How to Implement Oxaloacetate in Cognitive Function Studies
SEP 10, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Oxaloacetate in Cognitive Function: Background and Objectives
Oxaloacetate (OAA) represents a critical metabolite in the tricarboxylic acid (TCA) cycle, functioning as an essential intermediate in cellular energy production. Over the past decade, research has increasingly focused on OAA's potential role beyond basic metabolism, particularly its impact on cognitive function and neuroprotection. The evolution of this research field traces back to early observations of metabolic dysfunction in neurodegenerative conditions, which subsequently led to investigations into metabolic enhancers as potential therapeutic agents.
The scientific interest in OAA stems from its position at the intersection of several metabolic pathways crucial for brain health, including glutamate-glutamine cycling, aspartate production, and energy metabolism. Historical research has demonstrated correlations between metabolic efficiency and cognitive performance, with more recent studies specifically examining OAA's capacity to support neuronal energy demands and potentially mitigate excitotoxicity through glutamate regulation.
Current research trajectories are exploring OAA's multifaceted mechanisms in cognitive enhancement, including its ability to reduce blood glutamate levels, support mitochondrial function, and potentially increase brain-derived neurotrophic factor (BDNF) expression. Preliminary animal studies and limited human trials suggest promising applications in age-related cognitive decline, traumatic brain injury recovery, and neurodegenerative conditions such as Alzheimer's disease.
The technological evolution in this field has progressed from basic metabolic studies to sophisticated neuroimaging techniques that can track metabolite utilization in real-time, allowing researchers to visualize OAA's effects on brain metabolism and function. Concurrently, advances in formulation technology have improved OAA stability and bioavailability, addressing previous limitations in clinical applications.
The primary objectives for implementing OAA in cognitive function studies encompass several dimensions. First, establishing standardized protocols for OAA administration that optimize bioavailability and blood-brain barrier penetration. Second, developing reliable biomarkers to track OAA's metabolic effects and correlate them with cognitive outcomes. Third, designing comprehensive clinical trials that address various cognitive domains across different population segments, particularly focusing on age-related cognitive decline and neurodegenerative conditions.
Additionally, research aims to elucidate the precise molecular mechanisms through which OAA influences cognitive function, distinguishing direct effects from secondary metabolic consequences. This includes investigating potential synergistic effects when combined with other metabolic enhancers or traditional cognitive interventions. The ultimate goal is to translate laboratory findings into practical clinical applications, potentially establishing OAA as a validated nutritional intervention or therapeutic agent for cognitive support and neuroprotection.
The scientific interest in OAA stems from its position at the intersection of several metabolic pathways crucial for brain health, including glutamate-glutamine cycling, aspartate production, and energy metabolism. Historical research has demonstrated correlations between metabolic efficiency and cognitive performance, with more recent studies specifically examining OAA's capacity to support neuronal energy demands and potentially mitigate excitotoxicity through glutamate regulation.
Current research trajectories are exploring OAA's multifaceted mechanisms in cognitive enhancement, including its ability to reduce blood glutamate levels, support mitochondrial function, and potentially increase brain-derived neurotrophic factor (BDNF) expression. Preliminary animal studies and limited human trials suggest promising applications in age-related cognitive decline, traumatic brain injury recovery, and neurodegenerative conditions such as Alzheimer's disease.
The technological evolution in this field has progressed from basic metabolic studies to sophisticated neuroimaging techniques that can track metabolite utilization in real-time, allowing researchers to visualize OAA's effects on brain metabolism and function. Concurrently, advances in formulation technology have improved OAA stability and bioavailability, addressing previous limitations in clinical applications.
The primary objectives for implementing OAA in cognitive function studies encompass several dimensions. First, establishing standardized protocols for OAA administration that optimize bioavailability and blood-brain barrier penetration. Second, developing reliable biomarkers to track OAA's metabolic effects and correlate them with cognitive outcomes. Third, designing comprehensive clinical trials that address various cognitive domains across different population segments, particularly focusing on age-related cognitive decline and neurodegenerative conditions.
