Simulation-Driven Design vs Biomechanical Modeling: Precision
MAR 6, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Simulation-Driven Design and Biomechanical Modeling Background
Simulation-driven design emerged in the 1960s alongside the development of finite element analysis (FEA) and computational fluid dynamics (CFD). Initially confined to aerospace and automotive industries, this approach leveraged mathematical models and computational algorithms to predict product behavior under various conditions before physical prototyping. The methodology gained momentum with advances in computing power, enabling engineers to iterate designs rapidly while reducing development costs and time-to-market.
Biomechanical modeling has deeper historical roots, tracing back to Leonardo da Vinci's anatomical studies and Galileo's mechanical analysis of bone structures in the 17th century. Modern biomechanical modeling began in the 1970s with the application of engineering principles to biological systems. This field combines mechanical engineering, biology, and medicine to understand how living tissues respond to forces, loads, and environmental conditions.
The convergence of these two disciplines occurred in the 1990s when computational capabilities reached sufficient sophistication to handle complex biological geometries and material properties. This intersection created new possibilities for medical device design, prosthetics development, and surgical planning. The precision challenge emerged as practitioners recognized that while simulation-driven design excelled in controlled engineering environments, biological systems presented unique complexities requiring specialized modeling approaches.
The evolution toward precision-focused methodologies reflects growing demands in personalized medicine and patient-specific treatments. Traditional simulation-driven design assumes standardized material properties and loading conditions, whereas biomechanical modeling must account for individual anatomical variations, tissue heterogeneity, and dynamic biological responses. This fundamental difference has driven the development of hybrid approaches that combine the computational efficiency of simulation-driven design with the biological accuracy of biomechanical modeling.
Contemporary precision requirements have intensified due to regulatory demands, patient safety considerations, and the push toward minimally invasive procedures. The integration of artificial intelligence, machine learning, and advanced imaging technologies has further expanded the potential for achieving unprecedented precision levels in both simulation-driven design and biomechanical modeling applications.
Biomechanical modeling has deeper historical roots, tracing back to Leonardo da Vinci's anatomical studies and Galileo's mechanical analysis of bone structures in the 17th century. Modern biomechanical modeling began in the 1970s with the application of engineering principles to biological systems. This field combines mechanical engineering, biology, and medicine to understand how living tissues respond to forces, loads, and environmental conditions.
The convergence of these two disciplines occurred in the 1990s when computational capabilities reached sufficient sophistication to handle complex biological geometries and material properties. This intersection created new possibilities for medical device design, prosthetics development, and surgical planning. The precision challenge emerged as practitioners recognized that while simulation-driven design excelled in controlled engineering environments, biological systems presented unique complexities requiring specialized modeling approaches.
The evolution toward precision-focused methodologies reflects growing demands in personalized medicine and patient-specific treatments. Traditional simulation-driven design assumes standardized material properties and loading conditions, whereas biomechanical modeling must account for individual anatomical variations, tissue heterogeneity, and dynamic biological responses. This fundamental difference has driven the development of hybrid approaches that combine the computational efficiency of simulation-driven design with the biological accuracy of biomechanical modeling.
Contemporary precision requirements have intensified due to regulatory demands, patient safety considerations, and the push toward minimally invasive procedures. The integration of artificial intelligence, machine learning, and advanced imaging technologies has further expanded the potential for achieving unprecedented precision levels in both simulation-driven design and biomechanical modeling applications.
Market Demand for Precision Simulation Technologies
The global market for precision simulation technologies is experiencing unprecedented growth driven by increasing demands for accuracy and reliability across multiple industries. Healthcare sectors, particularly medical device development and surgical planning, represent the largest consumer segment for biomechanical modeling solutions. The pharmaceutical industry increasingly relies on precision simulation for drug delivery systems and tissue engineering applications, where traditional trial-and-error approaches prove both costly and time-consuming.
Automotive and aerospace industries constitute another major demand driver, where simulation-driven design methodologies have become essential for safety-critical applications. The need for precise biomechanical modeling in crash test simulations and ergonomic assessments has intensified as regulatory requirements become more stringent. These sectors require simulation tools capable of accurately predicting human body responses under various stress conditions.
