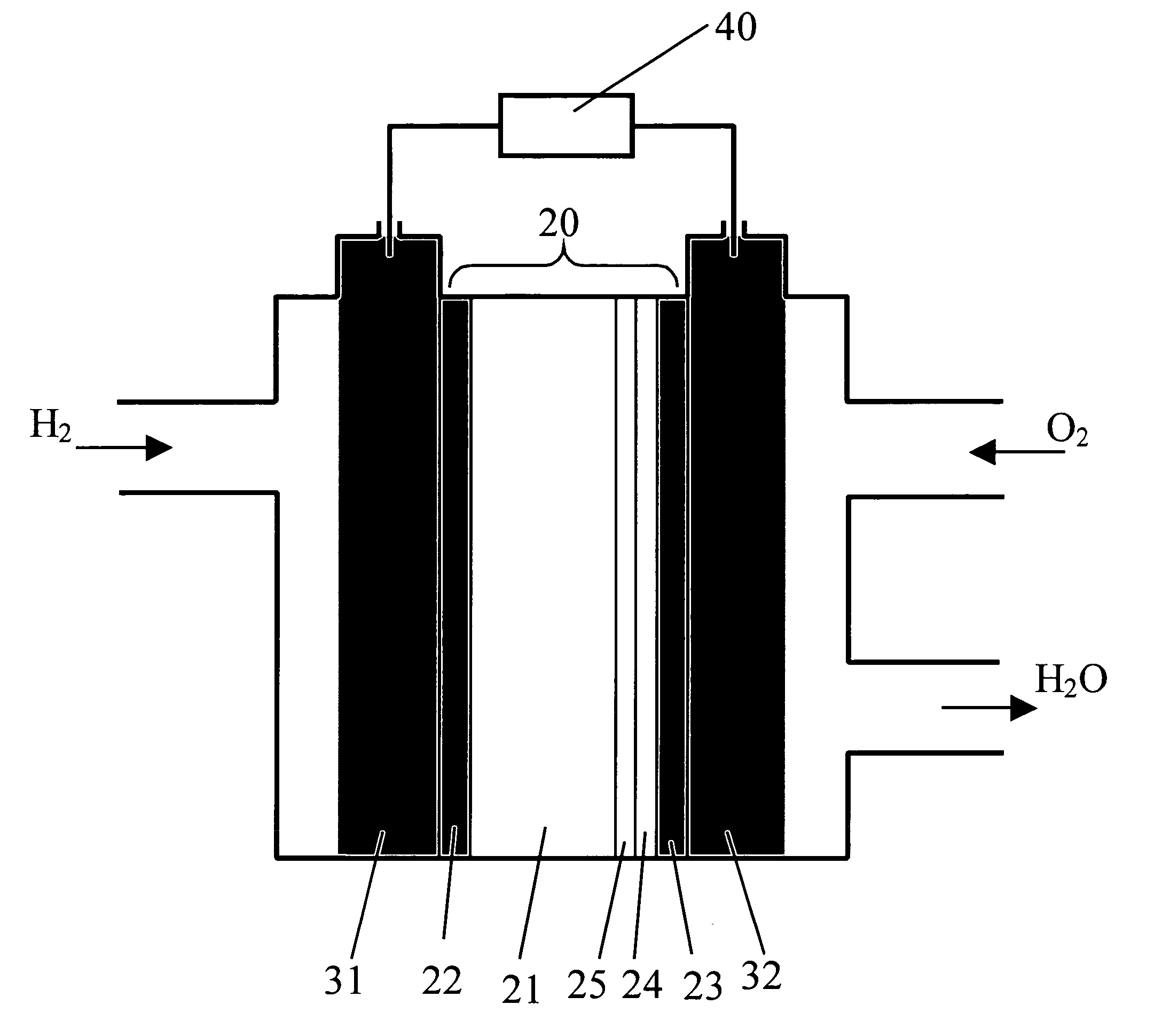
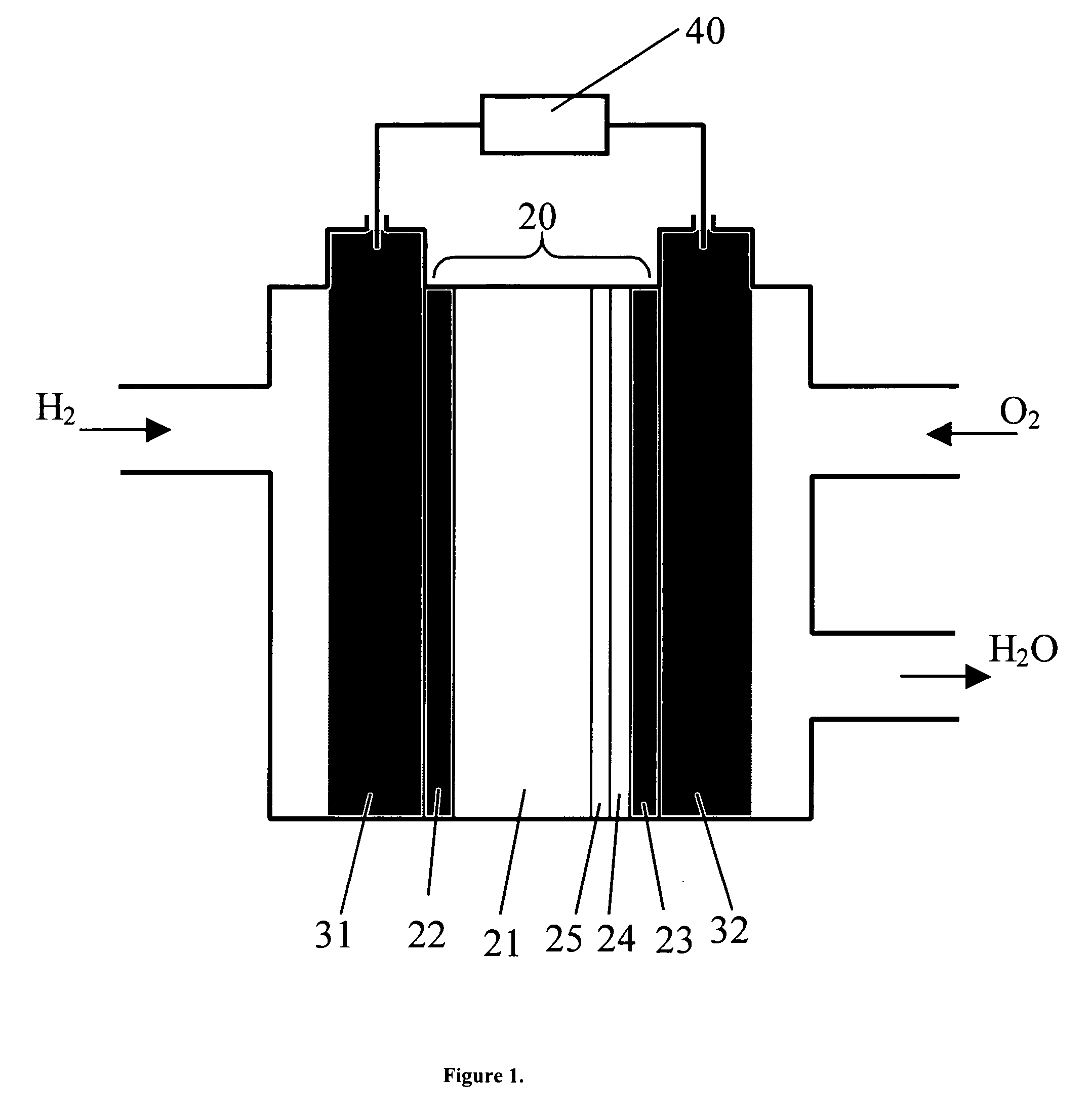
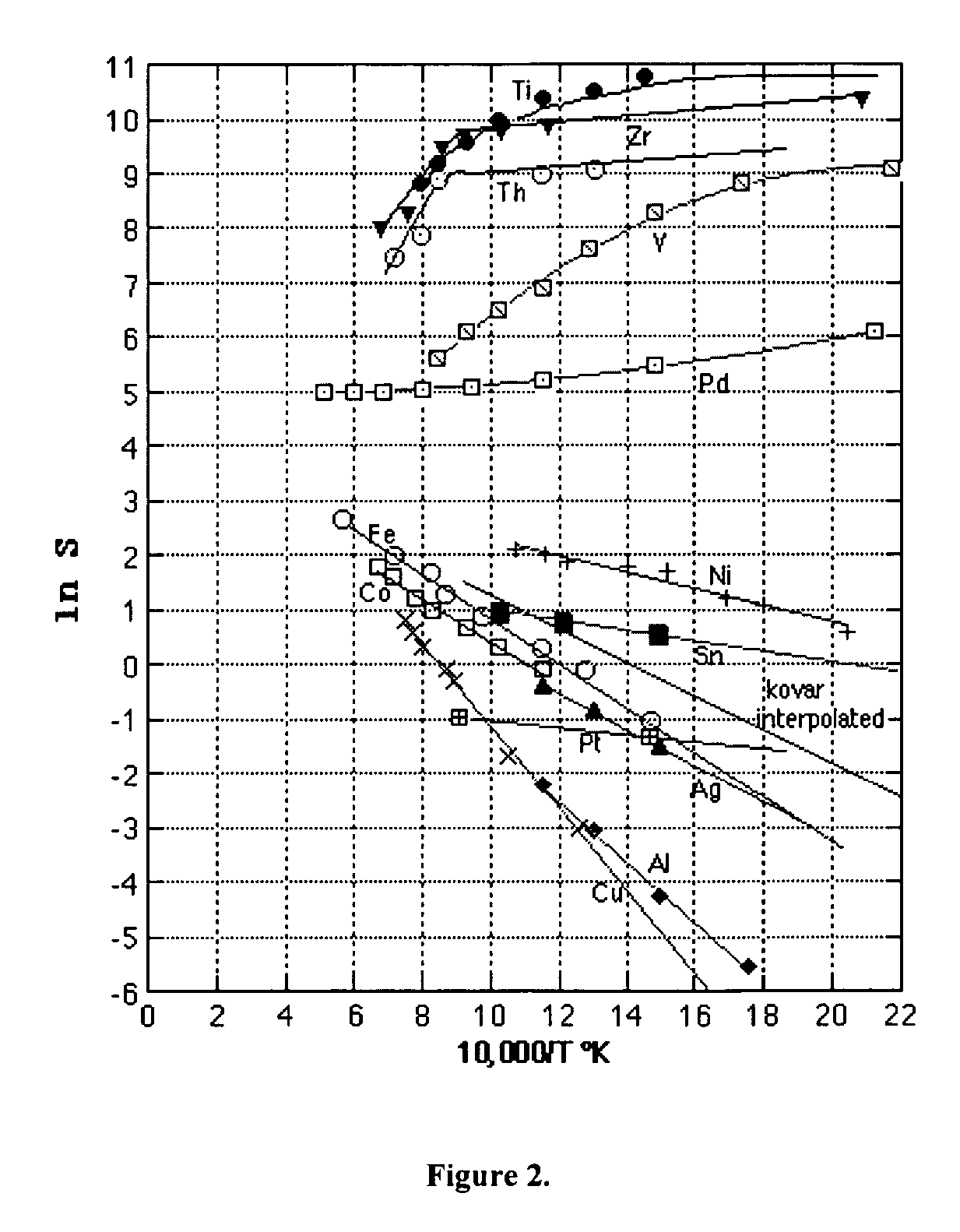
Solid state proton conductors for next generation fuel cells
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Solid State Proton Conductors Background and Objectives
Solid state proton conductors have emerged as a critical technology in the evolution of fuel cell systems, representing a significant departure from traditional liquid-based electrolyte solutions. The development trajectory of these materials spans several decades, beginning with the discovery of proton conduction in ceramic oxides in the 1960s and accelerating dramatically in the 1980s with the identification of high-temperature proton conductors based on perovskite-type oxides.
