How to Streamline Ionizing Ray Application Processes
MAR 16, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Ionizing Ray Tech Background and Goals
Ionizing radiation technology has evolved significantly since its discovery in the late 19th century, fundamentally transforming multiple industries through its unique ability to modify molecular structures and eliminate pathogens. The field encompasses various radiation sources including gamma rays, electron beams, and X-rays, each offering distinct advantages for specific applications. From Wilhelm Röntgen's initial X-ray discoveries to modern industrial sterilization systems, ionizing radiation has demonstrated remarkable versatility across medical device sterilization, food preservation, material modification, and pharmaceutical manufacturing.
The historical development trajectory reveals a consistent pattern of expanding applications driven by technological refinements and regulatory acceptance. Early medical applications in the 1900s established the foundation for therapeutic uses, while mid-20th century innovations introduced industrial-scale sterilization processes. The emergence of electron beam accelerators and cobalt-60 sources in the 1960s marked a pivotal transition toward commercial viability, enabling large-scale processing capabilities that previously remained economically unfeasible.
Contemporary ionizing radiation applications face increasing complexity as industries demand higher throughput, enhanced precision, and improved cost-effectiveness. Traditional batch processing methods, while reliable, often create bottlenecks that limit operational efficiency and increase processing costs. The challenge lies in balancing radiation dose uniformity, processing speed, and quality assurance requirements while maintaining regulatory compliance across diverse product categories.
Current technological objectives focus on developing streamlined application processes that minimize processing time, reduce operational complexity, and enhance dose distribution uniformity. Key goals include implementing automated dosimetry systems, optimizing radiation source utilization, and integrating real-time monitoring capabilities. Advanced process control systems aim to eliminate manual interventions while ensuring consistent treatment outcomes across varying product geometries and material compositions.
The strategic vision encompasses creating adaptive processing environments capable of handling diverse product portfolios without extensive reconfiguration requirements. This includes developing modular system architectures that accommodate varying throughput demands and implementing predictive maintenance protocols to minimize unplanned downtime. Integration of artificial intelligence and machine learning algorithms represents a crucial objective for optimizing processing parameters and predicting optimal treatment conditions based on product characteristics and quality specifications.
The historical development trajectory reveals a consistent pattern of expanding applications driven by technological refinements and regulatory acceptance. Early medical applications in the 1900s established the foundation for therapeutic uses, while mid-20th century innovations introduced industrial-scale sterilization processes. The emergence of electron beam accelerators and cobalt-60 sources in the 1960s marked a pivotal transition toward commercial viability, enabling large-scale processing capabilities that previously remained economically unfeasible.
Contemporary ionizing radiation applications face increasing complexity as industries demand higher throughput, enhanced precision, and improved cost-effectiveness. Traditional batch processing methods, while reliable, often create bottlenecks that limit operational efficiency and increase processing costs. The challenge lies in balancing radiation dose uniformity, processing speed, and quality assurance requirements while maintaining regulatory compliance across diverse product categories.
Current technological objectives focus on developing streamlined application processes that minimize processing time, reduce operational complexity, and enhance dose distribution uniformity. Key goals include implementing automated dosimetry systems, optimizing radiation source utilization, and integrating real-time monitoring capabilities. Advanced process control systems aim to eliminate manual interventions while ensuring consistent treatment outcomes across varying product geometries and material compositions.
The strategic vision encompasses creating adaptive processing environments capable of handling diverse product portfolios without extensive reconfiguration requirements. This includes developing modular system architectures that accommodate varying throughput demands and implementing predictive maintenance protocols to minimize unplanned downtime. Integration of artificial intelligence and machine learning algorithms represents a crucial objective for optimizing processing parameters and predicting optimal treatment conditions based on product characteristics and quality specifications.
Market Demand for Streamlined Ray Applications
The global ionizing radiation market demonstrates robust growth driven by expanding applications across healthcare, industrial processing, and food safety sectors. Healthcare applications, particularly in medical device sterilization and cancer treatment, represent the largest market segment. The increasing prevalence of chronic diseases and growing demand for sterile medical equipment fuel continuous market expansion. Radiation therapy equipment and radiopharmaceuticals constitute significant revenue streams within this sector.
Industrial applications encompass polymer modification, semiconductor manufacturing, and materials testing. The electronics industry's rapid evolution creates sustained demand for precise radiation processes in component manufacturing and quality assurance. Cross-linking of polymers through electron beam irradiation has become essential for producing high-performance materials used in automotive, aerospace, and construction industries.
Food irradiation markets show accelerating growth as consumers increasingly prioritize food safety and extended shelf life. Regulatory approvals for radiation treatment of various food products continue expanding globally, creating new market opportunities. The technology addresses critical challenges in food preservation, pathogen elimination, and international trade facilitation by meeting stringent safety standards.
Current market dynamics reveal significant inefficiencies in radiation application processes. Extended processing times, complex regulatory compliance procedures, and high operational costs create barriers to market penetration. Many potential users, particularly small and medium enterprises, face challenges accessing radiation technologies due to process complexity and resource requirements.