The sports and fitness industry has emerged as a significant growth area, with increasing demand for precision simulation in equipment design and performance optimization. Professional sports organizations and equipment manufacturers seek advanced biomechanical modeling capabilities to enhance athlete performance while minimizing injury risks. This trend extends to consumer fitness applications, where wearable technology integration demands sophisticated simulation algorithms.
Manufacturing sectors, particularly those involving human-machine interaction, demonstrate growing appetite for precision simulation technologies. Ergonomic workplace design and industrial safety assessments require accurate biomechanical models to optimize worker productivity and reduce occupational hazards. The integration of artificial intelligence with traditional simulation approaches has created new market opportunities in predictive maintenance and adaptive system design.
Academic and research institutions represent a stable demand base, driving innovation in fundamental simulation methodologies. Government funding for biomedical research and defense applications continues to support market expansion, particularly in areas requiring high-fidelity human performance modeling.
The convergence of virtual reality, augmented reality, and precision simulation technologies has opened new market segments in training and education. Medical training simulators, military applications, and industrial training programs increasingly demand realistic biomechanical modeling capabilities that can provide immediate feedback and assessment.
Market growth is further accelerated by the increasing availability of high-performance computing resources and cloud-based simulation platforms, making precision simulation technologies more accessible to smaller organizations and emerging markets.
Automotive and aerospace industries constitute another major demand driver, where simulation-driven design methodologies have become essential for safety-critical applications. The need for precise biomechanical modeling in crash test simulations and ergonomic assessments has intensified as regulatory requirements become more stringent. These sectors require simulation tools capable of accurately predicting human body responses under various stress conditions.
The sports and fitness industry has emerged as a significant growth area, with increasing demand for precision simulation in equipment design and performance optimization. Professional sports organizations and equipment manufacturers seek advanced biomechanical modeling capabilities to enhance athlete performance while minimizing injury risks. This trend extends to consumer fitness applications, where wearable technology integration demands sophisticated simulation algorithms.
Manufacturing sectors, particularly those involving human-machine interaction, demonstrate growing appetite for precision simulation technologies. Ergonomic workplace design and industrial safety assessments require accurate biomechanical models to optimize worker productivity and reduce occupational hazards. The integration of artificial intelligence with traditional simulation approaches has created new market opportunities in predictive maintenance and adaptive system design.
Academic and research institutions represent a stable demand base, driving innovation in fundamental simulation methodologies. Government funding for biomedical research and defense applications continues to support market expansion, particularly in areas requiring high-fidelity human performance modeling.
The convergence of virtual reality, augmented reality, and precision simulation technologies has opened new market segments in training and education. Medical training simulators, military applications, and industrial training programs increasingly demand realistic biomechanical modeling capabilities that can provide immediate feedback and assessment.
Market growth is further accelerated by the increasing availability of high-performance computing resources and cloud-based simulation platforms, making precision simulation technologies more accessible to smaller organizations and emerging markets.
Current State of Simulation vs Biomechanical Modeling Accuracy
The current landscape of simulation-driven design and biomechanical modeling presents a complex picture of varying accuracy levels across different application domains. Traditional finite element analysis (FEA) methods in simulation-driven design have achieved remarkable precision in structural mechanics, with error rates typically below 5% for linear elastic problems. However, when applied to biological systems, these conventional approaches often struggle with the inherent nonlinearity and multi-scale nature of living tissues.
Biomechanical modeling has evolved significantly over the past decade, with computational frameworks now capable of incorporating patient-specific anatomical data through advanced imaging techniques. Current state-of-the-art biomechanical models can achieve accuracy levels of 85-95% in predicting tissue deformation under controlled conditions. Nevertheless, this accuracy drops considerably when dealing with dynamic loading scenarios or long-term biological responses, where prediction errors can exceed 20-30%.
The integration of machine learning algorithms with traditional modeling approaches has emerged as a promising solution to bridge accuracy gaps. Hybrid models combining physics-based simulations with data-driven corrections have demonstrated improved precision, particularly in cardiovascular and orthopedic applications. Recent studies indicate that these hybrid approaches can reduce prediction errors by 15-25% compared to purely physics-based models.