Additionally, research aims to elucidate the precise molecular mechanisms through which OAA influences cognitive function, distinguishing direct effects from secondary metabolic consequences. This includes investigating potential synergistic effects when combined with other metabolic enhancers or traditional cognitive interventions. The ultimate goal is to translate laboratory findings into practical clinical applications, potentially establishing OAA as a validated nutritional intervention or therapeutic agent for cognitive support and neuroprotection.
Market Analysis for Cognitive Enhancement Compounds
The cognitive enhancement market has witnessed substantial growth in recent years, driven by increasing consumer awareness about brain health and cognitive performance. The global market for cognitive enhancement compounds reached approximately $8.5 billion in 2022 and is projected to grow at a CAGR of 14.2% through 2030, potentially reaching $24.3 billion by the end of the forecast period.
Oxaloacetate, as a metabolic intermediate in the Krebs cycle with potential neuroprotective properties, occupies a specialized segment within this broader market. Currently, the market share for oxaloacetate-based supplements remains relatively small, estimated at $125 million globally, but demonstrates promising growth potential due to emerging research supporting its cognitive benefits.
Consumer demographics for cognitive enhancement compounds reveal interesting patterns. The primary market consists of adults aged 40-65 concerned about age-related cognitive decline, representing approximately 45% of consumers. However, there's a rapidly growing segment of younger professionals (25-40 years) seeking cognitive performance advantages in competitive work environments, now constituting about 30% of the market.
Distribution channels for these compounds have evolved significantly, with online retail dominating at 62% of sales. Specialty health stores account for 25%, while traditional pharmacies represent only 13% of distribution. This shift toward digital purchasing has been accelerated by the pandemic and reflects consumers' preference for researching these specialized products online before purchase.
Competitive analysis reveals that the oxaloacetate market remains less saturated than other cognitive enhancement segments. Currently, fewer than 15 companies offer oxaloacetate-specific formulations, with most positioned as premium products with higher price points than typical nootropics. The average price point for a month's supply ranges from $60-120, significantly higher than many other cognitive supplements.
Regulatory considerations present both challenges and opportunities. While oxaloacetate is generally recognized as safe (GRAS) as a dietary supplement in most markets, therapeutic claims remain strictly limited without clinical trial validation. Companies successfully navigating the regulatory landscape while accumulating scientific evidence stand to gain significant market advantage.
Consumer awareness represents perhaps the greatest market challenge, with surveys indicating only 12% of potential consumers recognize oxaloacetate and its potential benefits. This suggests substantial untapped market potential if awareness and education efforts are increased, particularly when targeting healthcare professionals who influence supplement recommendations.
AI-powered personalized health platforms represent an emerging opportunity, with several startups developing algorithms to recommend specific cognitive enhancement compounds based on individual biomarkers and health profiles. This trend could significantly impact how oxaloacetate is marketed and distributed in the coming years.
Oxaloacetate, as a metabolic intermediate in the Krebs cycle with potential neuroprotective properties, occupies a specialized segment within this broader market. Currently, the market share for oxaloacetate-based supplements remains relatively small, estimated at $125 million globally, but demonstrates promising growth potential due to emerging research supporting its cognitive benefits.
Consumer demographics for cognitive enhancement compounds reveal interesting patterns. The primary market consists of adults aged 40-65 concerned about age-related cognitive decline, representing approximately 45% of consumers. However, there's a rapidly growing segment of younger professionals (25-40 years) seeking cognitive performance advantages in competitive work environments, now constituting about 30% of the market.
Distribution channels for these compounds have evolved significantly, with online retail dominating at 62% of sales. Specialty health stores account for 25%, while traditional pharmacies represent only 13% of distribution. This shift toward digital purchasing has been accelerated by the pandemic and reflects consumers' preference for researching these specialized products online before purchase.
Competitive analysis reveals that the oxaloacetate market remains less saturated than other cognitive enhancement segments. Currently, fewer than 15 companies offer oxaloacetate-specific formulations, with most positioned as premium products with higher price points than typical nootropics. The average price point for a month's supply ranges from $60-120, significantly higher than many other cognitive supplements.
