High-capacity sulfur cathode materials explained
FEB 14, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Sulfur Cathode Development Background and Objectives
The development of lithium-sulfur batteries represents a paradigm shift in energy storage technology, driven by the urgent need for next-generation power sources that surpass the limitations of conventional lithium-ion systems. Sulfur cathodes have emerged as a compelling alternative due to their exceptional theoretical specific capacity of 1675 mAh/g and theoretical energy density of 2600 Wh/kg, which are approximately five times higher than traditional lithium-ion cathode materials. This remarkable energy storage potential, combined with sulfur's natural abundance, low cost, and environmental benignity, positions sulfur-based cathodes as a transformative solution for applications ranging from electric vehicles to grid-scale energy storage.
The historical trajectory of sulfur cathode research began in the 1960s, but significant momentum accelerated in the early 2000s as researchers recognized the critical need for high-energy-density batteries to support the electrification revolution. Early investigations revealed fundamental challenges including poor electronic conductivity of sulfur, substantial volume expansion during lithiation, and the notorious polysulfide shuttle effect that severely compromised cycle life and coulombic efficiency. These technical barriers initially hindered commercial viability despite the material's inherent advantages.
Contemporary research objectives focus on achieving multiple concurrent goals to realize practical high-capacity sulfur cathodes. The primary technical target involves maintaining stable specific capacities exceeding 1000 mAh/g over extended cycling periods, ideally surpassing 500 charge-discharge cycles with minimal capacity fade. Researchers aim to suppress polysulfide dissolution and migration through innovative cathode architectures, advanced carbon host materials, and functional interlayers that physically and chemically confine sulfur species within the cathode region.
Additional objectives encompass improving sulfur utilization efficiency beyond 70%, reducing the electrolyte-to-sulfur ratio to economically viable levels below 3 μL/mg, and increasing sulfur loading in cathodes to practical levels exceeding 4 mg/cm². These targets collectively address the gap between laboratory demonstrations and commercial requirements, ensuring that sulfur cathodes can deliver both high energy density and long-term reliability for real-world applications in next-generation energy storage systems.
The historical trajectory of sulfur cathode research began in the 1960s, but significant momentum accelerated in the early 2000s as researchers recognized the critical need for high-energy-density batteries to support the electrification revolution. Early investigations revealed fundamental challenges including poor electronic conductivity of sulfur, substantial volume expansion during lithiation, and the notorious polysulfide shuttle effect that severely compromised cycle life and coulombic efficiency. These technical barriers initially hindered commercial viability despite the material's inherent advantages.
Contemporary research objectives focus on achieving multiple concurrent goals to realize practical high-capacity sulfur cathodes. The primary technical target involves maintaining stable specific capacities exceeding 1000 mAh/g over extended cycling periods, ideally surpassing 500 charge-discharge cycles with minimal capacity fade. Researchers aim to suppress polysulfide dissolution and migration through innovative cathode architectures, advanced carbon host materials, and functional interlayers that physically and chemically confine sulfur species within the cathode region.
Additional objectives encompass improving sulfur utilization efficiency beyond 70%, reducing the electrolyte-to-sulfur ratio to economically viable levels below 3 μL/mg, and increasing sulfur loading in cathodes to practical levels exceeding 4 mg/cm². These targets collectively address the gap between laboratory demonstrations and commercial requirements, ensuring that sulfur cathodes can deliver both high energy density and long-term reliability for real-world applications in next-generation energy storage systems.
Market Demand for High-Energy Battery Systems
The global transition toward electrification and renewable energy integration has created unprecedented demand for high-energy battery systems across multiple sectors. Electric vehicles represent the most significant driver, with automotive manufacturers worldwide committing to aggressive electrification roadmaps that require substantial improvements in driving range and charging efficiency. Current lithium-ion battery technology, while mature, faces inherent energy density limitations that constrain vehicle performance and increase costs. High-capacity sulfur cathode materials offer a promising pathway to address these constraints, potentially delivering energy densities several times higher than conventional cathode chemistries.
Beyond transportation, the proliferation of portable electronics continues to demand lighter, longer-lasting power sources. Consumer expectations for smartphones, laptops, and wearable devices increasingly center on extended battery life without compromising device form factors. Similarly, the aerospace and defense sectors require lightweight, high-energy solutions for unmanned aerial vehicles, satellites, and military applications where weight reduction directly translates to operational advantages.
Grid-scale energy storage represents another critical application domain. As renewable energy sources like solar and wind become more prevalent, the need for cost-effective, high-capacity storage systems intensifies to manage intermittency and ensure grid stability. Sulfur-based cathode materials, leveraging abundant and inexpensive raw materials, could significantly reduce the levelized cost of energy storage while improving system energy density.