Multi-scale modeling represents another frontier where accuracy challenges persist. While molecular dynamics simulations can provide atomic-level precision, scaling up to tissue and organ levels introduces cumulative errors that significantly impact overall model fidelity. Current multi-scale frameworks typically maintain acceptable accuracy only within specific scale ranges, with cross-scale validation remaining a critical bottleneck.
Real-time simulation requirements further complicate accuracy considerations. Many clinical applications demand computational speeds that necessitate model simplifications, creating an inherent trade-off between precision and computational efficiency. Current real-time biomechanical models typically operate at 70-80% accuracy compared to their offline counterparts, highlighting the ongoing challenge of balancing speed and precision in practical implementations.
Biomechanical modeling has evolved significantly over the past decade, with computational frameworks now capable of incorporating patient-specific anatomical data through advanced imaging techniques. Current state-of-the-art biomechanical models can achieve accuracy levels of 85-95% in predicting tissue deformation under controlled conditions. Nevertheless, this accuracy drops considerably when dealing with dynamic loading scenarios or long-term biological responses, where prediction errors can exceed 20-30%.
The integration of machine learning algorithms with traditional modeling approaches has emerged as a promising solution to bridge accuracy gaps. Hybrid models combining physics-based simulations with data-driven corrections have demonstrated improved precision, particularly in cardiovascular and orthopedic applications. Recent studies indicate that these hybrid approaches can reduce prediction errors by 15-25% compared to purely physics-based models.
Multi-scale modeling represents another frontier where accuracy challenges persist. While molecular dynamics simulations can provide atomic-level precision, scaling up to tissue and organ levels introduces cumulative errors that significantly impact overall model fidelity. Current multi-scale frameworks typically maintain acceptable accuracy only within specific scale ranges, with cross-scale validation remaining a critical bottleneck.
Real-time simulation requirements further complicate accuracy considerations. Many clinical applications demand computational speeds that necessitate model simplifications, creating an inherent trade-off between precision and computational efficiency. Current real-time biomechanical models typically operate at 70-80% accuracy compared to their offline counterparts, highlighting the ongoing challenge of balancing speed and precision in practical implementations.
Existing Precision Enhancement Solutions in Modeling
01 Finite Element Analysis for Biomechanical Simulation
Finite element analysis (FEA) methods are employed to create detailed biomechanical models that simulate the mechanical behavior of biological structures under various loading conditions. These computational techniques enable precise prediction of stress distribution, deformation patterns, and structural responses in complex anatomical geometries. The simulation-driven approach allows for iterative design optimization before physical prototyping, significantly reducing development time and costs while improving the accuracy of biomechanical predictions.- Finite Element Analysis for Biomechanical Simulation: Finite element analysis (FEA) methods are employed to create detailed biomechanical models that simulate the mechanical behavior of biological structures under various loading conditions. These computational techniques enable precise prediction of stress distribution, deformation patterns, and structural responses in complex anatomical geometries. The simulation-driven approach allows for iterative design optimization before physical prototyping, significantly improving the accuracy of biomechanical predictions and reducing development time and costs.
- Patient-Specific Modeling Using Medical Imaging Data: Advanced biomechanical modeling techniques incorporate patient-specific anatomical data derived from medical imaging modalities such as CT, MRI, or ultrasound scans. These personalized models account for individual variations in geometry, material properties, and boundary conditions, enabling highly accurate simulations tailored to specific patients. The integration of imaging data with computational modeling frameworks enhances the precision of biomechanical analysis and supports personalized treatment planning and device design.
- Multi-Scale Modeling for Hierarchical Biomechanical Systems: Multi-scale modeling approaches integrate biomechanical phenomena across different spatial and temporal scales, from molecular and cellular levels to tissue and organ systems. These hierarchical models capture the complex interactions between microscopic material properties and macroscopic mechanical behavior, providing comprehensive insights into biomechanical performance. The simulation framework enables the analysis of how changes at one scale propagate through the system, improving the overall precision of biomechanical predictions.
- Real-Time Simulation and Interactive Design Optimization: Real-time computational methods enable interactive biomechanical simulations that provide immediate feedback during the design process. These systems utilize efficient numerical algorithms and parallel computing architectures to achieve rapid simulation speeds while maintaining modeling accuracy. The interactive nature of these tools allows designers to explore multiple design variations quickly, evaluate performance metrics in real-time, and make informed decisions that optimize biomechanical characteristics.
