How to Optimize Tangential Flow Filtration for High Yield
MAR 16, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
TFF Technology Background and Optimization Goals
Tangential Flow Filtration (TFF) emerged in the 1960s as a revolutionary membrane-based separation technology, fundamentally transforming bioprocessing and pharmaceutical manufacturing. Unlike traditional dead-end filtration, TFF operates by circulating feed solution parallel to the membrane surface, creating a tangential flow that continuously sweeps away accumulated particles and reduces membrane fouling. This cross-flow mechanism enables continuous operation and significantly extends membrane lifespan while maintaining consistent filtration performance.
The technology gained prominence in biotechnology applications during the 1980s and 1990s, particularly for protein purification, vaccine production, and monoclonal antibody processing. TFF systems utilize semi-permeable membranes with defined molecular weight cutoffs (MWCO) to achieve size-based separation, allowing smaller molecules to pass through as permeate while retaining larger target molecules in the retentate stream. This selective separation capability makes TFF indispensable for concentration, diafiltration, and buffer exchange operations.
Modern TFF systems have evolved to incorporate advanced membrane materials, including polyethersulfone (PES), regenerated cellulose, and modified polyethylene, each offering specific advantages for different applications. The technology now spans multiple scales, from laboratory benchtop units processing milliliters to industrial systems handling thousands of liters, maintaining scalability principles that ensure consistent performance across different operational volumes.
Current optimization challenges center on maximizing product yield while minimizing processing time and operational costs. Key performance indicators include flux rates, product recovery efficiency, membrane fouling resistance, and overall process robustness. The primary optimization goals focus on achieving yields exceeding 95% for target molecules while maintaining product quality and reducing membrane replacement frequency.
Contemporary research emphasizes developing predictive models for membrane performance, implementing real-time monitoring systems, and establishing optimal operating parameters including transmembrane pressure, crossflow velocity, and temperature control. These optimization efforts aim to enhance process efficiency, reduce manufacturing costs, and ensure consistent product quality in biopharmaceutical production environments.
The technology gained prominence in biotechnology applications during the 1980s and 1990s, particularly for protein purification, vaccine production, and monoclonal antibody processing. TFF systems utilize semi-permeable membranes with defined molecular weight cutoffs (MWCO) to achieve size-based separation, allowing smaller molecules to pass through as permeate while retaining larger target molecules in the retentate stream. This selective separation capability makes TFF indispensable for concentration, diafiltration, and buffer exchange operations.
Modern TFF systems have evolved to incorporate advanced membrane materials, including polyethersulfone (PES), regenerated cellulose, and modified polyethylene, each offering specific advantages for different applications. The technology now spans multiple scales, from laboratory benchtop units processing milliliters to industrial systems handling thousands of liters, maintaining scalability principles that ensure consistent performance across different operational volumes.
Current optimization challenges center on maximizing product yield while minimizing processing time and operational costs. Key performance indicators include flux rates, product recovery efficiency, membrane fouling resistance, and overall process robustness. The primary optimization goals focus on achieving yields exceeding 95% for target molecules while maintaining product quality and reducing membrane replacement frequency.
Contemporary research emphasizes developing predictive models for membrane performance, implementing real-time monitoring systems, and establishing optimal operating parameters including transmembrane pressure, crossflow velocity, and temperature control. These optimization efforts aim to enhance process efficiency, reduce manufacturing costs, and ensure consistent product quality in biopharmaceutical production environments.
Market Demand for High-Yield TFF Applications
The biopharmaceutical industry represents the primary driver for high-yield tangential flow filtration applications, with monoclonal antibody production constituting the largest market segment. Manufacturing facilities require TFF systems capable of processing large volumes while maintaining product integrity and maximizing recovery rates. The increasing complexity of biologics, including fusion proteins, antibody-drug conjugates, and gene therapies, demands more sophisticated filtration approaches that can handle diverse molecular weights and maintain biological activity throughout the concentration and purification processes.
Vaccine manufacturing has emerged as a critical application area, particularly following recent global health events that highlighted the need for rapid, scalable production capabilities. High-yield TFF systems enable manufacturers to process viral vectors, mRNA formulations, and protein-based vaccines with minimal product loss. The demand extends beyond traditional vaccines to include personalized medicine applications where small batch sizes require exceptionally efficient recovery rates to ensure economic viability.