The convergence of environmental regulations, consumer preferences, and technological advancement has created a market environment highly receptive to breakthrough battery technologies. Governments worldwide are implementing stricter emissions standards and offering incentives for clean energy adoption, further accelerating demand for superior energy storage solutions. This multifaceted market pressure establishes a compelling commercial case for advancing high-capacity sulfur cathode research from laboratory development toward industrial-scale implementation.
Beyond transportation, the proliferation of portable electronics continues to demand lighter, longer-lasting power sources. Consumer expectations for smartphones, laptops, and wearable devices increasingly center on extended battery life without compromising device form factors. Similarly, the aerospace and defense sectors require lightweight, high-energy solutions for unmanned aerial vehicles, satellites, and military applications where weight reduction directly translates to operational advantages.
Grid-scale energy storage represents another critical application domain. As renewable energy sources like solar and wind become more prevalent, the need for cost-effective, high-capacity storage systems intensifies to manage intermittency and ensure grid stability. Sulfur-based cathode materials, leveraging abundant and inexpensive raw materials, could significantly reduce the levelized cost of energy storage while improving system energy density.
The convergence of environmental regulations, consumer preferences, and technological advancement has created a market environment highly receptive to breakthrough battery technologies. Governments worldwide are implementing stricter emissions standards and offering incentives for clean energy adoption, further accelerating demand for superior energy storage solutions. This multifaceted market pressure establishes a compelling commercial case for advancing high-capacity sulfur cathode research from laboratory development toward industrial-scale implementation.
Current Status and Challenges of Sulfur Cathodes
Sulfur cathode materials have garnered significant attention in lithium-sulfur battery research due to their exceptional theoretical specific capacity of 1675 mAh/g and energy density of 2600 Wh/kg, which substantially exceed those of conventional lithium-ion cathode materials. Despite these promising attributes, the practical implementation of sulfur cathodes faces multiple critical challenges that have hindered their commercial viability. Current research efforts worldwide are intensively focused on addressing these fundamental obstacles while advancing material design and engineering strategies.
The primary technical challenge stems from the intrinsic insulating nature of sulfur, with electrical conductivity below 5×10⁻³⁰ S/cm at room temperature, severely limiting electron transport during electrochemical reactions. This poor conductivity necessitates the incorporation of conductive additives or host materials, which inevitably reduces the overall sulfur content and compromises the practical energy density of the cathode. Researchers have explored various carbon-based hosts, conductive polymers, and metal compounds to enhance electronic conductivity, yet achieving optimal balance between conductivity enhancement and sulfur loading remains problematic.
The polysulfide shuttle effect represents another major impediment to sulfur cathode performance. During discharge-charge cycles, intermediate lithium polysulfides (Li₂Sₓ, 4≤x≤8) dissolve readily in liquid electrolytes and migrate between electrodes, causing active material loss, self-discharge, and rapid capacity fade. This phenomenon typically results in capacity retention below 60% after 100 cycles in unoptimized systems. Current mitigation strategies include physical confinement through porous carbon structures, chemical adsorption using polar materials, and electrolyte modification, though complete suppression remains elusive.
Volume expansion during lithiation presents additional structural challenges. The conversion from sulfur to Li₂S involves approximately 80% volumetric change, inducing mechanical stress that leads to electrode pulverization, loss of electrical contact, and accelerated degradation. This expansion-contraction cycling also disrupts the solid electrolyte interphase formation, contributing to unstable electrochemical performance. Advanced binder systems and three-dimensional electrode architectures have shown promise in accommodating these volume changes, but scalable manufacturing solutions are still under development.
Furthermore, achieving high sulfur loading while maintaining acceptable rate capability and cycle stability presents a persistent challenge. Most laboratory demonstrations operate with sulfur loadings below 3 mg/cm², whereas practical applications require loadings exceeding 6 mg/cm² to compete with existing battery technologies. The trade-off between sulfur content, electrolyte quantity, and electrochemical performance continues to constrain the development of commercially viable high-capacity sulfur cathodes.
The primary technical challenge stems from the intrinsic insulating nature of sulfur, with electrical conductivity below 5×10⁻³⁰ S/cm at room temperature, severely limiting electron transport during electrochemical reactions. This poor conductivity necessitates the incorporation of conductive additives or host materials, which inevitably reduces the overall sulfur content and compromises the practical energy density of the cathode. Researchers have explored various carbon-based hosts, conductive polymers, and metal compounds to enhance electronic conductivity, yet achieving optimal balance between conductivity enhancement and sulfur loading remains problematic.