- Validation and Calibration Through Experimental Data Integration: Biomechanical modeling precision is enhanced through systematic validation and calibration processes that integrate experimental measurements with computational predictions. These methods involve comparing simulation results against physical testing data, adjusting model parameters to minimize discrepancies, and establishing confidence intervals for predictions. The iterative refinement process ensures that simulation-driven designs accurately reflect real-world biomechanical behavior and meet specified performance criteria.
02 Patient-Specific Modeling Using Medical Imaging Data
Advanced biomechanical modeling techniques utilize patient-specific anatomical data derived from medical imaging modalities to create personalized computational models. These models incorporate individual variations in geometry, material properties, and boundary conditions to enhance simulation accuracy. The integration of imaging data with computational frameworks enables precise representation of anatomical structures and their mechanical characteristics, leading to more reliable predictions for clinical applications and device design.Expand Specific Solutions03 Multi-Scale Biomechanical Modeling Approaches
Multi-scale modeling methodologies integrate biomechanical phenomena across different spatial and temporal scales, from cellular and tissue levels to organ and system levels. These hierarchical modeling frameworks capture the complex interactions between microscopic material properties and macroscopic mechanical responses. The multi-scale approach enhances the precision of biomechanical simulations by accounting for the heterogeneous nature of biological tissues and their scale-dependent mechanical behaviors.Expand Specific Solutions04 Real-Time Simulation and Interactive Design Optimization
Real-time computational methods enable interactive biomechanical simulations that provide immediate feedback during the design process. These techniques employ efficient numerical algorithms and parallel computing strategies to achieve rapid simulation results without compromising accuracy. The interactive nature of these systems allows designers to explore multiple design variations quickly, facilitating data-driven decision-making and accelerating the development of biomechanical devices and implants.Expand Specific Solutions05 Validation and Verification of Biomechanical Models
Comprehensive validation and verification protocols are essential for ensuring the accuracy and reliability of simulation-driven biomechanical models. These methodologies involve comparing computational predictions with experimental measurements, conducting sensitivity analyses, and assessing model convergence. Rigorous validation processes establish confidence in the predictive capabilities of biomechanical simulations and ensure that the models accurately represent the physical phenomena they are intended to simulate.Expand Specific Solutions
Key Players in Simulation Software and Biomechanical Tools
The simulation-driven design versus biomechanical modeling precision landscape represents a mature technological domain experiencing significant growth across multiple sectors. The market demonstrates substantial scale, driven by increasing demand for precision engineering in automotive, aerospace, medical devices, and manufacturing industries. Leading technology companies including Siemens AG, Bentley Systems, and FANUC Corp. have established sophisticated simulation platforms, while academic institutions like MIT, Jilin University, and Huazhong University of Science & Technology contribute fundamental research advances. The competitive environment shows high technological maturity, with established players like Boeing, Sony Group, and NEC Corp. integrating advanced biomechanical modeling capabilities into their product development cycles. Chinese institutions and companies, including China Automotive Engineering Research Institute and Beijing Institute of Technology, are rapidly advancing in this space, creating a globally distributed innovation ecosystem that spans from fundamental research to commercial applications.
Massachusetts Institute of Technology
Technical Solution: MIT has pioneered advanced biomechanical modeling techniques that combine computational mechanics with biological system analysis. Their research focuses on developing high-precision finite element models for human tissue mechanics, prosthetic design, and biomedical device optimization. The institute's approach integrates machine learning algorithms with traditional biomechanical modeling to improve prediction accuracy for complex biological systems. Their methodologies incorporate patient-specific anatomical data from medical imaging to create personalized biomechanical models, achieving remarkable precision in predicting tissue behavior and device performance in clinical applications.
Strengths: Cutting-edge research capabilities, strong academic-industry partnerships, innovative AI integration approaches. Weaknesses: Limited commercial scalability, research-focused rather than production-ready solutions.
Bentley Systems, Inc.
