How to Implement PET Scan Quality Control Protocols
MAR 2, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PET Scan QC Background and Objectives
Positron Emission Tomography (PET) scanning has evolved significantly since its inception in the 1970s, transforming from experimental research tools to essential clinical diagnostic instruments. The technology's foundation lies in the detection of gamma rays emitted by positron-annihilation events from administered radiopharmaceuticals, enabling visualization of metabolic processes within living tissues. This capability has revolutionized oncology, cardiology, and neurology by providing functional imaging that complements anatomical imaging modalities.
The development trajectory of PET technology demonstrates continuous advancement in detector sensitivity, spatial resolution, and image reconstruction algorithms. Early PET systems suffered from limited resolution and lengthy acquisition times, but technological innovations including time-of-flight capabilities, silicon photomultiplier detectors, and advanced reconstruction techniques have dramatically improved image quality and diagnostic accuracy. The integration of PET with computed tomography (PET/CT) and magnetic resonance imaging (PET/MRI) has further enhanced clinical utility by providing simultaneous functional and anatomical information.
Quality control protocols in PET imaging have become increasingly sophisticated as the technology matured and clinical applications expanded. The complexity of PET systems, involving cyclotron-produced radiopharmaceuticals, intricate detector arrays, and sophisticated data processing algorithms, necessitates comprehensive quality assurance programs to ensure consistent performance and reliable diagnostic results. Regulatory bodies and professional organizations have established guidelines recognizing that systematic quality control is fundamental to maintaining diagnostic confidence and patient safety.
The primary objective of implementing robust PET scan quality control protocols centers on ensuring consistent image quality, quantitative accuracy, and system reliability across all clinical examinations. These protocols aim to detect and correct performance degradations before they impact diagnostic capabilities, thereby maintaining the high standards required for accurate disease detection, staging, and treatment monitoring. Quality control measures must address hardware performance, software functionality, and procedural consistency to achieve optimal clinical outcomes.
Contemporary quality control objectives extend beyond basic system functionality to encompass standardization across multi-site clinical trials, harmonization of quantitative measurements, and optimization of radiation dose management. The increasing emphasis on quantitative PET imaging for treatment response assessment and personalized medicine demands stringent quality control measures that ensure measurement reproducibility and accuracy. These objectives align with broader healthcare quality initiatives focused on evidence-based medicine and patient-centered care delivery.
The development trajectory of PET technology demonstrates continuous advancement in detector sensitivity, spatial resolution, and image reconstruction algorithms. Early PET systems suffered from limited resolution and lengthy acquisition times, but technological innovations including time-of-flight capabilities, silicon photomultiplier detectors, and advanced reconstruction techniques have dramatically improved image quality and diagnostic accuracy. The integration of PET with computed tomography (PET/CT) and magnetic resonance imaging (PET/MRI) has further enhanced clinical utility by providing simultaneous functional and anatomical information.
Quality control protocols in PET imaging have become increasingly sophisticated as the technology matured and clinical applications expanded. The complexity of PET systems, involving cyclotron-produced radiopharmaceuticals, intricate detector arrays, and sophisticated data processing algorithms, necessitates comprehensive quality assurance programs to ensure consistent performance and reliable diagnostic results. Regulatory bodies and professional organizations have established guidelines recognizing that systematic quality control is fundamental to maintaining diagnostic confidence and patient safety.
The primary objective of implementing robust PET scan quality control protocols centers on ensuring consistent image quality, quantitative accuracy, and system reliability across all clinical examinations. These protocols aim to detect and correct performance degradations before they impact diagnostic capabilities, thereby maintaining the high standards required for accurate disease detection, staging, and treatment monitoring. Quality control measures must address hardware performance, software functionality, and procedural consistency to achieve optimal clinical outcomes.
Contemporary quality control objectives extend beyond basic system functionality to encompass standardization across multi-site clinical trials, harmonization of quantitative measurements, and optimization of radiation dose management. The increasing emphasis on quantitative PET imaging for treatment response assessment and personalized medicine demands stringent quality control measures that ensure measurement reproducibility and accuracy. These objectives align with broader healthcare quality initiatives focused on evidence-based medicine and patient-centered care delivery.
Market Demand for PET Imaging Quality Assurance
The global healthcare industry is experiencing unprecedented growth in medical imaging services, with PET imaging representing one of the fastest-expanding segments. Healthcare facilities worldwide are increasingly recognizing the critical importance of implementing robust quality assurance protocols to ensure diagnostic accuracy and patient safety. This growing awareness has created substantial market demand for comprehensive PET imaging quality control solutions.
