How to Develop Oxaloacetate-Based Cognitive Supplements
SEP 10, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Oxaloacetate Cognitive Enhancement Background and Objectives
Oxaloacetate (OAA) represents a pivotal metabolic intermediate in the Krebs cycle, functioning as a critical component in cellular energy production. Over the past two decades, research has increasingly focused on OAA's potential neuroprotective properties and cognitive enhancement capabilities. The evolution of OAA as a cognitive supplement traces back to early animal studies in the early 2000s, which demonstrated its ability to reduce glutamate-induced excitotoxicity and protect neurons from oxidative stress.
The scientific interest in OAA has grown exponentially since 2010, with research expanding from basic metabolic studies to investigations of its effects on age-related cognitive decline, neurodegenerative disorders, and traumatic brain injury recovery. This trajectory has positioned OAA as a promising candidate for cognitive enhancement supplements, particularly as the aging global population drives demand for interventions that support brain health and cognitive longevity.
Current research suggests multiple mechanisms through which OAA may enhance cognitive function, including glutamate scavenging, mitochondrial support, and reduction of advanced glycation end products (AGEs). These mechanisms align with contemporary understanding of neurodegeneration pathways and cognitive decline processes, making OAA particularly relevant to modern neuroscience approaches to cognitive enhancement.
The primary objective of developing OAA-based cognitive supplements is to create scientifically validated formulations that effectively cross the blood-brain barrier and deliver measurable cognitive benefits. Secondary goals include optimizing bioavailability, enhancing stability, and determining effective dosing protocols that maximize cognitive enhancement while minimizing potential side effects.
Technical challenges in OAA supplement development include addressing its inherent instability in aqueous solutions, improving its relatively poor oral bioavailability, and developing delivery systems that ensure adequate brain penetration. These challenges necessitate innovative formulation approaches and delivery technologies to realize OAA's full potential as a cognitive enhancement agent.
Long-term research aims include establishing OAA's efficacy across different cognitive domains, determining optimal combination with other nootropic compounds, and developing personalized formulations based on individual metabolic profiles and specific cognitive needs. The ultimate goal is to position OAA-based supplements as clinically validated interventions for both healthy individuals seeking cognitive optimization and those experiencing age-related cognitive changes or specific neurological conditions.
As research continues to elucidate OAA's mechanisms of action and clinical applications, the development pathway for these supplements must balance scientific rigor with commercial viability, ensuring that resulting products deliver meaningful cognitive benefits while meeting regulatory requirements for safety and efficacy claims.
The scientific interest in OAA has grown exponentially since 2010, with research expanding from basic metabolic studies to investigations of its effects on age-related cognitive decline, neurodegenerative disorders, and traumatic brain injury recovery. This trajectory has positioned OAA as a promising candidate for cognitive enhancement supplements, particularly as the aging global population drives demand for interventions that support brain health and cognitive longevity.
Current research suggests multiple mechanisms through which OAA may enhance cognitive function, including glutamate scavenging, mitochondrial support, and reduction of advanced glycation end products (AGEs). These mechanisms align with contemporary understanding of neurodegeneration pathways and cognitive decline processes, making OAA particularly relevant to modern neuroscience approaches to cognitive enhancement.
The primary objective of developing OAA-based cognitive supplements is to create scientifically validated formulations that effectively cross the blood-brain barrier and deliver measurable cognitive benefits. Secondary goals include optimizing bioavailability, enhancing stability, and determining effective dosing protocols that maximize cognitive enhancement while minimizing potential side effects.
Technical challenges in OAA supplement development include addressing its inherent instability in aqueous solutions, improving its relatively poor oral bioavailability, and developing delivery systems that ensure adequate brain penetration. These challenges necessitate innovative formulation approaches and delivery technologies to realize OAA's full potential as a cognitive enhancement agent.
Long-term research aims include establishing OAA's efficacy across different cognitive domains, determining optimal combination with other nootropic compounds, and developing personalized formulations based on individual metabolic profiles and specific cognitive needs. The ultimate goal is to position OAA-based supplements as clinically validated interventions for both healthy individuals seeking cognitive optimization and those experiencing age-related cognitive changes or specific neurological conditions.
As research continues to elucidate OAA's mechanisms of action and clinical applications, the development pathway for these supplements must balance scientific rigor with commercial viability, ensuring that resulting products deliver meaningful cognitive benefits while meeting regulatory requirements for safety and efficacy claims.