Streamlined radiation processes could unlock substantial market potential by reducing implementation barriers and operational costs. Simplified workflows would enable broader adoption across industries currently underserved by radiation technologies. The pharmaceutical sector, in particular, shows strong interest in accelerated sterilization processes that maintain product integrity while reducing time-to-market.
Emerging markets in Asia-Pacific and Latin America present significant growth opportunities for streamlined radiation applications. These regions demonstrate increasing industrial development and healthcare infrastructure expansion, creating demand for efficient radiation solutions. Market penetration in these areas depends heavily on process simplification and cost optimization.
The convergence of automation technologies with radiation processes creates new market segments focused on integrated solutions. Industries seek comprehensive systems that combine radiation treatment with automated handling, monitoring, and quality control capabilities.
Industrial applications encompass polymer modification, semiconductor manufacturing, and materials testing. The electronics industry's rapid evolution creates sustained demand for precise radiation processes in component manufacturing and quality assurance. Cross-linking of polymers through electron beam irradiation has become essential for producing high-performance materials used in automotive, aerospace, and construction industries.
Food irradiation markets show accelerating growth as consumers increasingly prioritize food safety and extended shelf life. Regulatory approvals for radiation treatment of various food products continue expanding globally, creating new market opportunities. The technology addresses critical challenges in food preservation, pathogen elimination, and international trade facilitation by meeting stringent safety standards.
Current market dynamics reveal significant inefficiencies in radiation application processes. Extended processing times, complex regulatory compliance procedures, and high operational costs create barriers to market penetration. Many potential users, particularly small and medium enterprises, face challenges accessing radiation technologies due to process complexity and resource requirements.
Streamlined radiation processes could unlock substantial market potential by reducing implementation barriers and operational costs. Simplified workflows would enable broader adoption across industries currently underserved by radiation technologies. The pharmaceutical sector, in particular, shows strong interest in accelerated sterilization processes that maintain product integrity while reducing time-to-market.
Emerging markets in Asia-Pacific and Latin America present significant growth opportunities for streamlined radiation applications. These regions demonstrate increasing industrial development and healthcare infrastructure expansion, creating demand for efficient radiation solutions. Market penetration in these areas depends heavily on process simplification and cost optimization.
The convergence of automation technologies with radiation processes creates new market segments focused on integrated solutions. Industries seek comprehensive systems that combine radiation treatment with automated handling, monitoring, and quality control capabilities.
Current Ionizing Ray Process Limitations
Current ionizing ray application processes face significant operational inefficiencies that hinder widespread adoption across industries. Traditional radiation processing systems require extensive manual intervention for parameter adjustment, sample positioning, and dose monitoring, leading to prolonged processing times and increased labor costs. The lack of automated feedback mechanisms forces operators to rely on time-consuming trial-and-error approaches to achieve optimal irradiation conditions.
Equipment utilization rates remain suboptimal due to lengthy setup and calibration procedures between different processing batches. Conventional systems often require 30-60 minutes for reconfiguration when switching between product types or dose requirements, resulting in substantial downtime. This inefficiency becomes particularly problematic in high-throughput applications such as medical device sterilization and food processing, where rapid turnaround times are critical for maintaining supply chain efficiency.
Dose uniformity challenges represent another major limitation in current ionizing ray processes. Achieving consistent radiation distribution across complex geometries or varying product densities requires sophisticated positioning systems and multiple exposure angles. Existing solutions often struggle with three-dimensional dose mapping, leading to over-irradiation in some areas and under-irradiation in others, which compromises product quality and increases material waste.
Quality assurance protocols in current systems are predominantly reactive rather than predictive. Real-time dose monitoring capabilities are limited, forcing operators to rely on post-processing dosimetry measurements to verify treatment effectiveness. This approach introduces delays in identifying processing deviations and increases the risk of batch failures, particularly in applications requiring precise dose control such as polymer modification or semiconductor processing.
Integration challenges with existing manufacturing workflows create additional bottlenecks. Many ionizing ray systems operate as isolated processing units with limited connectivity to enterprise resource planning systems or production management software. This disconnection hampers data collection, traceability, and process optimization efforts, making it difficult to implement continuous improvement strategies.
Regulatory compliance requirements add another layer of complexity to current processes. Documentation and validation procedures for ionizing ray applications often involve manual record-keeping and paper-based reporting systems, which are prone to errors and time-consuming to maintain. The lack of automated compliance monitoring systems increases the administrative burden on operators and extends the time required for regulatory submissions and audits.
Equipment utilization rates remain suboptimal due to lengthy setup and calibration procedures between different processing batches. Conventional systems often require 30-60 minutes for reconfiguration when switching between product types or dose requirements, resulting in substantial downtime. This inefficiency becomes particularly problematic in high-throughput applications such as medical device sterilization and food processing, where rapid turnaround times are critical for maintaining supply chain efficiency.
Dose uniformity challenges represent another major limitation in current ionizing ray processes. Achieving consistent radiation distribution across complex geometries or varying product densities requires sophisticated positioning systems and multiple exposure angles. Existing solutions often struggle with three-dimensional dose mapping, leading to over-irradiation in some areas and under-irradiation in others, which compromises product quality and increases material waste.
