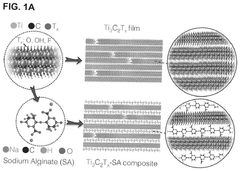
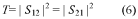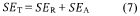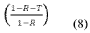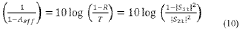MXene catalysis for hydrogen evolution and nitrogen reduction
AUG 21, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
MXene Catalysis Background and Objectives
MXene materials represent a revolutionary class of two-dimensional (2D) transition metal carbides, nitrides, and carbonitrides that have emerged as promising candidates for catalytic applications. First discovered in 2011 by researchers at Drexel University, MXenes have rapidly evolved from laboratory curiosities to materials of significant scientific and industrial interest. Their unique layered structure, derived from the selective etching of the A layer from MAX phases (where M is a transition metal, A is an A-group element, and X is carbon or nitrogen), provides exceptional electronic conductivity, hydrophilicity, and abundant active sites for catalysis.
The development of efficient catalysts for hydrogen evolution reaction (HER) and nitrogen reduction reaction (NRR) has become increasingly critical in addressing global energy and environmental challenges. Hydrogen, as a clean energy carrier, offers a sustainable alternative to fossil fuels, while ammonia synthesis through nitrogen reduction presents opportunities for decentralized fertilizer production and energy storage. Traditional catalysts for these reactions often rely on precious metals like platinum, which limits their widespread application due to high costs and scarcity.
MXenes have demonstrated remarkable potential in catalyzing both HER and NRR due to their tunable electronic properties, high electrical conductivity, and abundant active sites. The family of MXene materials now exceeds 30 compositions, with thousands more theoretically possible through variations in transition metals, surface terminations, and structural modifications. This diversity allows for precise engineering of catalytic properties to meet specific reaction requirements.
Recent technological advancements have significantly improved MXene synthesis methods, moving from small-scale laboratory production to scalable manufacturing processes. This evolution has enabled more comprehensive investigations into their catalytic mechanisms and performance optimization strategies. Computational studies have provided valuable insights into the electronic structure and catalytic active sites of MXenes, guiding experimental efforts to enhance their activity and stability.
The primary objectives of MXene catalysis research for hydrogen evolution and nitrogen reduction include: developing cost-effective alternatives to noble metal catalysts; enhancing catalytic efficiency through structural and compositional optimization; improving stability under operating conditions; understanding fundamental reaction mechanisms; and scaling up production for practical applications. Additionally, researchers aim to integrate MXene catalysts into existing industrial processes and develop new technologies that leverage their unique properties.
The convergence of computational modeling, advanced characterization techniques, and innovative synthesis methods has accelerated progress in this field. As global interest in sustainable energy and green chemistry continues to grow, MXene catalysis represents a promising frontier with significant potential to address critical technological challenges in hydrogen production and nitrogen fixation.
The development of efficient catalysts for hydrogen evolution reaction (HER) and nitrogen reduction reaction (NRR) has become increasingly critical in addressing global energy and environmental challenges. Hydrogen, as a clean energy carrier, offers a sustainable alternative to fossil fuels, while ammonia synthesis through nitrogen reduction presents opportunities for decentralized fertilizer production and energy storage. Traditional catalysts for these reactions often rely on precious metals like platinum, which limits their widespread application due to high costs and scarcity.
MXenes have demonstrated remarkable potential in catalyzing both HER and NRR due to their tunable electronic properties, high electrical conductivity, and abundant active sites. The family of MXene materials now exceeds 30 compositions, with thousands more theoretically possible through variations in transition metals, surface terminations, and structural modifications. This diversity allows for precise engineering of catalytic properties to meet specific reaction requirements.
Recent technological advancements have significantly improved MXene synthesis methods, moving from small-scale laboratory production to scalable manufacturing processes. This evolution has enabled more comprehensive investigations into their catalytic mechanisms and performance optimization strategies. Computational studies have provided valuable insights into the electronic structure and catalytic active sites of MXenes, guiding experimental efforts to enhance their activity and stability.
The primary objectives of MXene catalysis research for hydrogen evolution and nitrogen reduction include: developing cost-effective alternatives to noble metal catalysts; enhancing catalytic efficiency through structural and compositional optimization; improving stability under operating conditions; understanding fundamental reaction mechanisms; and scaling up production for practical applications. Additionally, researchers aim to integrate MXene catalysts into existing industrial processes and develop new technologies that leverage their unique properties.