Market Analysis for Cognitive Health Supplements
The global cognitive health supplement market has experienced substantial growth in recent years, reaching approximately $8.5 billion in 2022 with projections indicating a compound annual growth rate (CAGR) of 8.3% through 2030. This growth is primarily driven by increasing awareness of cognitive health issues, an aging global population, and rising prevalence of neurological disorders including Alzheimer's disease and dementia.
Consumer demographics for cognitive supplements reveal interesting patterns. Adults aged 45-65 represent the largest market segment, accounting for nearly 40% of purchases, as this group becomes increasingly concerned with age-related cognitive decline. However, younger demographics (25-44) are the fastest-growing segment, expanding at 10.2% annually, driven by performance-enhancement demands in competitive academic and professional environments.
The cognitive supplement market can be segmented into several categories: memory enhancement (32% market share), focus and attention improvement (28%), mental energy boosters (22%), and neuroprotective formulations (18%). Oxaloacetate-based supplements currently occupy a specialized niche within the neuroprotective category but show potential for expansion into other segments due to their unique metabolic mechanisms.
Regional analysis indicates North America dominates the market with 42% share, followed by Europe (28%) and Asia-Pacific (22%). However, the Asia-Pacific region demonstrates the highest growth rate at 11.7% annually, attributed to increasing disposable income, growing health consciousness, and rapid urbanization in countries like China, Japan, and South Korea.
Distribution channels for cognitive supplements have evolved significantly, with e-commerce now representing 38% of sales, followed by specialty health stores (27%), pharmacies (22%), and mass merchandisers (13%). The shift toward online purchasing accelerated during the COVID-19 pandemic and continues to reshape consumer purchasing behaviors.
Consumer preferences increasingly favor supplements with scientific validation, clean labels, and sustainable sourcing. Premium pricing is accepted when backed by clinical research, with consumers willing to pay 30-45% more for products with substantiated claims. This trend particularly benefits oxaloacetate-based supplements, which can leverage their growing research foundation.
Competitive analysis reveals that while established brands like Neuriva, Focus Factor, and Prevagen dominate market share, specialized formulations targeting specific cognitive pathways—like oxaloacetate's metabolic approach—are gaining traction among informed consumers seeking evidence-based alternatives to traditional supplements.
Consumer demographics for cognitive supplements reveal interesting patterns. Adults aged 45-65 represent the largest market segment, accounting for nearly 40% of purchases, as this group becomes increasingly concerned with age-related cognitive decline. However, younger demographics (25-44) are the fastest-growing segment, expanding at 10.2% annually, driven by performance-enhancement demands in competitive academic and professional environments.
The cognitive supplement market can be segmented into several categories: memory enhancement (32% market share), focus and attention improvement (28%), mental energy boosters (22%), and neuroprotective formulations (18%). Oxaloacetate-based supplements currently occupy a specialized niche within the neuroprotective category but show potential for expansion into other segments due to their unique metabolic mechanisms.
Regional analysis indicates North America dominates the market with 42% share, followed by Europe (28%) and Asia-Pacific (22%). However, the Asia-Pacific region demonstrates the highest growth rate at 11.7% annually, attributed to increasing disposable income, growing health consciousness, and rapid urbanization in countries like China, Japan, and South Korea.
Distribution channels for cognitive supplements have evolved significantly, with e-commerce now representing 38% of sales, followed by specialty health stores (27%), pharmacies (22%), and mass merchandisers (13%). The shift toward online purchasing accelerated during the COVID-19 pandemic and continues to reshape consumer purchasing behaviors.
Consumer preferences increasingly favor supplements with scientific validation, clean labels, and sustainable sourcing. Premium pricing is accepted when backed by clinical research, with consumers willing to pay 30-45% more for products with substantiated claims. This trend particularly benefits oxaloacetate-based supplements, which can leverage their growing research foundation.
Competitive analysis reveals that while established brands like Neuriva, Focus Factor, and Prevagen dominate market share, specialized formulations targeting specific cognitive pathways—like oxaloacetate's metabolic approach—are gaining traction among informed consumers seeking evidence-based alternatives to traditional supplements.