Quality assurance protocols in current systems are predominantly reactive rather than predictive. Real-time dose monitoring capabilities are limited, forcing operators to rely on post-processing dosimetry measurements to verify treatment effectiveness. This approach introduces delays in identifying processing deviations and increases the risk of batch failures, particularly in applications requiring precise dose control such as polymer modification or semiconductor processing.
Integration challenges with existing manufacturing workflows create additional bottlenecks. Many ionizing ray systems operate as isolated processing units with limited connectivity to enterprise resource planning systems or production management software. This disconnection hampers data collection, traceability, and process optimization efforts, making it difficult to implement continuous improvement strategies.
Regulatory compliance requirements add another layer of complexity to current processes. Documentation and validation procedures for ionizing ray applications often involve manual record-keeping and paper-based reporting systems, which are prone to errors and time-consuming to maintain. The lack of automated compliance monitoring systems increases the administrative burden on operators and extends the time required for regulatory submissions and audits.
Existing Ray Process Optimization Solutions
01 Ionizing radiation sterilization and disinfection processes
Ionizing radiation, such as gamma rays, electron beams, or X-rays, can be applied to sterilize and disinfect various materials, products, and equipment. This process is effective in eliminating microorganisms, bacteria, viruses, and other pathogens without leaving chemical residues. The radiation dose and exposure time are carefully controlled to achieve the desired level of sterilization while maintaining product integrity. This technology is widely used in medical device sterilization, food preservation, and pharmaceutical applications.- Ionizing radiation sterilization and disinfection processes: Ionizing radiation, such as gamma rays, electron beams, or X-rays, can be applied to sterilize and disinfect various materials, products, and equipment. This process is widely used in medical device manufacturing, pharmaceutical production, and food processing industries. The ionizing radiation effectively eliminates microorganisms, bacteria, viruses, and other pathogens without leaving chemical residues. The process parameters such as radiation dose, exposure time, and penetration depth are carefully controlled to ensure effective sterilization while maintaining product integrity.
- Material modification and surface treatment using ionizing radiation: Ionizing radiation can be utilized to modify material properties and treat surfaces of various substrates. This application includes cross-linking of polymers, improving mechanical strength, altering surface characteristics, and enhancing adhesion properties. The radiation-induced modifications can improve wear resistance, chemical resistance, and overall durability of materials. This process is particularly valuable in manufacturing industries for enhancing product performance and extending service life of components.
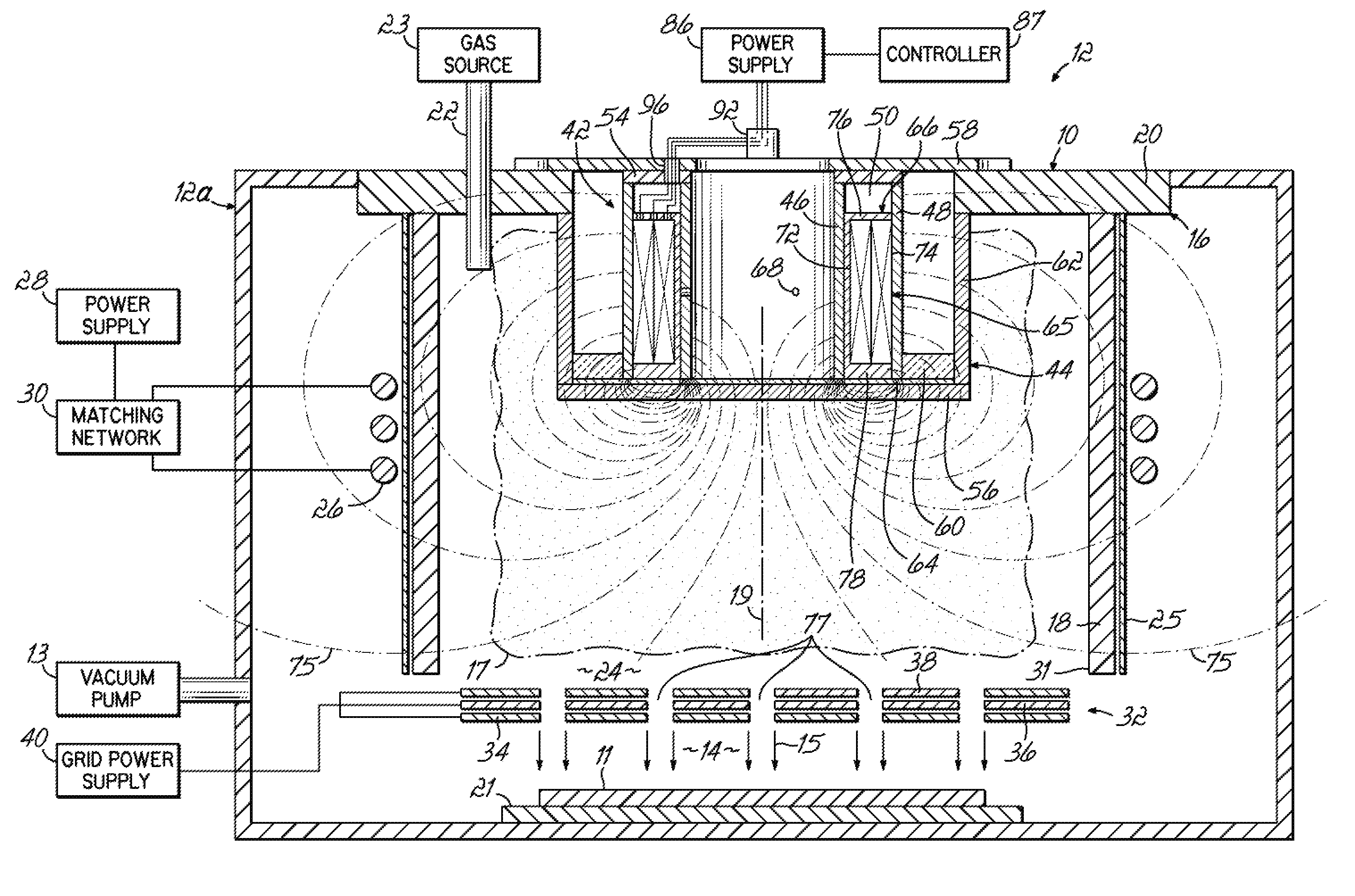
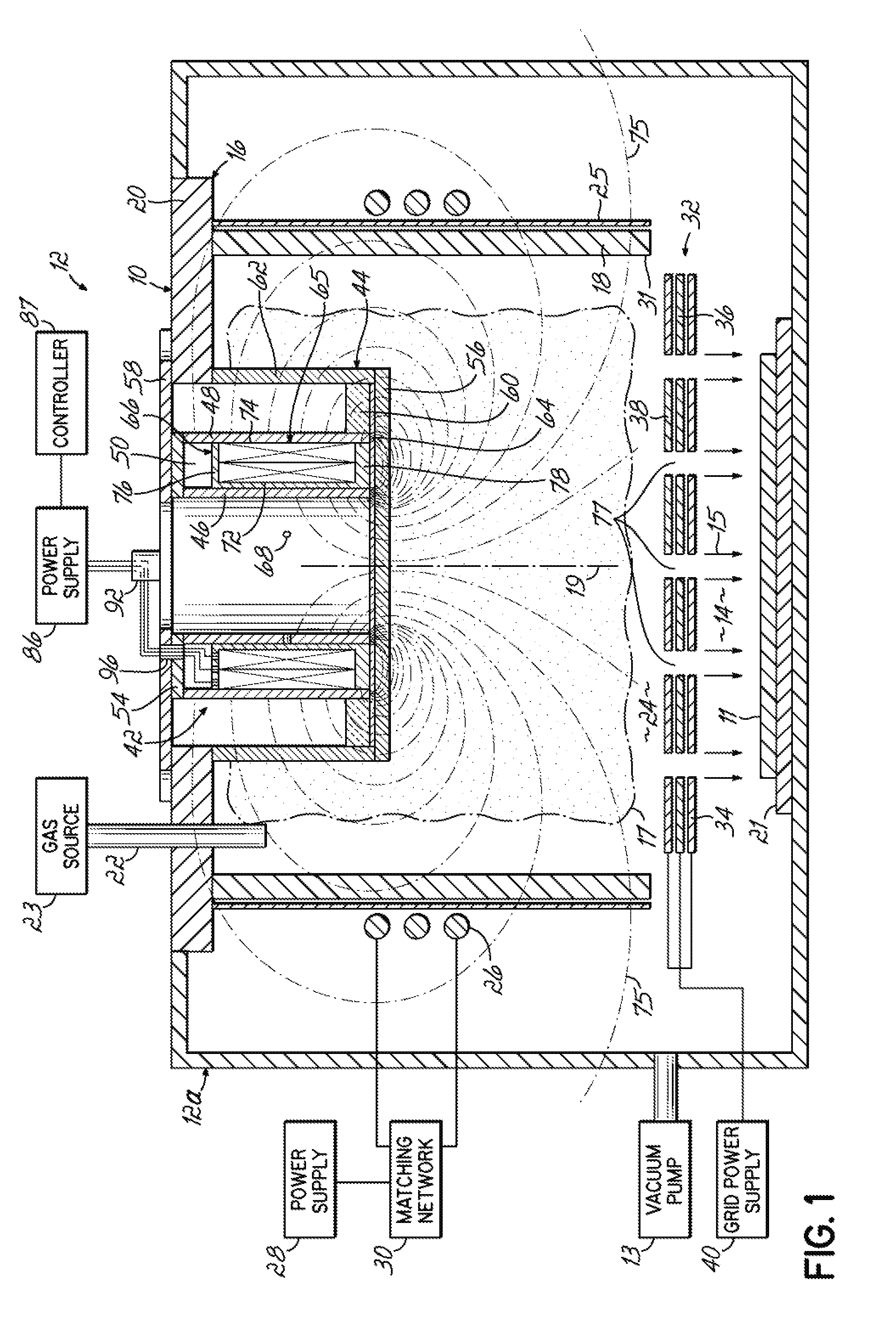
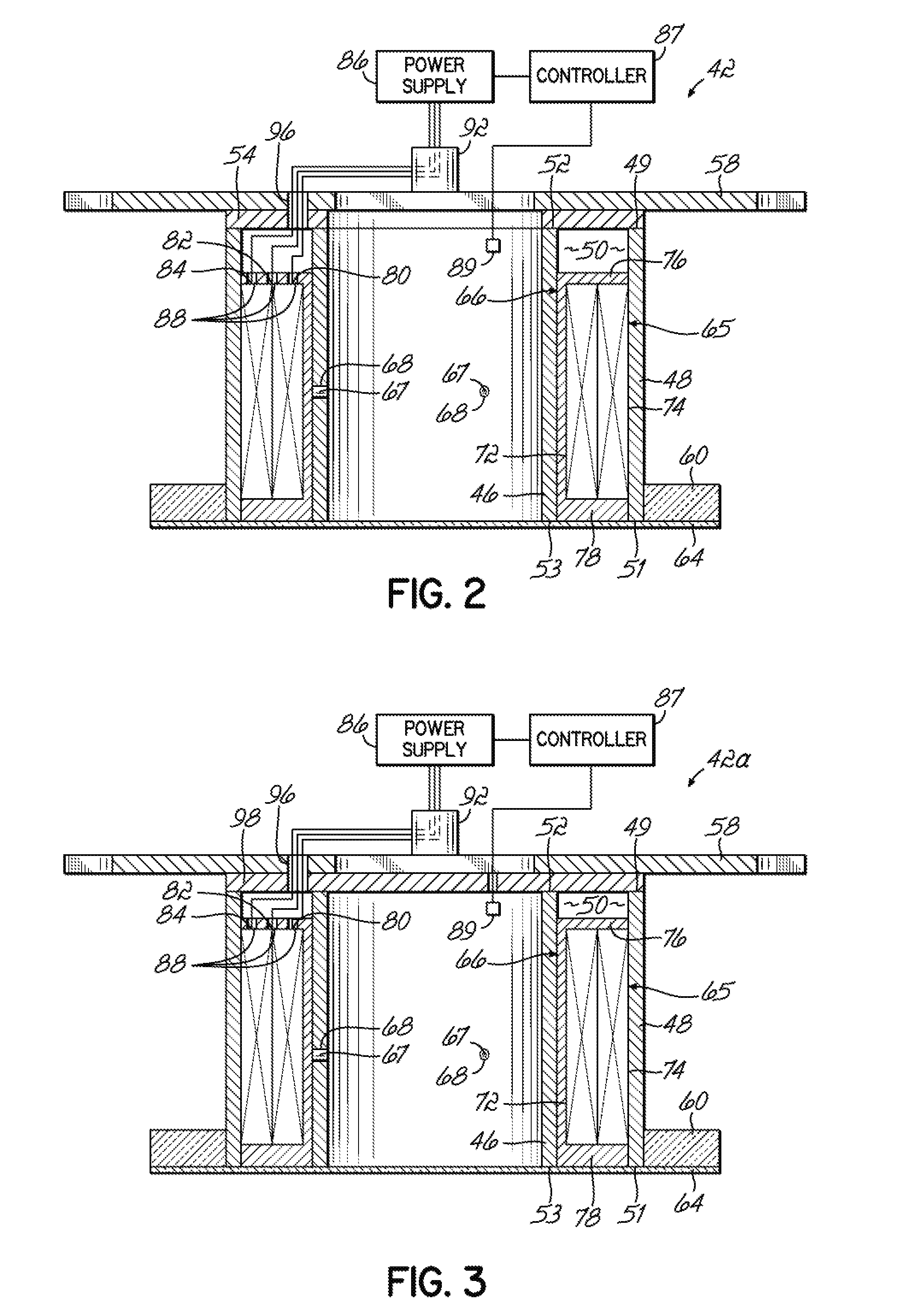
