of lithium-sulfur technology. These include optimized electrode configurations, advanced current collectors, and novel cell formats that accommodate the volume changes during cycling. System integration approaches focus on thermal management, pressure control, and electronic monitoring to ensure safe and efficient operation. These design innovations help translate the theoretical advantages of lithium-sulfur chemistry into practical battery systems with improved energy density and operational stability.
02 Electrolyte compositions for lithium-sulfur batteries
Electrolyte compositions significantly impact the performance of lithium-sulfur batteries. Specialized electrolytes are designed to suppress the dissolution of polysulfides and reduce the shuttle effect. Additives such as lithium nitrate and fluorinated compounds help form stable solid electrolyte interphases on the lithium anode. Ionic liquids and solid-state electrolytes are also being explored to enhance safety and cycling stability. The optimization of electrolyte formulations aims to balance ionic conductivity, electrochemical stability, and polysulfide solubility to maximize battery efficiency and lifespan.Expand Specific Solutions03 Anode protection strategies in lithium-sulfur batteries
Protecting the lithium metal anode is essential for improving lithium-sulfur battery functionality. Various strategies include the use of protective layers, artificial solid electrolyte interphases, and structured anodes to prevent dendrite formation and reduce side reactions with polysulfides. Composite anodes incorporating lithium alloys or silicon can enhance stability and safety. Advanced coating technologies using polymers, ceramics, or hybrid materials help to maintain anode integrity during cycling, leading to improved coulombic efficiency and extended battery life.Expand Specific Solutions04 Polysulfide shuttle effect mitigation techniques
Mitigating the polysulfide shuttle effect is crucial for enhancing lithium-sulfur battery performance. Techniques include the development of physical barriers and chemical traps to confine polysulfides within the cathode region. Functional separators with selective permeability help block polysulfide migration while allowing lithium ion transport. Interlayers placed between the cathode and separator act as additional barriers. Chemical approaches involve the use of polar materials that can adsorb or chemically bond with polysulfides, preventing their dissolution and migration to the anode.Expand Specific Solutions05 Advanced manufacturing and cell design for lithium-sulfur batteries
Advanced manufacturing techniques and cell designs are being developed to enhance lithium-sulfur battery functionality. These include novel electrode preparation methods, such as spray deposition and 3D printing, to achieve optimal sulfur loading and distribution. Cell architecture innovations focus on optimizing the cathode-to-anode ratio, electrolyte amount, and overall battery configuration to accommodate the unique characteristics of the lithium-sulfur chemistry. Encapsulation technologies and specialized packaging materials help manage volume changes during cycling and improve the practical energy density of lithium-sulfur batteries.Expand Specific Solutions
Leading Companies and Research Institutions in Li-S Battery Field
Nanotechnology in lithium-sulfur batteries is evolving rapidly, with the market currently in its growth phase as global demand for high-energy density storage solutions increases. The competitive landscape features established energy companies like LG Energy Solution, LG Chem, and Toyota Motor Corp. focusing on commercialization, while research institutions such as MIT, Cornell University, and Central South University drive fundamental innovations. Major chemical corporations including Sumitomo Electric, Arkema France, and China Petroleum & Chemical Corp. are developing specialized nanomaterials to address key challenges of sulfur cathodes. The technology is approaching commercial viability, with companies like EaglePicher Technologies and ULVAC working on manufacturing processes, though challenges in cycle life and production scaling remain before widespread adoption.
LG Chem Ltd.
Technical Solution: LG Chem has developed advanced nanostructured carbon-sulfur composites for lithium-sulfur batteries that address the polysulfide shuttle effect. Their approach involves encapsulating sulfur within mesoporous carbon frameworks with precisely controlled pore sizes (2-50 nm) to physically confine polysulfides while maintaining ionic conductivity. Additionally, they've implemented functional carbon coatings with nitrogen and oxygen doping to chemically bind polysulfides through polar interactions. LG Chem's technology also incorporates graphene-based interlayers between the cathode and separator that act as physical barriers to migrating polysulfides while enhancing electron transport. Their latest innovation includes core-shell nanostructures where sulfur is encapsulated in conductive polymer shells with functional groups that chemically interact with polysulfides, significantly improving cycling stability.
