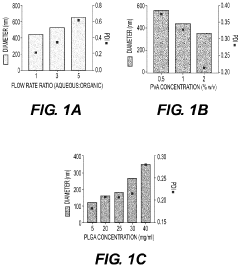
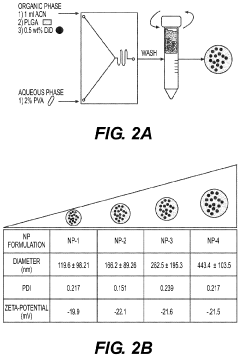
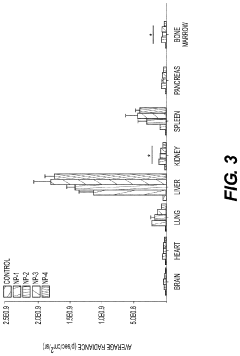
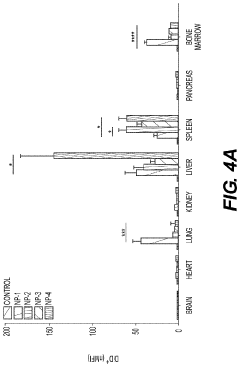
Nanoparticles in Bioengineering: Tissue Interaction Study
FEB 26, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Nanoparticle Bioengineering Background and Objectives
Nanoparticle bioengineering represents a revolutionary convergence of nanotechnology and biological sciences that has emerged as one of the most promising frontiers in modern medicine and biotechnology. This interdisciplinary field harnesses the unique properties of materials at the nanoscale, typically ranging from 1 to 100 nanometers, to create innovative solutions for complex biological challenges. The field has evolved from early theoretical concepts in the 1980s to sophisticated applications in drug delivery, tissue engineering, and regenerative medicine.
The historical development of nanoparticle bioengineering can be traced through several key phases. Initial research focused on understanding fundamental nanoparticle properties and their interactions with biological systems. The 1990s marked a significant advancement with the development of liposomal drug delivery systems, establishing the foundation for targeted therapeutic applications. The early 2000s witnessed exponential growth in synthesis techniques and surface functionalization methods, enabling precise control over nanoparticle behavior in biological environments.
Contemporary nanoparticle bioengineering encompasses diverse material platforms including polymeric nanoparticles, metallic nanoparticles, carbon-based nanomaterials, and hybrid systems. Each platform offers distinct advantages for specific biomedical applications, from imaging and diagnostics to therapeutic interventions and tissue regeneration. The field has progressively shifted toward developing multifunctional nanoparticles capable of simultaneous diagnosis and treatment, known as theranostic systems.
The primary objective of nanoparticle bioengineering in tissue interaction studies is to understand and optimize the complex interface between engineered nanomaterials and biological tissues. This involves investigating cellular uptake mechanisms, biodistribution patterns, and long-term biocompatibility profiles. Researchers aim to develop predictive models for nanoparticle behavior in various tissue environments, enabling rational design of next-generation therapeutic and diagnostic platforms.
Current technological goals focus on achieving precise spatiotemporal control over nanoparticle function within target tissues while minimizing off-target effects. This includes developing stimuli-responsive systems that can be activated by external triggers such as pH changes, temperature variations, or electromagnetic fields. Additionally, the field strives to establish comprehensive safety profiles and standardized evaluation protocols for clinical translation of nanoparticle-based technologies.
The historical development of nanoparticle bioengineering can be traced through several key phases. Initial research focused on understanding fundamental nanoparticle properties and their interactions with biological systems. The 1990s marked a significant advancement with the development of liposomal drug delivery systems, establishing the foundation for targeted therapeutic applications. The early 2000s witnessed exponential growth in synthesis techniques and surface functionalization methods, enabling precise control over nanoparticle behavior in biological environments.
Contemporary nanoparticle bioengineering encompasses diverse material platforms including polymeric nanoparticles, metallic nanoparticles, carbon-based nanomaterials, and hybrid systems. Each platform offers distinct advantages for specific biomedical applications, from imaging and diagnostics to therapeutic interventions and tissue regeneration. The field has progressively shifted toward developing multifunctional nanoparticles capable of simultaneous diagnosis and treatment, known as theranostic systems.