Healthcare providers face mounting pressure from regulatory bodies to maintain stringent quality standards in nuclear medicine departments. Accreditation organizations and government health agencies are implementing more rigorous inspection protocols, requiring facilities to demonstrate consistent adherence to quality control procedures. This regulatory environment has significantly amplified the demand for systematic quality assurance frameworks that can withstand scrutiny and ensure compliance.
The expansion of PET imaging applications beyond oncology into cardiology, neurology, and research applications has broadened the market scope for quality assurance solutions. Each clinical application requires specialized quality control protocols tailored to specific imaging parameters and diagnostic requirements. This diversification has created multiple market segments, each with distinct quality assurance needs and procurement patterns.
Large hospital networks and imaging centers are increasingly seeking standardized quality control protocols that can be implemented across multiple locations. This trend toward standardization has created demand for scalable quality assurance solutions that can maintain consistency while accommodating site-specific requirements. Healthcare organizations recognize that standardized protocols reduce training costs, improve operational efficiency, and minimize liability risks.
The integration of artificial intelligence and automated quality control systems has emerged as a significant market driver. Healthcare facilities are actively seeking solutions that can reduce manual quality control workload while improving detection of subtle quality issues. This technological evolution has created new market opportunities for vendors offering AI-powered quality assurance platforms.
Cost containment pressures in healthcare have paradoxically increased demand for quality control protocols, as facilities recognize that preventing quality issues is more cost-effective than addressing consequences of poor image quality. Healthcare administrators understand that robust quality assurance programs reduce repeat examinations, minimize diagnostic errors, and protect against potential litigation costs.
The shortage of qualified nuclear medicine technologists has intensified demand for quality control protocols that can be effectively implemented by staff with varying experience levels. Facilities require comprehensive training programs and standardized procedures that ensure consistent quality regardless of operator expertise, creating additional market demand for educational and procedural resources.
Healthcare providers face mounting pressure from regulatory bodies to maintain stringent quality standards in nuclear medicine departments. Accreditation organizations and government health agencies are implementing more rigorous inspection protocols, requiring facilities to demonstrate consistent adherence to quality control procedures. This regulatory environment has significantly amplified the demand for systematic quality assurance frameworks that can withstand scrutiny and ensure compliance.
The expansion of PET imaging applications beyond oncology into cardiology, neurology, and research applications has broadened the market scope for quality assurance solutions. Each clinical application requires specialized quality control protocols tailored to specific imaging parameters and diagnostic requirements. This diversification has created multiple market segments, each with distinct quality assurance needs and procurement patterns.
Large hospital networks and imaging centers are increasingly seeking standardized quality control protocols that can be implemented across multiple locations. This trend toward standardization has created demand for scalable quality assurance solutions that can maintain consistency while accommodating site-specific requirements. Healthcare organizations recognize that standardized protocols reduce training costs, improve operational efficiency, and minimize liability risks.
The integration of artificial intelligence and automated quality control systems has emerged as a significant market driver. Healthcare facilities are actively seeking solutions that can reduce manual quality control workload while improving detection of subtle quality issues. This technological evolution has created new market opportunities for vendors offering AI-powered quality assurance platforms.
Cost containment pressures in healthcare have paradoxically increased demand for quality control protocols, as facilities recognize that preventing quality issues is more cost-effective than addressing consequences of poor image quality. Healthcare administrators understand that robust quality assurance programs reduce repeat examinations, minimize diagnostic errors, and protect against potential litigation costs.
The shortage of qualified nuclear medicine technologists has intensified demand for quality control protocols that can be effectively implemented by staff with varying experience levels. Facilities require comprehensive training programs and standardized procedures that ensure consistent quality regardless of operator expertise, creating additional market demand for educational and procedural resources.
Current PET QC Challenges and Technical Barriers
PET scan quality control implementation faces significant technical barriers that stem from the complex nature of nuclear medicine imaging systems. The primary challenge lies in the inherent variability of radiotracer production and decay characteristics, which directly impacts image quality and quantitative accuracy. Manufacturing inconsistencies in radiopharmaceuticals, coupled with varying uptake periods and patient-specific metabolic factors, create substantial obstacles for standardized quality control protocols.
Scanner hardware limitations present another critical barrier. PET detectors experience performance degradation over time, with crystal efficiency declining and photomultiplier tube sensitivity varying across detector rings. These hardware inconsistencies result in non-uniform spatial resolution and sensitivity patterns that are difficult to predict and compensate for through conventional calibration methods. Additionally, the high cost of detector replacement and system maintenance creates economic constraints that often delay necessary hardware updates.
Data acquisition and reconstruction algorithms introduce computational challenges that complicate quality control implementation. Modern iterative reconstruction methods, while improving image quality, are highly sensitive to acquisition parameters and can mask or amplify certain types of artifacts. The lack of standardized reconstruction protocols across different vendor platforms creates inconsistencies in image characteristics, making it difficult to establish universal quality metrics and acceptance criteria.