Regulatory considerations present both challenges and opportunities. While oxaloacetate is generally recognized as safe (GRAS) as a dietary supplement in most markets, therapeutic claims remain strictly limited without clinical trial validation. Companies successfully navigating the regulatory landscape while accumulating scientific evidence stand to gain significant market advantage.
Consumer awareness represents perhaps the greatest market challenge, with surveys indicating only 12% of potential consumers recognize oxaloacetate and its potential benefits. This suggests substantial untapped market potential if awareness and education efforts are increased, particularly when targeting healthcare professionals who influence supplement recommendations.
AI-powered personalized health platforms represent an emerging opportunity, with several startups developing algorithms to recommend specific cognitive enhancement compounds based on individual biomarkers and health profiles. This trend could significantly impact how oxaloacetate is marketed and distributed in the coming years.
Current Research Status and Technical Challenges
The implementation of oxaloacetate in cognitive function studies represents an emerging frontier in neuroscience research, with significant developments occurring globally. Current research indicates that oxaloacetate, a key intermediate in the Krebs cycle, may play a crucial role in neuroprotection and cognitive enhancement through multiple mechanisms including glutamate scavenging and mitochondrial energy production optimization.
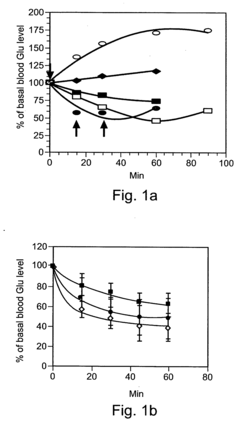
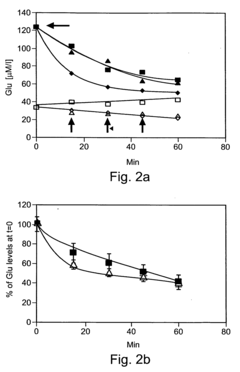
Laboratory studies have demonstrated promising results regarding oxaloacetate's potential to protect neurons from excitotoxicity and oxidative stress, which are common pathological features in various neurodegenerative conditions. Animal models have shown improvements in cognitive performance metrics following oxaloacetate supplementation, particularly in contexts of induced cognitive impairment. However, translation to human clinical applications remains in early stages, with limited controlled trials published to date.
The primary technical challenges in this field center around several critical factors. First, the blood-brain barrier (BBB) permeability of oxaloacetate presents a significant obstacle, as the molecule's hydrophilic nature limits its passive diffusion across this protective interface. Various delivery systems including nanoparticle encapsulation and prodrug approaches are being explored, though none has achieved optimal efficiency for clinical application.
Dosage optimization represents another substantial challenge, with current research showing considerable variability in effective concentrations across different experimental models. The therapeutic window appears narrow, with potential neurotoxicity at higher doses counteracting beneficial effects, necessitating precise administration protocols that remain to be standardized.
Stability issues further complicate implementation, as oxaloacetate demonstrates rapid degradation under physiological conditions. Current formulations exhibit limited shelf-life and variable bioavailability, requiring advanced stabilization techniques that add complexity to experimental design and clinical applications.
Measurement methodologies present additional technical hurdles. Quantifying oxaloacetate's cognitive effects requires sophisticated neuroimaging, biochemical analysis, and standardized cognitive assessment tools. The integration of these diverse measurement approaches into cohesive research protocols remains challenging, particularly for longitudinal studies tracking cognitive changes over extended periods.
Regulatory considerations also impact research progress, with varying requirements across jurisdictions regarding the use of oxaloacetate in human subjects. The compound's dual classification as both a metabolic intermediate and potential therapeutic agent creates regulatory ambiguity that researchers must navigate.
Despite these challenges, technological advancements in neuroimaging, metabolomics, and cognitive assessment tools are gradually enabling more sophisticated implementation strategies. Multi-disciplinary collaborations between neuroscientists, biochemists, and clinical researchers are increasingly addressing these technical barriers through innovative experimental designs and analytical approaches.