The primary objective of nanoparticle bioengineering in tissue interaction studies is to understand and optimize the complex interface between engineered nanomaterials and biological tissues. This involves investigating cellular uptake mechanisms, biodistribution patterns, and long-term biocompatibility profiles. Researchers aim to develop predictive models for nanoparticle behavior in various tissue environments, enabling rational design of next-generation therapeutic and diagnostic platforms.
Current technological goals focus on achieving precise spatiotemporal control over nanoparticle function within target tissues while minimizing off-target effects. This includes developing stimuli-responsive systems that can be activated by external triggers such as pH changes, temperature variations, or electromagnetic fields. Additionally, the field strives to establish comprehensive safety profiles and standardized evaluation protocols for clinical translation of nanoparticle-based technologies.
Market Demand for Nanoparticle-Based Biomedical Applications
The global biomedical nanoparticle market demonstrates robust growth driven by increasing prevalence of chronic diseases, aging populations, and rising demand for personalized medicine. Cancer treatment applications represent the largest market segment, with nanoparticle-based drug delivery systems offering enhanced therapeutic efficacy and reduced side effects compared to conventional treatments. The growing incidence of cancer worldwide has created substantial demand for innovative therapeutic approaches that can overcome traditional treatment limitations.
Cardiovascular disease management constitutes another significant market driver, as nanoparticles enable targeted delivery of therapeutic agents to specific cardiac tissues while minimizing systemic toxicity. The ability to engineer nanoparticles for precise tissue interaction has opened new possibilities for treating previously challenging cardiovascular conditions, creating substantial market opportunities for bioengineering companies.
Regenerative medicine and tissue engineering applications are experiencing accelerated market growth, particularly in orthopedic and wound healing sectors. Nanoparticles designed for optimal tissue interaction can enhance cellular regeneration, promote tissue integration, and provide controlled release of growth factors. This capability addresses the increasing demand for advanced wound care products and bone repair solutions in aging populations.
Diagnostic applications represent a rapidly expanding market segment, with nanoparticle-based contrast agents and biosensors offering superior imaging capabilities and early disease detection. The growing emphasis on preventive healthcare and early intervention strategies has increased demand for advanced diagnostic tools that can provide accurate, real-time tissue analysis.
Neurological disorder treatment presents an emerging high-value market opportunity, as engineered nanoparticles capable of crossing the blood-brain barrier offer potential solutions for previously untreatable conditions. The rising prevalence of neurodegenerative diseases has created urgent demand for innovative therapeutic delivery systems.
Market demand is further amplified by regulatory agencies' increasing acceptance of nanoparticle-based therapies, with several products receiving approval for clinical use. This regulatory progress has encouraged investment in nanoparticle bioengineering research and accelerated commercial development timelines.
The shift toward precision medicine has created demand for customizable nanoparticle platforms that can be tailored to individual patient needs and specific tissue interaction requirements, driving innovation in bioengineering approaches.
Cardiovascular disease management constitutes another significant market driver, as nanoparticles enable targeted delivery of therapeutic agents to specific cardiac tissues while minimizing systemic toxicity. The ability to engineer nanoparticles for precise tissue interaction has opened new possibilities for treating previously challenging cardiovascular conditions, creating substantial market opportunities for bioengineering companies.
Regenerative medicine and tissue engineering applications are experiencing accelerated market growth, particularly in orthopedic and wound healing sectors. Nanoparticles designed for optimal tissue interaction can enhance cellular regeneration, promote tissue integration, and provide controlled release of growth factors. This capability addresses the increasing demand for advanced wound care products and bone repair solutions in aging populations.
Diagnostic applications represent a rapidly expanding market segment, with nanoparticle-based contrast agents and biosensors offering superior imaging capabilities and early disease detection. The growing emphasis on preventive healthcare and early intervention strategies has increased demand for advanced diagnostic tools that can provide accurate, real-time tissue analysis.