Strengths: Superior polysulfide containment through both physical confinement and chemical bonding, resulting in enhanced cycling stability and capacity retention. Their multi-layered approach addresses multiple failure mechanisms simultaneously. Weaknesses: The complex nanostructured materials likely involve costly and potentially difficult-to-scale manufacturing processes, and the increased inactive material content (carbon frameworks) may reduce overall energy density.
Massachusetts Institute of Technology
Technical Solution: MIT has developed a groundbreaking approach to lithium-sulfur battery technology through their work on nanostructured sulfur cathodes. Their research focuses on creating yolk-shell nanoarchitectures where sulfur particles are encapsulated within conductive carbon shells with precisely engineered void spaces to accommodate the 80% volume expansion during lithiation. MIT researchers have pioneered the use of two-dimensional MXene nanosheets as sulfur hosts and polysulfide traps, leveraging their metallic conductivity and abundant surface functional groups. Their technology also incorporates a novel electrolyte design featuring fluorinated ether solvents and lithium nitrate additives that form a stable solid-electrolyte interphase on the lithium anode while suppressing polysulfide shuttling. Additionally, MIT has developed self-healing polymeric binders with dynamic bonds that can accommodate the mechanical stress during cycling while maintaining electrode integrity and electrical contact.
Strengths: Highly innovative nanoarchitectures that effectively address multiple failure mechanisms simultaneously, particularly the volume expansion and polysulfide shuttling issues. Their comprehensive approach integrating materials science with electrochemistry provides solutions across multiple battery components. Weaknesses: The sophisticated materials and structures may face challenges in scaling to commercial production volumes, and the specialized components could increase manufacturing complexity and cost.
Key Nanostructure Innovations for Sulfur Cathodes
Lithium-sulfur cell and method for manufacturing
PatentInactiveUS20110151335A1
Innovation
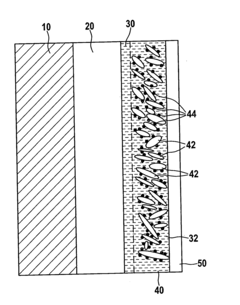
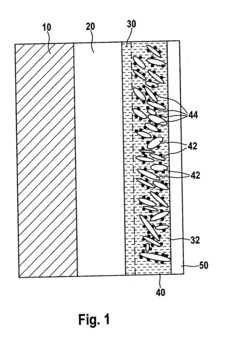
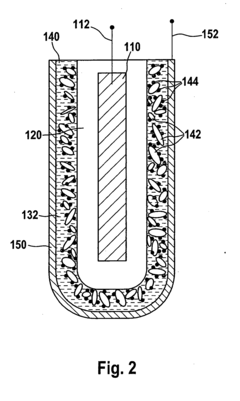
- A lithium-sulfur battery design featuring a continuous layer of nanotubes or nanowires as the cathode structure, where sulfur particles are distributed, providing mechanical integrity and preventing sulfide dissolution, while maintaining high ion mobility and electronic conductivity.
Positive electrode for lithium-sulfur battery, method for manufacturing same, and lithium-sulfur battery including same
PatentWO2023128592A1
Innovation
- A positive electrode for lithium-sulfur batteries is developed, comprising a sulfur-containing material, graphene, and carbon nanotubes, where the carbon nanotubes and graphene form a network, and the sulfur-containing material is embedded within, enhancing ion conductivity and electrochemical characteristics. The manufacturing method involves preparing a binder solution, mixing it with the active material, and forming pellets under controlled pressure and temperature to improve the electrode's structure and performance.
Environmental Impact and Sustainability Assessment
The integration of nanotechnology in lithium-sulfur batteries presents significant environmental and sustainability implications that warrant careful assessment. Conventional lithium-ion batteries contain materials like cobalt and nickel that pose substantial environmental concerns due to resource scarcity and extraction impacts. In contrast, lithium-sulfur batteries utilize sulfur, an abundant by-product of petroleum refining, potentially reducing the environmental footprint associated with raw material acquisition.