Patient-related factors constitute a significant technical barrier, particularly in clinical environments where protocol adherence varies. Patient motion, breathing artifacts, and physiological variations in tracer uptake create dynamic quality control challenges that cannot be addressed through static calibration procedures. The integration of motion correction algorithms and respiratory gating systems adds complexity to quality assurance workflows while introducing new potential failure modes.
Regulatory compliance requirements further complicate quality control implementation. Different international standards and local regulations often conflict or overlap, creating confusion about mandatory testing procedures and acceptance criteria. The documentation burden associated with comprehensive quality control programs requires substantial administrative resources and specialized training that many facilities struggle to maintain consistently.
Integration challenges between PET systems and existing hospital information technology infrastructure create additional technical barriers. Quality control data management, automated analysis workflows, and real-time monitoring systems require sophisticated software solutions that must interface with multiple vendor platforms and legacy systems, often resulting in fragmented and inefficient quality assurance processes.
Scanner hardware limitations present another critical barrier. PET detectors experience performance degradation over time, with crystal efficiency declining and photomultiplier tube sensitivity varying across detector rings. These hardware inconsistencies result in non-uniform spatial resolution and sensitivity patterns that are difficult to predict and compensate for through conventional calibration methods. Additionally, the high cost of detector replacement and system maintenance creates economic constraints that often delay necessary hardware updates.
Data acquisition and reconstruction algorithms introduce computational challenges that complicate quality control implementation. Modern iterative reconstruction methods, while improving image quality, are highly sensitive to acquisition parameters and can mask or amplify certain types of artifacts. The lack of standardized reconstruction protocols across different vendor platforms creates inconsistencies in image characteristics, making it difficult to establish universal quality metrics and acceptance criteria.
Patient-related factors constitute a significant technical barrier, particularly in clinical environments where protocol adherence varies. Patient motion, breathing artifacts, and physiological variations in tracer uptake create dynamic quality control challenges that cannot be addressed through static calibration procedures. The integration of motion correction algorithms and respiratory gating systems adds complexity to quality assurance workflows while introducing new potential failure modes.
Regulatory compliance requirements further complicate quality control implementation. Different international standards and local regulations often conflict or overlap, creating confusion about mandatory testing procedures and acceptance criteria. The documentation burden associated with comprehensive quality control programs requires substantial administrative resources and specialized training that many facilities struggle to maintain consistently.
Integration challenges between PET systems and existing hospital information technology infrastructure create additional technical barriers. Quality control data management, automated analysis workflows, and real-time monitoring systems require sophisticated software solutions that must interface with multiple vendor platforms and legacy systems, often resulting in fragmented and inefficient quality assurance processes.
Existing PET Quality Control Protocol Solutions
01 Automated quality control systems for PET imaging
Automated quality control protocols utilize computer-based systems to monitor and assess PET scan quality parameters. These systems can automatically detect artifacts, evaluate image quality metrics, and generate quality control reports without manual intervention. The automation reduces human error and ensures consistent quality assessment across multiple scans and facilities.- Automated quality control systems for PET imaging: Automated quality control protocols utilize computer-based systems to monitor and assess PET scan quality parameters. These systems can automatically detect artifacts, evaluate image quality metrics, and generate quality control reports without manual intervention. The automation reduces human error and ensures consistent quality assessment across multiple scans and facilities.
- Calibration and standardization methods for PET scanners: Quality control protocols include regular calibration procedures using phantom devices and standardized test objects to ensure accurate quantification and image quality. These methods verify scanner performance parameters such as spatial resolution, sensitivity, and uniformity. Calibration protocols help maintain consistency across different scanning sessions and enable comparison of results over time.
- Image reconstruction quality assessment techniques: Quality control measures are applied during and after image reconstruction to evaluate the integrity of reconstructed PET images. These techniques assess noise levels, contrast resolution, and reconstruction artifacts. Advanced algorithms analyze reconstructed images to identify potential issues that may affect diagnostic accuracy and ensure optimal image quality for clinical interpretation.
- Real-time monitoring and feedback systems: Real-time quality control systems continuously monitor PET scanner performance during acquisition and provide immediate feedback to operators. These systems track critical parameters such as detector efficiency, coincidence timing, and count rates. Immediate alerts enable prompt corrective actions when deviations from acceptable quality standards are detected, minimizing the need for repeat scans.
- Data validation and compliance protocols: Quality control frameworks incorporate comprehensive data validation procedures to ensure compliance with regulatory standards and clinical guidelines. These protocols verify data integrity, proper patient identification, and adherence to scanning protocols. Documentation systems track quality control activities and maintain audit trails for regulatory compliance and accreditation purposes.