Neurological disorder treatment presents an emerging high-value market opportunity, as engineered nanoparticles capable of crossing the blood-brain barrier offer potential solutions for previously untreatable conditions. The rising prevalence of neurodegenerative diseases has created urgent demand for innovative therapeutic delivery systems.
Market demand is further amplified by regulatory agencies' increasing acceptance of nanoparticle-based therapies, with several products receiving approval for clinical use. This regulatory progress has encouraged investment in nanoparticle bioengineering research and accelerated commercial development timelines.
The shift toward precision medicine has created demand for customizable nanoparticle platforms that can be tailored to individual patient needs and specific tissue interaction requirements, driving innovation in bioengineering approaches.
Current State of Nanoparticle-Tissue Interaction Research
The field of nanoparticle-tissue interaction research has experienced remarkable growth over the past two decades, driven by advances in nanotechnology and bioengineering. Current research encompasses a broad spectrum of investigations ranging from fundamental cellular uptake mechanisms to complex tissue-level responses. The primary focus areas include understanding biocompatibility, biodistribution patterns, and the long-term effects of various nanoparticle formulations on different tissue types.
Internationally, research efforts are concentrated in several key regions, with the United States, European Union, and East Asian countries leading in both publication volume and technological innovation. American institutions excel in translational research and clinical applications, while European research centers demonstrate strength in regulatory frameworks and safety assessment protocols. Asian countries, particularly China, Japan, and South Korea, have made significant contributions to synthesis methodologies and characterization techniques.
The current technological landscape reveals significant disparities in research capabilities and regulatory approaches across different regions. Developed nations possess advanced characterization facilities and standardized testing protocols, enabling comprehensive evaluation of nanoparticle behavior in biological systems. However, developing countries face challenges in accessing sophisticated analytical equipment and establishing robust safety evaluation frameworks.
Major technical challenges persist in achieving predictable and controllable nanoparticle-tissue interactions. The complexity of biological systems creates substantial obstacles in understanding how surface modifications, particle size, and material composition influence tissue responses. Researchers struggle with reproducibility issues, as slight variations in synthesis conditions can dramatically alter biological outcomes. Additionally, the lack of standardized characterization methods across laboratories hampers comparative studies and meta-analyses.
Current research methodologies predominantly rely on in vitro cell culture models and animal studies to evaluate nanoparticle-tissue interactions. Advanced imaging techniques, including confocal microscopy, electron microscopy, and real-time fluorescence tracking, provide insights into cellular uptake pathways and intracellular distribution patterns. However, translating these findings to human applications remains challenging due to species-specific differences and the complexity of human tissue architecture.
The regulatory landscape presents another significant constraint, with agencies worldwide still developing comprehensive guidelines for nanoparticle-based biomedical applications. This regulatory uncertainty creates hesitation among researchers and industry partners, potentially slowing the translation of promising laboratory findings into clinical applications.
Internationally, research efforts are concentrated in several key regions, with the United States, European Union, and East Asian countries leading in both publication volume and technological innovation. American institutions excel in translational research and clinical applications, while European research centers demonstrate strength in regulatory frameworks and safety assessment protocols. Asian countries, particularly China, Japan, and South Korea, have made significant contributions to synthesis methodologies and characterization techniques.
The current technological landscape reveals significant disparities in research capabilities and regulatory approaches across different regions. Developed nations possess advanced characterization facilities and standardized testing protocols, enabling comprehensive evaluation of nanoparticle behavior in biological systems. However, developing countries face challenges in accessing sophisticated analytical equipment and establishing robust safety evaluation frameworks.
Major technical challenges persist in achieving predictable and controllable nanoparticle-tissue interactions. The complexity of biological systems creates substantial obstacles in understanding how surface modifications, particle size, and material composition influence tissue responses. Researchers struggle with reproducibility issues, as slight variations in synthesis conditions can dramatically alter biological outcomes. Additionally, the lack of standardized characterization methods across laboratories hampers comparative studies and meta-analyses.
