Quantifying Enol's Tautomerization Rate Using NMR Spectroscopy
MAR 6, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Enol Tautomerization Background and Research Objectives
Enol-keto tautomerization represents one of the most fundamental chemical equilibria in organic chemistry, involving the rapid interconversion between enol and keto forms of carbonyl compounds. This dynamic process has been extensively studied since the early 20th century, with initial investigations focusing on simple β-dicarbonyl compounds where enol forms exhibit significant stability. The phenomenon gained prominence through pioneering work on acetoacetic ester and malonic ester derivatives, establishing the theoretical foundation for understanding tautomeric equilibria.
The evolution of analytical techniques has dramatically enhanced our understanding of tautomerization kinetics. Early studies relied primarily on chemical trapping methods and UV-visible spectroscopy, which provided limited temporal resolution and often required harsh conditions that could perturb the natural equilibrium. The advent of nuclear magnetic resonance spectroscopy in the 1960s revolutionized this field by enabling real-time observation of tautomeric interconversion under mild conditions.
Modern NMR spectroscopy offers unprecedented capabilities for quantifying tautomerization rates through various methodologies including dynamic NMR, magnetization transfer experiments, and exchange spectroscopy techniques. Variable temperature NMR studies have become particularly valuable for determining activation parameters and understanding the mechanistic pathways governing these transformations. Advanced pulse sequences such as EXSY and saturation transfer experiments now allow precise measurement of exchange rates spanning several orders of magnitude.
The primary research objective centers on developing robust NMR-based methodologies for accurate quantification of enol tautomerization kinetics across diverse molecular systems. This encompasses establishing standardized protocols for rate constant determination, validating measurement accuracy through complementary analytical approaches, and extending applicability to complex polyfunctional molecules where multiple tautomeric equilibria may coexist.
Secondary objectives include investigating solvent effects on tautomerization dynamics, elucidating the role of intramolecular hydrogen bonding in stabilizing enol forms, and developing predictive models correlating molecular structure with tautomerization rates. Understanding these relationships is crucial for rational design of molecules with tailored tautomeric properties for pharmaceutical and materials science applications.
The ultimate goal involves creating a comprehensive framework for tautomerization rate analysis that bridges fundamental mechanistic understanding with practical applications in drug discovery, catalyst design, and functional materials development.
The evolution of analytical techniques has dramatically enhanced our understanding of tautomerization kinetics. Early studies relied primarily on chemical trapping methods and UV-visible spectroscopy, which provided limited temporal resolution and often required harsh conditions that could perturb the natural equilibrium. The advent of nuclear magnetic resonance spectroscopy in the 1960s revolutionized this field by enabling real-time observation of tautomeric interconversion under mild conditions.
Modern NMR spectroscopy offers unprecedented capabilities for quantifying tautomerization rates through various methodologies including dynamic NMR, magnetization transfer experiments, and exchange spectroscopy techniques. Variable temperature NMR studies have become particularly valuable for determining activation parameters and understanding the mechanistic pathways governing these transformations. Advanced pulse sequences such as EXSY and saturation transfer experiments now allow precise measurement of exchange rates spanning several orders of magnitude.
The primary research objective centers on developing robust NMR-based methodologies for accurate quantification of enol tautomerization kinetics across diverse molecular systems. This encompasses establishing standardized protocols for rate constant determination, validating measurement accuracy through complementary analytical approaches, and extending applicability to complex polyfunctional molecules where multiple tautomeric equilibria may coexist.
Secondary objectives include investigating solvent effects on tautomerization dynamics, elucidating the role of intramolecular hydrogen bonding in stabilizing enol forms, and developing predictive models correlating molecular structure with tautomerization rates. Understanding these relationships is crucial for rational design of molecules with tailored tautomeric properties for pharmaceutical and materials science applications.
The ultimate goal involves creating a comprehensive framework for tautomerization rate analysis that bridges fundamental mechanistic understanding with practical applications in drug discovery, catalyst design, and functional materials development.
Market Demand for Advanced NMR Quantification Methods
The pharmaceutical industry represents the primary market driver for advanced NMR quantification methods, particularly for tautomerization studies. Drug discovery and development processes increasingly require precise characterization of molecular dynamics and equilibrium states, as tautomeric forms can exhibit dramatically different biological activities, pharmacokinetic properties, and toxicity profiles. Pharmaceutical companies are investing heavily in analytical technologies that can provide real-time, quantitative insights into tautomeric equilibria to optimize lead compound selection and reduce late-stage development failures.