The cell and gene therapy sector presents unique challenges that drive demand for optimized TFF solutions. These applications often involve processing precious cellular materials or viral vectors where even minor losses can significantly impact treatment availability and costs. Manufacturers require filtration systems that can achieve near-complete recovery while maintaining cell viability and vector infectivity. The growing pipeline of approved cell therapies continues to expand market requirements for specialized high-yield filtration equipment.
Industrial biotechnology applications, including enzyme production, fermentation-derived chemicals, and food ingredients, represent an expanding market segment. These applications often operate on larger scales where yield optimization directly translates to significant cost savings and improved profitability. The trend toward sustainable manufacturing processes has increased demand for efficient separation technologies that minimize waste generation and resource consumption.
Emerging applications in personalized medicine and point-of-care manufacturing create new market opportunities for compact, high-efficiency TFF systems. These applications require equipment that can deliver pharmaceutical-grade results in smaller footprints while maintaining the yield performance traditionally associated with large-scale manufacturing. The decentralization of biomanufacturing continues to drive innovation in portable, automated TFF solutions that can operate with minimal operator intervention while achieving optimal recovery rates.
Vaccine manufacturing has emerged as a critical application area, particularly following recent global health events that highlighted the need for rapid, scalable production capabilities. High-yield TFF systems enable manufacturers to process viral vectors, mRNA formulations, and protein-based vaccines with minimal product loss. The demand extends beyond traditional vaccines to include personalized medicine applications where small batch sizes require exceptionally efficient recovery rates to ensure economic viability.