- Ionizing radiation equipment and apparatus design: Specialized equipment and apparatus are designed for delivering ionizing radiation in controlled industrial processes. These systems include radiation sources, shielding mechanisms, conveyor systems, and dosimetry monitoring devices. The equipment is engineered to ensure uniform radiation distribution, operator safety, and process efficiency. Advanced designs incorporate automated controls, real-time monitoring systems, and safety interlocks to optimize the radiation application process while minimizing exposure risks.
- Radiation dose control and measurement systems: Precise control and measurement of radiation doses are critical for effective ionizing ray applications. Advanced dosimetry systems and monitoring technologies are employed to ensure accurate delivery of prescribed radiation levels. These systems utilize various detection methods and calibration techniques to verify dose uniformity and consistency throughout the treatment process. Real-time monitoring and feedback mechanisms enable process optimization and quality assurance in radiation-based applications.
- Industrial-scale ionizing radiation processing systems: Large-scale industrial systems are developed for high-throughput ionizing radiation processing applications. These systems are designed to handle continuous or batch processing of products with consistent quality and efficiency. The infrastructure includes material handling systems, radiation chambers, safety systems, and process control automation. Such installations are optimized for specific applications including food irradiation, polymer processing, and large-volume sterilization operations, ensuring economic viability and regulatory compliance.