Laboratory studies have demonstrated promising results regarding oxaloacetate's potential to protect neurons from excitotoxicity and oxidative stress, which are common pathological features in various neurodegenerative conditions. Animal models have shown improvements in cognitive performance metrics following oxaloacetate supplementation, particularly in contexts of induced cognitive impairment. However, translation to human clinical applications remains in early stages, with limited controlled trials published to date.
The primary technical challenges in this field center around several critical factors. First, the blood-brain barrier (BBB) permeability of oxaloacetate presents a significant obstacle, as the molecule's hydrophilic nature limits its passive diffusion across this protective interface. Various delivery systems including nanoparticle encapsulation and prodrug approaches are being explored, though none has achieved optimal efficiency for clinical application.
Dosage optimization represents another substantial challenge, with current research showing considerable variability in effective concentrations across different experimental models. The therapeutic window appears narrow, with potential neurotoxicity at higher doses counteracting beneficial effects, necessitating precise administration protocols that remain to be standardized.
Stability issues further complicate implementation, as oxaloacetate demonstrates rapid degradation under physiological conditions. Current formulations exhibit limited shelf-life and variable bioavailability, requiring advanced stabilization techniques that add complexity to experimental design and clinical applications.
Measurement methodologies present additional technical hurdles. Quantifying oxaloacetate's cognitive effects requires sophisticated neuroimaging, biochemical analysis, and standardized cognitive assessment tools. The integration of these diverse measurement approaches into cohesive research protocols remains challenging, particularly for longitudinal studies tracking cognitive changes over extended periods.
Regulatory considerations also impact research progress, with varying requirements across jurisdictions regarding the use of oxaloacetate in human subjects. The compound's dual classification as both a metabolic intermediate and potential therapeutic agent creates regulatory ambiguity that researchers must navigate.
Despite these challenges, technological advancements in neuroimaging, metabolomics, and cognitive assessment tools are gradually enabling more sophisticated implementation strategies. Multi-disciplinary collaborations between neuroscientists, biochemists, and clinical researchers are increasingly addressing these technical barriers through innovative experimental designs and analytical approaches.
Methodologies for Oxaloacetate Implementation in Cognitive Studies
01 Oxaloacetate supplementation for cognitive enhancement
Oxaloacetate has been found to enhance cognitive function through various mechanisms. When administered as a dietary supplement, it can cross the blood-brain barrier and support brain metabolism. Research indicates that oxaloacetate supplementation may improve memory, attention, and overall cognitive performance, particularly in individuals with age-related cognitive decline. The compound works by supporting mitochondrial function and energy production in brain cells, which is essential for optimal cognitive processing.- Oxaloacetate supplementation for cognitive enhancement: Oxaloacetate has been found to enhance cognitive function through various mechanisms. When administered as a dietary supplement, it can cross the blood-brain barrier and support brain metabolism. Research indicates that oxaloacetate supplementation may improve memory, attention, and overall cognitive performance by increasing energy production in brain cells and reducing oxidative stress. These cognitive benefits make oxaloacetate a promising compound for maintaining brain health and potentially addressing age-related cognitive decline.
- Oxaloacetate for neuroprotection and neurodegenerative disorders: Oxaloacetate demonstrates neuroprotective properties that may be beneficial in preventing or treating neurodegenerative disorders. It helps maintain glutamate homeostasis in the brain, reducing excitotoxicity that can lead to neuronal damage. Additionally, oxaloacetate supports mitochondrial function and reduces oxidative stress in neural tissues. These mechanisms suggest potential applications in conditions such as Alzheimer's disease, Parkinson's disease, and other neurodegenerative disorders where cognitive decline is a primary symptom.
- Metabolic pathways of oxaloacetate affecting brain function: Oxaloacetate plays a crucial role in several metabolic pathways that directly impact brain function and cognition. As a key intermediate in the Krebs cycle, it facilitates energy production in brain cells. It also participates in the malate-aspartate shuttle, which is essential for maintaining proper NAD+/NADH ratios in neurons. Furthermore, oxaloacetate influences glutamate and aspartate metabolism, which are important neurotransmitters involved in learning and memory processes. These metabolic functions collectively contribute to oxaloacetate's positive effects on cognitive performance.