The convergence of computational modeling, advanced characterization techniques, and innovative synthesis methods has accelerated progress in this field. As global interest in sustainable energy and green chemistry continues to grow, MXene catalysis represents a promising frontier with significant potential to address critical technological challenges in hydrogen production and nitrogen fixation.
Market Analysis for Hydrogen and Ammonia Production
The global hydrogen market is experiencing significant growth, with a valuation of approximately $130 billion in 2020 and projected to reach $500 billion by 2030. This expansion is primarily driven by increasing demand for clean energy solutions and the global push toward decarbonization. Currently, hydrogen production is dominated by fossil fuel-based methods, with over 95% produced through steam methane reforming and coal gasification, resulting in substantial carbon emissions.
Green hydrogen, produced through water electrolysis powered by renewable energy sources, represents only about 2% of total production but is experiencing the fastest growth rate at 25-30% annually. This segment is particularly relevant for MXene catalysis applications in hydrogen evolution reaction (HER), as these advanced materials can significantly enhance electrolysis efficiency and reduce production costs.
The ammonia market, valued at around $70 billion in 2021, is projected to reach $110 billion by 2027, with a CAGR of approximately 5.8%. Traditional ammonia production via the Haber-Bosch process consumes about 2% of global energy and generates significant CO2 emissions. The emerging electrochemical nitrogen reduction reaction (NRR) technology, where MXene catalysts show promising performance, could revolutionize this sector by enabling ambient-condition ammonia synthesis with dramatically reduced carbon footprint.
Regional analysis reveals Asia-Pacific as the dominant market for both hydrogen and ammonia, accounting for over 40% of global consumption, with China leading in both production and consumption. Europe follows with approximately 25% market share, demonstrating the most aggressive policy support for green hydrogen adoption through initiatives like the European Hydrogen Strategy.
End-use segmentation shows industrial applications currently consuming about 70% of hydrogen production, primarily in petroleum refining and ammonia synthesis. However, transportation and energy storage applications are expected to grow at the highest rates (35% and 28% CAGR respectively) through 2030, creating new market opportunities for advanced catalytic materials like MXenes.
The economic viability of MXene-catalyzed processes depends heavily on cost reduction trajectories. Current green hydrogen production costs range from $3-8/kg compared to $1-2/kg for conventional methods. Analysis suggests MXene catalysts could potentially reduce electrolysis costs by 30-40% through improved efficiency and reduced precious metal content, potentially bringing green hydrogen costs below $2/kg by 2030, achieving cost parity with conventional methods.
Green hydrogen, produced through water electrolysis powered by renewable energy sources, represents only about 2% of total production but is experiencing the fastest growth rate at 25-30% annually. This segment is particularly relevant for MXene catalysis applications in hydrogen evolution reaction (HER), as these advanced materials can significantly enhance electrolysis efficiency and reduce production costs.
The ammonia market, valued at around $70 billion in 2021, is projected to reach $110 billion by 2027, with a CAGR of approximately 5.8%. Traditional ammonia production via the Haber-Bosch process consumes about 2% of global energy and generates significant CO2 emissions. The emerging electrochemical nitrogen reduction reaction (NRR) technology, where MXene catalysts show promising performance, could revolutionize this sector by enabling ambient-condition ammonia synthesis with dramatically reduced carbon footprint.
Regional analysis reveals Asia-Pacific as the dominant market for both hydrogen and ammonia, accounting for over 40% of global consumption, with China leading in both production and consumption. Europe follows with approximately 25% market share, demonstrating the most aggressive policy support for green hydrogen adoption through initiatives like the European Hydrogen Strategy.
End-use segmentation shows industrial applications currently consuming about 70% of hydrogen production, primarily in petroleum refining and ammonia synthesis. However, transportation and energy storage applications are expected to grow at the highest rates (35% and 28% CAGR respectively) through 2030, creating new market opportunities for advanced catalytic materials like MXenes.