The cell and gene therapy sector presents unique challenges that drive demand for optimized TFF solutions. These applications often involve processing precious cellular materials or viral vectors where even minor losses can significantly impact treatment availability and costs. Manufacturers require filtration systems that can achieve near-complete recovery while maintaining cell viability and vector infectivity. The growing pipeline of approved cell therapies continues to expand market requirements for specialized high-yield filtration equipment.
Industrial biotechnology applications, including enzyme production, fermentation-derived chemicals, and food ingredients, represent an expanding market segment. These applications often operate on larger scales where yield optimization directly translates to significant cost savings and improved profitability. The trend toward sustainable manufacturing processes has increased demand for efficient separation technologies that minimize waste generation and resource consumption.
Emerging applications in personalized medicine and point-of-care manufacturing create new market opportunities for compact, high-efficiency TFF systems. These applications require equipment that can deliver pharmaceutical-grade results in smaller footprints while maintaining the yield performance traditionally associated with large-scale manufacturing. The decentralization of biomanufacturing continues to drive innovation in portable, automated TFF solutions that can operate with minimal operator intervention while achieving optimal recovery rates.
Current TFF Performance Limitations and Challenges
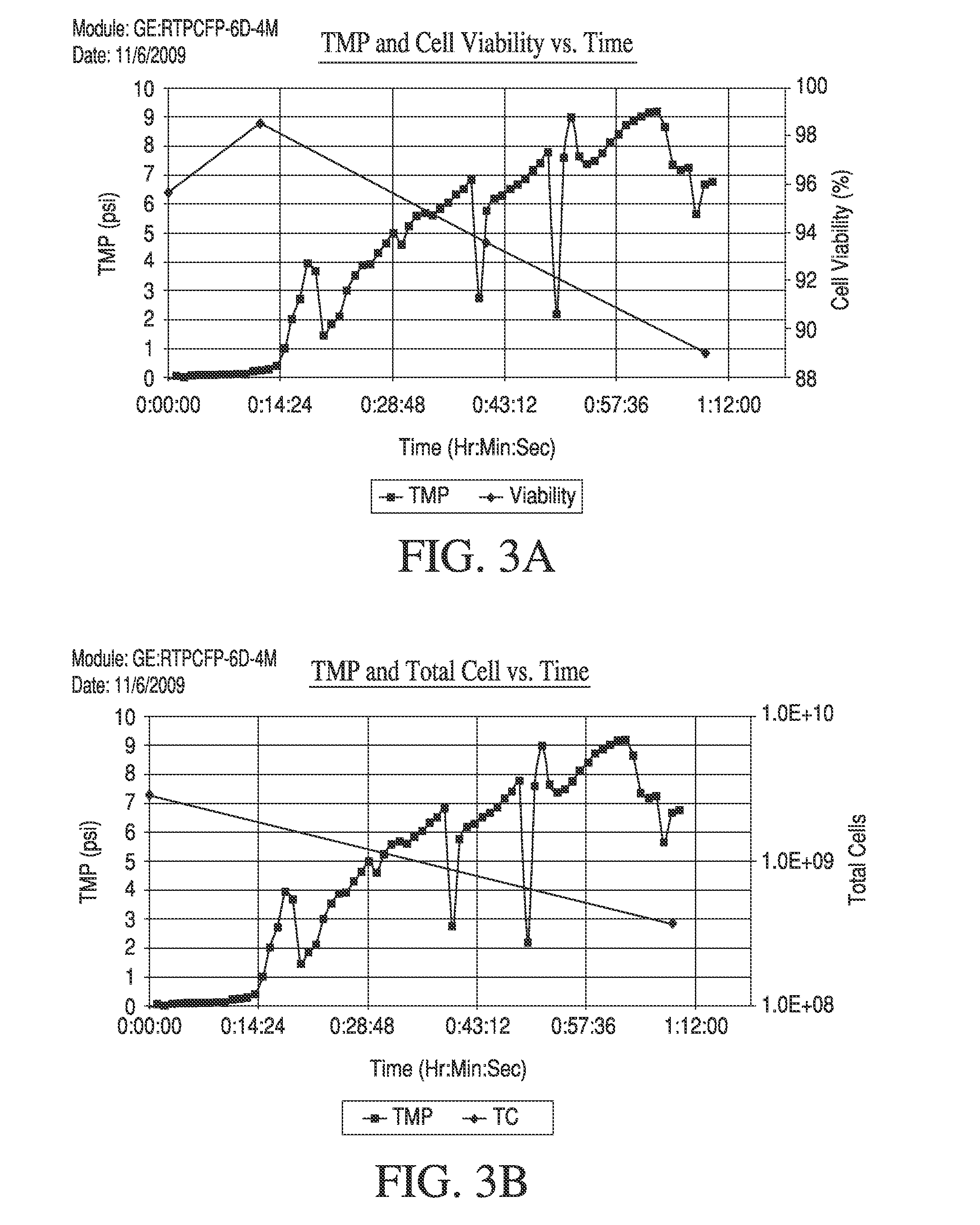
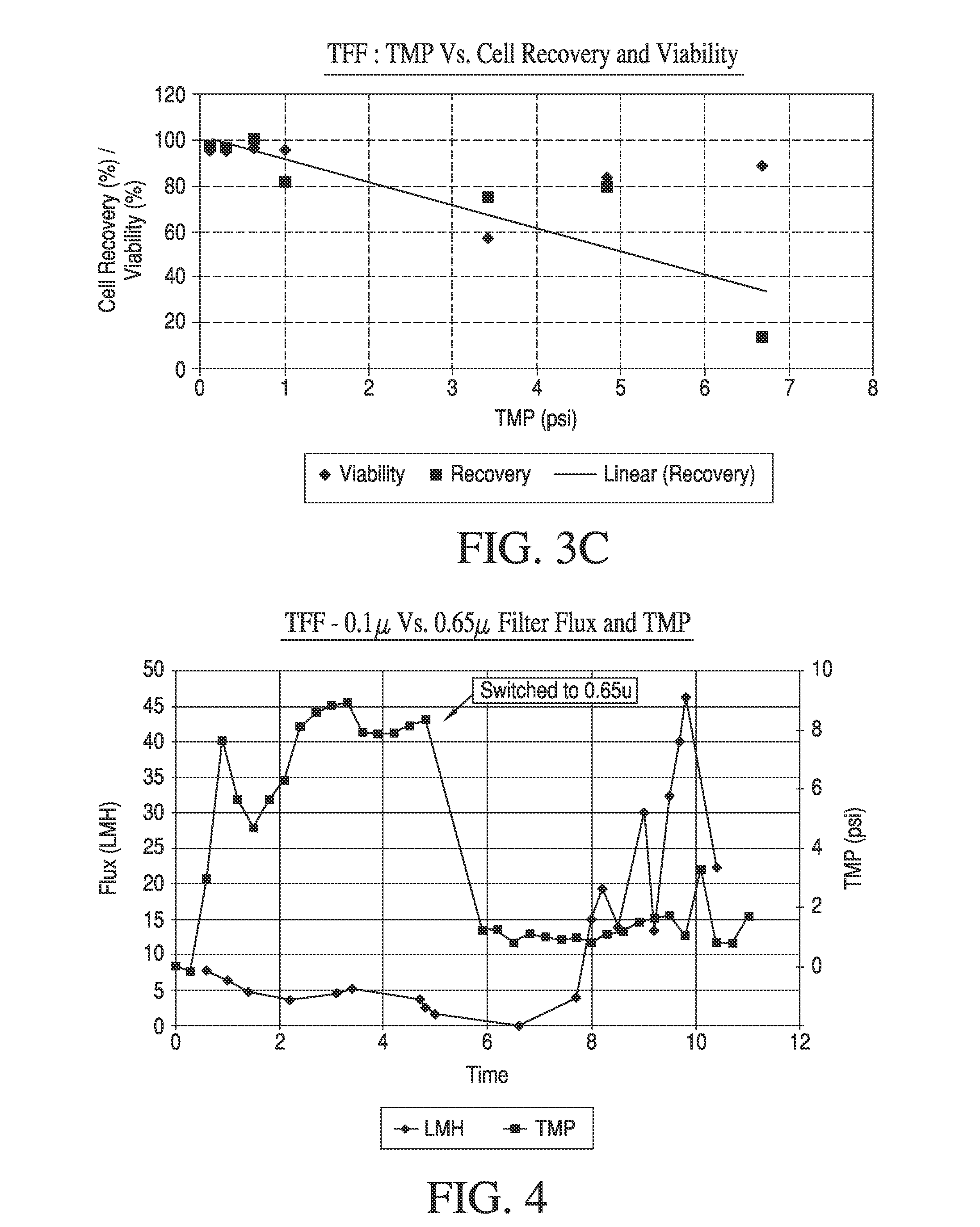
Tangential Flow Filtration systems face significant performance limitations that directly impact yield optimization across various bioprocessing applications. The most prominent challenge lies in concentration polarization, where retained molecules accumulate at the membrane surface, creating a secondary filtration layer that progressively reduces permeate flux. This phenomenon becomes particularly pronounced when processing high-concentration protein solutions or complex biological mixtures, leading to substantial yield losses over extended operation periods.