02 Calibration and standardization methods for PET scanners
Quality control protocols include regular calibration procedures using phantom devices and standardized test objects to ensure accurate quantification and image quality. These methods verify scanner performance parameters such as spatial resolution, sensitivity, and uniformity. Calibration protocols help maintain consistency in imaging results over time and across different scanning systems.Expand Specific Solutions03 Image reconstruction quality assessment techniques
Quality control measures evaluate the reconstruction algorithms and parameters used in PET imaging to ensure optimal image quality. These techniques assess factors such as noise levels, contrast resolution, and reconstruction artifacts. Advanced assessment methods can identify and correct issues in the reconstruction process to improve diagnostic accuracy.Expand Specific Solutions04 Real-time monitoring and feedback systems
Quality control protocols incorporate real-time monitoring capabilities that provide immediate feedback during PET scan acquisition. These systems can detect technical issues, patient motion, or equipment malfunctions as they occur, allowing for immediate corrective action. Real-time monitoring helps prevent the need for repeat scans and improves overall workflow efficiency.Expand Specific Solutions05 Data validation and compliance verification protocols
Quality control frameworks include comprehensive data validation procedures to ensure compliance with regulatory standards and clinical requirements. These protocols verify data integrity, proper documentation, and adherence to established quality metrics. Validation systems can track quality control results over time and generate compliance reports for regulatory purposes.Expand Specific Solutions
Major Players in PET QC Solutions Market
The PET scan quality control protocols market represents a mature yet evolving sector within medical imaging, driven by increasing demand for precise diagnostic capabilities and regulatory compliance requirements. The industry has reached a consolidation phase where established players like Siemens Medical Solutions USA, Canon Medical Systems Corp., and Toshiba Medical Systems dominate through comprehensive imaging solutions and extensive service networks. Technology maturity varies significantly across market segments, with traditional hardware manufacturers like United Imaging Healthcare and MinFound Medical Systems advancing integrated PET/CT systems, while emerging players such as Positrigo AG and Cerebriu A/S introduce innovative AI-driven quality assurance platforms. Academic institutions including Washington University in St. Louis, Cleveland Clinic Foundation, and Huazhong University of Science & Technology contribute essential research and protocol development. The market demonstrates strong growth potential, particularly in automated quality control solutions and cloud-based monitoring systems, as healthcare providers seek to enhance diagnostic accuracy while reducing operational costs and ensuring regulatory compliance across diverse clinical environments.
Shanghai United Imaging Healthcare Co., Ltd.
Technical Solution: United Imaging Healthcare has developed integrated quality control protocols for their uEXPLORER total-body PET/CT system, featuring automated daily QC procedures including detector uniformity assessment, spatial resolution testing, and sensitivity verification. Their uWS-MI workstation provides comprehensive QC analysis tools with automated phantom positioning and measurement protocols. The system incorporates real-time monitoring of detector performance, automatic gain adjustment, and standardized calibration procedures. Their quality control framework includes daily blank scans, weekly uniformity checks, and monthly spatial resolution assessments using standardized phantoms, all integrated into their uCloud platform for remote monitoring and data analysis.
Strengths: Advanced total-body imaging capabilities, integrated cloud-based monitoring, cost-effective solutions. Weaknesses: Limited global service network, newer technology with less clinical validation, regional market focus.
Siemens Medical Solutions USA, Inc.
Technical Solution: Siemens implements comprehensive PET scan quality control protocols through their syngo.via platform, which provides automated daily quality assurance testing including detector uniformity checks, spatial resolution verification, and sensitivity measurements. Their Biograph PET/CT systems feature built-in quality control phantoms and automated calibration procedures that ensure consistent image quality. The system performs automatic detector normalization, dead-time correction, and scatter correction algorithms. Siemens also provides standardized protocols for routine maintenance including detector crystal calibration, timing alignment verification, and system performance monitoring through their eHealth cloud-based remote monitoring services.
Strengths: Comprehensive automated QC procedures, integrated cloud monitoring, established clinical workflows. Weaknesses: High system complexity, requires specialized training, expensive maintenance contracts.