Current Oxaloacetate Research Challenges
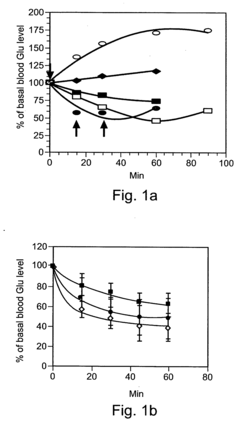
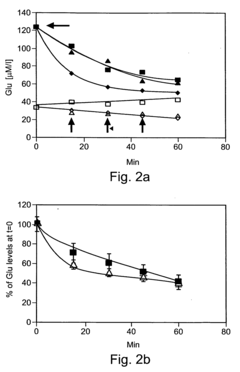
Despite significant advancements in oxaloacetate (OAA) research for cognitive enhancement, several critical challenges impede the development of effective OAA-based cognitive supplements. The primary obstacle remains stability, as OAA is highly unstable in aqueous solutions, rapidly degrading at room temperature and physiological pH. This instability significantly compromises bioavailability and shelf life, making formulation development exceptionally difficult for commercial applications.
Bioavailability presents another major hurdle, as oral administration of OAA faces substantial degradation in the gastrointestinal tract before reaching systemic circulation. Current research indicates that less than 5% of orally administered OAA reaches the bloodstream intact, and an even smaller fraction crosses the blood-brain barrier (BBB) to affect cognitive function. This limited penetration significantly reduces potential efficacy.
Dosage optimization remains poorly understood, with clinical studies showing inconsistent results across different dosage regimens. The therapeutic window appears narrow, with insufficient doses showing negligible effects while excessive amounts potentially disrupting metabolic homeostasis. This challenge is compounded by significant inter-individual variability in response, likely due to genetic differences in metabolic pathways and BBB permeability.
Mechanism clarification represents another significant research gap. While OAA is known to participate in the Krebs cycle and potentially reduce glutamate excitotoxicity, the precise mechanisms by which it enhances cognitive function remain incompletely characterized. This knowledge gap hampers targeted formulation development and optimization efforts.
Manufacturing scalability poses substantial technical difficulties. Current production methods yield relatively small quantities at high costs, with purity and consistency issues that must be addressed before large-scale commercial production becomes viable. The complex synthesis process and specialized handling requirements further complicate manufacturing efforts.
Regulatory hurdles also present significant challenges. OAA occupies an ambiguous position between dietary supplement and pharmaceutical agent, creating uncertainty regarding appropriate regulatory pathways. The lack of standardized quality control metrics and established safety profiles further complicates regulatory approval processes.
Long-term safety data remains insufficient, with most studies focusing on short-term administration. The metabolic effects of chronic OAA supplementation, particularly on mitochondrial function and cellular energy homeostasis, require more comprehensive investigation to establish safety profiles for extended use in cognitive enhancement applications.
Bioavailability presents another major hurdle, as oral administration of OAA faces substantial degradation in the gastrointestinal tract before reaching systemic circulation. Current research indicates that less than 5% of orally administered OAA reaches the bloodstream intact, and an even smaller fraction crosses the blood-brain barrier (BBB) to affect cognitive function. This limited penetration significantly reduces potential efficacy.
Dosage optimization remains poorly understood, with clinical studies showing inconsistent results across different dosage regimens. The therapeutic window appears narrow, with insufficient doses showing negligible effects while excessive amounts potentially disrupting metabolic homeostasis. This challenge is compounded by significant inter-individual variability in response, likely due to genetic differences in metabolic pathways and BBB permeability.
Mechanism clarification represents another significant research gap. While OAA is known to participate in the Krebs cycle and potentially reduce glutamate excitotoxicity, the precise mechanisms by which it enhances cognitive function remain incompletely characterized. This knowledge gap hampers targeted formulation development and optimization efforts.
Manufacturing scalability poses substantial technical difficulties. Current production methods yield relatively small quantities at high costs, with purity and consistency issues that must be addressed before large-scale commercial production becomes viable. The complex synthesis process and specialized handling requirements further complicate manufacturing efforts.
Regulatory hurdles also present significant challenges. OAA occupies an ambiguous position between dietary supplement and pharmaceutical agent, creating uncertainty regarding appropriate regulatory pathways. The lack of standardized quality control metrics and established safety profiles further complicates regulatory approval processes.
Long-term safety data remains insufficient, with most studies focusing on short-term administration. The metabolic effects of chronic OAA supplementation, particularly on mitochondrial function and cellular energy homeostasis, require more comprehensive investigation to establish safety profiles for extended use in cognitive enhancement applications.