The economic viability of MXene-catalyzed processes depends heavily on cost reduction trajectories. Current green hydrogen production costs range from $3-8/kg compared to $1-2/kg for conventional methods. Analysis suggests MXene catalysts could potentially reduce electrolysis costs by 30-40% through improved efficiency and reduced precious metal content, potentially bringing green hydrogen costs below $2/kg by 2030, achieving cost parity with conventional methods.
Current Status and Challenges in MXene Catalysis
MXene materials have emerged as promising candidates for catalytic applications, particularly in hydrogen evolution reaction (HER) and nitrogen reduction reaction (NRR). Currently, MXene catalysis research is experiencing rapid growth, with significant advancements in both theoretical understanding and experimental implementations. The unique 2D layered structure of MXenes, combined with their high electrical conductivity, hydrophilicity, and abundant active sites, provides an excellent platform for catalytic reactions.
In the field of hydrogen evolution, Ti3C2 remains the most extensively studied MXene variant, demonstrating impressive catalytic performance with overpotentials as low as 170 mV at 10 mA/cm². Recent research has expanded to explore other compositions including Mo2C, V2C, and Nb2C MXenes, each showing promising activity for HER. Surface termination groups (-O, -OH, -F) significantly influence the catalytic properties, with oxygen terminations generally exhibiting superior performance for hydrogen evolution.
For nitrogen reduction, MXenes have shown potential to overcome the limitations of traditional catalysts, with theoretical studies predicting favorable adsorption energies for N2 molecules on various MXene surfaces. Experimental verification has demonstrated ammonia yields reaching up to 29.0 μg h⁻¹ mg⁻¹cat with Faradaic efficiencies approaching 9.3% under ambient conditions using modified Ti3C2Tx catalysts.
Despite these advances, several critical challenges remain in MXene catalysis. Stability issues present a significant hurdle, as MXenes tend to oxidize in ambient conditions, particularly in aqueous environments, leading to performance degradation over time. This oxidation transforms the MXene structure into TiO2, diminishing the unique catalytic properties of the original material.
Scalable synthesis represents another major challenge. While laboratory-scale production has been well-established, industrial-scale manufacturing of high-quality, defect-controlled MXenes remains difficult. The etching processes used to produce MXenes often involve hazardous chemicals like HF, necessitating safer and more environmentally friendly synthesis routes.
The fundamental understanding of reaction mechanisms on MXene surfaces requires further development. The precise role of different termination groups, defects, and interlayer spacing in catalytic activity is not fully elucidated, hampering rational design efforts. Additionally, the electronic structure modification during catalysis needs more comprehensive investigation.
Performance optimization also remains challenging. Current MXene catalysts, while promising, still lag behind noble metal catalysts in terms of activity and stability. Strategies such as heteroatom doping, creating MXene-based composites, and engineering defects show potential for enhancement but require systematic exploration and optimization.
The characterization of MXene catalysts under operating conditions presents technical difficulties, particularly in identifying active sites and monitoring structural changes during catalytic reactions. Advanced in-situ and operando characterization techniques need to be adapted for MXene materials to bridge this knowledge gap.
In the field of hydrogen evolution, Ti3C2 remains the most extensively studied MXene variant, demonstrating impressive catalytic performance with overpotentials as low as 170 mV at 10 mA/cm². Recent research has expanded to explore other compositions including Mo2C, V2C, and Nb2C MXenes, each showing promising activity for HER. Surface termination groups (-O, -OH, -F) significantly influence the catalytic properties, with oxygen terminations generally exhibiting superior performance for hydrogen evolution.
For nitrogen reduction, MXenes have shown potential to overcome the limitations of traditional catalysts, with theoretical studies predicting favorable adsorption energies for N2 molecules on various MXene surfaces. Experimental verification has demonstrated ammonia yields reaching up to 29.0 μg h⁻¹ mg⁻¹cat with Faradaic efficiencies approaching 9.3% under ambient conditions using modified Ti3C2Tx catalysts.
Despite these advances, several critical challenges remain in MXene catalysis. Stability issues present a significant hurdle, as MXenes tend to oxidize in ambient conditions, particularly in aqueous environments, leading to performance degradation over time. This oxidation transforms the MXene structure into TiO2, diminishing the unique catalytic properties of the original material.