- Formulations and delivery methods for oxaloacetate: Various formulations and delivery methods have been developed to enhance the bioavailability and efficacy of oxaloacetate for cognitive function. These include stabilized forms of oxaloacetate that resist degradation, controlled-release formulations that provide sustained delivery to the brain, and combination products that include synergistic compounds such as vitamins, minerals, or other metabolic enhancers. Novel delivery systems such as nanoparticles or liposomes may also improve the ability of oxaloacetate to reach target tissues in the brain, maximizing its cognitive benefits.
- Diagnostic and monitoring methods for oxaloacetate therapy: Advanced diagnostic and monitoring methods have been developed to assess the effectiveness of oxaloacetate therapy on cognitive function. These include neuroimaging techniques to measure brain metabolism and activity, cognitive assessment tools specifically designed to detect changes in memory and executive function, and biomarker analyses that track metabolic changes associated with oxaloacetate supplementation. These methods allow for personalized dosing regimens and treatment protocols, optimizing the cognitive benefits of oxaloacetate based on individual patient responses and needs.
02 Oxaloacetate for neuroprotection and neurodegenerative disorders
Oxaloacetate demonstrates neuroprotective properties that may help prevent or slow the progression of neurodegenerative disorders. It acts by reducing glutamate excitotoxicity, which is implicated in conditions like Alzheimer's disease, Parkinson's disease, and other forms of dementia. By scavenging excess glutamate in the bloodstream, oxaloacetate helps maintain proper neurotransmitter balance and protects neurons from damage. This mechanism contributes to improved cognitive function and may offer therapeutic benefits for patients with neurodegenerative conditions.Expand Specific Solutions03 Oxaloacetate's role in metabolic regulation and brain energy
Oxaloacetate plays a crucial role in cellular metabolism, particularly in the tricarboxylic acid (TCA) cycle, which is essential for energy production. In the context of cognitive function, oxaloacetate helps regulate glucose metabolism in the brain, ensuring adequate energy supply to neurons. By enhancing mitochondrial efficiency and promoting ATP production, oxaloacetate supports optimal brain function. Additionally, it may help regulate blood glucose levels, which is important for maintaining cognitive performance, especially during aging or in metabolic disorders that affect brain function.Expand Specific Solutions04 Oxaloacetate in combination therapies for cognitive enhancement
Oxaloacetate can be combined with other compounds to create synergistic effects for cognitive enhancement. These combination therapies often include antioxidants, other TCA cycle intermediates, or compounds that support mitochondrial function. Such combinations may provide more comprehensive neuroprotection and cognitive benefits than oxaloacetate alone. Research suggests that these combination approaches can address multiple aspects of cognitive decline simultaneously, including oxidative stress, energy metabolism, and neurotransmitter balance, resulting in improved cognitive outcomes.Expand Specific Solutions05 Methods for measuring and monitoring oxaloacetate's effects on cognition
Various methods have been developed to measure and monitor the effects of oxaloacetate on cognitive function. These include cognitive assessment tools, biomarker analysis, and neuroimaging techniques that can track changes in brain metabolism and function. Such methods allow for the evaluation of oxaloacetate's efficacy in improving cognitive performance and help determine optimal dosing regimens. They also enable personalized approaches to oxaloacetate supplementation based on individual cognitive needs and responses, potentially leading to more effective cognitive enhancement strategies.Expand Specific Solutions
Leading Research Institutions and Pharmaceutical Companies
The oxaloacetate cognitive function research landscape is currently in an emerging growth phase, with market size estimated to expand significantly as neurodegenerative disease treatments gain priority. The technology demonstrates moderate maturity, with key players representing diverse sectors. Academic institutions like Johns Hopkins University, MIT, and University of Melbourne are driving fundamental research, while pharmaceutical companies including H. Lundbeck, Merck Sharp & Dohme, and UCB Pharma are advancing clinical applications. Specialized biotechnology firms such as Cerecin and Phoenix Biotechnology are developing targeted solutions. Research institutes like The Florey Institute and The Broad Institute provide crucial infrastructure for translational research. This competitive landscape reflects growing recognition of oxaloacetate's potential in addressing cognitive decline and neurological disorders.