Academic research institutions constitute another significant market segment, driven by fundamental research needs in organic chemistry, biochemistry, and materials science. Universities and research centers require sophisticated NMR quantification capabilities to advance understanding of molecular behavior, reaction mechanisms, and structure-activity relationships. The growing emphasis on interdisciplinary research combining chemistry, biology, and physics has expanded demand for versatile analytical platforms capable of handling complex tautomeric systems.
The chemical manufacturing sector presents substantial opportunities for advanced NMR quantification methods, particularly in quality control and process optimization applications. Chemical companies need to monitor tautomeric compositions in real-time during synthesis processes, as different tautomeric forms can affect product stability, reactivity, and performance characteristics. This demand is particularly pronounced in specialty chemicals, agrochemicals, and advanced materials manufacturing.
Biotechnology companies focusing on enzyme engineering and biocatalysis represent an emerging market segment. These organizations require precise quantification of substrate and product tautomeric states to optimize enzymatic processes and understand biocatalytic mechanisms. The growing biotechnology sector's emphasis on sustainable manufacturing processes has increased interest in analytical methods that can monitor complex molecular transformations.
Contract research organizations and analytical service providers form a crucial market component, offering specialized NMR quantification services to clients lacking in-house capabilities. These organizations require robust, high-throughput analytical platforms to serve diverse client needs across multiple industries. The outsourcing trend in analytical chemistry has strengthened demand for advanced, automated NMR quantification systems.
Regulatory requirements across pharmaceutical and chemical industries are driving market growth, as regulatory agencies increasingly demand comprehensive characterization of molecular forms and their interconversion kinetics. This regulatory pressure creates sustained demand for validated, reliable quantification methods capable of meeting stringent documentation and reproducibility standards.
Academic research institutions constitute another significant market segment, driven by fundamental research needs in organic chemistry, biochemistry, and materials science. Universities and research centers require sophisticated NMR quantification capabilities to advance understanding of molecular behavior, reaction mechanisms, and structure-activity relationships. The growing emphasis on interdisciplinary research combining chemistry, biology, and physics has expanded demand for versatile analytical platforms capable of handling complex tautomeric systems.
The chemical manufacturing sector presents substantial opportunities for advanced NMR quantification methods, particularly in quality control and process optimization applications. Chemical companies need to monitor tautomeric compositions in real-time during synthesis processes, as different tautomeric forms can affect product stability, reactivity, and performance characteristics. This demand is particularly pronounced in specialty chemicals, agrochemicals, and advanced materials manufacturing.
Biotechnology companies focusing on enzyme engineering and biocatalysis represent an emerging market segment. These organizations require precise quantification of substrate and product tautomeric states to optimize enzymatic processes and understand biocatalytic mechanisms. The growing biotechnology sector's emphasis on sustainable manufacturing processes has increased interest in analytical methods that can monitor complex molecular transformations.
Contract research organizations and analytical service providers form a crucial market component, offering specialized NMR quantification services to clients lacking in-house capabilities. These organizations require robust, high-throughput analytical platforms to serve diverse client needs across multiple industries. The outsourcing trend in analytical chemistry has strengthened demand for advanced, automated NMR quantification systems.
Regulatory requirements across pharmaceutical and chemical industries are driving market growth, as regulatory agencies increasingly demand comprehensive characterization of molecular forms and their interconversion kinetics. This regulatory pressure creates sustained demand for validated, reliable quantification methods capable of meeting stringent documentation and reproducibility standards.
Current NMR Limitations in Tautomerization Rate Studies
Nuclear Magnetic Resonance spectroscopy faces significant temporal resolution constraints when applied to tautomerization rate studies, particularly for enol-keto equilibria. Traditional NMR techniques operate on timescales that often exceed the rapid interconversion rates characteristic of many tautomeric systems. The fundamental limitation stems from the relationship between chemical exchange rates and NMR observability, where exchange processes faster than the chemical shift difference between tautomers result in averaged signals that obscure individual species identification.
The sensitivity threshold of conventional NMR instrumentation presents another critical barrier in quantifying enol tautomerization rates. Enol forms typically exist as minor components in tautomeric equilibria, often comprising less than 1% of the total population under ambient conditions. Standard proton NMR detection limits struggle to accurately quantify such low-abundance species, particularly when attempting to monitor their dynamic behavior in real-time. This sensitivity challenge is compounded by the need for sufficient signal-to-noise ratios to enable reliable kinetic measurements.
Temperature-dependent studies, essential for determining activation parameters in tautomerization processes, introduce additional complications in NMR-based rate quantification. Variable temperature NMR experiments often suffer from baseline instabilities, shimming difficulties, and thermal equilibration issues that can introduce systematic errors in rate measurements. The temperature range accessible in standard NMR probes may also be insufficient to achieve the necessary dynamic range for accurate kinetic parameter determination.