Current research methodologies predominantly rely on in vitro cell culture models and animal studies to evaluate nanoparticle-tissue interactions. Advanced imaging techniques, including confocal microscopy, electron microscopy, and real-time fluorescence tracking, provide insights into cellular uptake pathways and intracellular distribution patterns. However, translating these findings to human applications remains challenging due to species-specific differences and the complexity of human tissue architecture.
The regulatory landscape presents another significant constraint, with agencies worldwide still developing comprehensive guidelines for nanoparticle-based biomedical applications. This regulatory uncertainty creates hesitation among researchers and industry partners, potentially slowing the translation of promising laboratory findings into clinical applications.
Current Nanoparticle-Tissue Interface Solutions
01 Nanoparticle-based drug delivery systems for tissue targeting
Nanoparticles can be engineered as carriers for therapeutic agents to achieve targeted delivery to specific tissues. These systems utilize surface modifications and size optimization to enhance cellular uptake and tissue penetration. The nanoparticles can be designed to respond to specific tissue microenvironments, improving drug bioavailability and reducing systemic toxicity. Various polymeric and lipid-based nanoparticles have been developed to facilitate controlled release and enhanced therapeutic efficacy at target tissue sites.- Nanoparticle-based drug delivery systems for tissue targeting: Nanoparticles can be engineered as carriers for therapeutic agents to achieve targeted delivery to specific tissues. These systems utilize surface modifications and size optimization to enhance cellular uptake and tissue penetration. The nanoparticles can be designed to respond to specific tissue microenvironments, improving drug bioavailability and reducing systemic side effects. Various polymeric and lipid-based nanoparticles have been developed to facilitate controlled release and enhanced therapeutic efficacy at target tissue sites.
- Biocompatibility and tissue response to nanoparticles: The interaction between nanoparticles and biological tissues involves complex cellular and molecular responses that determine biocompatibility. Surface properties, particle size, and composition influence tissue inflammation, immune responses, and cellular toxicity. Studies focus on evaluating tissue integration, foreign body reactions, and long-term safety profiles of nanoparticles. Understanding these interactions is crucial for developing safe nanomaterials for biomedical applications.
- Nanoparticles for tissue imaging and diagnostics: Nanoparticles serve as contrast agents and imaging probes for enhanced visualization of tissue structures and pathological conditions. These materials can be functionalized with targeting ligands to accumulate in specific tissues, enabling early disease detection and monitoring. Various nanoparticle types including quantum dots, magnetic nanoparticles, and gold nanoparticles have been utilized for multimodal imaging applications. The interaction between these nanoparticles and tissues allows for improved spatial resolution and diagnostic accuracy.
- Nanoparticle penetration and distribution in tissue matrices: The ability of nanoparticles to penetrate and distribute within tissue matrices depends on physicochemical properties and tissue architecture. Factors such as particle size, surface charge, and hydrophobicity affect diffusion through extracellular matrices and cellular barriers. Research focuses on optimizing nanoparticle characteristics to achieve uniform tissue distribution and overcome biological barriers. Understanding tissue-nanoparticle interactions at the microscopic level is essential for therapeutic and diagnostic applications.
- Nanoparticles for tissue regeneration and engineering: Nanoparticles play a significant role in tissue engineering by providing scaffolds, delivering growth factors, and modulating cellular behavior. These materials can be incorporated into biomaterials to enhance mechanical properties and promote cell adhesion, proliferation, and differentiation. Nanoparticle-tissue interactions facilitate the formation of functional tissue constructs for regenerative medicine applications. The controlled release of bioactive molecules from nanoparticles supports tissue repair and regeneration processes.