The polysulfide shuttle effect represents another major impediment to sulfur cathode performance. During discharge-charge cycles, intermediate lithium polysulfides (Li₂Sₓ, 4≤x≤8) dissolve readily in liquid electrolytes and migrate between electrodes, causing active material loss, self-discharge, and rapid capacity fade. This phenomenon typically results in capacity retention below 60% after 100 cycles in unoptimized systems. Current mitigation strategies include physical confinement through porous carbon structures, chemical adsorption using polar materials, and electrolyte modification, though complete suppression remains elusive.
Volume expansion during lithiation presents additional structural challenges. The conversion from sulfur to Li₂S involves approximately 80% volumetric change, inducing mechanical stress that leads to electrode pulverization, loss of electrical contact, and accelerated degradation. This expansion-contraction cycling also disrupts the solid electrolyte interphase formation, contributing to unstable electrochemical performance. Advanced binder systems and three-dimensional electrode architectures have shown promise in accommodating these volume changes, but scalable manufacturing solutions are still under development.
Furthermore, achieving high sulfur loading while maintaining acceptable rate capability and cycle stability presents a persistent challenge. Most laboratory demonstrations operate with sulfur loadings below 3 mg/cm², whereas practical applications require loadings exceeding 6 mg/cm² to compete with existing battery technologies. The trade-off between sulfur content, electrolyte quantity, and electrochemical performance continues to constrain the development of commercially viable high-capacity sulfur cathodes.
Mainstream Sulfur Cathode Design Solutions
01 Carbon-based composite materials for sulfur cathodes
Sulfur cathode materials can be combined with various carbon-based materials such as graphene, carbon nanotubes, or porous carbon to improve electrical conductivity and capacity retention. The carbon matrix provides a conductive framework that accommodates volume expansion during cycling and prevents polysulfide dissolution. These composite structures enhance the utilization of sulfur active material and improve the overall electrochemical performance of the cathode.- Carbon-based composite materials for sulfur cathodes: Sulfur cathode materials can be combined with various carbon-based materials such as graphene, carbon nanotubes, or porous carbon to improve electrical conductivity and capacity retention. The carbon matrix provides a conductive framework that accommodates volume expansion during cycling and prevents polysulfide dissolution. These composite structures enhance the utilization of sulfur active material and improve the overall electrochemical performance of the cathode.
- Metal oxide coatings and additives for capacity enhancement: Metal oxides can be incorporated into sulfur cathode materials to suppress polysulfide shuttling and improve capacity stability. These materials act as physical and chemical barriers that trap polysulfides within the cathode region. The metal oxide additives also provide additional active sites for sulfur immobilization, leading to enhanced capacity retention and cycling stability.
- Nanostructured sulfur cathode architectures: Designing sulfur cathodes with nanostructured architectures such as hollow spheres, core-shell structures, or hierarchical porous frameworks can significantly improve capacity performance. These nanostructures provide high surface area for sulfur loading, short ion diffusion pathways, and effective volume accommodation during charge-discharge cycles. The optimized morphology enhances sulfur utilization and maintains structural integrity during operation.
- Conductive polymer binders and interlayers: Utilizing conductive polymers as binders or interlayers in sulfur cathodes can improve both mechanical stability and electrical conductivity. These polymers facilitate electron transport throughout the cathode structure while providing flexible binding that accommodates volume changes. The polymer components also help to trap polysulfides through chemical interactions, thereby improving capacity retention and coulombic efficiency.
- Electrolyte optimization and separator modification: Modifying the electrolyte composition or incorporating functional separators can enhance sulfur cathode capacity by reducing polysulfide crossover and improving ion transport. Specialized electrolyte additives or modified separators with selective permeability can effectively confine polysulfides to the cathode side while maintaining high ionic conductivity. These approaches result in improved active material utilization and extended cycle life.