Solvent effects represent a fundamental challenge in NMR-based tautomerization studies, as the choice of deuterated solvents can significantly influence both the equilibrium position and exchange kinetics. Protic solvents, which often provide the most physiologically relevant conditions, are incompatible with standard NMR measurements due to signal overlap and exchange broadening effects. This limitation forces researchers to rely on aprotic deuterated solvents that may not accurately reflect the tautomeric behavior under biologically or industrially relevant conditions.
The complexity of overlapping multipicity patterns in crowded spectral regions further complicates accurate integration and peak assignment necessary for rate determination. Enol protons often appear in regions with significant spectral congestion, making baseline definition and peak integration challenging. Additionally, the coupling patterns of enol protons can change upon tautomerization, leading to complex lineshape analysis requirements that exceed the capabilities of standard NMR processing software.
The sensitivity threshold of conventional NMR instrumentation presents another critical barrier in quantifying enol tautomerization rates. Enol forms typically exist as minor components in tautomeric equilibria, often comprising less than 1% of the total population under ambient conditions. Standard proton NMR detection limits struggle to accurately quantify such low-abundance species, particularly when attempting to monitor their dynamic behavior in real-time. This sensitivity challenge is compounded by the need for sufficient signal-to-noise ratios to enable reliable kinetic measurements.
Temperature-dependent studies, essential for determining activation parameters in tautomerization processes, introduce additional complications in NMR-based rate quantification. Variable temperature NMR experiments often suffer from baseline instabilities, shimming difficulties, and thermal equilibration issues that can introduce systematic errors in rate measurements. The temperature range accessible in standard NMR probes may also be insufficient to achieve the necessary dynamic range for accurate kinetic parameter determination.
Solvent effects represent a fundamental challenge in NMR-based tautomerization studies, as the choice of deuterated solvents can significantly influence both the equilibrium position and exchange kinetics. Protic solvents, which often provide the most physiologically relevant conditions, are incompatible with standard NMR measurements due to signal overlap and exchange broadening effects. This limitation forces researchers to rely on aprotic deuterated solvents that may not accurately reflect the tautomeric behavior under biologically or industrially relevant conditions.
The complexity of overlapping multipicity patterns in crowded spectral regions further complicates accurate integration and peak assignment necessary for rate determination. Enol protons often appear in regions with significant spectral congestion, making baseline definition and peak integration challenging. Additionally, the coupling patterns of enol protons can change upon tautomerization, leading to complex lineshape analysis requirements that exceed the capabilities of standard NMR processing software.
Existing NMR Methods for Tautomerization Rate Measurement
01 Dynamic NMR spectroscopy techniques for measuring tautomerization kinetics
Dynamic NMR spectroscopy methods can be employed to determine tautomerization rates by analyzing temperature-dependent spectral changes and line-shape variations. These techniques allow for the measurement of exchange rates between tautomeric forms through variable temperature NMR experiments and coalescence temperature analysis. The methods provide quantitative data on activation energies and rate constants for tautomeric equilibria.- Dynamic NMR spectroscopy techniques for measuring tautomerization kinetics: Dynamic NMR spectroscopy methods can be employed to determine tautomerization rates by analyzing temperature-dependent spectral changes and coalescence phenomena. These techniques involve variable temperature NMR experiments where the exchange rates between tautomeric forms are calculated from line shape analysis and coalescence temperature measurements. The activation energies and rate constants for tautomeric interconversion can be derived from the temperature dependence of the NMR signals.
- Two-dimensional NMR methods for tautomer identification and exchange analysis: Two-dimensional NMR techniques such as EXSY (Exchange Spectroscopy) and NOESY can be utilized to identify tautomeric species and measure their interconversion rates. These methods provide cross-peaks that correlate exchanging nuclei, allowing for the determination of exchange rates and equilibrium constants. The intensity of cross-peaks in 2D spectra can be analyzed to quantify the kinetics of tautomerization processes.
- Solvent and pH effects on tautomerization rates studied by NMR: NMR spectroscopy can be used to investigate how different solvents and pH conditions affect tautomerization rates. By conducting NMR experiments in various solvent systems and at different pH values, the influence of environmental factors on tautomeric equilibria and exchange kinetics can be determined. This approach helps in understanding the mechanism of tautomerization and the role of solvent-solute interactions.
- Quantitative analysis of tautomeric populations using NMR integration: The relative populations of different tautomeric forms can be quantified through integration of NMR signals corresponding to each tautomer. This method allows for the determination of equilibrium constants and thermodynamic parameters associated with tautomerization. By monitoring signal intensities as a function of experimental conditions, the distribution of tautomeric species and their interconversion dynamics can be characterized.