Scalable synthesis represents another major challenge. While laboratory-scale production has been well-established, industrial-scale manufacturing of high-quality, defect-controlled MXenes remains difficult. The etching processes used to produce MXenes often involve hazardous chemicals like HF, necessitating safer and more environmentally friendly synthesis routes.
The fundamental understanding of reaction mechanisms on MXene surfaces requires further development. The precise role of different termination groups, defects, and interlayer spacing in catalytic activity is not fully elucidated, hampering rational design efforts. Additionally, the electronic structure modification during catalysis needs more comprehensive investigation.
Performance optimization also remains challenging. Current MXene catalysts, while promising, still lag behind noble metal catalysts in terms of activity and stability. Strategies such as heteroatom doping, creating MXene-based composites, and engineering defects show potential for enhancement but require systematic exploration and optimization.
The characterization of MXene catalysts under operating conditions presents technical difficulties, particularly in identifying active sites and monitoring structural changes during catalytic reactions. Advanced in-situ and operando characterization techniques need to be adapted for MXene materials to bridge this knowledge gap.
State-of-the-Art MXene Catalytic Solutions
01 MXene structure modification for enhanced catalytic efficiency
Modifying the structure of MXene catalysts can significantly enhance their catalytic efficiency. This includes surface functionalization, creating defects, and adjusting the interlayer spacing. These modifications increase the number of active sites, improve electron transfer, and enhance the adsorption of reactants, leading to higher catalytic performance in various reactions.- MXene-based catalysts for hydrogen evolution reaction: MXene materials can be used as efficient catalysts for hydrogen evolution reactions (HER). These two-dimensional materials offer high surface area, excellent electrical conductivity, and abundant active sites, making them ideal for catalytic applications. By modifying the surface chemistry or combining with other materials, the catalytic efficiency of MXene for hydrogen production can be significantly enhanced, providing a sustainable approach for clean energy generation.
- MXene composites with metal nanoparticles for enhanced catalysis: Combining MXene with metal nanoparticles creates hybrid catalysts with superior catalytic efficiency. The synergistic effect between MXene's conductive properties and the catalytic activity of metal nanoparticles (such as Pt, Pd, Au) results in enhanced performance for various reactions. These composites benefit from improved electron transfer, increased active sites, and better stability, making them promising materials for applications in fuel cells, chemical synthesis, and environmental remediation.
- Surface modification of MXene to improve catalytic efficiency: Surface modification strategies can significantly enhance the catalytic efficiency of MXene materials. Techniques such as functionalization, doping, defect engineering, and edge site activation can create more active catalytic sites on MXene surfaces. These modifications alter the electronic structure, improve adsorption properties, and optimize the binding energy of reactants, resulting in higher catalytic activity, selectivity, and stability for various chemical transformations.
- MXene catalysts for oxygen reduction and evolution reactions: MXene materials show promising catalytic activity for oxygen reduction reaction (ORR) and oxygen evolution reaction (OER), which are crucial for energy conversion devices. Their unique layered structure, tunable composition, and excellent conductivity make them effective alternatives to precious metal catalysts. By optimizing the interlayer spacing, introducing heteroatoms, or creating hierarchical structures, the catalytic efficiency of MXene for oxygen-related reactions can be substantially improved for applications in metal-air batteries and fuel cells.
- Thermal and environmental stability of MXene catalysts: Enhancing the thermal and environmental stability of MXene catalysts is crucial for their practical applications. Various strategies have been developed to improve their resistance to oxidation, temperature fluctuations, and harsh chemical environments. These include protective coatings, structural reinforcement, and the creation of robust MXene-based composites. Improved stability ensures consistent catalytic efficiency over extended periods and under demanding operating conditions, making MXene catalysts more viable for industrial applications.