Membrane fouling represents another critical limitation affecting TFF performance. Irreversible fouling occurs when proteins, cells, or other particulates permanently adhere to membrane surfaces through hydrophobic interactions, electrostatic binding, or pore blocking mechanisms. This fouling not only reduces filtration efficiency but also necessitates frequent membrane replacement, increasing operational costs and downtime. The severity of fouling varies significantly depending on feed composition, pH conditions, and ionic strength.
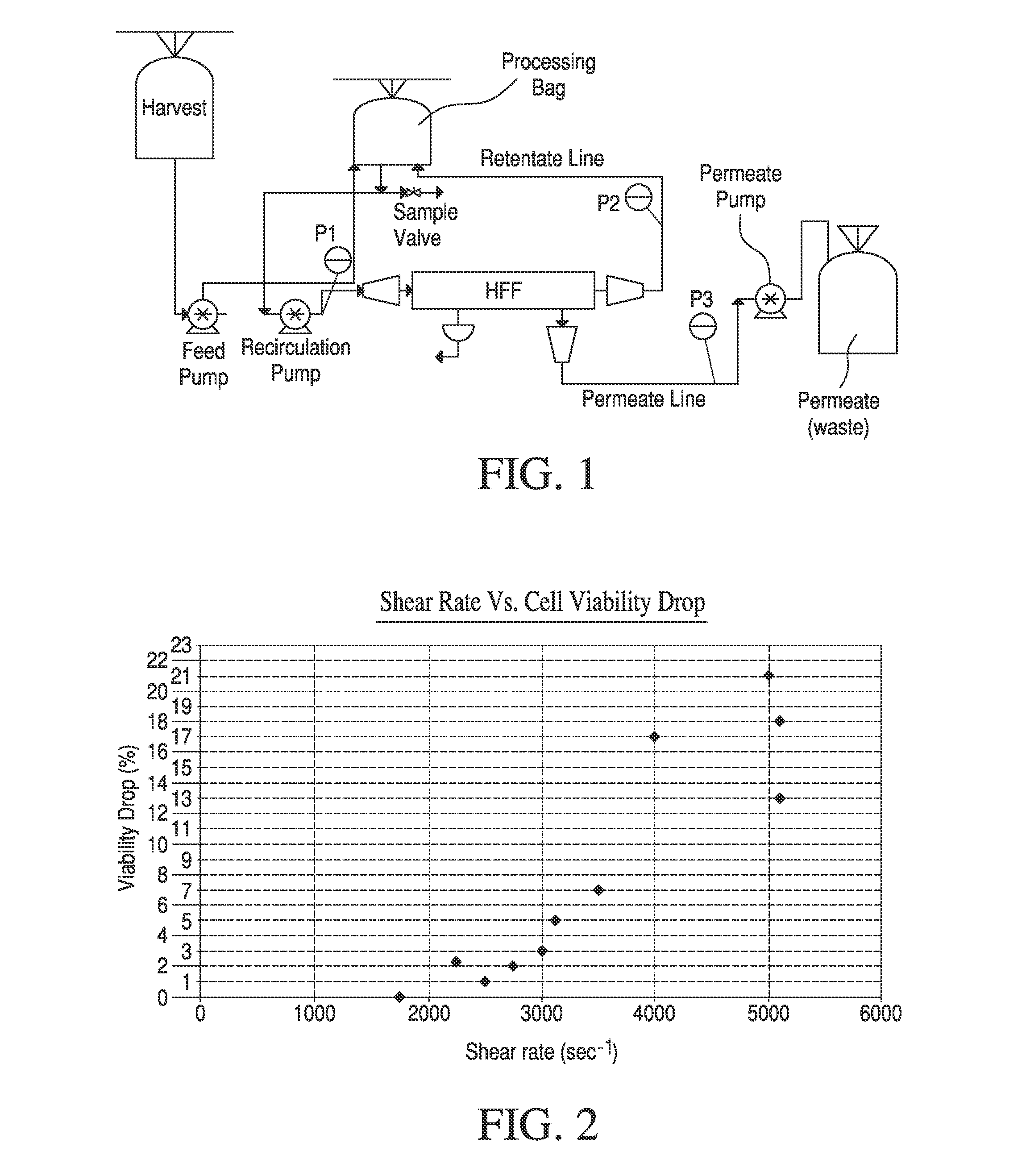
Shear stress-induced damage poses a substantial challenge when processing shear-sensitive biologics such as monoclonal antibodies, viral vectors, or cell-based products. Conventional TFF systems often operate at high crossflow velocities to minimize concentration polarization, but this approach can compromise product integrity and reduce overall yield. The balance between maintaining adequate crossflow rates and preserving product quality remains a persistent optimization challenge.
Temperature control limitations during extended filtration runs create additional performance constraints. Heat generation from pump operation and membrane friction can elevate process temperatures, potentially affecting product stability and membrane selectivity. Inadequate temperature management becomes particularly problematic during concentration steps where processing times extend over several hours.
Membrane selectivity challenges emerge when processing complex mixtures containing molecules with similar molecular weights. Traditional size-based separation mechanisms may inadequately distinguish between target products and closely related impurities, resulting in either product loss or insufficient purification. This limitation becomes more pronounced in applications requiring high purity standards, such as pharmaceutical manufacturing.