02 Biocompatibility and tissue response to nanoparticles
The interaction between nanoparticles and biological tissues involves complex cellular and molecular responses. Surface properties, particle size, and composition significantly influence tissue compatibility and inflammatory responses. Studies focus on evaluating cytotoxicity, immune responses, and long-term tissue integration of various nanoparticle formulations. Understanding these interactions is crucial for developing safe nanomaterials for biomedical applications and minimizing adverse tissue reactions.Expand Specific Solutions03 Nanoparticles for tissue imaging and diagnostics
Nanoparticles serve as contrast agents and imaging probes for enhanced tissue visualization and disease detection. These materials can be functionalized with targeting ligands to accumulate in specific tissues, enabling improved diagnostic accuracy. Various nanoparticle types including quantum dots, magnetic nanoparticles, and gold nanoparticles are utilized for multimodal imaging applications. The technology allows for real-time monitoring of tissue pathology and treatment responses.Expand Specific Solutions04 Nanoparticle penetration and distribution in tissue matrices
The ability of nanoparticles to penetrate and distribute within tissue structures depends on physicochemical properties and tissue architecture. Research addresses barriers to nanoparticle transport including extracellular matrix density, vascular permeability, and interstitial pressure. Strategies to enhance tissue penetration involve optimizing particle size, surface charge, and incorporating tissue-penetrating peptides. Understanding distribution patterns is essential for maximizing therapeutic efficacy and minimizing off-target effects.Expand Specific Solutions05 Nanoparticle-mediated tissue regeneration and repair
Nanoparticles are employed to promote tissue regeneration by delivering growth factors, genes, or stem cells to damaged tissues. These systems can provide scaffolding support and create favorable microenvironments for tissue healing. The technology enables controlled release of bioactive molecules that stimulate cellular proliferation and differentiation. Applications include wound healing, bone regeneration, and neural tissue repair through enhanced cell-material interactions.Expand Specific Solutions
Key Players in Nanoparticle Bioengineering Industry
The nanoparticles in bioengineering field represents a rapidly evolving sector transitioning from early research to clinical applications, with substantial market growth driven by increasing demand for targeted drug delivery and tissue engineering solutions. The competitive landscape features diverse players ranging from leading academic institutions like Johns Hopkins University, Yale University, and Shanghai Jiao Tong University conducting foundational research, to established healthcare giants such as General Electric Company and Koninklijke Philips NV leveraging their technological expertise. Technology maturity varies significantly across applications, with companies like Liquidia Technologies and Osiris Therapeutics advancing specialized nanoparticle therapeutics through clinical trials, while research foundations like Purdue Research Foundation and biotech firms such as BICO Group AB focus on platform development and commercialization of bioengineering tools for tissue interaction studies.
The Regents of the University of California
Technical Solution: UC system has developed comprehensive nanoparticle platforms for tissue engineering applications, focusing on biocompatible polymer-based nanocarriers for targeted drug delivery and tissue regeneration. Their research encompasses functionalized nanoparticles with surface modifications to enhance cellular uptake and reduce immunogenicity. The university has pioneered work in hydrogel-encapsulated nanoparticles for controlled release in tissue scaffolds, demonstrating improved integration with native tissue architecture. Their multidisciplinary approach combines materials science with bioengineering to create nanoparticles that can modulate cellular behavior and promote tissue repair through controlled presentation of growth factors and bioactive molecules.
Strengths: Extensive research infrastructure, multidisciplinary expertise, strong publication record in nanobioengineering. Weaknesses: Academic focus may limit immediate commercial translation, regulatory pathway challenges for novel nanomaterials.
Koninklijke Philips NV
Technical Solution: Philips has developed integrated nanoparticle-based solutions for medical imaging and therapeutic applications in tissue engineering. Their technology platform combines nanoparticle contrast agents with advanced imaging systems to enable real-time monitoring of tissue-nanoparticle interactions. The company has created biocompatible nanoparticles optimized for various imaging modalities including MRI, CT, and ultrasound, allowing for comprehensive assessment of tissue integration and therapeutic efficacy. Their research encompasses smart nanoparticles that can provide both diagnostic information and therapeutic intervention, supporting precision medicine approaches in tissue engineering. Philips has also developed computational models to predict nanoparticle behavior in different tissue environments, enhancing the design and optimization of nanobioengineering applications.
Strengths: Strong imaging technology integration, commercial manufacturing capabilities, regulatory experience with medical devices. Weaknesses: Limited fundamental research in nanomaterial synthesis, dependence on external research partnerships.