02 Ionizing radiation for material modification and surface treatment
Ionizing radiation can be used to modify the properties of materials and treat surfaces. The process involves exposing materials to controlled doses of radiation to induce chemical and physical changes, such as cross-linking polymers, improving adhesion properties, or altering surface characteristics. This technique is applied in various industries including packaging, electronics, and automotive manufacturing to enhance material performance and durability.Expand Specific Solutions03 Radiation detection and monitoring systems
Advanced systems and devices are designed to detect, measure, and monitor ionizing radiation during application processes. These systems ensure proper dosage delivery, safety compliance, and quality control. They incorporate sensors, detectors, and real-time monitoring capabilities to track radiation levels and distribution patterns. Such monitoring systems are essential for maintaining process consistency and protecting operators and products from excessive radiation exposure.Expand Specific Solutions04 Ionizing radiation equipment and apparatus design
Specialized equipment and apparatus are developed for efficient delivery and application of ionizing radiation. These designs include radiation sources, shielding systems, conveyor mechanisms, and exposure chambers optimized for different materials and throughput requirements. The equipment incorporates safety features, automated controls, and precise positioning systems to ensure uniform radiation distribution and process repeatability across various industrial applications.Expand Specific Solutions05 Ionizing radiation processing for food and agricultural products
Ionizing radiation is applied to food and agricultural products for preservation, pest control, and shelf-life extension. The process reduces microbial contamination, inhibits sprouting, and delays ripening without significantly affecting nutritional value or sensory properties. Specific radiation doses are calibrated based on product type and desired outcomes. This technology provides an alternative to chemical treatments and thermal processing methods for ensuring food safety and quality.Expand Specific Solutions
Key Players in Ionizing Ray Industry
The ionizing ray application process market represents a mature technology sector experiencing steady growth, driven by expanding semiconductor manufacturing and advanced materials processing demands. The industry demonstrates a well-established competitive landscape with market leaders including Axcelis Technologies, Applied Materials, and Varian Semiconductor Equipment Associates dominating ion implantation systems. Technology maturity varies across segments, with companies like Sumitomo Heavy Industries Ion Technology and Nissin Ion Equipment advancing high-energy implanter capabilities, while Veeco Instruments and FEI Co. push precision deposition technologies. Research institutions such as Industrial Technology Research Institute and University of Science & Technology of China contribute to next-generation innovations. The market shows consolidation trends with established players leveraging decades of expertise, while emerging applications in quantum computing through companies like Quantinuum LLC indicate evolving technological frontiers beyond traditional semiconductor processing applications.
Axcelis Technologies, Inc.
Technical Solution: Axcelis specializes in high-current ion implantation systems with streamlined workflow management. Their Purion platform incorporates automated beam tuning, real-time process monitoring, and integrated metrology capabilities that reduce cycle times by 30%. The system features advanced source life management, predictive analytics for equipment health monitoring, and automated recipe optimization based on historical performance data. Smart scheduling algorithms optimize tool utilization while maintaining process quality standards. The platform includes comprehensive data logging and analysis tools for continuous process improvement and regulatory compliance in semiconductor device manufacturing.
Strengths: Excellent beam current stability and advanced process analytics capabilities. Weaknesses: Limited flexibility for non-standard applications and higher operating costs.
Sumitomo Heavy Industries Ion Technology Co., Ltd.
Technical Solution: Sumitomo Heavy Industries focuses on high-energy ion implantation systems with advanced process automation and control capabilities. Their systems integrate intelligent beam management, automated calibration procedures, and comprehensive process monitoring that enhances operational efficiency by 35%. The platform features advanced ion source technology, precise dose control mechanisms, and automated substrate handling systems. Real-time process optimization algorithms adjust parameters dynamically based on feedback from integrated sensors. The system includes comprehensive safety management, automated fault detection, and recovery procedures that minimize process interruptions and ensure consistent product quality in semiconductor manufacturing applications.
Strengths: Robust high-energy beam capabilities and excellent process repeatability. Weaknesses: Higher complexity requiring specialized operator training and significant floor space requirements.