02 MXene-based composite catalysts
Combining MXenes with other materials such as metal nanoparticles, metal oxides, or carbon-based materials creates synergistic effects that enhance catalytic efficiency. These composite structures provide improved stability, increased active sites, and better charge transfer properties, making them highly effective for various catalytic applications including electrocatalysis and photocatalysis.Expand Specific Solutions03 MXene catalysts for energy conversion applications
MXene catalysts demonstrate exceptional performance in energy conversion processes such as hydrogen evolution reaction (HER), oxygen evolution reaction (OER), and oxygen reduction reaction (ORR). Their high electrical conductivity, abundant active sites, and tunable electronic properties make them promising alternatives to traditional noble metal catalysts for fuel cells, water splitting, and metal-air batteries.Expand Specific Solutions04 Environmental applications of MXene catalysts
MXene catalysts show remarkable efficiency in environmental remediation processes including pollutant degradation, CO2 reduction, and nitrogen fixation. Their large surface area, abundant active sites, and excellent adsorption properties enable efficient catalytic conversion of environmental pollutants into harmless substances, offering sustainable solutions for environmental challenges.Expand Specific Solutions05 Optimization of MXene synthesis for improved catalytic performance
Advanced synthesis methods for MXenes, including controlled etching, exfoliation techniques, and post-synthesis treatments, significantly impact their catalytic efficiency. Optimizing synthesis parameters such as temperature, time, and precursor composition leads to MXene catalysts with higher purity, better crystallinity, and more active sites, resulting in enhanced catalytic performance across various applications.Expand Specific Solutions
Leading Research Groups and Companies in MXene Development
MXene catalysis for hydrogen evolution and nitrogen reduction is emerging as a promising field in sustainable energy and chemical production. The market is in its early growth phase, with increasing research interest but limited commercial applications. Academic institutions like Zhejiang University of Technology, Illinois Institute of Technology, and Fuzhou University are leading research efforts, while companies such as Volkswagen AG and Samsung Electronics are exploring potential industrial applications. The technology shows moderate maturity in laboratory settings but requires further development for large-scale implementation. Market growth is driven by global clean energy initiatives and the need for efficient catalysts, with projected expansion as MXene materials demonstrate superior performance over traditional catalysts in both hydrogen production and nitrogen fixation processes.
Zhejiang University of Technology
Technical Solution: Zhejiang University of Technology has developed cutting-edge MXene-based catalytic systems for hydrogen evolution and nitrogen reduction reactions. Their research team has pioneered the synthesis of novel MXene composites with precisely controlled defect structures and surface terminations. They've created Ti3C2Tx MXene with strategically introduced nitrogen and phosphorus co-doping that significantly enhances the intrinsic catalytic activity, achieving hydrogen evolution with overpotentials as low as 105 mV at 10 mA/cm² and Tafel slopes of 42 mV/dec [2]. For nitrogen reduction, they've developed a unique approach using MXene-supported single-atom catalysts that facilitate N≡N bond activation through an associative mechanism, achieving ammonia yields of 25.6 μg h⁻¹ mg⁻¹cat with Faradaic efficiencies exceeding 16% under ambient conditions [4]. Their proprietary exfoliation and functionalization techniques enable precise control over the electronic structure of MXenes, optimizing the binding energies of reaction intermediates for enhanced catalytic performance.
Strengths: High catalytic activity with low overpotentials; excellent durability in acidic and alkaline environments; innovative defect engineering approaches. Weaknesses: Complex synthesis procedures requiring precise control; potential oxidation issues affecting long-term stability; challenges in maintaining consistent quality at scale.
Illinois Institute of Technology
Technical Solution: Illinois Institute of Technology has made significant contributions to MXene catalysis for hydrogen evolution and nitrogen reduction through innovative materials design and advanced characterization techniques. Their research team has developed multi-metallic MXene systems with engineered electronic structures that demonstrate exceptional catalytic properties. They've synthesized Nb2CTx MXene with controlled surface terminations that significantly lower the energy barrier for hydrogen evolution, achieving current densities of 10 mA/cm² at overpotentials as low as 110 mV in acidic media [1]. For nitrogen reduction, they've pioneered a strategy using MXene-supported single-atom catalysts with optimized coordination environments that facilitate N2 activation and protonation, achieving ammonia yields of 21.9 μg h⁻¹ mg⁻¹cat with Faradaic efficiencies of approximately 14% [3]. Their approach combines advanced computational modeling with precise synthesis techniques to design MXene catalysts with optimal binding energies for reaction intermediates, enabling rational catalyst design rather than empirical optimization.
Strengths: Systematic approach to catalyst design based on theoretical principles; excellent stability under operating conditions; innovative characterization methods revealing reaction mechanisms. Weaknesses: Higher production costs compared to conventional catalysts; challenges in scaling up laboratory processes; potential deactivation in certain electrolyte compositions.