Scale-up difficulties represent a significant operational challenge, as TFF performance characteristics often change unpredictably when transitioning from laboratory to production scales. Factors such as membrane area scaling, flow distribution uniformity, and pressure drop variations can substantially impact yield optimization strategies developed at smaller scales.
Membrane fouling represents another critical limitation affecting TFF performance. Irreversible fouling occurs when proteins, cells, or other particulates permanently adhere to membrane surfaces through hydrophobic interactions, electrostatic binding, or pore blocking mechanisms. This fouling not only reduces filtration efficiency but also necessitates frequent membrane replacement, increasing operational costs and downtime. The severity of fouling varies significantly depending on feed composition, pH conditions, and ionic strength.
Shear stress-induced damage poses a substantial challenge when processing shear-sensitive biologics such as monoclonal antibodies, viral vectors, or cell-based products. Conventional TFF systems often operate at high crossflow velocities to minimize concentration polarization, but this approach can compromise product integrity and reduce overall yield. The balance between maintaining adequate crossflow rates and preserving product quality remains a persistent optimization challenge.
Temperature control limitations during extended filtration runs create additional performance constraints. Heat generation from pump operation and membrane friction can elevate process temperatures, potentially affecting product stability and membrane selectivity. Inadequate temperature management becomes particularly problematic during concentration steps where processing times extend over several hours.
Membrane selectivity challenges emerge when processing complex mixtures containing molecules with similar molecular weights. Traditional size-based separation mechanisms may inadequately distinguish between target products and closely related impurities, resulting in either product loss or insufficient purification. This limitation becomes more pronounced in applications requiring high purity standards, such as pharmaceutical manufacturing.