Core Technologies in PET QC Implementation
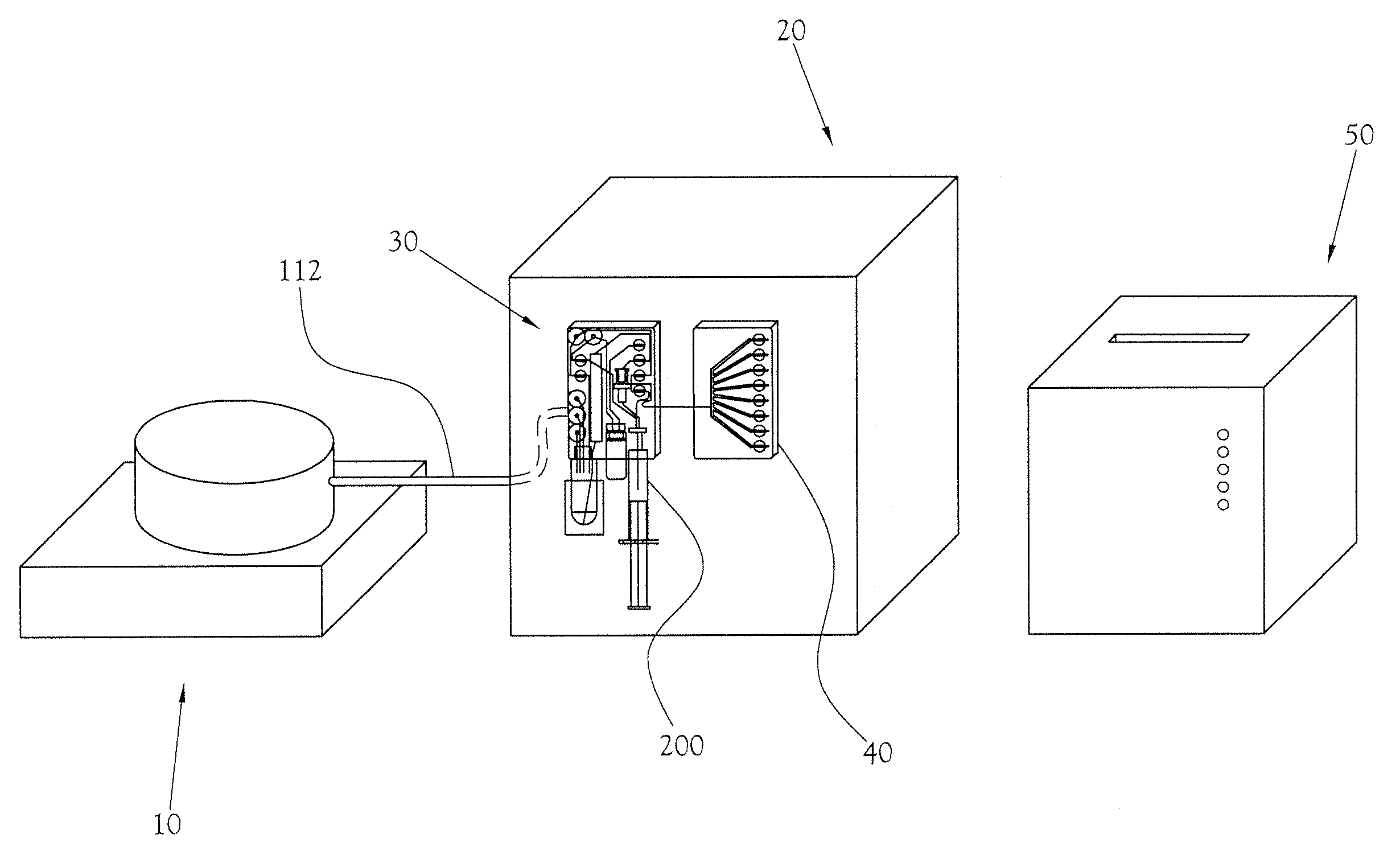
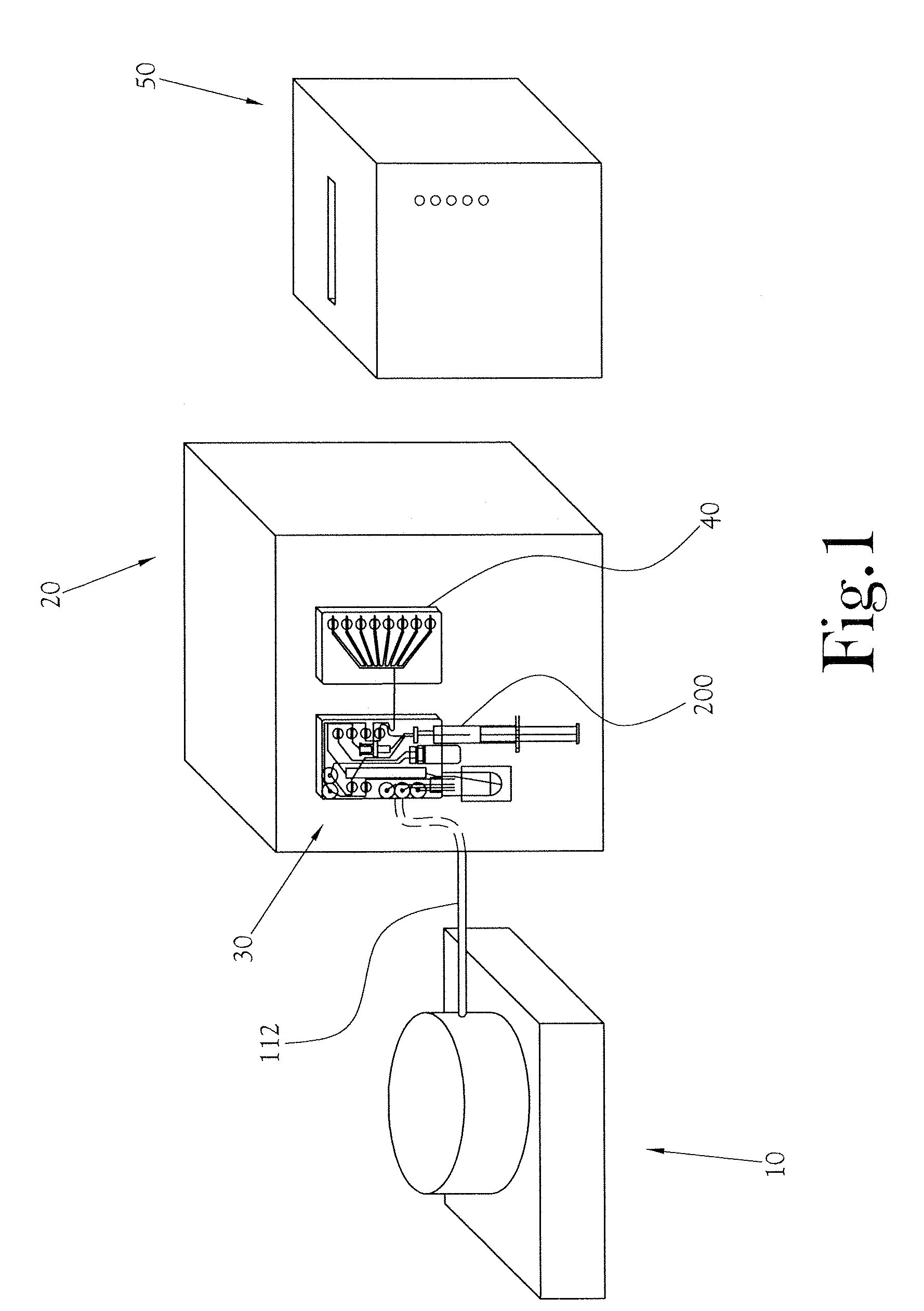
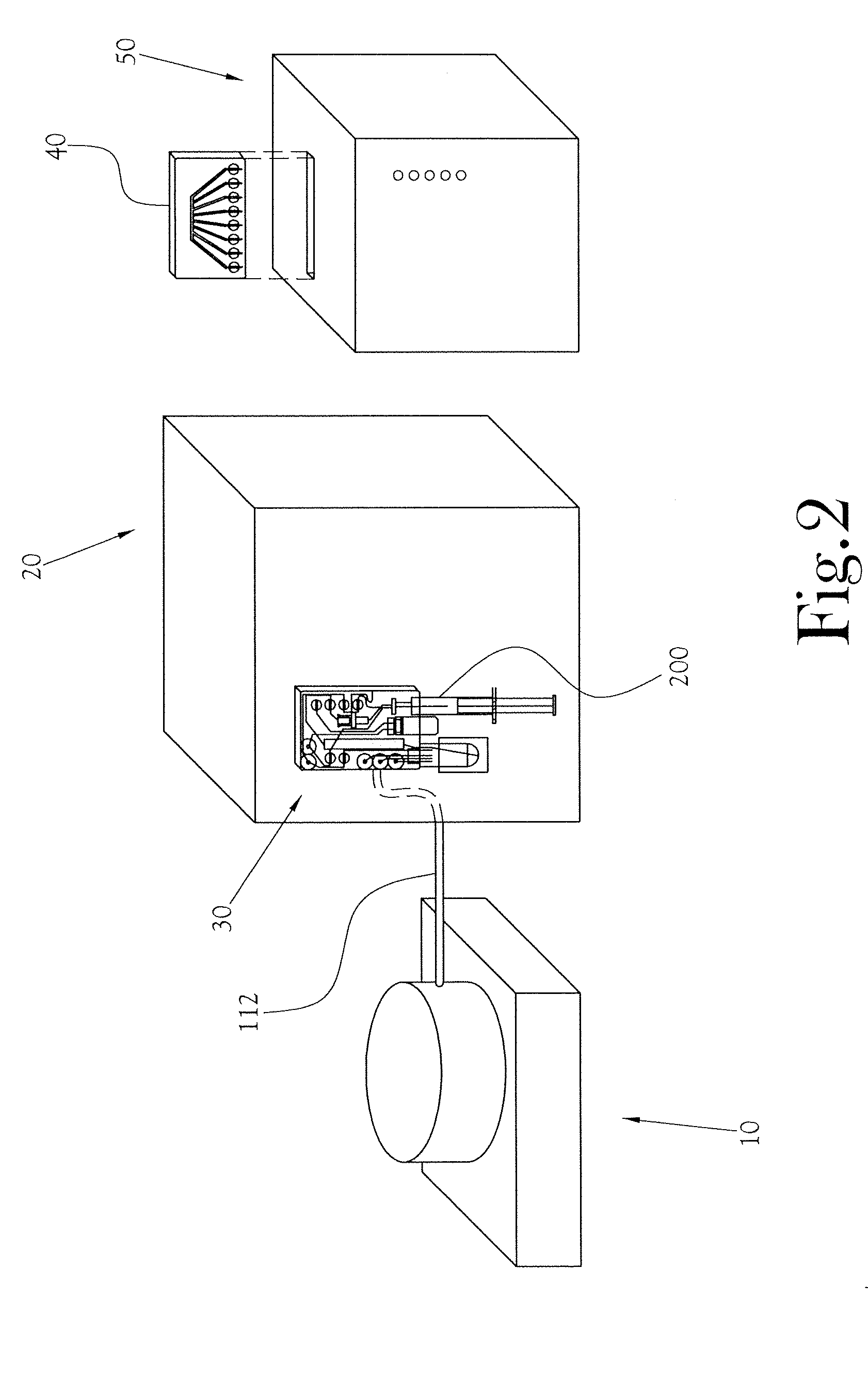
Quality Control Module for Biomarker Generator System
PatentInactiveUS20110070158A1
Innovation
- A compact PET biomarker production system incorporating a small, low-power cyclotron, microfluidic chemical production module, and automated quality control module that allows for on-site production and rapid testing of unit doses, reducing the need for transportation and shortening quality control times.
Method and system for automated quality control platform
PatentWO2013012798A1
Innovation
- An automated quality control system that uniquely identifies each radiopharmaceutical and implements tailored quality control programs, performing multiple quality control tests automatically and collecting information to ensure precise and efficient production, while also enabling remote data collection and real-time monitoring.
Regulatory Standards for PET QC Compliance
PET scan quality control protocols must adhere to stringent regulatory frameworks established by multiple governing bodies worldwide. The Nuclear Regulatory Commission (NRC) in the United States provides fundamental guidelines for radioactive material handling and facility operations, while the Food and Drug Administration (FDA) oversees radiopharmaceutical quality standards and imaging device approvals. These regulations form the backbone of PET facility compliance requirements.