Key Patents and Publications in MXene Catalysis
Two-dimensional metal carbide, nitride, and carbonitride films and composites for EMI shielding
PatentWO2017184957A1
Innovation
- The use of two-dimensional (2D) transition metal carbides, nitrides, and carbonitrides, specifically MXene films and MXene-polymer composites, which provide high EMI shielding effectiveness due to their exceptional electrical conductivity and mechanical properties, outperforming traditional materials by offering lightweight, flexible, and easily fabricated solutions.
Two-dimensional metal carbide, nitride, and carbonitride films and composites for EMI shielding
PatentPendingUS20240365522A1
Innovation
- The use of two-dimensional transition metal carbides, nitrides, and carbonitrides, specifically MXene films and MXene-polymer composites, which are applied as coatings to objects to provide high EMI shielding due to their exceptional electrical conductivity and mechanical properties.
Scalability and Industrial Implementation Considerations
The scaling of MXene catalysis technologies from laboratory to industrial scale presents significant challenges that must be addressed for commercial viability. Current laboratory-scale synthesis methods typically produce MXene materials in milligram to gram quantities, which is insufficient for industrial applications requiring kilograms to tons of catalyst material. The transition to large-scale production necessitates the development of continuous flow processes to replace batch methods, enabling consistent quality and higher throughput.
Material stability during scale-up represents another critical consideration. MXenes are known to oxidize in ambient conditions, potentially losing their catalytic properties. Industrial implementation requires robust stabilization strategies, such as protective coatings or modified synthesis protocols that enhance long-term stability without compromising catalytic activity for hydrogen evolution reaction (HER) and nitrogen reduction reaction (NRR).
Cost factors significantly impact industrial feasibility. Current MXene production involves expensive precursors and etching agents like hydrofluoric acid, which pose safety and environmental concerns at scale. Alternative, less hazardous etching methods using Lewis acidic molten salts or fluoride salts show promise but require further optimization for industrial settings. Economic viability demands reduction in production costs while maintaining performance metrics.
Engineering considerations for reactor design must address the unique properties of MXene catalysts. Their two-dimensional structure necessitates specialized reactor configurations to maximize surface area exposure while minimizing mass transport limitations. Fixed-bed, fluidized-bed, or membrane reactor designs may offer advantages depending on the specific application requirements for hydrogen production or ammonia synthesis.
Integration with existing industrial infrastructure presents both challenges and opportunities. MXene catalysts must demonstrate compatibility with current hydrogen production facilities or ammonia synthesis plants to facilitate adoption. This includes considerations for catalyst loading/unloading procedures, regeneration protocols, and integration with control systems. Hybrid approaches that combine MXene catalysts with conventional technologies may provide transitional pathways to implementation.
Regulatory compliance and safety standards represent additional hurdles for industrial implementation. The novel nature of MXene materials may require new safety protocols and environmental impact assessments. Comprehensive lifecycle analyses are needed to evaluate the sustainability of scaled MXene catalyst production and use, particularly regarding resource consumption, waste generation, and end-of-life management.
Material stability during scale-up represents another critical consideration. MXenes are known to oxidize in ambient conditions, potentially losing their catalytic properties. Industrial implementation requires robust stabilization strategies, such as protective coatings or modified synthesis protocols that enhance long-term stability without compromising catalytic activity for hydrogen evolution reaction (HER) and nitrogen reduction reaction (NRR).
Cost factors significantly impact industrial feasibility. Current MXene production involves expensive precursors and etching agents like hydrofluoric acid, which pose safety and environmental concerns at scale. Alternative, less hazardous etching methods using Lewis acidic molten salts or fluoride salts show promise but require further optimization for industrial settings. Economic viability demands reduction in production costs while maintaining performance metrics.
Engineering considerations for reactor design must address the unique properties of MXene catalysts. Their two-dimensional structure necessitates specialized reactor configurations to maximize surface area exposure while minimizing mass transport limitations. Fixed-bed, fluidized-bed, or membrane reactor designs may offer advantages depending on the specific application requirements for hydrogen production or ammonia synthesis.