Technical Solution: Bentley Systems develops comprehensive simulation-driven design solutions for infrastructure and construction projects, integrating structural analysis with environmental modeling. Their MicroStation and STAAD platforms combine traditional engineering simulation with biomechanical principles for human-structure interaction analysis. The company's approach emphasizes digital twin technology that continuously updates simulation models with real-world sensor data, enabling precise prediction of structural behavior and human comfort factors. Their solutions incorporate advanced algorithms for modeling complex interactions between built environments and human occupants, achieving high precision in predicting structural performance and occupant safety under various loading conditions.
Strengths: Strong infrastructure focus, digital twin integration, comprehensive project lifecycle support. Weaknesses: Less specialized in pure biomechanical applications, primarily focused on civil engineering domains.
Core Algorithms for High-Precision Biomechanical Simulation
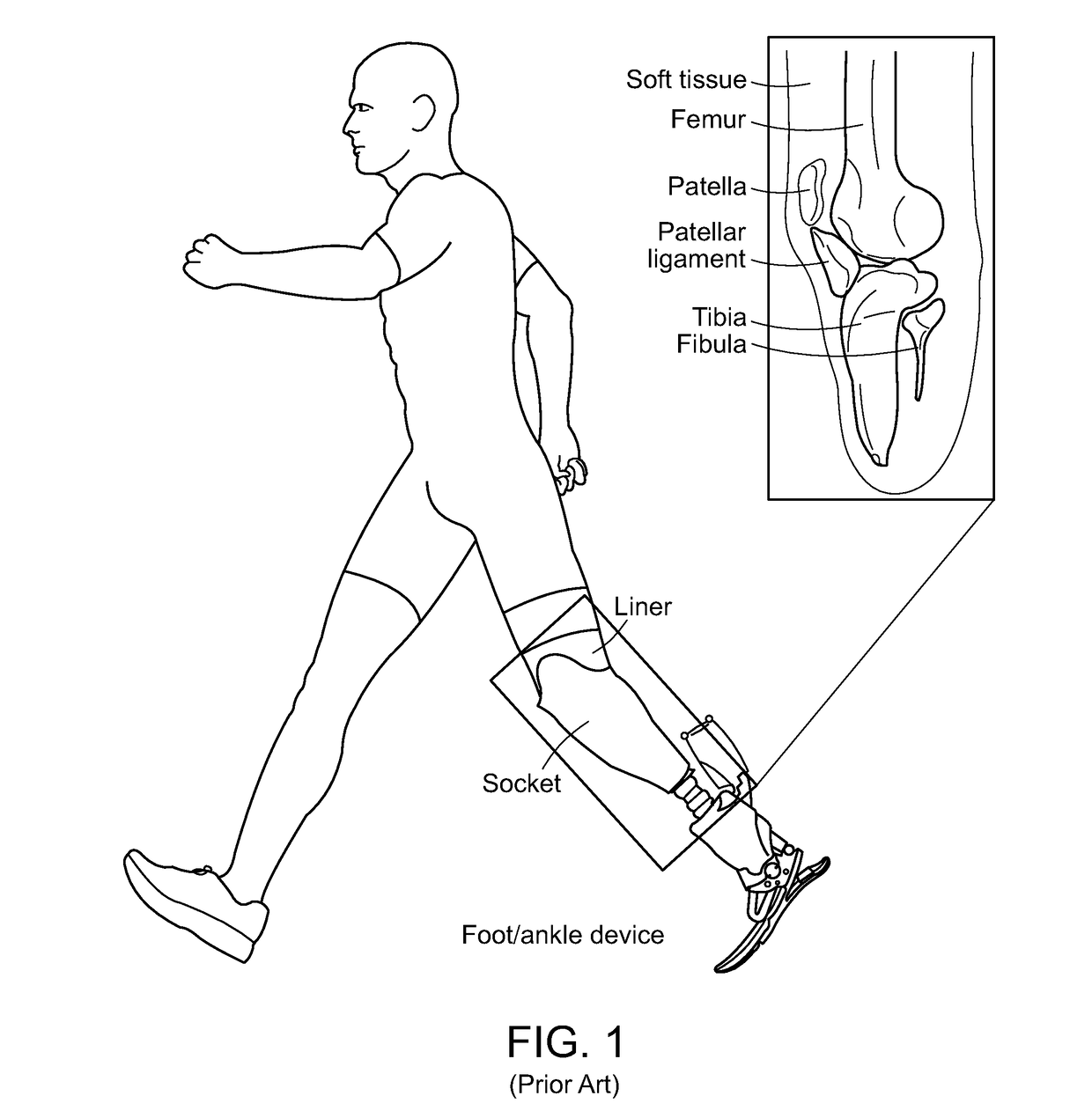
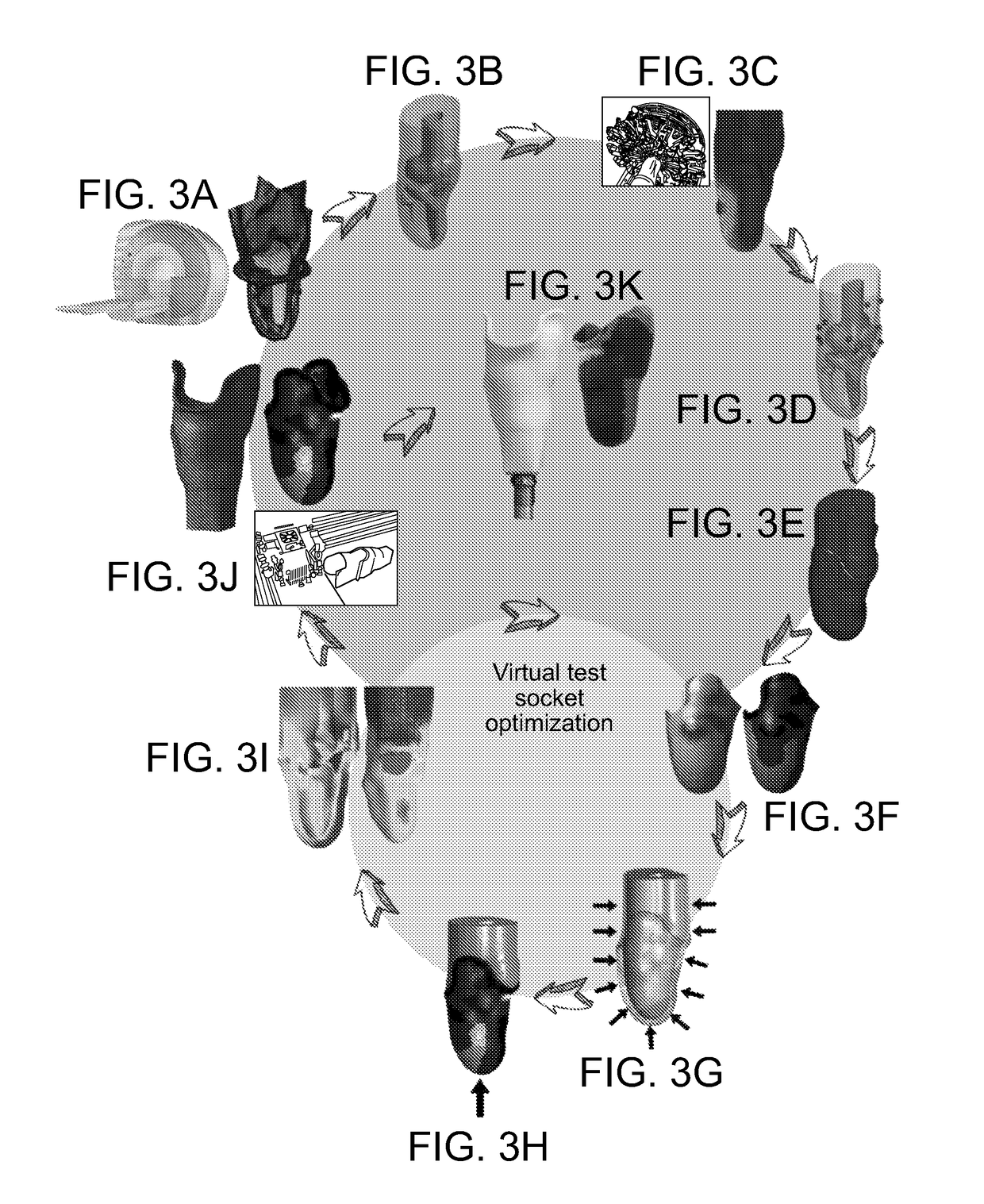
Method And System For Designing A Biomechanical Interface Contacting A Biological Body Segment
PatentPendingUS20190021880A1
Innovation
- A data-driven, automated computational framework that uses non-invasive imaging and finite element analysis (FEA) to create subject-specific biomechanical interfaces, accounting for tissue geometry and material properties to optimize socket design and reduce user involvement through iterative virtual prototyping.