Scale-up difficulties represent a significant operational challenge, as TFF performance characteristics often change unpredictably when transitioning from laboratory to production scales. Factors such as membrane area scaling, flow distribution uniformity, and pressure drop variations can substantially impact yield optimization strategies developed at smaller scales.
Existing TFF Optimization Solutions and Methods
01 Optimization of tangential flow filtration membrane configuration and operating parameters
Tangential flow filtration yield can be improved by optimizing membrane configuration, including membrane material selection, pore size, and surface area. Operating parameters such as transmembrane pressure, crossflow velocity, and feed flow rate are critical factors that affect filtration efficiency and product recovery. Proper control of these parameters helps minimize membrane fouling and maximize permeate flux, thereby increasing overall yield.- Optimization of membrane configuration and operating parameters: Tangential flow filtration yield can be improved by optimizing membrane configuration, including membrane material selection, pore size, and surface area. Operating parameters such as transmembrane pressure, crossflow velocity, and feed flow rate are critical factors that affect filtration efficiency and product recovery. Proper control of these parameters helps minimize membrane fouling and maximize permeate flux, thereby increasing overall yield.
- Multi-stage filtration process design: Implementing multi-stage tangential flow filtration systems can significantly enhance yield by separating the filtration process into concentration and diafiltration steps. This approach allows for better control of product concentration while removing impurities and buffer exchange. Sequential filtration stages with different membrane cutoffs enable more efficient separation and higher product recovery rates.
- Membrane fouling prevention and cleaning strategies: Yield improvement in tangential flow filtration can be achieved through effective fouling prevention and membrane cleaning protocols. This includes pre-filtration of feed streams, optimization of solution pH and ionic strength, and implementation of periodic backflushing or cleaning-in-place procedures. Proper membrane maintenance and regeneration techniques help maintain consistent filtration performance and extend membrane lifetime.
- Buffer and formulation optimization: The composition of buffers and formulation conditions significantly impact tangential flow filtration yield. Optimizing buffer pH, ionic strength, and the presence of stabilizing agents can reduce protein aggregation and membrane interactions. Proper formulation design minimizes product loss due to adsorption and precipitation, while maintaining product stability throughout the filtration process.
- Process monitoring and control systems: Advanced process monitoring and automated control systems enhance tangential flow filtration yield by enabling real-time adjustment of operating parameters. Integration of sensors for pressure, flow rate, conductivity, and turbidity allows for continuous process optimization. Automated feedback control systems can maintain optimal filtration conditions and prevent process deviations that may reduce yield.
02 Use of diafiltration and buffer exchange strategies
Diafiltration techniques can be employed during tangential flow filtration to enhance product purity and yield by continuously replacing the filtrate with fresh buffer. This approach allows for effective removal of contaminants and concentration of target molecules while maintaining product integrity. Buffer exchange strategies help optimize the ionic strength and pH conditions to improve filtration performance and recovery rates.Expand Specific Solutions03 Implementation of multi-stage filtration processes
Multi-stage tangential flow filtration systems can significantly improve yield by employing sequential filtration steps with different membrane cutoffs. This cascading approach allows for progressive concentration and purification of target products while minimizing product loss. The staged process enables better control over selectivity and can be tailored to specific product characteristics and process requirements.Expand Specific Solutions04 Application of membrane cleaning and regeneration protocols
Regular membrane cleaning and regeneration procedures are essential for maintaining high filtration yields over extended operation periods. Chemical cleaning agents and protocols can be optimized to remove fouling layers and restore membrane permeability without damaging the membrane structure. Proper maintenance schedules and cleaning validation help ensure consistent performance and extend membrane lifetime, contributing to improved overall process yield.Expand Specific Solutions05 Integration of process monitoring and control systems
Advanced process monitoring and control systems enable real-time optimization of tangential flow filtration operations to maximize yield. Sensors and analytical tools can track key parameters such as pressure, flow rates, and product concentration throughout the filtration process. Automated feedback control systems can adjust operating conditions dynamically to maintain optimal performance and prevent yield losses due to process deviations or membrane fouling.Expand Specific Solutions
Key Players in TFF Equipment and Membrane Industry
The tangential flow filtration optimization market is experiencing robust growth driven by increasing biopharmaceutical manufacturing demands and the need for higher process yields. The industry is in a mature development stage with established players like Repligen Corp., EMD Millipore Corp., and Sartorius Stedim Biotech GmbH leading innovation through their ATF systems and advanced filtration technologies. The market demonstrates strong technical maturity, evidenced by companies like Cytiva (Global Life Sciences Solutions) and specialized filtration manufacturers such as Hangzhou Cobetter and InnovaPrep offering comprehensive solutions. Competition spans from large multinational corporations including Siemens AG and Bristol Myers Squibb to specialized biotechnology firms, indicating a diverse ecosystem supporting both equipment manufacturing and end-user applications across pharmaceutical, biotechnology, and research sectors.