- Advanced NMR pulse sequences for slow exchange tautomerization studies: Specialized NMR pulse sequences and magnetization transfer experiments can be applied to study slow tautomerization processes where conventional methods are insufficient. These techniques include saturation transfer, selective inversion recovery, and other magnetization transfer methods that enhance sensitivity to slow exchange phenomena. Such approaches enable the measurement of very slow tautomerization rates that would otherwise be difficult to detect.
02 Two-dimensional NMR methods for tautomer identification and rate determination
Two-dimensional NMR techniques including EXSY (Exchange Spectroscopy) and NOESY experiments enable the detection and quantification of tautomeric exchange processes. These methods provide cross-peaks that correlate exchanging species and allow calculation of exchange rates from peak intensities and mixing times. Advanced 2D NMR approaches offer improved sensitivity for studying slow to intermediate exchange rates in tautomeric systems.Expand Specific Solutions03 Solvent and pH effects on tautomerization rates studied by NMR
NMR spectroscopy can be utilized to investigate how different solvents and pH conditions influence tautomerization kinetics. Studies examine the impact of solvent polarity, hydrogen bonding capabilities, and ionic strength on the equilibrium position and interconversion rates between tautomers. These investigations provide insights into the mechanisms of proton transfer and the role of environmental factors in tautomeric processes.Expand Specific Solutions04 Computational methods combined with NMR for tautomerization rate analysis
Integration of computational chemistry approaches with experimental NMR data enhances the understanding of tautomerization mechanisms and rate predictions. Quantum chemical calculations complement NMR measurements by providing theoretical chemical shifts, energy barriers, and transition state structures. This combined approach allows for validation of experimental findings and prediction of tautomeric behavior in complex molecular systems.Expand Specific Solutions05 Specialized NMR pulse sequences for slow tautomerization rate measurements
Advanced NMR pulse sequences and magnetization transfer experiments have been developed specifically for measuring slow tautomerization rates that are challenging to detect by conventional methods. These techniques include saturation transfer, selective inversion recovery, and spin-lock experiments that enhance sensitivity to slow exchange processes. The methods enable accurate determination of rate constants over a wide range of timescales relevant to tautomeric equilibria.Expand Specific Solutions
Key Players in NMR Instrumentation and Software Industry
The competitive landscape for quantifying enol's tautomerization rate using NMR spectroscopy reflects an emerging research area with significant industrial potential. The field is in its early development stage, primarily driven by academic institutions and research organizations rather than established commercial markets. Key players include major pharmaceutical companies like Novartis AG, Daiichi Sankyo, and AstraZeneca AB, who recognize the importance of tautomerization studies in drug development. Chemical industry giants such as China Petroleum & Chemical Corp., ExxonMobil Chemical Patents, and Mitsui Chemicals are exploring applications in petrochemical processes. Leading research institutions including Max Planck Gesellschaft, Johns Hopkins University, and National University of Singapore are advancing fundamental understanding. The technology remains in nascent stages with limited commercial deployment, suggesting substantial growth opportunities as NMR-based tautomerization quantification methods mature and find broader industrial applications across pharmaceutical, chemical, and materials science sectors.
Max Planck Gesellschaft zur Förderung der Wissenschaften eV
Technical Solution: Max Planck Institute has pioneered fundamental research in NMR-based tautomerization studies, developing novel pulse sequences and analytical methods for quantifying enol-keto equilibria. Their approach incorporates advanced multidimensional NMR techniques, including real-time NMR monitoring and sophisticated mathematical modeling frameworks. The institute has contributed significantly to understanding the theoretical foundations of tautomeric exchange processes and developed innovative experimental protocols for measuring fast exchange rates using line-shape analysis and relaxation-based methods.
Strengths: Cutting-edge fundamental research capabilities and development of novel NMR methodologies. Weaknesses: Academic focus may limit immediate industrial application and scalability.
Novartis AG
Technical Solution: Novartis has developed advanced NMR spectroscopy methodologies for studying tautomeric equilibria in pharmaceutical compounds. Their approach utilizes variable temperature NMR experiments combined with dynamic NMR (DNMR) techniques to quantify enol-keto tautomerization rates. The company employs sophisticated pulse sequences and mathematical modeling to extract kinetic parameters from line-shape analysis and magnetization transfer experiments, enabling precise determination of activation energies and rate constants for tautomeric interconversion processes in drug molecules.
Strengths: Extensive pharmaceutical expertise and access to high-field NMR instruments. Weaknesses: Focus primarily on drug-related applications may limit broader chemical scope.