Biomechanical model for predicting deformations of a patient body
PatentPendingUS20240225738A1
Innovation
- A computer-implemented method that generates a pre-calculated, parametrized biomechanical model through forward simulations, allowing for rapid optimization and alignment of pre-operative data with intra-operative data using multivariate interpolation or mode decomposition, enabling fast and accurate deformation prediction even with sparse data.
Validation Standards for Computational Modeling Precision
The establishment of robust validation standards for computational modeling precision represents a critical foundation for advancing both simulation-driven design and biomechanical modeling methodologies. Current validation frameworks must address the inherent complexity of biological systems while maintaining the rigor required for engineering applications. These standards serve as the bridge between theoretical computational models and their practical implementation in medical device development, surgical planning, and therapeutic interventions.
Verification and validation protocols form the cornerstone of computational modeling precision assessment. Verification ensures that mathematical models are correctly implemented in software, while validation confirms that these models accurately represent real-world biological phenomena. The distinction becomes particularly crucial when comparing simulation-driven design approaches, which often prioritize computational efficiency and parametric optimization, against biomechanical modeling methods that emphasize physiological accuracy and biological fidelity.
Quantitative metrics for model accuracy assessment include statistical measures such as root mean square error, correlation coefficients, and confidence intervals. These metrics must be contextualized within the specific application domain, as acceptable precision levels vary significantly between different biomedical applications. For instance, cardiovascular flow simulations may require different accuracy thresholds compared to orthopedic implant stress analysis or neural tissue modeling.
Experimental validation benchmarks establish reference standards through controlled laboratory studies, clinical data collection, and standardized test protocols. These benchmarks provide ground truth data against which computational predictions can be evaluated. The development of standardized datasets and validation cases enables consistent comparison across different modeling approaches and research groups, fostering reproducibility and scientific rigor.
Regulatory compliance considerations increasingly influence validation standard development, particularly for medical applications. Organizations such as the FDA and ISO have begun establishing guidelines for computational modeling validation in medical device approval processes. These regulatory frameworks emphasize the need for comprehensive documentation of model assumptions, limitations, and validation evidence.
Cross-platform validation protocols ensure that modeling precision standards remain consistent across different software implementations and computational environments. This standardization becomes essential as the field moves toward integrated workflows combining multiple modeling tools and methodologies, requiring seamless data exchange and consistent accuracy assessment across diverse computational platforms.
Verification and validation protocols form the cornerstone of computational modeling precision assessment. Verification ensures that mathematical models are correctly implemented in software, while validation confirms that these models accurately represent real-world biological phenomena. The distinction becomes particularly crucial when comparing simulation-driven design approaches, which often prioritize computational efficiency and parametric optimization, against biomechanical modeling methods that emphasize physiological accuracy and biological fidelity.
Quantitative metrics for model accuracy assessment include statistical measures such as root mean square error, correlation coefficients, and confidence intervals. These metrics must be contextualized within the specific application domain, as acceptable precision levels vary significantly between different biomedical applications. For instance, cardiovascular flow simulations may require different accuracy thresholds compared to orthopedic implant stress analysis or neural tissue modeling.
Experimental validation benchmarks establish reference standards through controlled laboratory studies, clinical data collection, and standardized test protocols. These benchmarks provide ground truth data against which computational predictions can be evaluated. The development of standardized datasets and validation cases enables consistent comparison across different modeling approaches and research groups, fostering reproducibility and scientific rigor.
Regulatory compliance considerations increasingly influence validation standard development, particularly for medical applications. Organizations such as the FDA and ISO have begun establishing guidelines for computational modeling validation in medical device approval processes. These regulatory frameworks emphasize the need for comprehensive documentation of model assumptions, limitations, and validation evidence.