02 Metal oxide coatings and additives for capacity enhancement
Metal oxides can be incorporated into sulfur cathode materials to suppress polysulfide shuttling and improve capacity stability. These materials act as physical and chemical barriers that trap polysulfides within the cathode region. The metal oxide additives also provide additional active sites for sulfur immobilization, leading to enhanced capacity retention and cycling stability.Expand Specific Solutions03 Nanostructured sulfur cathode architectures
Designing sulfur cathodes with nanostructured architectures such as hollow spheres, core-shell structures, or hierarchical porous frameworks can significantly improve capacity performance. These nanostructures provide high surface area for sulfur loading, short ion diffusion pathways, and effective volume accommodation during charge-discharge cycles. The optimized morphology enhances sulfur utilization and maintains structural integrity during operation.Expand Specific Solutions04 Polymer binders and conductive additives optimization
The selection and optimization of polymer binders and conductive additives play a crucial role in improving sulfur cathode capacity. Advanced binders with strong polysulfide affinity and flexible mechanical properties help maintain electrode integrity and prevent active material loss. Conductive additives enhance electron transport throughout the cathode structure, leading to improved capacity utilization and rate performance.Expand Specific Solutions05 Electrolyte additives and interface modification
Modifying the cathode-electrolyte interface through specialized additives or surface treatments can enhance sulfur cathode capacity. These modifications help form stable solid electrolyte interphase layers that reduce polysulfide dissolution and shuttle effect. Interface engineering also improves lithium ion transport kinetics and reduces polarization, resulting in higher accessible capacity and better cycling performance.Expand Specific Solutions
Major Players in Sulfur Battery Industry
The high-capacity sulfur cathode materials sector represents an emerging yet rapidly maturing field within next-generation battery technology, currently transitioning from laboratory research to early commercialization stages. The market demonstrates significant growth potential driven by demand for higher energy density solutions in electric vehicles and energy storage systems. Technology maturity varies considerably across players, with established leaders like Sion Power Corp. and Honeycomb Battery Co. advancing commercial-scale Licerion® technology achieving 500 Wh/kg energy density, while companies such as Ionic Materials Inc. and Sionic Energy focus on electrolyte innovations to enhance sulfur cathode performance. Academic institutions including MIT, Central South University, and Drexel University contribute fundamental research breakthroughs, whereas industrial giants like Robert Bosch GmbH and Toyota Motor Co. integrate these materials into broader automotive applications, creating a competitive landscape characterized by strategic partnerships between material innovators and end-user manufacturers.
Nanotek Instruments, Inc.
Technical Solution: Nanotek Instruments specializes in graphene-enhanced sulfur cathode materials for lithium-sulfur batteries. Their technology utilizes functionalized graphene sheets as conductive hosts for sulfur, creating a three-dimensional porous network structure that achieves sulfur content of 60-75 wt% with initial specific capacity exceeding 1350 mAh/g. The graphene framework provides dual functionality: excellent electrical conductivity (>1000 S/m) and physical confinement of polysulfides through surface chemistry modifications. Their manufacturing process involves scalable solution-based methods for graphene-sulfur composite synthesis, incorporating heteroatom doping (nitrogen, oxygen) to enhance polysulfide adsorption. The company has developed cathode formulations maintaining over 80% capacity retention after 200 cycles at C/5 rate.
Strengths: Advanced graphene-based conductive framework provides superior electron transport, scalable production methods, strong intellectual property portfolio in carbon nanomaterial applications. Weaknesses: Graphene production costs remain relatively high, long-term cycling performance needs further improvement for commercial viability.
Sion Power Corp.
Technical Solution: Sion Power has developed advanced lithium-sulfur battery technology with high-capacity sulfur cathode materials featuring proprietary protective membrane systems. Their technology achieves sulfur cathode loading exceeding 4 mAh/cm² with specific capacity reaching 1200-1400 mAh/g. The company employs a unique encapsulation strategy to address polysulfide dissolution issues, utilizing specialized interlayers and electrolyte formulations to enhance cycling stability. Their cathode architecture incorporates conductive carbon frameworks with optimized sulfur distribution, enabling efficient electron transport while maintaining structural integrity during charge-discharge cycles. Sion Power's approach focuses on scalable manufacturing processes suitable for commercial production.
Strengths: Proven commercial-scale production capability, excellent cycling stability with proprietary membrane technology, high sulfur loading achieving superior energy density. Weaknesses: Complex manufacturing process increases production costs, limited public disclosure of detailed technical specifications.
Core Patents in High-Capacity Sulfur Cathodes
ZNP2 modified separator to improve the absorption and conversion kinetic of polysulfides for metal-sulphur batteries
PatentPendingIN202211018239A
Innovation
- A ZnP2 modified separator with surface anchoring groups is developed, synthesized using a vacuum sealed tube method, and applied as a coating composition with a carbon source and binder, enhancing polysulfide conversion kinetics and preventing ion shuttling between electrodes.