Core NMR Innovations for Enol Tautomerization Quantification
NMR spectroscopy using spin states with long lifetimes
PatentInactiveUS7474095B2
Innovation
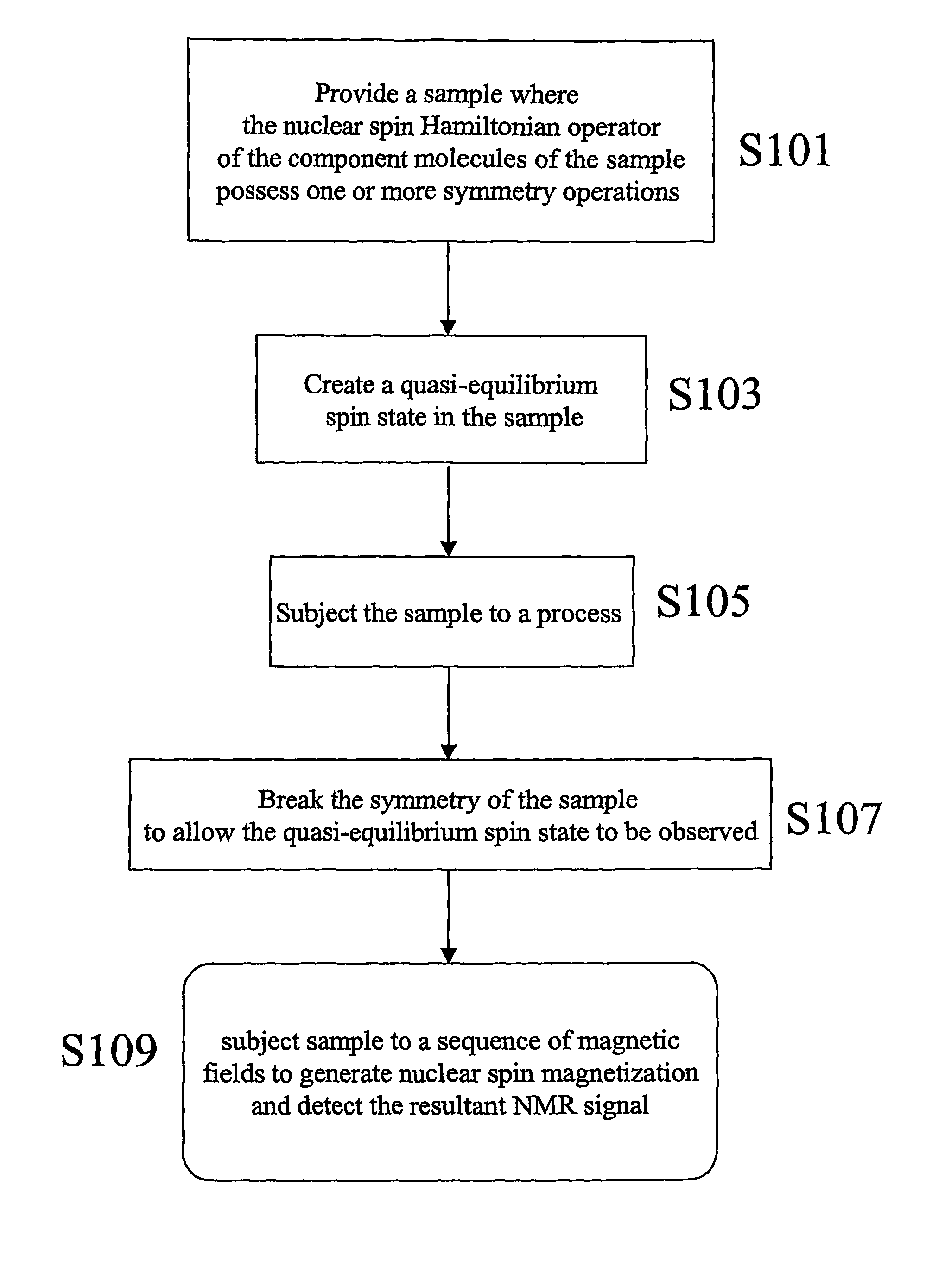
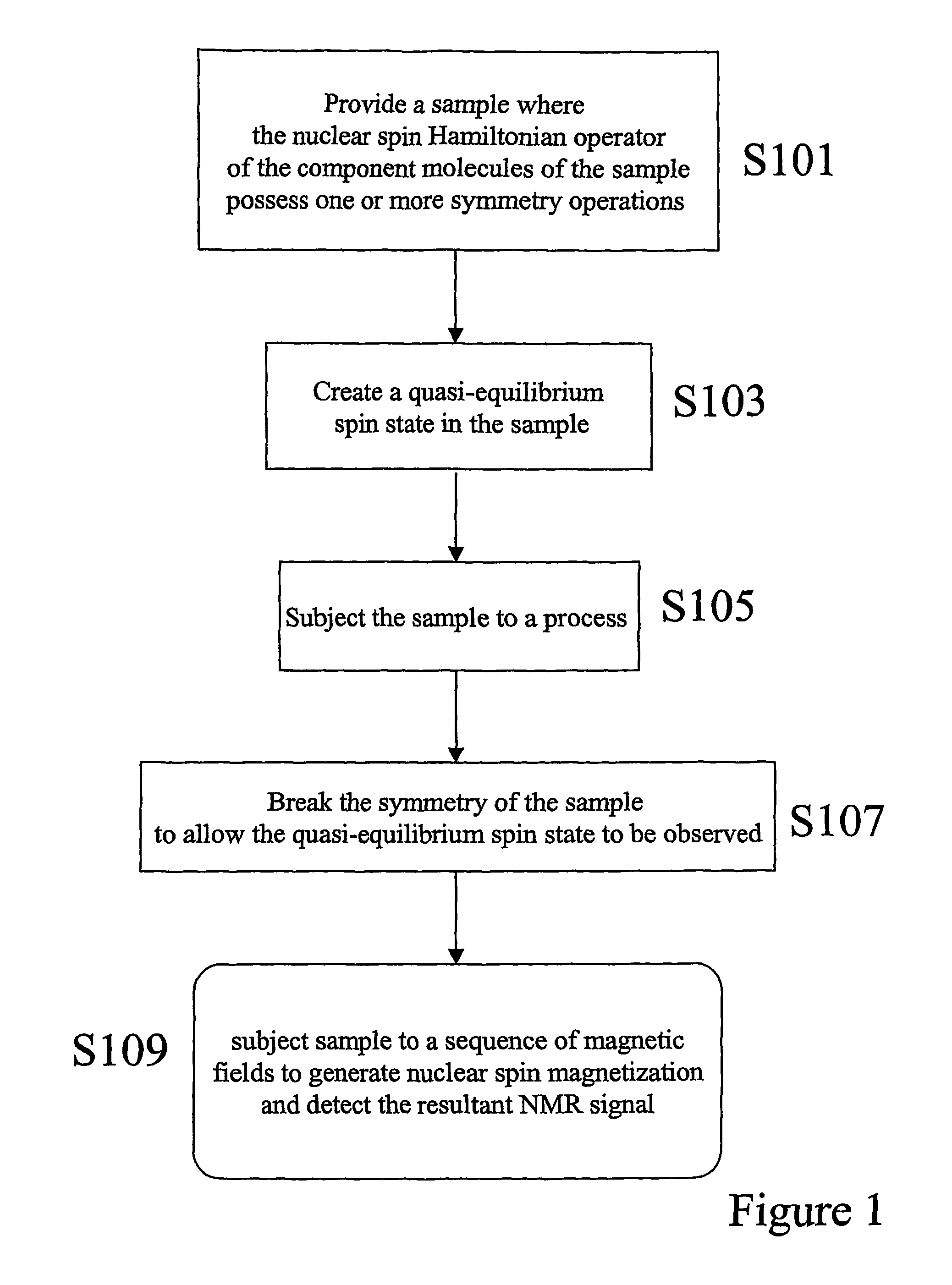
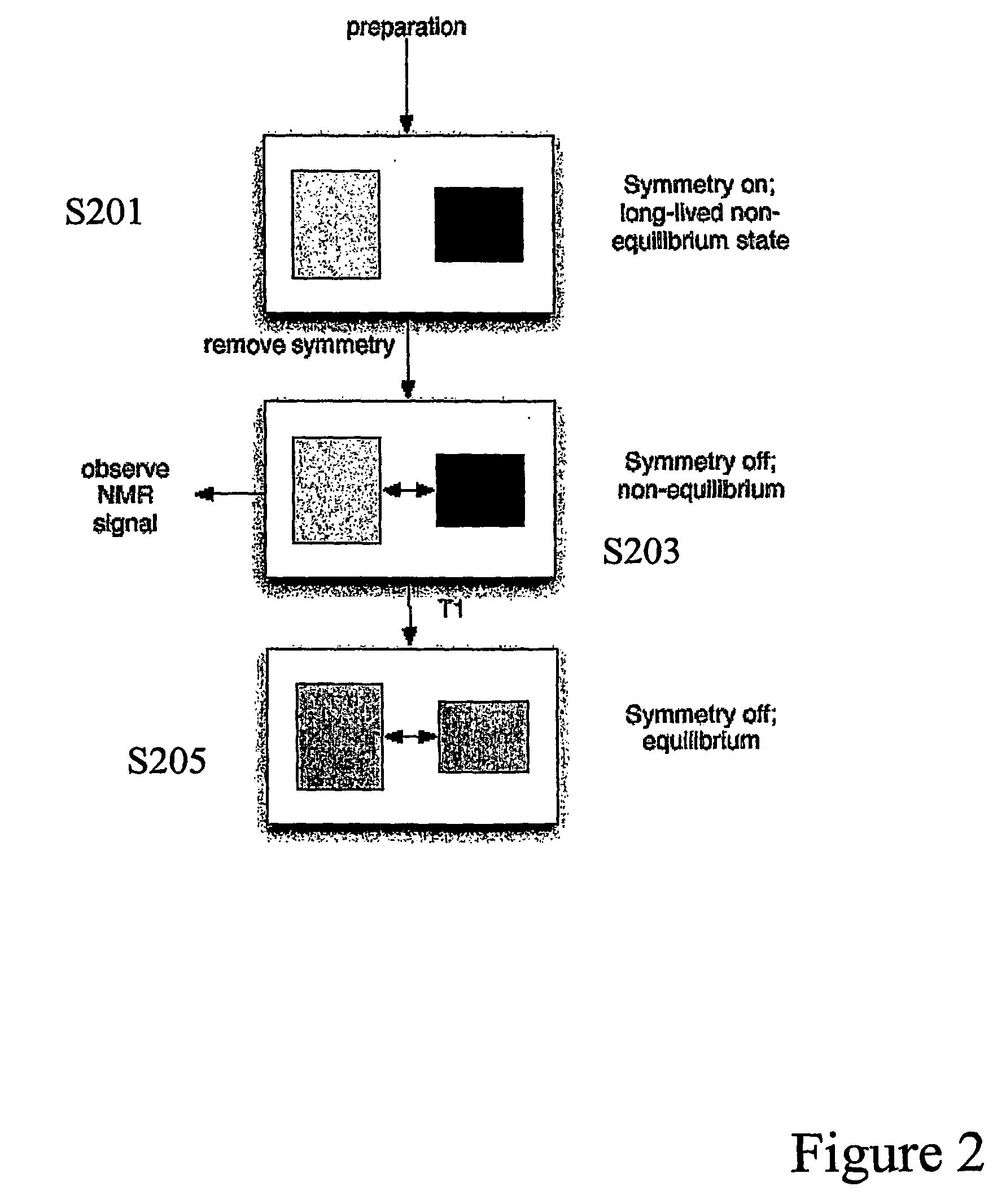
- The method involves creating quasi-equilibrium nuclear spin ensemble states by manipulating the symmetry of the nuclear spin Hamiltonian, allowing these states to persist for times greater than 3T1, enabling the generation of nuclear magnetic resonance signals through the application of magnetic fields and detection techniques.
Systems and methods for fast quantitative NMR spectrum acquisition
PatentActiveEP4502636A1
Innovation
- A computer-implemented method that uses a combination of long and short delay intervals between NMR scans to allow for near-full relaxation of the sample, significantly reducing the total acquisition time while maintaining high accuracy.
Safety Regulations for NMR Laboratory Operations