Nanotechnology applications in lithium-sulfur batteries can further enhance their environmental profile through several mechanisms. Nanostructured materials enable more efficient sulfur utilization and improved cycle life, effectively reducing the frequency of battery replacement and associated waste generation. The extended lifespan achieved through nano-enhanced components—such as carbon nanotubes, graphene, and metal oxide nanoparticles—translates directly into reduced material consumption and waste production over the battery's operational lifetime.
Life cycle assessment (LCA) studies indicate that nano-enhanced lithium-sulfur batteries may offer up to 30% lower carbon footprint compared to conventional lithium-ion technologies when accounting for manufacturing, use, and end-of-life phases. This reduction stems primarily from decreased energy requirements during production and the elimination of energy-intensive mining operations associated with cobalt and nickel extraction.
Water consumption represents another critical environmental consideration. Preliminary research suggests that manufacturing processes for nano-enhanced lithium-sulfur batteries could potentially reduce water usage by 20-25% compared to conventional battery production, though this advantage depends significantly on the specific nanomaterials and synthesis methods employed.
End-of-life management presents both challenges and opportunities. The diverse nanomaterials incorporated into lithium-sulfur batteries may complicate recycling processes, requiring specialized techniques to safely recover and separate components. However, the higher concentration of valuable materials in nano-enhanced designs could improve the economic viability of recycling operations, creating incentives for closed-loop material systems.
Toxicity concerns remain an area requiring further investigation. While sulfur itself presents minimal toxicological risks, certain nanomaterials used as additives or catalysts may pose novel environmental hazards if released during production, use, or disposal. Comprehensive risk assessment frameworks specifically addressing nanomaterial behavior in environmental systems are essential to ensure responsible development of this technology.
From a sustainability perspective, the reduced reliance on critical minerals positions lithium-sulfur battery technology as a potentially important contributor to circular economy objectives and resource security strategies. The technology's alignment with renewable energy storage applications further amplifies its positive environmental impact potential by enabling greater integration of intermittent renewable energy sources into electrical grids.
Nanotechnology applications in lithium-sulfur batteries can further enhance their environmental profile through several mechanisms. Nanostructured materials enable more efficient sulfur utilization and improved cycle life, effectively reducing the frequency of battery replacement and associated waste generation. The extended lifespan achieved through nano-enhanced components—such as carbon nanotubes, graphene, and metal oxide nanoparticles—translates directly into reduced material consumption and waste production over the battery's operational lifetime.
Life cycle assessment (LCA) studies indicate that nano-enhanced lithium-sulfur batteries may offer up to 30% lower carbon footprint compared to conventional lithium-ion technologies when accounting for manufacturing, use, and end-of-life phases. This reduction stems primarily from decreased energy requirements during production and the elimination of energy-intensive mining operations associated with cobalt and nickel extraction.
Water consumption represents another critical environmental consideration. Preliminary research suggests that manufacturing processes for nano-enhanced lithium-sulfur batteries could potentially reduce water usage by 20-25% compared to conventional battery production, though this advantage depends significantly on the specific nanomaterials and synthesis methods employed.
End-of-life management presents both challenges and opportunities. The diverse nanomaterials incorporated into lithium-sulfur batteries may complicate recycling processes, requiring specialized techniques to safely recover and separate components. However, the higher concentration of valuable materials in nano-enhanced designs could improve the economic viability of recycling operations, creating incentives for closed-loop material systems.
Toxicity concerns remain an area requiring further investigation. While sulfur itself presents minimal toxicological risks, certain nanomaterials used as additives or catalysts may pose novel environmental hazards if released during production, use, or disposal. Comprehensive risk assessment frameworks specifically addressing nanomaterial behavior in environmental systems are essential to ensure responsible development of this technology.
From a sustainability perspective, the reduced reliance on critical minerals positions lithium-sulfur battery technology as a potentially important contributor to circular economy objectives and resource security strategies. The technology's alignment with renewable energy storage applications further amplifies its positive environmental impact potential by enabling greater integration of intermittent renewable energy sources into electrical grids.