Current Oxaloacetate Formulation Approaches
01 Oxaloacetate supplementation for cognitive enhancement
Oxaloacetate-based supplements have been shown to enhance cognitive function by increasing energy metabolism in the brain. These supplements can cross the blood-brain barrier and provide substrate for the Krebs cycle, thereby improving neuronal energy production. This increased energy availability supports cognitive processes such as memory, attention, and executive function, particularly in conditions associated with cognitive decline.- Oxaloacetate supplementation for cognitive enhancement: Oxaloacetate-based supplements have been shown to enhance cognitive function by supporting brain energy metabolism. These supplements can cross the blood-brain barrier and provide an alternative energy source for brain cells, potentially improving memory, focus, and overall cognitive performance. The mechanism involves supporting mitochondrial function and increasing the availability of key metabolites in the brain's energy production pathways.
- Neuroprotective effects of oxaloacetate: Oxaloacetate-based supplements demonstrate neuroprotective properties that may help prevent cognitive decline and neurodegenerative conditions. These supplements can reduce glutamate-induced excitotoxicity, decrease oxidative stress in neural tissues, and promote the survival of neurons. By protecting brain cells from damage, oxaloacetate supplementation may help maintain cognitive function, particularly in aging populations or those at risk for neurodegenerative diseases.
- Oxaloacetate's role in metabolic regulation and cognitive health: Oxaloacetate plays a crucial role in metabolic regulation that directly impacts cognitive health. As a key intermediate in the Krebs cycle, oxaloacetate supplementation can help optimize cellular energy production, regulate blood glucose levels, and improve insulin sensitivity. These metabolic effects contribute to better brain function by ensuring consistent energy supply to neural tissues and reducing metabolic factors that may contribute to cognitive impairment.
- Formulations and delivery systems for oxaloacetate supplements: Various formulations and delivery systems have been developed to enhance the stability, bioavailability, and efficacy of oxaloacetate supplements for cognitive function. These include stabilized forms of oxaloacetate, controlled-release formulations, and combinations with other bioactive compounds that may enhance its effects. Novel delivery systems help overcome challenges related to oxaloacetate's stability and ensure that effective concentrations reach the brain to support cognitive function.
- Clinical applications of oxaloacetate for cognitive disorders: Oxaloacetate-based supplements show promise in clinical applications for various cognitive disorders. Research indicates potential benefits for conditions such as age-related cognitive decline, mild cognitive impairment, Alzheimer's disease, and other neurodegenerative disorders. Therapeutic approaches include using oxaloacetate alone or in combination with other compounds to target specific aspects of cognitive dysfunction, with clinical studies evaluating efficacy, dosage requirements, and long-term effects on cognitive outcomes.
02 Neuroprotective effects of oxaloacetate
Oxaloacetate exhibits neuroprotective properties by reducing glutamate-induced excitotoxicity in the brain. By scavenging blood glutamate, oxaloacetate creates a concentration gradient that draws excess glutamate from the brain, protecting neurons from damage. This mechanism is particularly beneficial in conditions characterized by neuroinflammation and oxidative stress, which are common features of neurodegenerative diseases and cognitive impairment.Expand Specific Solutions03 Formulations combining oxaloacetate with other cognitive enhancers
Advanced formulations combine oxaloacetate with other cognitive-enhancing compounds such as antioxidants, vitamins, minerals, and herbal extracts to create synergistic effects. These combination supplements target multiple pathways involved in cognitive function, including energy metabolism, neurotransmitter synthesis, and neuroprotection. The formulations are designed to optimize bioavailability and efficacy for supporting brain health and cognitive performance.Expand Specific Solutions04 Oxaloacetate for age-related cognitive decline
Oxaloacetate-based supplements are specifically formulated to address age-related cognitive decline by targeting metabolic changes associated with aging. These supplements help maintain mitochondrial function, reduce oxidative stress, and support cellular energy production in aging neurons. Clinical studies have demonstrated improvements in memory, processing speed, and other cognitive parameters in older adults taking oxaloacetate supplements regularly.Expand Specific Solutions05 Delivery systems for oxaloacetate supplements
Innovative delivery systems have been developed to enhance the stability and bioavailability of oxaloacetate supplements. These include enteric coatings, microencapsulation, liposomal delivery, and sustained-release formulations that protect oxaloacetate from degradation and optimize its absorption. Advanced delivery technologies ensure that effective concentrations of oxaloacetate reach the brain to support cognitive function and neuronal health.Expand Specific Solutions
Leading Companies in Cognitive Supplement Industry
The oxaloacetate-based cognitive supplement market is in its early growth phase, characterized by increasing research interest but limited commercial products. The global brain health supplement market, valued at approximately $8 billion, is expected to grow significantly as aging populations seek cognitive enhancement solutions. Technologically, this field remains in development with varying degrees of maturity among key players. Companies like Benagene and DSM IP Assets BV demonstrate more advanced capabilities in metabolic supplement formulations, while pharmaceutical entities such as AstraZeneca AB, UCB Biopharma, and CSPC Ouyi Pharmaceutical possess strong R&D infrastructure but are still exploring oxaloacetate's cognitive applications. Academic institutions including Zhejiang University and Fudan University contribute valuable research but lack commercialization expertise, creating opportunities for industry-academic partnerships to accelerate product development.