NMR laboratory operations involving tautomerization rate studies require stringent safety protocols due to the unique hazards associated with both high-field magnetic environments and chemical handling procedures. The superconducting magnets used in modern NMR spectrometers operate at extremely low temperatures using liquid helium and nitrogen, creating risks of cryogenic burns, asphyxiation from displaced oxygen, and potential quench events that can rapidly release stored magnetic energy.
Personnel working with enol tautomerization experiments must undergo specialized training in magnetic field safety, as ferromagnetic objects can become dangerous projectiles when brought near the magnet. All individuals entering NMR facilities must be screened for metallic implants, pacemakers, and other medical devices that could malfunction or cause injury in strong magnetic fields. Emergency procedures must be established for magnet quench scenarios, including immediate evacuation protocols and ventilation system activation.
Chemical safety protocols are particularly critical when handling enol-containing compounds and deuterated solvents commonly used in tautomerization studies. Many enol substrates are air-sensitive and require inert atmosphere handling techniques. Deuterated solvents, while generally less toxic than their protiated counterparts, still pose health risks and require proper storage in moisture-free environments. Sample preparation areas must be equipped with appropriate fume hoods and spill containment systems.
Temperature-controlled experiments studying tautomerization kinetics introduce additional safety considerations. Variable temperature NMR operations require careful monitoring of cryogenic gas flow rates and pressure systems. Personnel must be trained in the proper handling of temperature control units and emergency shutdown procedures to prevent equipment damage and ensure operator safety.
Radiation safety protocols, while less stringent than for radioactive materials, must address potential RF exposure from high-power pulse sequences used in dynamic NMR experiments. Regular calibration of RF power levels and implementation of interlock systems prevent accidental exposure to harmful electromagnetic radiation levels during extended kinetic measurements.