Scalability and Manufacturing Considerations
The transition from laboratory-scale prototypes to commercial-scale production represents one of the most significant challenges in lithium-sulfur (Li-S) battery development enhanced by nanotechnology. Current manufacturing processes for conventional lithium-ion batteries cannot be directly applied to Li-S systems due to fundamental differences in electrode materials and cell chemistry. The integration of nanomaterials introduces additional complexity to manufacturing workflows, requiring specialized equipment and handling protocols.
Scalability issues primarily stem from the difficulty in maintaining consistent nanomaterial properties during mass production. Precise control over nanostructure dimensions, morphology, and surface characteristics becomes increasingly challenging as production volumes increase. For instance, carbon nanotubes and graphene-based host materials that effectively trap polysulfides in laboratory settings may exhibit significant batch-to-batch variations when synthesized at industrial scales, leading to inconsistent battery performance.
Manufacturing considerations must address the cost implications of nanomaterial synthesis. While nanotechnology offers remarkable performance enhancements, the production of high-quality nanomaterials often involves expensive precursors, energy-intensive processes, and specialized equipment. Economic viability requires optimization of synthesis routes to reduce costs while maintaining the functional properties that make these materials valuable for Li-S batteries.
Environmental and safety concerns also factor prominently in scaling considerations. Nanomaterials present unique handling challenges due to their small size and potential toxicity. Manufacturing facilities must implement robust containment systems and worker protection protocols. Additionally, sustainable manufacturing practices must be developed to minimize environmental impact, including the recycling and recovery of valuable materials from production waste and end-of-life batteries.
Quality control represents another critical aspect of scalable manufacturing. Advanced characterization techniques must be adapted for high-throughput inspection to ensure nanomaterial consistency. This includes developing in-line monitoring systems capable of detecting deviations in critical parameters such as particle size distribution, surface area, and defect density during continuous production processes.
Recent advances in roll-to-roll processing and solution-based deposition techniques show promise for scaling up the production of nanostructured electrodes. These approaches enable continuous manufacturing of electrode materials with controlled architectures at significantly higher throughput than batch processes. Integration with existing battery manufacturing infrastructure would facilitate more rapid commercialization and reduce capital investment requirements for new production facilities.
Scalability issues primarily stem from the difficulty in maintaining consistent nanomaterial properties during mass production. Precise control over nanostructure dimensions, morphology, and surface characteristics becomes increasingly challenging as production volumes increase. For instance, carbon nanotubes and graphene-based host materials that effectively trap polysulfides in laboratory settings may exhibit significant batch-to-batch variations when synthesized at industrial scales, leading to inconsistent battery performance.
Manufacturing considerations must address the cost implications of nanomaterial synthesis. While nanotechnology offers remarkable performance enhancements, the production of high-quality nanomaterials often involves expensive precursors, energy-intensive processes, and specialized equipment. Economic viability requires optimization of synthesis routes to reduce costs while maintaining the functional properties that make these materials valuable for Li-S batteries.
Environmental and safety concerns also factor prominently in scaling considerations. Nanomaterials present unique handling challenges due to their small size and potential toxicity. Manufacturing facilities must implement robust containment systems and worker protection protocols. Additionally, sustainable manufacturing practices must be developed to minimize environmental impact, including the recycling and recovery of valuable materials from production waste and end-of-life batteries.
Quality control represents another critical aspect of scalable manufacturing. Advanced characterization techniques must be adapted for high-throughput inspection to ensure nanomaterial consistency. This includes developing in-line monitoring systems capable of detecting deviations in critical parameters such as particle size distribution, surface area, and defect density during continuous production processes.
Recent advances in roll-to-roll processing and solution-based deposition techniques show promise for scaling up the production of nanostructured electrodes. These approaches enable continuous manufacturing of electrode materials with controlled architectures at significantly higher throughput than batch processes. Integration with existing battery manufacturing infrastructure would facilitate more rapid commercialization and reduce capital investment requirements for new production facilities.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!