International standards organizations play equally critical roles in establishing comprehensive QC frameworks. The International Atomic Energy Agency (IAEA) publishes technical reports and safety standards that serve as global benchmarks for nuclear medicine practices. The International Electrotechnical Commission (IEC) develops specific standards for medical electrical equipment, including PET scanners, addressing safety, performance, and electromagnetic compatibility requirements.
Professional medical organizations contribute specialized guidance tailored to clinical practice. The American College of Radiology (ACR) maintains accreditation programs that define minimum quality standards for PET facilities, including personnel qualifications, equipment specifications, and quality assurance procedures. The Society of Nuclear Medicine and Molecular Imaging (SNMMI) provides practice guidelines that complement regulatory requirements with evidence-based recommendations for optimal imaging protocols.
European regulatory frameworks operate under the Medical Device Regulation (MDR) and the European Medicines Agency (EMA) oversight, establishing parallel but distinct compliance pathways. These regulations emphasize post-market surveillance, clinical evidence requirements, and harmonized quality management systems across member states.
State and local regulatory bodies often impose additional requirements beyond federal standards. State radiation control programs typically mandate facility licensing, personnel certification, and regular inspection schedules. These multi-layered regulatory structures create complex compliance matrices that facilities must navigate systematically.
Emerging regulatory trends focus on risk-based approaches to quality management, emphasizing continuous improvement processes rather than purely prescriptive compliance measures. Recent updates to international standards incorporate advanced imaging technologies and artificial intelligence applications, requiring facilities to adapt their QC protocols accordingly while maintaining fundamental safety and quality principles.
International standards organizations play equally critical roles in establishing comprehensive QC frameworks. The International Atomic Energy Agency (IAEA) publishes technical reports and safety standards that serve as global benchmarks for nuclear medicine practices. The International Electrotechnical Commission (IEC) develops specific standards for medical electrical equipment, including PET scanners, addressing safety, performance, and electromagnetic compatibility requirements.