Cross-platform validation protocols ensure that modeling precision standards remain consistent across different software implementations and computational environments. This standardization becomes essential as the field moves toward integrated workflows combining multiple modeling tools and methodologies, requiring seamless data exchange and consistent accuracy assessment across diverse computational platforms.
Computational Resource Requirements and Optimization
The computational demands of simulation-driven design and biomechanical modeling present distinct resource allocation challenges that significantly impact precision outcomes. Simulation-driven design typically requires substantial parallel processing capabilities, with computational loads scaling exponentially as model complexity increases. Modern finite element analysis workflows demand high-performance computing clusters with distributed memory architectures, often requiring 64-256 CPU cores for complex geometries. Memory requirements frequently exceed 128GB for detailed structural simulations, while storage needs can reach terabytes for comprehensive design optimization cycles.
Biomechanical modeling exhibits different computational characteristics, with resource requirements varying dramatically based on modeling approach. Continuum mechanics models demand intensive matrix operations and iterative solvers, requiring specialized numerical libraries and optimized linear algebra routines. Multi-scale modeling approaches, incorporating cellular and molecular interactions, necessitate hybrid computing architectures combining CPU and GPU resources. Real-time biomechanical simulations for clinical applications require careful optimization to maintain computational efficiency while preserving model fidelity.
Resource optimization strategies differ fundamentally between these approaches. Simulation-driven design benefits from adaptive mesh refinement techniques and parallel domain decomposition methods, enabling efficient utilization of distributed computing resources. Model order reduction techniques can decrease computational complexity by 2-3 orders of magnitude while maintaining acceptable accuracy levels. Cloud-based computing platforms offer scalable solutions, allowing dynamic resource allocation based on simulation complexity and timeline requirements.
Biomechanical modeling optimization focuses on algorithmic efficiency and problem-specific acceleration techniques. GPU computing provides significant advantages for particle-based simulations and molecular dynamics calculations, achieving 10-50x speedup over traditional CPU implementations. Machine learning-assisted surrogate modeling emerges as a promising optimization approach, reducing computational overhead while maintaining predictive accuracy for specific biomechanical phenomena.
The precision-performance trade-off requires careful consideration of computational budget constraints. High-fidelity simulations demand substantial resources but provide enhanced accuracy for critical design decisions. Conversely, reduced-order models enable rapid iteration cycles essential for design optimization workflows. Effective resource management strategies must balance computational cost against precision requirements, considering both immediate simulation needs and long-term research objectives.
Biomechanical modeling exhibits different computational characteristics, with resource requirements varying dramatically based on modeling approach. Continuum mechanics models demand intensive matrix operations and iterative solvers, requiring specialized numerical libraries and optimized linear algebra routines. Multi-scale modeling approaches, incorporating cellular and molecular interactions, necessitate hybrid computing architectures combining CPU and GPU resources. Real-time biomechanical simulations for clinical applications require careful optimization to maintain computational efficiency while preserving model fidelity.
Resource optimization strategies differ fundamentally between these approaches. Simulation-driven design benefits from adaptive mesh refinement techniques and parallel domain decomposition methods, enabling efficient utilization of distributed computing resources. Model order reduction techniques can decrease computational complexity by 2-3 orders of magnitude while maintaining acceptable accuracy levels. Cloud-based computing platforms offer scalable solutions, allowing dynamic resource allocation based on simulation complexity and timeline requirements.
Biomechanical modeling optimization focuses on algorithmic efficiency and problem-specific acceleration techniques. GPU computing provides significant advantages for particle-based simulations and molecular dynamics calculations, achieving 10-50x speedup over traditional CPU implementations. Machine learning-assisted surrogate modeling emerges as a promising optimization approach, reducing computational overhead while maintaining predictive accuracy for specific biomechanical phenomena.
The precision-performance trade-off requires careful consideration of computational budget constraints. High-fidelity simulations demand substantial resources but provide enhanced accuracy for critical design decisions. Conversely, reduced-order models enable rapid iteration cycles essential for design optimization workflows. Effective resource management strategies must balance computational cost against precision requirements, considering both immediate simulation needs and long-term research objectives.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!