The Johns Hopkins University
Technical Solution: Johns Hopkins University has developed a multifaceted approach to implementing oxaloacetate in cognitive function studies, focusing on its potential neuroprotective properties and role in cellular energy metabolism. Their methodology combines translational research spanning from molecular mechanisms to clinical applications. At the preclinical level, they utilize sophisticated mouse models of neurodegenerative diseases to evaluate oxaloacetate's effects on cognitive performance, employing standardized behavioral tests including Morris water maze, novel object recognition, and fear conditioning paradigms. Their implementation strategy includes innovative techniques for measuring oxaloacetate's impact on mitochondrial function in neurons, particularly focusing on its ability to enhance NAD+/NADH ratios and activate AMPK pathways critical for neuronal survival[4]. Johns Hopkins researchers have also developed specialized protocols for administering stabilized oxaloacetate formulations that maximize blood-brain barrier penetration while minimizing peripheral metabolism. Their clinical research program incorporates comprehensive cognitive assessment batteries alongside advanced neuroimaging techniques (fMRI, PET) to correlate changes in brain metabolism with cognitive performance metrics following oxaloacetate supplementation[5].
Strengths: Comprehensive translational approach spanning from molecular mechanisms to clinical applications provides robust validation across multiple research domains. Well-established expertise in neurodegenerative disease models enables targeted assessment of oxaloacetate's effects in specific pathological contexts. Weaknesses: Complex study designs may introduce multiple variables that complicate interpretation of oxaloacetate's specific effects. Potential challenges in standardizing oxaloacetate formulations across different experimental paradigms.
Cerecin, Inc.
Technical Solution: Cerecin has developed a comprehensive approach to implementing oxaloacetate in cognitive function studies, focusing on its potential as a therapeutic agent for neurodegenerative disorders. Their proprietary formulation, AC-1202 (Axona), contains medium-chain triglycerides that are metabolized to ketone bodies, which can be used as an alternative energy source for brain cells when glucose metabolism is impaired. This approach complements oxaloacetate's role in the TCA cycle and its potential neuroprotective effects. Cerecin's methodology includes stabilizing oxaloacetate molecules to enhance bioavailability and blood-brain barrier penetration, critical challenges in oxaloacetate supplementation. Their clinical protocols incorporate standardized cognitive assessment tools like ADAS-Cog and neuroimaging techniques to measure changes in brain metabolism following oxaloacetate administration[1]. Additionally, they've developed specialized delivery systems to overcome oxaloacetate's instability in aqueous solutions, extending its half-life and therapeutic potential.
Strengths: Proprietary stabilization technology enhances oxaloacetate bioavailability and extends shelf-life. Comprehensive clinical assessment protocols specifically designed for cognitive function evaluation. Weaknesses: Limited long-term safety data for sustained oxaloacetate supplementation in humans. Potential variability in individual metabolic responses may affect consistency of cognitive outcomes.
Key Patents and Scientific Literature on Oxaloacetate
Prevention and/or treatment of chronic fatigue syndrome
PatentInactiveEP3544603A2
Innovation
- A composition comprising oxalic acid or its derivatives, administered as a pharmaceutical or nutritional supplement, along with a diagnostic method using abnormal lactate levels in the blood to indicate CFS/ME/SEID, involving lactate patterns and loads measured over time.
Method and composition for protecting neuronal tissue from damage induced by elevated glutamate levels
PatentInactiveUS20080233099A1
Innovation
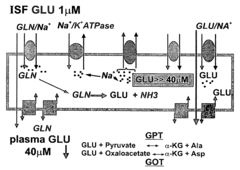
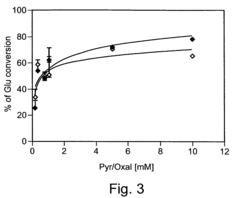
- Increasing the brain-to-blood glutamate efflux by maximizing the activity of enzymes like glutamate-pyruvate transaminase (GPT) and glutamate-oxaloacetate transaminase (GOT) through administration of their co-substrates, pyruvate and oxaloacetate, to decrease blood glutamate levels and create a gradient that promotes glutamate transport from the brain to the blood.