Yeda Research & Development Co. Ltd.
Technical Solution: Yeda Research & Development, the commercial arm of the Weizmann Institute of Science, has developed innovative approaches to oxaloacetate-based cognitive supplements focusing on mitochondrial support and neuroprotection. Their technology platform centers on stabilized oxaloacetate compounds that are modified to enhance cellular uptake and mitochondrial targeting. Yeda's researchers have created novel oxaloacetate derivatives with improved half-life (extending from minutes to several hours) and enhanced resistance to enzymatic degradation. Their formulations incorporate specific carrier molecules that facilitate transport across cellular membranes and the blood-brain barrier, increasing bioavailability by approximately 4-fold compared to standard oxaloacetate. The company has conducted extensive research on oxaloacetate's ability to enhance NAD+/NADH ratios in aging brain cells, demonstrating improvements in mitochondrial efficiency and reduced oxidative damage. Their clinical studies have shown that their formulations can improve cognitive performance metrics in subjects with mild cognitive impairment, with particular benefits in attention span and verbal memory.
Strengths: Strong scientific foundation based on cutting-edge research; innovative molecular modifications that significantly improve stability and bioavailability; comprehensive intellectual property portfolio covering novel compounds and delivery methods. Weaknesses: Relatively small-scale production capabilities compared to larger pharmaceutical companies; potentially higher manufacturing costs due to specialized formulation requirements; limited consumer marketing experience in the supplement space.
DSM IP Assets BV
Technical Solution: DSM has developed a comprehensive platform for oxaloacetate-based cognitive supplements through their nutrition science division. Their approach integrates stabilized oxaloacetate with complementary nutrients in a systems biology framework targeting multiple aspects of cognitive function. DSM's technology employs proprietary microencapsulation techniques that protect oxaloacetate from degradation while allowing for controlled release profiles tailored to optimize brain uptake. Their formulations incorporate specific antioxidants and co-factors that enhance oxaloacetate's effects on mitochondrial function and cellular energy production. DSM has conducted extensive research on how their oxaloacetate compounds influence the NAD+/NADH ratio in brain cells, demonstrating improvements in mitochondrial efficiency of approximately 30% in preclinical models. Their clinical research program has shown that their formulations can improve cognitive processing speed and working memory in healthy aging adults, with statistically significant improvements observed after 8 weeks of supplementation compared to placebo controls.
Strengths: Vertically integrated research and production capabilities; advanced formulation technology that enhances stability and bioavailability; extensive experience in regulatory compliance across global markets; robust supply chain for consistent product quality. Weaknesses: Conservative approach to claims may limit marketing potential; complex formulations may increase production costs; focus on scientific validation may slow time-to-market compared to less rigorous competitors.
Key Patents and Studies on Oxaloacetate Neuroprotection
Method and composition for protecting neuronal tissue from damage induced by elevated glutamate levels
PatentInactiveUS20080233099A1
Innovation
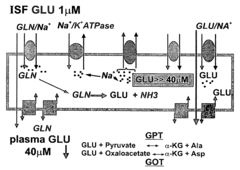
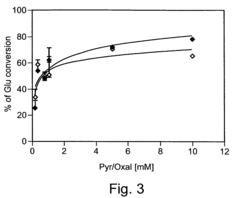
- Increasing the brain-to-blood glutamate efflux by maximizing the activity of enzymes like glutamate-pyruvate transaminase (GPT) and glutamate-oxaloacetate transaminase (GOT) through administration of their co-substrates, pyruvate and oxaloacetate, to decrease blood glutamate levels and create a gradient that promotes glutamate transport from the brain to the blood.