The technological progression has been marked by continuous improvements in conductivity, stability, and operating temperature ranges. Early solid proton conductors exhibited modest conductivity values of 10^-4 S/cm, whereas contemporary materials can achieve 10^-2 S/cm under optimal conditions, approaching the performance requirements for commercial viability. This evolution reflects the field's growing understanding of proton transport mechanisms in solid materials, including the Grotthuss mechanism, vehicle mechanism, and quantum tunneling effects.
Current research trends are focused on several promising material classes: perovskite-type oxides (BaCeO3, BaZrO3), rare-earth ortho-niobates and ortho-tantalates, phosphates and phosphoric acid derivatives, and coordination polymers incorporating proton-conducting pathways. Each material class presents distinct advantages and limitations regarding conductivity, chemical stability, mechanical properties, and fabrication complexity.
The primary objective of solid state proton conductor research is to develop materials that simultaneously achieve high proton conductivity (>10^-2 S/cm), excellent chemical stability in fuel cell operating environments, mechanical robustness, and compatibility with electrode materials. These materials must maintain performance across intermediate temperature ranges (200-600°C) to enable more efficient and durable fuel cell systems compared to current technologies.
Additional technical goals include reducing interfacial resistance between electrolyte and electrode materials, enhancing long-term operational stability under thermal cycling, and developing cost-effective fabrication methods suitable for mass production. Researchers are particularly focused on eliminating the trade-off between conductivity and stability that has historically limited the commercial implementation of solid state proton conductors.
The ultimate aim is to enable next-generation fuel cells that operate at intermediate temperatures with higher efficiency, longer lifetimes, and simpler system designs than current technologies. Such advancements would position fuel cells as a more competitive and versatile clean energy technology for applications ranging from distributed power generation to transportation and portable electronics, contributing significantly to global decarbonization efforts.
The technological progression has been marked by continuous improvements in conductivity, stability, and operating temperature ranges. Early solid proton conductors exhibited modest conductivity values of 10^-4 S/cm, whereas contemporary materials can achieve 10^-2 S/cm under optimal conditions, approaching the performance requirements for commercial viability. This evolution reflects the field's growing understanding of proton transport mechanisms in solid materials, including the Grotthuss mechanism, vehicle mechanism, and quantum tunneling effects.
Current research trends are focused on several promising material classes: perovskite-type oxides (BaCeO3, BaZrO3), rare-earth ortho-niobates and ortho-tantalates, phosphates and phosphoric acid derivatives, and coordination polymers incorporating proton-conducting pathways. Each material class presents distinct advantages and limitations regarding conductivity, chemical stability, mechanical properties, and fabrication complexity.
The primary objective of solid state proton conductor research is to develop materials that simultaneously achieve high proton conductivity (>10^-2 S/cm), excellent chemical stability in fuel cell operating environments, mechanical robustness, and compatibility with electrode materials. These materials must maintain performance across intermediate temperature ranges (200-600°C) to enable more efficient and durable fuel cell systems compared to current technologies.
Additional technical goals include reducing interfacial resistance between electrolyte and electrode materials, enhancing long-term operational stability under thermal cycling, and developing cost-effective fabrication methods suitable for mass production. Researchers are particularly focused on eliminating the trade-off between conductivity and stability that has historically limited the commercial implementation of solid state proton conductors.
The ultimate aim is to enable next-generation fuel cells that operate at intermediate temperatures with higher efficiency, longer lifetimes, and simpler system designs than current technologies. Such advancements would position fuel cells as a more competitive and versatile clean energy technology for applications ranging from distributed power generation to transportation and portable electronics, contributing significantly to global decarbonization efforts.