Repligen Corp.
Technical Solution: Repligen has developed advanced TFF systems focusing on single-use technologies and process optimization. Their approach emphasizes automated control systems that monitor transmembrane pressure (TMP) and crossflow velocity in real-time to maintain optimal filtration conditions. The company's TFF cassettes feature proprietary membrane chemistry designed to minimize fouling and maximize product recovery. Their systems incorporate predictive algorithms that adjust operating parameters dynamically based on feed concentration and product characteristics, achieving recovery rates exceeding 95% for monoclonal antibodies and other biologics.
Strengths: Industry-leading single-use technology, excellent product recovery rates, strong automation capabilities. Weaknesses: Higher cost compared to traditional systems, limited scalability for very large volumes.
EMD Millipore Corp.
Technical Solution: EMD Millipore has developed comprehensive TFF optimization strategies centered on their Pellicon cassette technology and process development expertise. Their approach involves systematic evaluation of membrane selection, operating conditions, and buffer optimization to maximize yield. The company utilizes advanced membrane materials with controlled pore structures and surface modifications to reduce protein adsorption and improve selectivity. Their optimization protocols include detailed flux decline analysis, concentration polarization modeling, and multi-stage diafiltration strategies that can achieve over 90% product recovery while maintaining product quality and reducing processing time by up to 40%.
Strengths: Extensive membrane portfolio, proven track record in bioprocessing, comprehensive technical support. Weaknesses: Complex optimization procedures, requires significant process development time.
Core Innovations in High-Yield TFF Systems
High yield method and apparatus for volume reduction and washing of therapeutic cells using tangential flow filtration
PatentInactiveUS20120294836A1
Innovation
- The use of Tangential Flow Filtration (TFF) with optimized parameters such as low trans-membrane pressure and shear rates to concentrate and wash live mammalian cells, achieving high yields and viability while minimizing processing time and residual contaminants, using a completely closed and scalable system.
Tangential Flow Filtration Systems and Methods
PatentPendingUS20230372870A1
Innovation
- A TFF system comprising a module with a backpressure regulator and sensors that adjust transmembrane pressure dynamically to maintain constant flow rates, allowing automated membrane switching and parallel operation with feed generation, and promoting membrane recovery through buffer streams.
Process Validation Requirements for TFF Systems
Process validation for tangential flow filtration systems represents a critical regulatory requirement that ensures consistent performance, product quality, and patient safety in biopharmaceutical manufacturing. The validation framework encompasses comprehensive documentation, testing protocols, and performance qualification studies that demonstrate the TFF system's capability to consistently deliver high-yield outcomes within predetermined specifications.
The validation process begins with installation qualification, which verifies that all TFF system components are correctly installed according to manufacturer specifications and design requirements. This phase includes documentation of equipment specifications, utility connections, safety systems, and calibration certificates for critical instruments such as pressure transmitters, flow meters, and temperature sensors.
Operational qualification follows, focusing on demonstrating that the TFF system operates within design parameters across its intended operating ranges. Key parameters include transmembrane pressure control, crossflow velocity maintenance, temperature stability, and automated system responses to alarm conditions. This phase requires extensive testing of process control systems, data acquisition capabilities, and cleaning-in-place procedures.
Performance qualification represents the most critical validation phase, requiring demonstration of consistent high-yield performance using actual process conditions or validated process simulants. This involves multiple consecutive runs under worst-case scenarios, including maximum processing volumes, extended processing times, and challenging feed stream compositions. Statistical analysis of yield data must demonstrate process capability and reproducibility.
Documentation requirements encompass validation master plans, detailed test protocols, executed test records, deviation investigations, and comprehensive validation reports. Risk assessment methodologies, such as FMEA, must identify critical process parameters and establish appropriate monitoring and control strategies. Change control procedures ensure that any system modifications undergo appropriate revalidation activities.
Regulatory compliance extends beyond initial validation to include ongoing performance monitoring, periodic revalidation, and continuous process verification programs that maintain validated status throughout the system's operational lifecycle.