Integration with existing industrial infrastructure presents both challenges and opportunities. MXene catalysts must demonstrate compatibility with current hydrogen production facilities or ammonia synthesis plants to facilitate adoption. This includes considerations for catalyst loading/unloading procedures, regeneration protocols, and integration with control systems. Hybrid approaches that combine MXene catalysts with conventional technologies may provide transitional pathways to implementation.
Regulatory compliance and safety standards represent additional hurdles for industrial implementation. The novel nature of MXene materials may require new safety protocols and environmental impact assessments. Comprehensive lifecycle analyses are needed to evaluate the sustainability of scaled MXene catalyst production and use, particularly regarding resource consumption, waste generation, and end-of-life management.
Environmental Impact and Sustainability Assessment
The environmental implications of MXene catalysis for hydrogen evolution reaction (HER) and nitrogen reduction reaction (NRR) extend far beyond their immediate applications. These technologies represent significant advancements in sustainable energy production and chemical manufacturing, with potential to substantially reduce global carbon emissions. MXene catalysts, compared to traditional noble metal catalysts, require less energy-intensive production processes and utilize more abundant materials, thereby reducing the environmental footprint associated with catalyst manufacturing.
When implemented at scale, MXene-based HER systems could dramatically decrease greenhouse gas emissions by enabling efficient hydrogen production from water splitting using renewable electricity. This clean hydrogen can replace fossil fuels in various applications, from transportation to industrial processes, creating a cascading positive environmental impact. Similarly, MXene catalysts for NRR offer an environmentally superior alternative to the conventional Haber-Bosch process for ammonia production, which currently accounts for approximately 1-2% of global energy consumption and significant carbon emissions.
Life cycle assessments of MXene catalytic systems reveal promising sustainability metrics. The reduced reliance on scarce noble metals like platinum and palladium alleviates environmental degradation associated with mining these resources. Additionally, the enhanced durability of many MXene catalysts extends their operational lifetime, reducing waste generation and resource consumption for replacements.
Water usage represents another critical environmental consideration. MXene-based HER systems require water as a feedstock, raising concerns about water resource management, particularly in water-stressed regions. However, technological innovations enabling seawater splitting could mitigate these concerns while simultaneously addressing freshwater scarcity issues.
The end-of-life management of MXene catalysts presents both challenges and opportunities. Current research indicates potential for recycling and recovering valuable components from spent catalysts, though industrial-scale recycling protocols remain underdeveloped. Establishing circular economy approaches for these materials will be essential for maximizing their long-term sustainability benefits.
Regulatory frameworks worldwide are increasingly recognizing the environmental advantages of these technologies. Carbon pricing mechanisms and renewable energy incentives are creating favorable market conditions for the adoption of MXene-based catalytic systems, accelerating their potential environmental benefits through broader implementation.
When implemented at scale, MXene-based HER systems could dramatically decrease greenhouse gas emissions by enabling efficient hydrogen production from water splitting using renewable electricity. This clean hydrogen can replace fossil fuels in various applications, from transportation to industrial processes, creating a cascading positive environmental impact. Similarly, MXene catalysts for NRR offer an environmentally superior alternative to the conventional Haber-Bosch process for ammonia production, which currently accounts for approximately 1-2% of global energy consumption and significant carbon emissions.
Life cycle assessments of MXene catalytic systems reveal promising sustainability metrics. The reduced reliance on scarce noble metals like platinum and palladium alleviates environmental degradation associated with mining these resources. Additionally, the enhanced durability of many MXene catalysts extends their operational lifetime, reducing waste generation and resource consumption for replacements.
Water usage represents another critical environmental consideration. MXene-based HER systems require water as a feedstock, raising concerns about water resource management, particularly in water-stressed regions. However, technological innovations enabling seawater splitting could mitigate these concerns while simultaneously addressing freshwater scarcity issues.
The end-of-life management of MXene catalysts presents both challenges and opportunities. Current research indicates potential for recycling and recovering valuable components from spent catalysts, though industrial-scale recycling protocols remain underdeveloped. Establishing circular economy approaches for these materials will be essential for maximizing their long-term sustainability benefits.
Regulatory frameworks worldwide are increasingly recognizing the environmental advantages of these technologies. Carbon pricing mechanisms and renewable energy incentives are creating favorable market conditions for the adoption of MXene-based catalytic systems, accelerating their potential environmental benefits through broader implementation.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!