Market Analysis for Next Generation Fuel Cells
The global fuel cell market is experiencing significant growth, projected to reach $13.7 billion by 2026, with a compound annual growth rate of 26.4% from 2021. Next generation fuel cells, particularly those utilizing solid state proton conductors, represent a rapidly expanding segment within this market. The transportation sector currently dominates fuel cell applications, accounting for approximately 65% of the market share, followed by stationary power generation at 25% and portable applications at 10%.
Regionally, Asia-Pacific leads the market with Japan and South Korea at the forefront of fuel cell technology adoption. North America follows closely, with significant investments in fuel cell research and development, particularly in the United States. Europe is rapidly catching up, driven by stringent environmental regulations and substantial government support for clean energy technologies.
The demand for next generation fuel cells is primarily driven by increasing environmental concerns, government initiatives to reduce carbon emissions, and the growing need for reliable and efficient power generation solutions. The automotive industry represents a particularly promising market, with major manufacturers investing heavily in fuel cell electric vehicles (FCEVs). Toyota, Hyundai, and Honda have already commercialized FCEVs, with others announcing plans to enter the market.
Consumer demand for FCEVs is growing steadily, particularly in regions with developed hydrogen infrastructure. Japan aims to have 800,000 FCEVs on its roads by 2030, while California targets 1 million by the same year. This increasing adoption is expected to significantly drive the demand for advanced solid state proton conductors.
The stationary power generation sector also presents substantial opportunities, with fuel cells increasingly being deployed for backup power, combined heat and power (CHP) systems, and distributed generation. The telecommunications industry, data centers, and healthcare facilities are key end-users in this segment, valuing the reliability and efficiency of fuel cell systems.
Market barriers include high initial costs, limited hydrogen infrastructure, and competition from battery electric vehicles. However, technological advancements in solid state proton conductors are expected to address many of these challenges by improving efficiency, durability, and cost-effectiveness.
Investors are showing growing interest in the sector, with venture capital funding for fuel cell technologies reaching $1.5 billion in 2020, a 40% increase from the previous year. Strategic partnerships between technology developers, material suppliers, and end-users are becoming increasingly common, accelerating commercialization efforts and market penetration.
Regionally, Asia-Pacific leads the market with Japan and South Korea at the forefront of fuel cell technology adoption. North America follows closely, with significant investments in fuel cell research and development, particularly in the United States. Europe is rapidly catching up, driven by stringent environmental regulations and substantial government support for clean energy technologies.
The demand for next generation fuel cells is primarily driven by increasing environmental concerns, government initiatives to reduce carbon emissions, and the growing need for reliable and efficient power generation solutions. The automotive industry represents a particularly promising market, with major manufacturers investing heavily in fuel cell electric vehicles (FCEVs). Toyota, Hyundai, and Honda have already commercialized FCEVs, with others announcing plans to enter the market.
Consumer demand for FCEVs is growing steadily, particularly in regions with developed hydrogen infrastructure. Japan aims to have 800,000 FCEVs on its roads by 2030, while California targets 1 million by the same year. This increasing adoption is expected to significantly drive the demand for advanced solid state proton conductors.
The stationary power generation sector also presents substantial opportunities, with fuel cells increasingly being deployed for backup power, combined heat and power (CHP) systems, and distributed generation. The telecommunications industry, data centers, and healthcare facilities are key end-users in this segment, valuing the reliability and efficiency of fuel cell systems.
Market barriers include high initial costs, limited hydrogen infrastructure, and competition from battery electric vehicles. However, technological advancements in solid state proton conductors are expected to address many of these challenges by improving efficiency, durability, and cost-effectiveness.
Investors are showing growing interest in the sector, with venture capital funding for fuel cell technologies reaching $1.5 billion in 2020, a 40% increase from the previous year. Strategic partnerships between technology developers, material suppliers, and end-users are becoming increasingly common, accelerating commercialization efforts and market penetration.