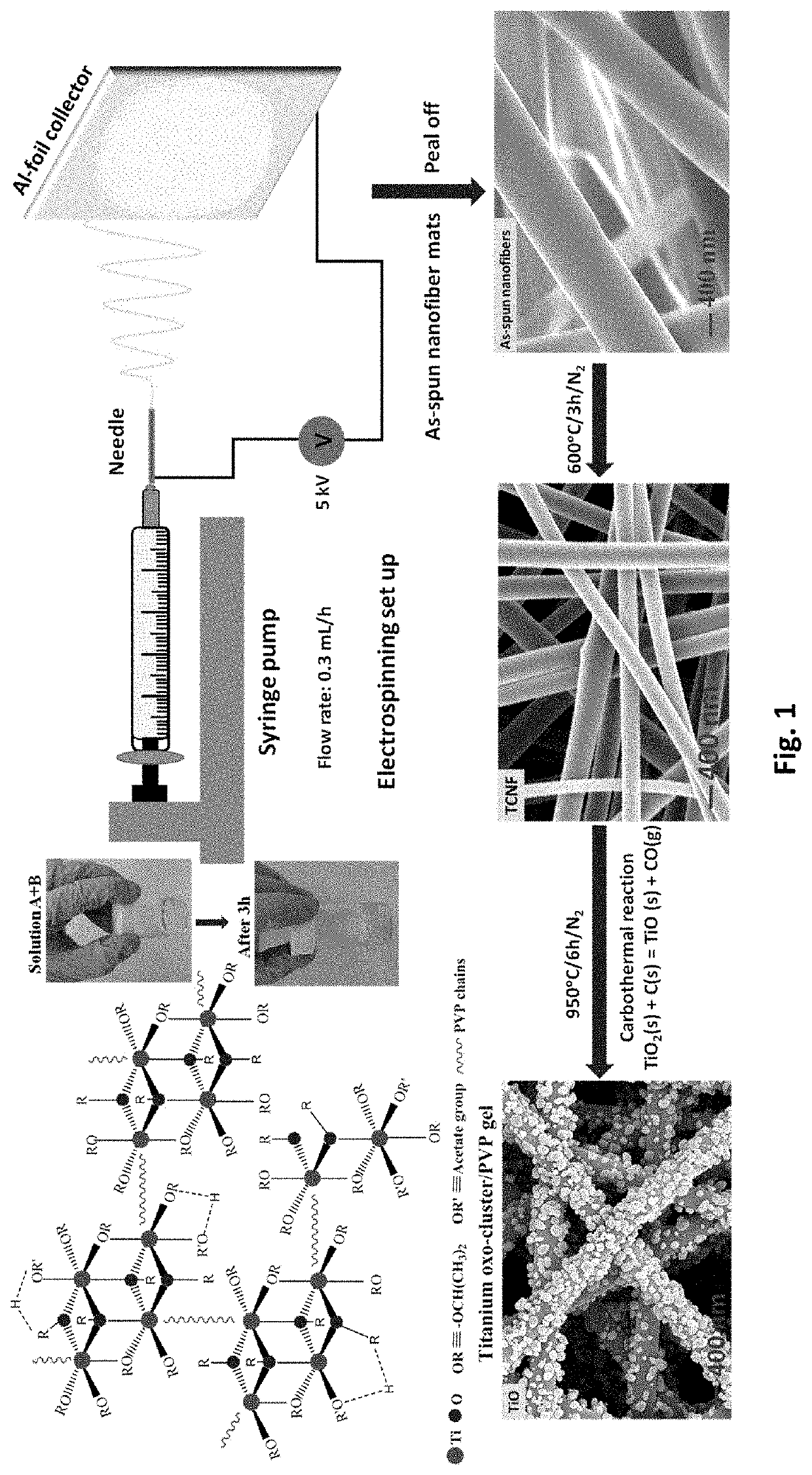
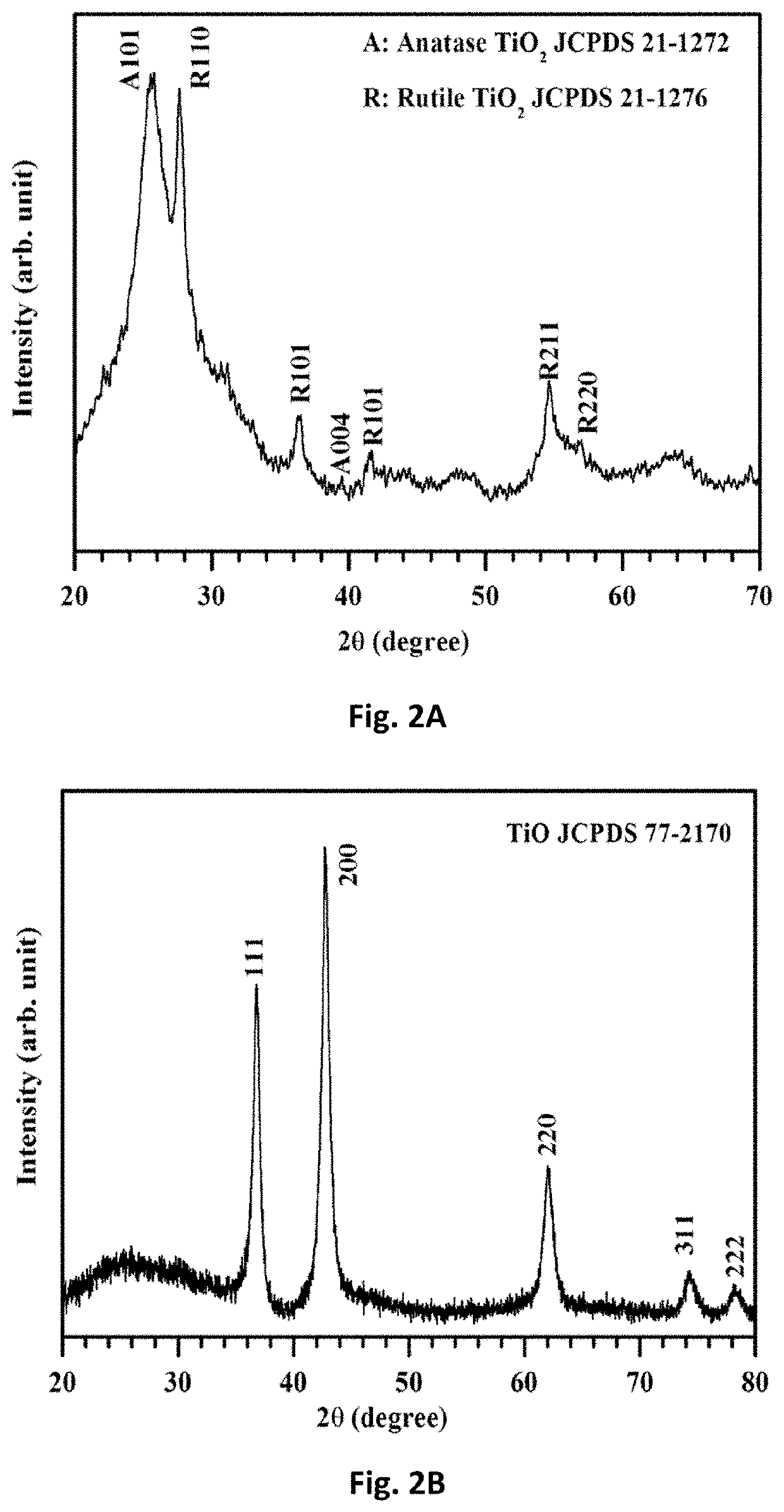
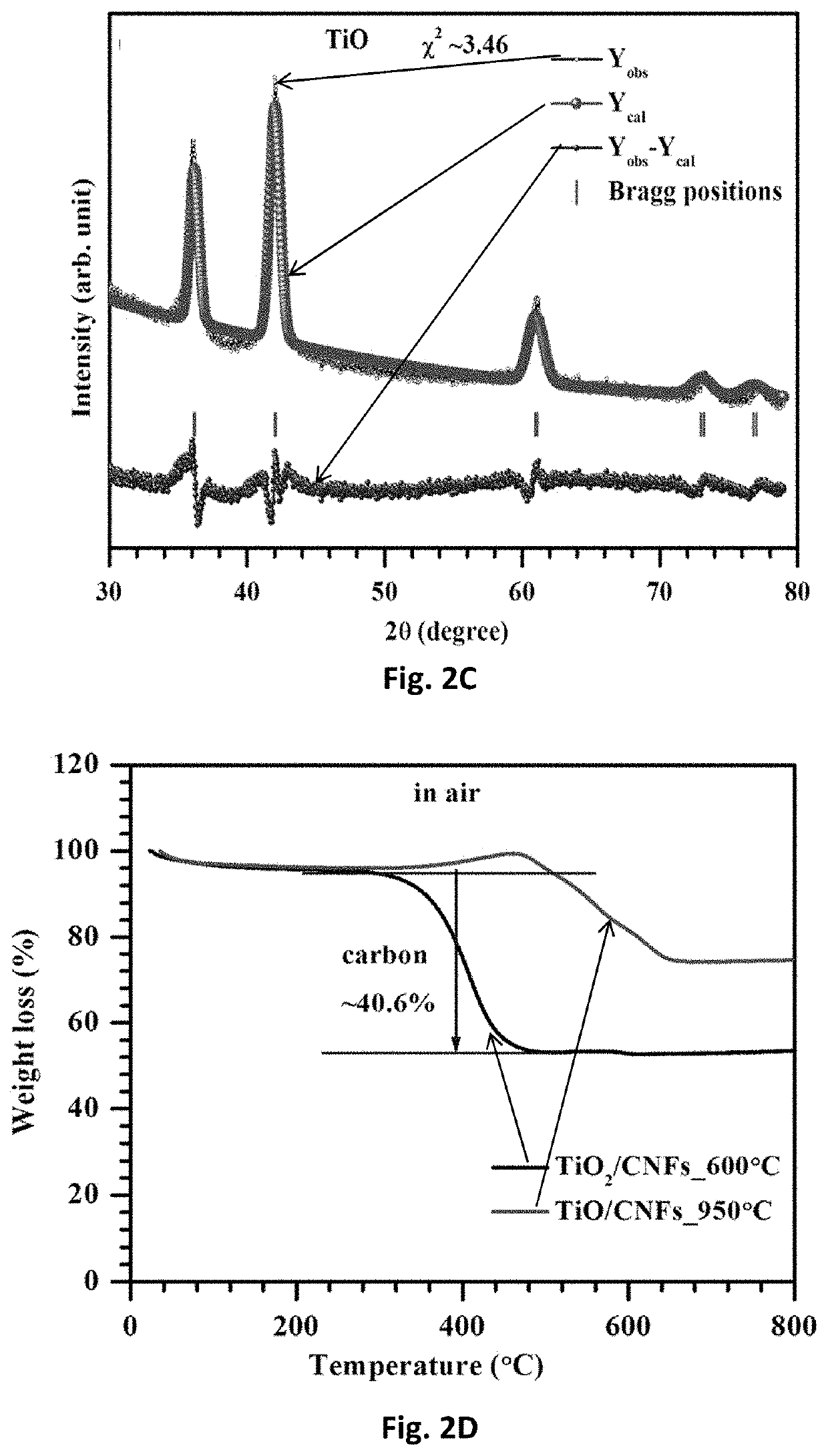
Free-standing, binder-free metal monoxide/suboxide nanofiber as cathodes or anodes for batteries
PatentActiveUS20210111390A1
Innovation
- The development of free-standing, binder-free nanofiber mats with high surface area and conductive metal oxide nanofibers, produced through electrospinning and carbothermal processes, which allow rapid sulfur diffusion and eliminate the need for harsh slurry casting, providing a robust 3D conducting network for uninterrupted electron supply and strong interactions with lithium polysulfides.
Safety Standards for Sulfur-Based Batteries
The development of high-capacity sulfur cathode materials has necessitated the establishment of comprehensive safety standards for sulfur-based batteries to ensure their reliable and secure deployment in commercial applications. Currently, the regulatory framework for these emerging energy storage systems remains fragmented, with various international organizations and national bodies working to address the unique safety challenges posed by lithium-sulfur and other sulfur-based battery chemistries. Unlike conventional lithium-ion batteries, sulfur cathode systems present distinct safety considerations including polysulfide dissolution, volume expansion during cycling, and potential thermal runaway mechanisms that differ fundamentally from traditional battery technologies.