The validation process begins with installation qualification, which verifies that all TFF system components are correctly installed according to manufacturer specifications and design requirements. This phase includes documentation of equipment specifications, utility connections, safety systems, and calibration certificates for critical instruments such as pressure transmitters, flow meters, and temperature sensors.
Operational qualification follows, focusing on demonstrating that the TFF system operates within design parameters across its intended operating ranges. Key parameters include transmembrane pressure control, crossflow velocity maintenance, temperature stability, and automated system responses to alarm conditions. This phase requires extensive testing of process control systems, data acquisition capabilities, and cleaning-in-place procedures.
Performance qualification represents the most critical validation phase, requiring demonstration of consistent high-yield performance using actual process conditions or validated process simulants. This involves multiple consecutive runs under worst-case scenarios, including maximum processing volumes, extended processing times, and challenging feed stream compositions. Statistical analysis of yield data must demonstrate process capability and reproducibility.
Documentation requirements encompass validation master plans, detailed test protocols, executed test records, deviation investigations, and comprehensive validation reports. Risk assessment methodologies, such as FMEA, must identify critical process parameters and establish appropriate monitoring and control strategies. Change control procedures ensure that any system modifications undergo appropriate revalidation activities.
Regulatory compliance extends beyond initial validation to include ongoing performance monitoring, periodic revalidation, and continuous process verification programs that maintain validated status throughout the system's operational lifecycle.
Economic Impact of TFF Yield Optimization
The economic implications of TFF yield optimization extend far beyond immediate production metrics, fundamentally reshaping the cost structure of biopharmaceutical manufacturing. Enhanced yield performance directly translates to reduced raw material consumption per unit of final product, creating substantial savings in expensive upstream components such as cell culture media, growth factors, and specialized buffers. This efficiency gain becomes particularly significant in large-scale commercial production where even marginal improvements can generate millions in annual cost reductions.
Capital expenditure optimization represents another critical economic dimension. Higher TFF yields enable manufacturers to achieve target production volumes with smaller equipment footprints, reducing initial infrastructure investments and ongoing facility maintenance costs. The ability to process larger quantities of product through existing systems extends equipment lifecycle value and delays costly capacity expansion projects, providing significant return on investment for optimization initiatives.
Operational efficiency improvements cascade throughout the entire manufacturing value chain. Optimized TFF processes typically demonstrate greater consistency and predictability, reducing batch failure rates and associated waste disposal costs. This reliability enhancement minimizes expensive reprocessing activities and reduces quality control overhead, while simultaneously improving production scheduling accuracy and resource allocation efficiency.
The competitive positioning benefits of TFF yield optimization create substantial market value. Companies achieving superior yield performance can offer more competitive pricing while maintaining healthy profit margins, potentially capturing increased market share in price-sensitive therapeutic segments. Additionally, improved process efficiency enables faster response to market demand fluctuations and reduces time-to-market for new product launches.
Risk mitigation represents a significant but often undervalued economic benefit. Optimized TFF systems typically exhibit greater robustness against process variations and equipment failures, reducing the financial impact of production disruptions. This stability translates to more predictable revenue streams and reduced insurance costs, while also minimizing regulatory compliance risks that could result in costly manufacturing delays or product recalls.
Capital expenditure optimization represents another critical economic dimension. Higher TFF yields enable manufacturers to achieve target production volumes with smaller equipment footprints, reducing initial infrastructure investments and ongoing facility maintenance costs. The ability to process larger quantities of product through existing systems extends equipment lifecycle value and delays costly capacity expansion projects, providing significant return on investment for optimization initiatives.
Operational efficiency improvements cascade throughout the entire manufacturing value chain. Optimized TFF processes typically demonstrate greater consistency and predictability, reducing batch failure rates and associated waste disposal costs. This reliability enhancement minimizes expensive reprocessing activities and reduces quality control overhead, while simultaneously improving production scheduling accuracy and resource allocation efficiency.
The competitive positioning benefits of TFF yield optimization create substantial market value. Companies achieving superior yield performance can offer more competitive pricing while maintaining healthy profit margins, potentially capturing increased market share in price-sensitive therapeutic segments. Additionally, improved process efficiency enables faster response to market demand fluctuations and reduces time-to-market for new product launches.
Risk mitigation represents a significant but often undervalued economic benefit. Optimized TFF systems typically exhibit greater robustness against process variations and equipment failures, reducing the financial impact of production disruptions. This stability translates to more predictable revenue streams and reduced insurance costs, while also minimizing regulatory compliance risks that could result in costly manufacturing delays or product recalls.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!