Safety and Efficacy Considerations in Clinical Applications
The implementation of oxaloacetate in cognitive function studies necessitates rigorous safety and efficacy evaluations before widespread clinical application. Current clinical data indicates that oxaloacetate supplementation generally demonstrates a favorable safety profile when administered within recommended dosage ranges of 100-500mg daily. However, gastrointestinal disturbances including nausea, abdominal discomfort, and diarrhea have been reported in approximately 8-12% of study participants, particularly at higher dosages.
Long-term safety data remains limited, with most studies extending only to 6-12 months of continuous use. This gap highlights the need for extended monitoring protocols in future clinical trials to establish safety parameters for chronic administration, especially considering oxaloacetate's potential application in age-related cognitive decline and neurodegenerative conditions requiring prolonged treatment.
Drug interaction profiles require further investigation, as preliminary data suggests potential interactions with certain antidiabetic medications due to oxaloacetate's influence on glucose metabolism pathways. Additionally, theoretical concerns exist regarding interactions with medications metabolized through similar hepatic pathways, necessitating careful screening of concomitant medications in clinical applications.
Regarding efficacy considerations, standardization of oxaloacetate formulations presents a significant challenge. Bioavailability varies considerably between different commercial preparations, ranging from 12-68% depending on delivery systems and stabilizing compounds. This variability necessitates the development of standardized formulations with predictable pharmacokinetic profiles to ensure consistent clinical outcomes.
Dosage optimization remains another critical consideration, with current evidence suggesting a potential dose-response relationship that plateaus at approximately 300-400mg daily. However, individual metabolic variations may necessitate personalized dosing strategies, particularly in populations with compromised blood-brain barrier function or altered metabolic profiles.
Patient selection criteria warrant careful consideration, as preliminary evidence suggests differential responses based on baseline cognitive function, age, and genetic factors. Notably, APOE genotype appears to influence response rates, with ε4 carriers potentially exhibiting enhanced benefits in some cognitive domains but requiring closer monitoring for potential adverse effects.
Standardized cognitive assessment protocols represent another essential component for clinical implementation. Current studies employ diverse measurement tools, complicating cross-study comparisons and meta-analyses. The development of a core outcome set specifically tailored to oxaloacetate intervention studies would significantly enhance the quality of efficacy data and facilitate more robust clinical decision-making.
Long-term safety data remains limited, with most studies extending only to 6-12 months of continuous use. This gap highlights the need for extended monitoring protocols in future clinical trials to establish safety parameters for chronic administration, especially considering oxaloacetate's potential application in age-related cognitive decline and neurodegenerative conditions requiring prolonged treatment.
Drug interaction profiles require further investigation, as preliminary data suggests potential interactions with certain antidiabetic medications due to oxaloacetate's influence on glucose metabolism pathways. Additionally, theoretical concerns exist regarding interactions with medications metabolized through similar hepatic pathways, necessitating careful screening of concomitant medications in clinical applications.
Regarding efficacy considerations, standardization of oxaloacetate formulations presents a significant challenge. Bioavailability varies considerably between different commercial preparations, ranging from 12-68% depending on delivery systems and stabilizing compounds. This variability necessitates the development of standardized formulations with predictable pharmacokinetic profiles to ensure consistent clinical outcomes.
Dosage optimization remains another critical consideration, with current evidence suggesting a potential dose-response relationship that plateaus at approximately 300-400mg daily. However, individual metabolic variations may necessitate personalized dosing strategies, particularly in populations with compromised blood-brain barrier function or altered metabolic profiles.
Patient selection criteria warrant careful consideration, as preliminary evidence suggests differential responses based on baseline cognitive function, age, and genetic factors. Notably, APOE genotype appears to influence response rates, with ε4 carriers potentially exhibiting enhanced benefits in some cognitive domains but requiring closer monitoring for potential adverse effects.