The International Electrotechnical Commission (IEC) and Underwriters Laboratories (UL) have initiated preliminary efforts to extend existing battery safety standards to encompass sulfur-based systems, though dedicated specifications remain under development. Key safety parameters being evaluated include thermal stability thresholds, short-circuit resistance, mechanical abuse tolerance, and containment of sulfur-containing compounds during normal operation and failure modes. The Chinese national standard GB/T series and the United Nations Manual of Tests and Criteria for transport regulations are also being adapted to address sulfur battery-specific hazards.
Critical safety concerns center on the flammability of elemental sulfur and certain polysulfide intermediates, the corrosive nature of dissolved polysulfides to cell components, and the potential for hydrogen sulfide gas generation under abuse conditions. Testing protocols are being developed to assess cell-level and pack-level responses to overcharge, over-discharge, external heating, nail penetration, and crush scenarios specific to sulfur cathode architectures. Particular attention is directed toward evaluating the effectiveness of separator materials and electrolyte formulations in preventing internal short circuits and managing thermal events.
Manufacturing standards are emerging to control sulfur purity, cathode loading uniformity, and electrolyte composition consistency, as these factors significantly influence both performance and safety margins. Quality control measures for detecting micro-defects in sulfur cathode structures and ensuring proper sealing against moisture ingress are becoming standardized practices. As the technology matures toward commercialization, harmonized international safety standards will be essential for facilitating market acceptance and enabling cross-border trade of sulfur-based battery products.
The International Electrotechnical Commission (IEC) and Underwriters Laboratories (UL) have initiated preliminary efforts to extend existing battery safety standards to encompass sulfur-based systems, though dedicated specifications remain under development. Key safety parameters being evaluated include thermal stability thresholds, short-circuit resistance, mechanical abuse tolerance, and containment of sulfur-containing compounds during normal operation and failure modes. The Chinese national standard GB/T series and the United Nations Manual of Tests and Criteria for transport regulations are also being adapted to address sulfur battery-specific hazards.