Core Innovations in Ray Process Automation
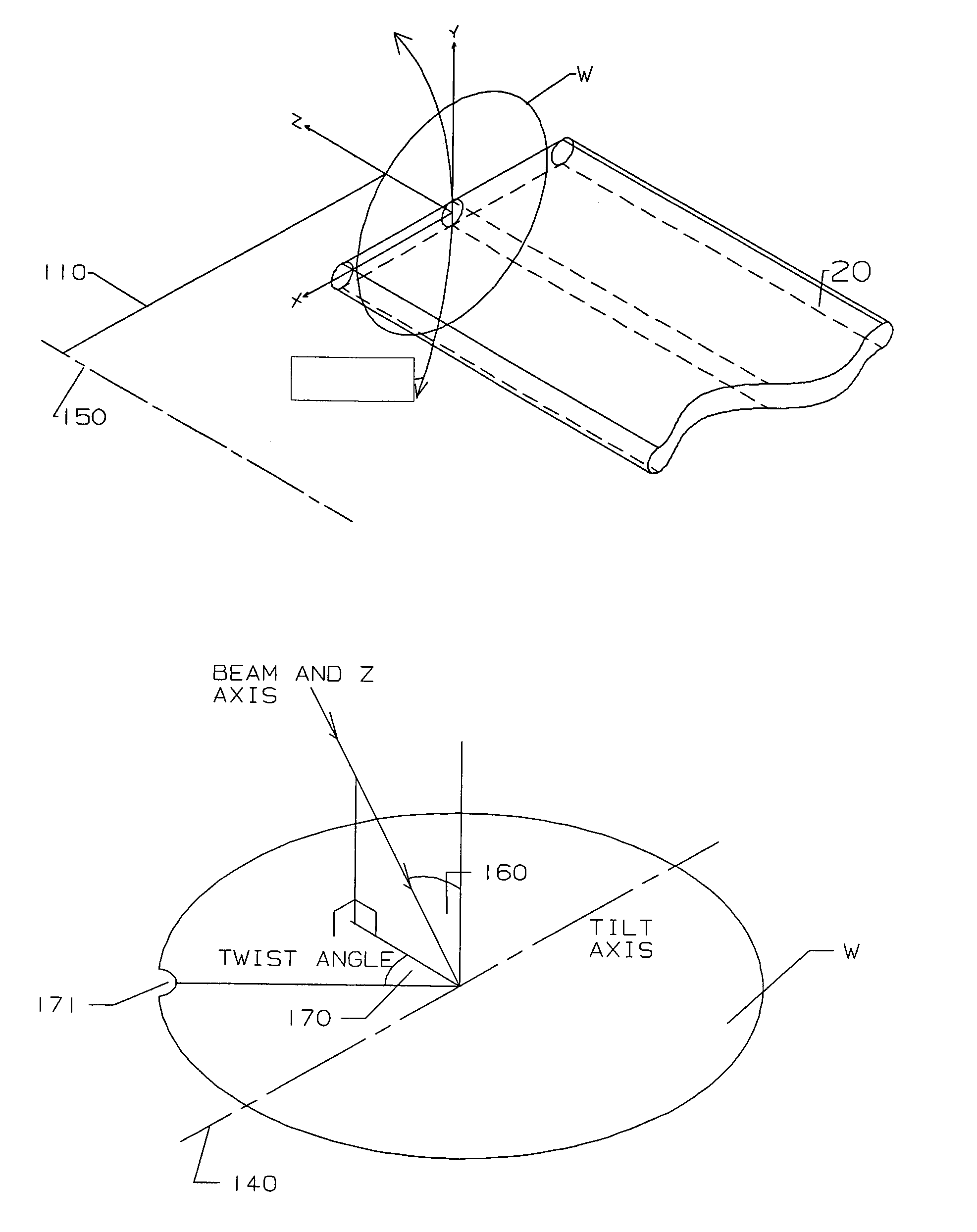
Radial scan arm and collimator for serial processing of semiconductor wafers with ribbon beams
PatentInactiveUS7057192B2
Innovation
- A radial scan arm system that uses a pivot-mounted chuck to control the angle of the wafer relative to the ion beam, combined with electrostatic or magnetic lenses to maintain parallel ion trajectories, and a mechanism to adjust current density proportionally to distance from the pivot axis, allowing for uniform ion implantation across an arc-shaped path.
Ion sources and methods for generating an ion beam with a controllable ion current density distribution
PatentInactiveUS20070194245A1
Innovation
- An ion source with a discharge chamber and an electromagnet assembly that generates a magnetic field to tailor the plasma density distribution, allowing for optimized ion flux and current density distribution by adjusting the magnetic field strength and configuration.
Radiation Safety Regulatory Framework
The radiation safety regulatory framework represents a complex multi-layered system designed to govern ionizing radiation applications across various industries. This framework encompasses international standards, national regulations, and local implementation guidelines that collectively ensure safe utilization of ionizing radiation technologies while facilitating legitimate commercial and research applications.
At the international level, the International Atomic Energy Agency (IAEA) establishes fundamental safety standards that serve as the foundation for national regulatory systems. These standards provide comprehensive guidance on radiation protection principles, dose limits, and safety management systems. The International Commission on Radiological Protection (ICRP) contributes essential recommendations on radiological protection philosophy and dose limitation frameworks that influence regulatory development worldwide.
National regulatory bodies, such as the Nuclear Regulatory Commission in the United States, the Office for Nuclear Regulation in the United Kingdom, and equivalent agencies in other countries, translate international standards into enforceable domestic regulations. These agencies maintain licensing systems, conduct inspections, and enforce compliance measures specific to their jurisdictions. The regulatory scope typically covers medical applications, industrial radiography, nuclear gauging, food irradiation, and research activities.
The licensing process constitutes a critical component of the regulatory framework, requiring applicants to demonstrate technical competence, adequate safety measures, and proper waste management protocols. License conditions often specify equipment specifications, personnel qualifications, operational procedures, and reporting requirements. Regular renewals and amendments ensure continued compliance with evolving safety standards.
Regulatory harmonization efforts aim to reduce inconsistencies between different jurisdictions while maintaining safety standards. International cooperation initiatives facilitate mutual recognition of certifications, standardized training programs, and coordinated emergency response protocols. These harmonization efforts are particularly important for multinational organizations and cross-border transportation of radioactive materials.