Personnel working with enol tautomerization experiments must undergo specialized training in magnetic field safety, as ferromagnetic objects can become dangerous projectiles when brought near the magnet. All individuals entering NMR facilities must be screened for metallic implants, pacemakers, and other medical devices that could malfunction or cause injury in strong magnetic fields. Emergency procedures must be established for magnet quench scenarios, including immediate evacuation protocols and ventilation system activation.
Chemical safety protocols are particularly critical when handling enol-containing compounds and deuterated solvents commonly used in tautomerization studies. Many enol substrates are air-sensitive and require inert atmosphere handling techniques. Deuterated solvents, while generally less toxic than their protiated counterparts, still pose health risks and require proper storage in moisture-free environments. Sample preparation areas must be equipped with appropriate fume hoods and spill containment systems.
Temperature-controlled experiments studying tautomerization kinetics introduce additional safety considerations. Variable temperature NMR operations require careful monitoring of cryogenic gas flow rates and pressure systems. Personnel must be trained in the proper handling of temperature control units and emergency shutdown procedures to prevent equipment damage and ensure operator safety.
Radiation safety protocols, while less stringent than for radioactive materials, must address potential RF exposure from high-power pulse sequences used in dynamic NMR experiments. Regular calibration of RF power levels and implementation of interlock systems prevent accidental exposure to harmful electromagnetic radiation levels during extended kinetic measurements.
Standardization Challenges in Tautomerization Rate Reporting
The quantification of enol tautomerization rates using NMR spectroscopy faces significant standardization challenges that hinder reproducibility and cross-laboratory comparisons. Currently, there is no universally accepted protocol for reporting kinetic parameters, leading to inconsistent methodologies across research groups and making it difficult to establish reliable databases for tautomerization rate constants.
One of the primary standardization issues lies in the diversity of experimental conditions employed across different studies. Temperature control represents a critical variable, as tautomerization rates are highly temperature-dependent, yet many publications fail to report precise temperature measurements or thermal equilibration protocols. Similarly, solvent selection and purity specifications vary considerably, with some researchers using deuterated solvents while others employ protonated systems, creating incomparable datasets.
The lack of standardized reference compounds further complicates rate comparisons. Different research groups often select distinct internal standards or reference molecules, making it challenging to normalize results across studies. Additionally, the absence of agreed-upon calibration procedures for NMR instruments introduces systematic errors that can significantly impact measured rate constants.
Reporting formats present another substantial challenge. Some researchers report half-lives, others use first-order rate constants, and still others present activation energies without providing sufficient experimental details for reproduction. The inconsistent use of units, significant figures, and uncertainty estimates makes quantitative comparisons nearly impossible.
Sample preparation protocols also lack standardization, particularly regarding concentration ranges, degassing procedures, and moisture control. These variables can dramatically influence tautomerization kinetics, yet they are often inadequately documented or vary significantly between laboratories.
The absence of round-robin testing programs or interlaboratory validation studies has prevented the development of consensus methods. Without collaborative efforts to establish standard operating procedures, the field continues to struggle with reproducibility issues that limit the practical application of NMR-based tautomerization rate measurements in pharmaceutical and chemical industries.
One of the primary standardization issues lies in the diversity of experimental conditions employed across different studies. Temperature control represents a critical variable, as tautomerization rates are highly temperature-dependent, yet many publications fail to report precise temperature measurements or thermal equilibration protocols. Similarly, solvent selection and purity specifications vary considerably, with some researchers using deuterated solvents while others employ protonated systems, creating incomparable datasets.
The lack of standardized reference compounds further complicates rate comparisons. Different research groups often select distinct internal standards or reference molecules, making it challenging to normalize results across studies. Additionally, the absence of agreed-upon calibration procedures for NMR instruments introduces systematic errors that can significantly impact measured rate constants.
Reporting formats present another substantial challenge. Some researchers report half-lives, others use first-order rate constants, and still others present activation energies without providing sufficient experimental details for reproduction. The inconsistent use of units, significant figures, and uncertainty estimates makes quantitative comparisons nearly impossible.
Sample preparation protocols also lack standardization, particularly regarding concentration ranges, degassing procedures, and moisture control. These variables can dramatically influence tautomerization kinetics, yet they are often inadequately documented or vary significantly between laboratories.
The absence of round-robin testing programs or interlaboratory validation studies has prevented the development of consensus methods. Without collaborative efforts to establish standard operating procedures, the field continues to struggle with reproducibility issues that limit the practical application of NMR-based tautomerization rate measurements in pharmaceutical and chemical industries.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!