Current Status and Technical Barriers in Proton Conductors
Solid state proton conductors have emerged as critical components for next-generation fuel cells, with significant research progress achieved globally over the past decade. Current state-of-the-art proton conductors can be categorized into several material families: perovskite-type oxides, rare-earth ortho-niobates and ortho-tantalates, pyrochlores, and solid acid electrolytes. Among these, BaZrO₃-based perovskites doped with trivalent elements (particularly Y-doped BaZrO₃) have demonstrated the most promising conductivity values, reaching 10⁻² S/cm at intermediate temperatures (400-600°C).
Despite these advances, several technical barriers persist in the development of practical solid-state proton conductors. The most significant challenge remains the trade-off between proton conductivity and chemical stability. Materials with high conductivity often suffer from poor stability in CO₂ and H₂O-containing environments, while chemically stable materials typically exhibit insufficient conductivity for practical applications. This fundamental contradiction has hindered widespread commercialization.
Grain boundary resistance represents another major obstacle, particularly in polycrystalline ceramic electrolytes. The total conductivity of these materials is often dominated by highly resistive grain boundaries, which can be orders of magnitude less conductive than the bulk material. This necessitates high sintering temperatures (>1600°C) to reduce grain boundary density, complicating manufacturing processes and increasing production costs.
Mechanical integrity issues also plague current proton conductors. Many promising materials exhibit brittleness and poor mechanical strength, making them vulnerable to cracking during thermal cycling or mechanical stress. This fragility compromises long-term operational reliability and limits practical application in real-world fuel cell systems.
Processing challenges further complicate development efforts. The fabrication of dense, thin electrolyte films (required for reduced ohmic losses) with consistent properties remains technically demanding. Current manufacturing methods struggle to produce uniform, defect-free electrolytes at scales necessary for commercial viability.
The operational temperature window presents another significant barrier. Most current proton conductors achieve optimal performance either at high temperatures (>600°C), where material degradation accelerates, or at low temperatures (<300°C), where conductivity becomes insufficient. The intermediate temperature range (300-600°C) represents an ideal operational window, but few materials maintain adequate conductivity and stability throughout this range.
Finally, integration challenges with electrode materials create additional complications. The chemical and thermal expansion compatibility between electrolytes and electrodes remains problematic, often leading to delamination, cracking, or formation of resistive interfacial phases during operation, significantly reducing cell performance and longevity.
Despite these advances, several technical barriers persist in the development of practical solid-state proton conductors. The most significant challenge remains the trade-off between proton conductivity and chemical stability. Materials with high conductivity often suffer from poor stability in CO₂ and H₂O-containing environments, while chemically stable materials typically exhibit insufficient conductivity for practical applications. This fundamental contradiction has hindered widespread commercialization.
Grain boundary resistance represents another major obstacle, particularly in polycrystalline ceramic electrolytes. The total conductivity of these materials is often dominated by highly resistive grain boundaries, which can be orders of magnitude less conductive than the bulk material. This necessitates high sintering temperatures (>1600°C) to reduce grain boundary density, complicating manufacturing processes and increasing production costs.
Mechanical integrity issues also plague current proton conductors. Many promising materials exhibit brittleness and poor mechanical strength, making them vulnerable to cracking during thermal cycling or mechanical stress. This fragility compromises long-term operational reliability and limits practical application in real-world fuel cell systems.
Processing challenges further complicate development efforts. The fabrication of dense, thin electrolyte films (required for reduced ohmic losses) with consistent properties remains technically demanding. Current manufacturing methods struggle to produce uniform, defect-free electrolytes at scales necessary for commercial viability.
The operational temperature window presents another significant barrier. Most current proton conductors achieve optimal performance either at high temperatures (>600°C), where material degradation accelerates, or at low temperatures (<300°C), where conductivity becomes insufficient. The intermediate temperature range (300-600°C) represents an ideal operational window, but few materials maintain adequate conductivity and stability throughout this range.
Finally, integration challenges with electrode materials create additional complications. The chemical and thermal expansion compatibility between electrolytes and electrodes remains problematic, often leading to delamination, cracking, or formation of resistive interfacial phases during operation, significantly reducing cell performance and longevity.