Core Patents in Nanoparticle Biocompatibility Research
Nanoparticles for selective tissue or cellular uptake
PatentPendingUS20220339294A1
Innovation
- Development of biodegradable polymeric nanoparticles with controlled sizes between 70 nm and 220 nm, manufactured using a microfluidic system, which are selectively taken up by lung cells and bone marrow cells without the need for targeting agents, allowing for the delivery of therapeutic, diagnostic, and prophylactic agents.
Materials, monitoring, and controlling tissue growth using magnetic nanoparticles
PatentWO2012105984A1
Innovation
- Incorporating magnetic nanoparticles into scaffold materials and cells, which can be ruptured by a magnetic field to release biological factors and enhance imaging contrast, allowing for controlled tissue growth and evaluation through magnetic resonance imaging systems.
Regulatory Framework for Nanomedicine Applications
The regulatory landscape for nanomedicine applications represents one of the most complex and evolving areas in biotechnology governance. As nanoparticles demonstrate unique properties that differ significantly from their bulk counterparts, traditional regulatory frameworks require substantial adaptation to address the novel challenges posed by nanoscale materials in biomedical applications.
The United States Food and Drug Administration has established specific guidance documents for nanotechnology products, emphasizing the importance of characterizing nanomaterials based on their size-dependent properties rather than solely on chemical composition. The FDA's approach focuses on a case-by-case evaluation, recognizing that nanoparticles may exhibit altered pharmacokinetics, biodistribution, and toxicity profiles compared to conventional therapeutics.
European regulatory authorities, particularly the European Medicines Agency, have developed parallel frameworks that emphasize comprehensive risk assessment protocols. The European approach mandates extensive characterization of nanoparticle physicochemical properties, including size distribution, surface chemistry, and stability profiles under physiological conditions. These requirements reflect growing recognition that nanomaterial behavior in biological systems cannot be predicted from conventional drug development paradigms.
International harmonization efforts through organizations such as the International Council for Harmonisation have begun addressing standardization of testing protocols for nanomedicines. These initiatives aim to establish consistent methodologies for evaluating nanoparticle safety, efficacy, and quality across different regulatory jurisdictions, facilitating global development and commercialization pathways.
Current regulatory challenges include establishing appropriate animal models that accurately predict human responses to nanoparticles, developing standardized analytical methods for nanomaterial characterization, and creating guidelines for manufacturing consistency at the nanoscale. The dynamic nature of nanoparticle-tissue interactions necessitates novel assessment strategies that account for temporal changes in particle properties following administration.
Emerging regulatory trends indicate movement toward adaptive frameworks that can accommodate rapid technological advances while maintaining rigorous safety standards. These evolving approaches recognize the need for flexible regulatory pathways that can address the diverse applications of nanoparticles in tissue engineering, drug delivery, and diagnostic applications while ensuring patient safety and therapeutic efficacy.
The United States Food and Drug Administration has established specific guidance documents for nanotechnology products, emphasizing the importance of characterizing nanomaterials based on their size-dependent properties rather than solely on chemical composition. The FDA's approach focuses on a case-by-case evaluation, recognizing that nanoparticles may exhibit altered pharmacokinetics, biodistribution, and toxicity profiles compared to conventional therapeutics.
European regulatory authorities, particularly the European Medicines Agency, have developed parallel frameworks that emphasize comprehensive risk assessment protocols. The European approach mandates extensive characterization of nanoparticle physicochemical properties, including size distribution, surface chemistry, and stability profiles under physiological conditions. These requirements reflect growing recognition that nanomaterial behavior in biological systems cannot be predicted from conventional drug development paradigms.
International harmonization efforts through organizations such as the International Council for Harmonisation have begun addressing standardization of testing protocols for nanomedicines. These initiatives aim to establish consistent methodologies for evaluating nanoparticle safety, efficacy, and quality across different regulatory jurisdictions, facilitating global development and commercialization pathways.
Current regulatory challenges include establishing appropriate animal models that accurately predict human responses to nanoparticles, developing standardized analytical methods for nanomaterial characterization, and creating guidelines for manufacturing consistency at the nanoscale. The dynamic nature of nanoparticle-tissue interactions necessitates novel assessment strategies that account for temporal changes in particle properties following administration.