Critical safety concerns center on the flammability of elemental sulfur and certain polysulfide intermediates, the corrosive nature of dissolved polysulfides to cell components, and the potential for hydrogen sulfide gas generation under abuse conditions. Testing protocols are being developed to assess cell-level and pack-level responses to overcharge, over-discharge, external heating, nail penetration, and crush scenarios specific to sulfur cathode architectures. Particular attention is directed toward evaluating the effectiveness of separator materials and electrolyte formulations in preventing internal short circuits and managing thermal events.
Manufacturing standards are emerging to control sulfur purity, cathode loading uniformity, and electrolyte composition consistency, as these factors significantly influence both performance and safety margins. Quality control measures for detecting micro-defects in sulfur cathode structures and ensuring proper sealing against moisture ingress are becoming standardized practices. As the technology matures toward commercialization, harmonized international safety standards will be essential for facilitating market acceptance and enabling cross-border trade of sulfur-based battery products.
Environmental Impact of Sulfur Cathode Production
The production of high-capacity sulfur cathode materials presents significant environmental considerations that must be addressed to ensure sustainable development of lithium-sulfur battery technology. While sulfur itself is abundant and relatively benign as a raw material, often sourced as a byproduct from petroleum refining and natural gas processing, the manufacturing processes and associated chemical treatments introduce various environmental concerns that warrant careful examination.
The synthesis of sulfur cathode materials typically involves energy-intensive processes including heating, grinding, and chemical modification steps. Carbon-sulfur composite fabrication, which is essential for improving conductivity and cycling stability, often requires high-temperature carbonization processes that consume substantial energy and may generate greenhouse gas emissions. Additionally, the use of organic solvents in slurry preparation and coating processes poses risks of volatile organic compound emissions if not properly managed through recovery systems.
Chemical additives and binders used in cathode production, such as polyvinylidene fluoride and various conductive agents, introduce additional environmental burdens. The manufacturing of these auxiliary materials involves complex chemical synthesis routes with their own environmental footprints. Furthermore, waste streams generated during production, including off-specification materials and processing residues, require appropriate treatment and disposal protocols to prevent environmental contamination.
Water consumption represents another environmental consideration, particularly in washing and purification steps required to remove impurities from sulfur materials. Wastewater treatment becomes critical when dealing with sulfur-containing effluents that could lead to acidification if released untreated. The potential formation of hydrogen sulfide during certain processing conditions also necessitates proper ventilation and gas treatment systems to protect both workers and the surrounding environment.
Compared to conventional cathode materials like lithium cobalt oxide or nickel-manganese-cobalt compounds, sulfur cathodes offer inherent environmental advantages due to the non-toxic nature of sulfur and elimination of heavy metal mining requirements. However, realizing these benefits requires implementing clean production technologies, optimizing energy efficiency, establishing closed-loop solvent recovery systems, and developing comprehensive waste management strategies throughout the manufacturing lifecycle.
The synthesis of sulfur cathode materials typically involves energy-intensive processes including heating, grinding, and chemical modification steps. Carbon-sulfur composite fabrication, which is essential for improving conductivity and cycling stability, often requires high-temperature carbonization processes that consume substantial energy and may generate greenhouse gas emissions. Additionally, the use of organic solvents in slurry preparation and coating processes poses risks of volatile organic compound emissions if not properly managed through recovery systems.
Chemical additives and binders used in cathode production, such as polyvinylidene fluoride and various conductive agents, introduce additional environmental burdens. The manufacturing of these auxiliary materials involves complex chemical synthesis routes with their own environmental footprints. Furthermore, waste streams generated during production, including off-specification materials and processing residues, require appropriate treatment and disposal protocols to prevent environmental contamination.
Water consumption represents another environmental consideration, particularly in washing and purification steps required to remove impurities from sulfur materials. Wastewater treatment becomes critical when dealing with sulfur-containing effluents that could lead to acidification if released untreated. The potential formation of hydrogen sulfide during certain processing conditions also necessitates proper ventilation and gas treatment systems to protect both workers and the surrounding environment.
Compared to conventional cathode materials like lithium cobalt oxide or nickel-manganese-cobalt compounds, sulfur cathodes offer inherent environmental advantages due to the non-toxic nature of sulfur and elimination of heavy metal mining requirements. However, realizing these benefits requires implementing clean production technologies, optimizing energy efficiency, establishing closed-loop solvent recovery systems, and developing comprehensive waste management strategies throughout the manufacturing lifecycle.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!