Professional medical organizations contribute specialized guidance tailored to clinical practice. The American College of Radiology (ACR) maintains accreditation programs that define minimum quality standards for PET facilities, including personnel qualifications, equipment specifications, and quality assurance procedures. The Society of Nuclear Medicine and Molecular Imaging (SNMMI) provides practice guidelines that complement regulatory requirements with evidence-based recommendations for optimal imaging protocols.
European regulatory frameworks operate under the Medical Device Regulation (MDR) and the European Medicines Agency (EMA) oversight, establishing parallel but distinct compliance pathways. These regulations emphasize post-market surveillance, clinical evidence requirements, and harmonized quality management systems across member states.
State and local regulatory bodies often impose additional requirements beyond federal standards. State radiation control programs typically mandate facility licensing, personnel certification, and regular inspection schedules. These multi-layered regulatory structures create complex compliance matrices that facilities must navigate systematically.
Emerging regulatory trends focus on risk-based approaches to quality management, emphasizing continuous improvement processes rather than purely prescriptive compliance measures. Recent updates to international standards incorporate advanced imaging technologies and artificial intelligence applications, requiring facilities to adapt their QC protocols accordingly while maintaining fundamental safety and quality principles.
Risk Management in PET QC Protocol Design
Risk management in PET quality control protocol design represents a critical framework for ensuring patient safety, diagnostic accuracy, and regulatory compliance throughout the imaging process. The systematic identification and mitigation of potential hazards requires a comprehensive approach that addresses technical, operational, and clinical risk factors inherent in PET scanning procedures.
The primary risk categories in PET QC protocols encompass radiation safety concerns, equipment malfunction scenarios, radiopharmaceutical quality issues, and human error factors. Radiation exposure risks demand stringent monitoring protocols for both patients and staff, with established dose limits and ALARA principles guiding protocol development. Equipment-related risks include detector calibration failures, scanner performance degradation, and software malfunctions that could compromise image quality or patient safety.
Radiopharmaceutical risks constitute another significant concern, involving potential contamination, incorrect dosing, or chemical impurities that could affect both diagnostic accuracy and patient wellbeing. These risks necessitate robust quality assurance procedures for radiopharmaceutical preparation, storage, and administration, including regular purity testing and sterility verification protocols.
Human factors represent a substantial risk component, encompassing operator errors in protocol execution, inadequate training, and communication breakdowns between departments. Effective risk management strategies must address these vulnerabilities through standardized procedures, comprehensive training programs, and clear communication protocols between nuclear medicine technologists, physicians, and support staff.
Risk assessment methodologies in PET QC protocol design typically employ failure mode and effects analysis (FMEA) to systematically evaluate potential failure points and their consequences. This approach enables prioritization of risk mitigation efforts based on severity, occurrence probability, and detectability factors. Critical control points are established throughout the imaging workflow, from patient preparation through image reconstruction and interpretation.
Mitigation strategies include redundant safety systems, automated monitoring capabilities, and real-time alert mechanisms that can detect deviations from established parameters. Regular protocol reviews and incident analysis contribute to continuous improvement of risk management frameworks, ensuring adaptation to emerging technologies and evolving regulatory requirements while maintaining the highest standards of patient care and diagnostic excellence.
The primary risk categories in PET QC protocols encompass radiation safety concerns, equipment malfunction scenarios, radiopharmaceutical quality issues, and human error factors. Radiation exposure risks demand stringent monitoring protocols for both patients and staff, with established dose limits and ALARA principles guiding protocol development. Equipment-related risks include detector calibration failures, scanner performance degradation, and software malfunctions that could compromise image quality or patient safety.
Radiopharmaceutical risks constitute another significant concern, involving potential contamination, incorrect dosing, or chemical impurities that could affect both diagnostic accuracy and patient wellbeing. These risks necessitate robust quality assurance procedures for radiopharmaceutical preparation, storage, and administration, including regular purity testing and sterility verification protocols.
Human factors represent a substantial risk component, encompassing operator errors in protocol execution, inadequate training, and communication breakdowns between departments. Effective risk management strategies must address these vulnerabilities through standardized procedures, comprehensive training programs, and clear communication protocols between nuclear medicine technologists, physicians, and support staff.
Risk assessment methodologies in PET QC protocol design typically employ failure mode and effects analysis (FMEA) to systematically evaluate potential failure points and their consequences. This approach enables prioritization of risk mitigation efforts based on severity, occurrence probability, and detectability factors. Critical control points are established throughout the imaging workflow, from patient preparation through image reconstruction and interpretation.
Mitigation strategies include redundant safety systems, automated monitoring capabilities, and real-time alert mechanisms that can detect deviations from established parameters. Regular protocol reviews and incident analysis contribute to continuous improvement of risk management frameworks, ensuring adaptation to emerging technologies and evolving regulatory requirements while maintaining the highest standards of patient care and diagnostic excellence.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!