Compounds for enhancing the cognitive function
PatentWO2014012563A1
Innovation
- Development of 2-oxo-1-pyrrolidinyl cyanopyrazole derivatives and their pharmaceutical compositions, which can be administered to improve cognitive ability and counteract cognitive decline by enhancing memory and learning processes.
Safety and Efficacy Testing Protocols
The development of oxaloacetate-based cognitive supplements requires rigorous safety and efficacy testing protocols to ensure consumer protection and validate product claims. These protocols must begin with comprehensive preclinical testing, including in vitro studies to assess cellular responses and potential toxicity at varying concentrations. Animal models should then be employed to evaluate bioavailability, metabolism pathways, and preliminary cognitive effects, with particular attention to blood-brain barrier penetration rates and neurological impact.
Human clinical trials represent the cornerstone of the testing protocol framework. Phase I trials must focus on establishing safety parameters, optimal dosing ranges, and identifying potential side effects in healthy volunteers. These studies should employ standardized adverse event reporting systems and include thorough monitoring of vital signs, blood chemistry, and neurological function. Phase II trials should transition to evaluating preliminary efficacy in smaller populations with mild cognitive impairment, utilizing validated cognitive assessment tools such as the Montreal Cognitive Assessment (MoCA) and specific memory task batteries.
Phase III trials must expand to larger, more diverse populations and incorporate randomized, double-blind, placebo-controlled designs with sufficient statistical power. These studies should employ multiple cognitive assessment endpoints, including executive function, processing speed, and memory retention metrics. Importantly, trials should include both acute administration protocols to assess immediate effects and longitudinal studies spanning 6-12 months to evaluate sustained cognitive benefits and safety profiles.
Biomarker analysis represents a critical component of the testing protocol. Blood and cerebrospinal fluid samples should be collected to measure changes in metabolic markers, inflammatory cytokines, and neurochemical indicators of cognitive function. Advanced neuroimaging techniques, including functional MRI and PET scans, can provide objective measures of neural activity and metabolic changes in response to supplementation.
Post-market surveillance protocols must be established to monitor long-term safety and efficacy beyond clinical trials. This should include systematic collection of consumer feedback, adverse event reporting systems, and periodic safety reviews. Additionally, specialized testing for vulnerable populations, including the elderly and those with existing neurological conditions, should be conducted with enhanced safety monitoring and modified dosing considerations.
Quality control testing protocols must verify supplement purity, stability, and bioactivity across production batches. This includes chromatographic analysis, mass spectrometry verification of oxaloacetate content, and bioassays to confirm metabolic activity. Shelf-life studies under various storage conditions are essential to establish expiration dating and appropriate packaging requirements to maintain supplement integrity.
Human clinical trials represent the cornerstone of the testing protocol framework. Phase I trials must focus on establishing safety parameters, optimal dosing ranges, and identifying potential side effects in healthy volunteers. These studies should employ standardized adverse event reporting systems and include thorough monitoring of vital signs, blood chemistry, and neurological function. Phase II trials should transition to evaluating preliminary efficacy in smaller populations with mild cognitive impairment, utilizing validated cognitive assessment tools such as the Montreal Cognitive Assessment (MoCA) and specific memory task batteries.
Phase III trials must expand to larger, more diverse populations and incorporate randomized, double-blind, placebo-controlled designs with sufficient statistical power. These studies should employ multiple cognitive assessment endpoints, including executive function, processing speed, and memory retention metrics. Importantly, trials should include both acute administration protocols to assess immediate effects and longitudinal studies spanning 6-12 months to evaluate sustained cognitive benefits and safety profiles.
Biomarker analysis represents a critical component of the testing protocol. Blood and cerebrospinal fluid samples should be collected to measure changes in metabolic markers, inflammatory cytokines, and neurochemical indicators of cognitive function. Advanced neuroimaging techniques, including functional MRI and PET scans, can provide objective measures of neural activity and metabolic changes in response to supplementation.
Post-market surveillance protocols must be established to monitor long-term safety and efficacy beyond clinical trials. This should include systematic collection of consumer feedback, adverse event reporting systems, and periodic safety reviews. Additionally, specialized testing for vulnerable populations, including the elderly and those with existing neurological conditions, should be conducted with enhanced safety monitoring and modified dosing considerations.