Emerging technologies and applications continuously challenge existing regulatory frameworks, necessitating adaptive approaches that balance innovation with safety requirements. Regulatory agencies increasingly adopt risk-informed decision-making processes and performance-based standards to accommodate technological advances while maintaining protective measures for workers, the public, and the environment.
At the international level, the International Atomic Energy Agency (IAEA) establishes fundamental safety standards that serve as the foundation for national regulatory systems. These standards provide comprehensive guidance on radiation protection principles, dose limits, and safety management systems. The International Commission on Radiological Protection (ICRP) contributes essential recommendations on radiological protection philosophy and dose limitation frameworks that influence regulatory development worldwide.
National regulatory bodies, such as the Nuclear Regulatory Commission in the United States, the Office for Nuclear Regulation in the United Kingdom, and equivalent agencies in other countries, translate international standards into enforceable domestic regulations. These agencies maintain licensing systems, conduct inspections, and enforce compliance measures specific to their jurisdictions. The regulatory scope typically covers medical applications, industrial radiography, nuclear gauging, food irradiation, and research activities.
The licensing process constitutes a critical component of the regulatory framework, requiring applicants to demonstrate technical competence, adequate safety measures, and proper waste management protocols. License conditions often specify equipment specifications, personnel qualifications, operational procedures, and reporting requirements. Regular renewals and amendments ensure continued compliance with evolving safety standards.
Regulatory harmonization efforts aim to reduce inconsistencies between different jurisdictions while maintaining safety standards. International cooperation initiatives facilitate mutual recognition of certifications, standardized training programs, and coordinated emergency response protocols. These harmonization efforts are particularly important for multinational organizations and cross-border transportation of radioactive materials.
Emerging technologies and applications continuously challenge existing regulatory frameworks, necessitating adaptive approaches that balance innovation with safety requirements. Regulatory agencies increasingly adopt risk-informed decision-making processes and performance-based standards to accommodate technological advances while maintaining protective measures for workers, the public, and the environment.
Quality Assurance in Ray Applications
Quality assurance in ionizing ray applications represents a critical framework that ensures consistent performance, safety compliance, and optimal outcomes across diverse industrial and medical implementations. The establishment of robust QA protocols has become increasingly vital as ionizing radiation technologies expand into new sectors, demanding standardized approaches that can adapt to varying operational requirements while maintaining stringent safety and efficacy standards.
The foundation of effective quality assurance lies in comprehensive calibration protocols that verify equipment performance against established benchmarks. Modern QA systems incorporate automated monitoring capabilities that continuously track radiation output, beam uniformity, and dose delivery accuracy. These systems utilize advanced dosimetry techniques, including real-time feedback mechanisms that can detect deviations from predetermined parameters and trigger immediate corrective actions.
Regulatory compliance forms another cornerstone of quality assurance, with international standards such as ISO 11137 for sterilization and IEC 60601 for medical equipment providing structured frameworks for validation and ongoing monitoring. These standards mandate regular performance verification, documentation protocols, and traceability requirements that ensure consistent application quality across different facilities and operators.
Personnel training and certification programs constitute essential elements of comprehensive QA systems. Qualified radiation safety officers and trained technicians must demonstrate competency in equipment operation, safety procedures, and emergency response protocols. Regular recertification ensures that staff maintain current knowledge of evolving best practices and regulatory requirements.
Documentation and record-keeping systems enable continuous improvement through trend analysis and performance tracking. Modern QA programs implement digital platforms that integrate equipment logs, maintenance records, and performance metrics, facilitating data-driven decision-making and predictive maintenance strategies.
Risk assessment methodologies identify potential failure modes and establish preventive measures that minimize the likelihood of quality deviations. These assessments consider equipment reliability, environmental factors, and human error potential, resulting in comprehensive mitigation strategies that enhance overall system reliability and application consistency.
The foundation of effective quality assurance lies in comprehensive calibration protocols that verify equipment performance against established benchmarks. Modern QA systems incorporate automated monitoring capabilities that continuously track radiation output, beam uniformity, and dose delivery accuracy. These systems utilize advanced dosimetry techniques, including real-time feedback mechanisms that can detect deviations from predetermined parameters and trigger immediate corrective actions.
Regulatory compliance forms another cornerstone of quality assurance, with international standards such as ISO 11137 for sterilization and IEC 60601 for medical equipment providing structured frameworks for validation and ongoing monitoring. These standards mandate regular performance verification, documentation protocols, and traceability requirements that ensure consistent application quality across different facilities and operators.
Personnel training and certification programs constitute essential elements of comprehensive QA systems. Qualified radiation safety officers and trained technicians must demonstrate competency in equipment operation, safety procedures, and emergency response protocols. Regular recertification ensures that staff maintain current knowledge of evolving best practices and regulatory requirements.
Documentation and record-keeping systems enable continuous improvement through trend analysis and performance tracking. Modern QA programs implement digital platforms that integrate equipment logs, maintenance records, and performance metrics, facilitating data-driven decision-making and predictive maintenance strategies.
Risk assessment methodologies identify potential failure modes and establish preventive measures that minimize the likelihood of quality deviations. These assessments consider equipment reliability, environmental factors, and human error potential, resulting in comprehensive mitigation strategies that enhance overall system reliability and application consistency.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!