Emerging regulatory trends indicate movement toward adaptive frameworks that can accommodate rapid technological advances while maintaining rigorous safety standards. These evolving approaches recognize the need for flexible regulatory pathways that can address the diverse applications of nanoparticles in tissue engineering, drug delivery, and diagnostic applications while ensuring patient safety and therapeutic efficacy.
Safety Assessment Protocols for Nanoparticle Biointeractions
The establishment of comprehensive safety assessment protocols for nanoparticle biointeractions represents a critical foundation for advancing bioengineering applications while ensuring patient safety and regulatory compliance. These protocols must address the unique challenges posed by nanoscale materials, which exhibit distinct physicochemical properties that can significantly influence their biological behavior and potential toxicity profiles.
Current safety assessment frameworks integrate multiple evaluation methodologies, beginning with in vitro cytotoxicity screening using standardized cell culture models. These initial assessments examine cellular viability, membrane integrity, and metabolic function following nanoparticle exposure across various concentrations and time points. Advanced protocols incorporate multi-endpoint assays that simultaneously evaluate oxidative stress markers, inflammatory responses, and genotoxicity indicators to provide comprehensive toxicity profiles.
Standardized characterization protocols form the backbone of safety assessment, requiring detailed analysis of nanoparticle size distribution, surface charge, morphology, and chemical composition both before and after biological exposure. Dynamic light scattering, electron microscopy, and spectroscopic techniques provide essential data for correlating physicochemical properties with observed biological effects. These characterization studies must account for protein corona formation and agglomeration behavior in physiological media.
In vivo safety evaluation protocols follow tiered approaches, progressing from acute toxicity studies to chronic exposure assessments. These protocols incorporate biodistribution analysis using advanced imaging techniques and quantitative tissue analysis to track nanoparticle accumulation and clearance pathways. Histopathological examination of target organs, particularly liver, spleen, and kidneys, provides crucial insights into potential systemic toxicity and long-term safety implications.
Regulatory harmonization efforts have led to the development of standardized testing guidelines that align with international safety standards while addressing nanoparticle-specific considerations. These protocols emphasize dose-response relationships, exposure route optimization, and species-specific sensitivity factors. Integration of computational modeling approaches enhances predictive capabilities and reduces reliance on extensive animal testing while maintaining rigorous safety standards for clinical translation.
Current safety assessment frameworks integrate multiple evaluation methodologies, beginning with in vitro cytotoxicity screening using standardized cell culture models. These initial assessments examine cellular viability, membrane integrity, and metabolic function following nanoparticle exposure across various concentrations and time points. Advanced protocols incorporate multi-endpoint assays that simultaneously evaluate oxidative stress markers, inflammatory responses, and genotoxicity indicators to provide comprehensive toxicity profiles.
Standardized characterization protocols form the backbone of safety assessment, requiring detailed analysis of nanoparticle size distribution, surface charge, morphology, and chemical composition both before and after biological exposure. Dynamic light scattering, electron microscopy, and spectroscopic techniques provide essential data for correlating physicochemical properties with observed biological effects. These characterization studies must account for protein corona formation and agglomeration behavior in physiological media.
In vivo safety evaluation protocols follow tiered approaches, progressing from acute toxicity studies to chronic exposure assessments. These protocols incorporate biodistribution analysis using advanced imaging techniques and quantitative tissue analysis to track nanoparticle accumulation and clearance pathways. Histopathological examination of target organs, particularly liver, spleen, and kidneys, provides crucial insights into potential systemic toxicity and long-term safety implications.
Regulatory harmonization efforts have led to the development of standardized testing guidelines that align with international safety standards while addressing nanoparticle-specific considerations. These protocols emphasize dose-response relationships, exposure route optimization, and species-specific sensitivity factors. Integration of computational modeling approaches enhances predictive capabilities and reduces reliance on extensive animal testing while maintaining rigorous safety standards for clinical translation.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!