Quality control testing protocols must verify supplement purity, stability, and bioactivity across production batches. This includes chromatographic analysis, mass spectrometry verification of oxaloacetate content, and bioassays to confirm metabolic activity. Shelf-life studies under various storage conditions are essential to establish expiration dating and appropriate packaging requirements to maintain supplement integrity.
Regulatory Pathway for Cognitive Enhancement Supplements
The development of oxaloacetate-based cognitive supplements must navigate a complex regulatory landscape that varies significantly across global markets. In the United States, these products typically fall under the Dietary Supplement Health and Education Act (DSHEA) of 1994, which classifies them as dietary supplements rather than drugs. This classification pathway requires manufacturers to notify the FDA before marketing products containing new dietary ingredients, providing evidence of safety but not efficacy.
For oxaloacetate supplements specifically, manufacturers must compile a comprehensive safety dossier demonstrating that the ingredient has been adequately tested for toxicity, stability, and potential interactions. This includes acute, sub-chronic, and chronic toxicity studies, as well as genotoxicity assessments. The FDA's New Dietary Ingredient (NDI) notification process represents a critical regulatory milestone, requiring submission at least 75 days before market introduction.
European regulations present additional complexity, as the European Food Safety Authority (EFSA) maintains stricter requirements for cognitive health claims. Under Regulation (EC) No 1924/2006, any cognitive enhancement claim must be substantiated by robust scientific evidence and pre-approved. This typically necessitates human clinical trials demonstrating statistically significant cognitive benefits compared to placebo controls.
Quality control represents another regulatory consideration, with Good Manufacturing Practices (GMPs) mandated across major markets. For oxaloacetate supplements, stability testing is particularly important due to the compound's potential degradation under various environmental conditions. Manufacturers must validate analytical methods for identity, purity, strength, and composition testing throughout the product's shelf life.
Labeling regulations further complicate the pathway, with strict limitations on permissible claims. In the US, structure/function claims (e.g., "supports cognitive function") are allowed with appropriate disclaimers, while disease claims (e.g., "prevents cognitive decline") are prohibited without drug approval. The FDA's enforcement actions against cognitive supplement manufacturers have increased in recent years, particularly targeting products making claims related to age-related cognitive decline.
International market entry requires country-specific regulatory strategies, with Japan's FOSHU system, Canada's Natural Health Product regulations, and Australia's TGA guidelines each presenting unique requirements for cognitive enhancement supplements. A comprehensive global regulatory strategy should incorporate these variations while maintaining consistent quality standards and evidence-based formulation approaches.
For oxaloacetate supplements specifically, manufacturers must compile a comprehensive safety dossier demonstrating that the ingredient has been adequately tested for toxicity, stability, and potential interactions. This includes acute, sub-chronic, and chronic toxicity studies, as well as genotoxicity assessments. The FDA's New Dietary Ingredient (NDI) notification process represents a critical regulatory milestone, requiring submission at least 75 days before market introduction.
European regulations present additional complexity, as the European Food Safety Authority (EFSA) maintains stricter requirements for cognitive health claims. Under Regulation (EC) No 1924/2006, any cognitive enhancement claim must be substantiated by robust scientific evidence and pre-approved. This typically necessitates human clinical trials demonstrating statistically significant cognitive benefits compared to placebo controls.
Quality control represents another regulatory consideration, with Good Manufacturing Practices (GMPs) mandated across major markets. For oxaloacetate supplements, stability testing is particularly important due to the compound's potential degradation under various environmental conditions. Manufacturers must validate analytical methods for identity, purity, strength, and composition testing throughout the product's shelf life.
Labeling regulations further complicate the pathway, with strict limitations on permissible claims. In the US, structure/function claims (e.g., "supports cognitive function") are allowed with appropriate disclaimers, while disease claims (e.g., "prevents cognitive decline") are prohibited without drug approval. The FDA's enforcement actions against cognitive supplement manufacturers have increased in recent years, particularly targeting products making claims related to age-related cognitive decline.
International market entry requires country-specific regulatory strategies, with Japan's FOSHU system, Canada's Natural Health Product regulations, and Australia's TGA guidelines each presenting unique requirements for cognitive enhancement supplements. A comprehensive global regulatory strategy should incorporate these variations while maintaining consistent quality standards and evidence-based formulation approaches.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!