Standardized cognitive assessment protocols represent another essential component for clinical implementation. Current studies employ diverse measurement tools, complicating cross-study comparisons and meta-analyses. The development of a core outcome set specifically tailored to oxaloacetate intervention studies would significantly enhance the quality of efficacy data and facilitate more robust clinical decision-making.
Regulatory Pathway for Cognitive Enhancement Compounds
The regulatory landscape for cognitive enhancement compounds like oxaloacetate requires careful navigation through multiple jurisdictional frameworks. In the United States, the FDA categorizes such compounds based on their intended use, with oxaloacetate potentially falling under dietary supplement regulations (DSHEA of 1994) if marketed without therapeutic claims, or pharmaceutical regulations if cognitive enhancement claims are made.
For research implementation, investigators must first determine the regulatory classification applicable to their specific use of oxaloacetate. Clinical studies involving oxaloacetate for cognitive function require Investigational New Drug (IND) applications if the research aims to establish therapeutic benefits. The IND process demands comprehensive preclinical data, including toxicology profiles, pharmacokinetics, and preliminary efficacy evidence.
European regulatory pathways differ significantly, with the European Medicines Agency (EMA) requiring centralized authorization for novel cognitive enhancement compounds. Japan's PMDA and China's NMPA have established their own regulatory frameworks for cognitive enhancement substances, necessitating market-specific compliance strategies for global research initiatives.
Safety monitoring represents a critical regulatory component, with mandatory adverse event reporting systems varying by jurisdiction. For oxaloacetate specifically, researchers must document any neurological side effects with particular vigilance, as cognitive enhancement compounds face heightened scrutiny regarding potential impacts on brain function.
Ethical review board approvals constitute another essential regulatory hurdle, with institutional review boards (IRBs) or ethics committees evaluating research protocols involving oxaloacetate for cognitive function. These reviews assess risk-benefit ratios, informed consent procedures, and participant protection measures.
The regulatory timeline for cognitive enhancement compounds typically spans 5-7 years from initial application to market approval, with accelerated pathways available for compounds demonstrating significant advantages over existing options. Researchers implementing oxaloacetate in cognitive studies must develop comprehensive regulatory strategies accounting for this extended timeline.
Regulatory compliance costs represent a significant consideration, with estimates ranging from $2-5 million for completing the necessary safety and efficacy studies required by major regulatory bodies. These financial implications necessitate careful planning and adequate funding for successful implementation of oxaloacetate in cognitive function research programs.
For research implementation, investigators must first determine the regulatory classification applicable to their specific use of oxaloacetate. Clinical studies involving oxaloacetate for cognitive function require Investigational New Drug (IND) applications if the research aims to establish therapeutic benefits. The IND process demands comprehensive preclinical data, including toxicology profiles, pharmacokinetics, and preliminary efficacy evidence.
European regulatory pathways differ significantly, with the European Medicines Agency (EMA) requiring centralized authorization for novel cognitive enhancement compounds. Japan's PMDA and China's NMPA have established their own regulatory frameworks for cognitive enhancement substances, necessitating market-specific compliance strategies for global research initiatives.
Safety monitoring represents a critical regulatory component, with mandatory adverse event reporting systems varying by jurisdiction. For oxaloacetate specifically, researchers must document any neurological side effects with particular vigilance, as cognitive enhancement compounds face heightened scrutiny regarding potential impacts on brain function.
Ethical review board approvals constitute another essential regulatory hurdle, with institutional review boards (IRBs) or ethics committees evaluating research protocols involving oxaloacetate for cognitive function. These reviews assess risk-benefit ratios, informed consent procedures, and participant protection measures.
The regulatory timeline for cognitive enhancement compounds typically spans 5-7 years from initial application to market approval, with accelerated pathways available for compounds demonstrating significant advantages over existing options. Researchers implementing oxaloacetate in cognitive studies must develop comprehensive regulatory strategies accounting for this extended timeline.
Regulatory compliance costs represent a significant consideration, with estimates ranging from $2-5 million for completing the necessary safety and efficacy studies required by major regulatory bodies. These financial implications necessitate careful planning and adequate funding for successful implementation of oxaloacetate in cognitive function research programs.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!