State-of-the-Art Solid State Proton Conductor Solutions
01 Polymer-based proton conductors
Polymer-based materials serve as effective solid-state proton conductors. These include modified polymers with functional groups that facilitate proton transport through hydrogen bonding networks. Polymers such as sulfonated polyimides, perfluorosulfonic acid polymers, and polymer composites with inorganic additives exhibit enhanced proton conductivity under various temperature and humidity conditions. These materials are particularly valuable for fuel cell applications due to their mechanical flexibility and processability.- Polymer-based solid state proton conductors: Polymer-based materials serve as effective solid-state proton conductors in various applications. These include modified polymers with functional groups that facilitate proton transport, polymer electrolyte membranes, and polymer composites. The incorporation of specific functional groups enhances proton conductivity through hydrogen bonding networks. These materials often demonstrate good mechanical stability while maintaining high proton conductivity under various operating conditions.
- Metal-organic frameworks for proton conduction: Metal-organic frameworks (MOFs) represent an emerging class of solid-state proton conductors with tunable properties. These crystalline materials feature metal ions coordinated to organic ligands, creating porous structures that can facilitate proton transport. The proton conductivity in MOFs can be enhanced by incorporating acidic functional groups, water molecules in the pores, or by designing specific proton-conducting pathways within the framework structure.
- Ceramic and inorganic oxide proton conductors: Ceramic and inorganic oxide materials exhibit excellent proton conductivity at elevated temperatures. These include perovskite-type oxides, phosphates, and other crystalline structures that allow proton transport through oxygen vacancies or hydrogen bonding networks. The proton conductivity mechanism often involves proton hopping between oxygen sites in the crystal lattice. These materials typically offer high thermal stability and are suitable for high-temperature applications such as fuel cells.
- Composite and hybrid proton conductors: Composite and hybrid materials combine the advantages of different components to achieve enhanced proton conductivity. These typically consist of an inorganic component (such as metal oxides or nanoparticles) dispersed within an organic matrix (such as a polymer). The interface between the components often creates additional proton conduction pathways. These materials can exhibit improved mechanical properties, thermal stability, and proton conductivity compared to single-component systems.
- Proton-conducting membranes for fuel cell applications: Specialized proton-conducting membranes are designed specifically for fuel cell applications. These membranes feature optimized proton conductivity, gas barrier properties, and mechanical durability under operating conditions. Various approaches to enhance proton conductivity include the incorporation of acidic groups, hydrophilic domains, and nano-structured architectures that create efficient proton transport channels while maintaining dimensional stability and durability during fuel cell operation.
02 Metal-organic frameworks for proton conduction
Metal-organic frameworks (MOFs) represent an emerging class of solid-state proton conductors with tunable pore structures and functionalities. These crystalline materials feature metal ions coordinated to organic ligands, creating channels that facilitate proton transport. The proton conductivity in MOFs can be enhanced by incorporating acidic functional groups, water molecules in the pores, or other proton carriers. Their modular nature allows for precise engineering of proton conduction pathways and properties.Expand Specific Solutions03 Ceramic and inorganic oxide proton conductors
Ceramic and inorganic oxide materials exhibit excellent proton conductivity at elevated temperatures. These include perovskite-type oxides, rare-earth doped materials, and various metal oxides with oxygen vacancies that facilitate proton transport. The proton conduction mechanism typically involves proton hopping between oxygen sites in the crystal lattice. These materials offer high thermal stability and are suitable for high-temperature applications such as solid oxide fuel cells and hydrogen separation membranes.Expand Specific Solutions04 Composite and heterogeneous proton conductors
Composite materials combining organic and inorganic components demonstrate enhanced proton conductivity through synergistic effects. These heterogeneous systems often feature an inorganic phase (such as metal oxides or phosphates) dispersed within a polymer matrix, or vice versa. The interfaces between different phases create additional proton conduction pathways, while the composite structure provides improved mechanical properties and stability. These materials can be tailored to operate effectively across a wide temperature range.Expand Specific Solutions05 Acid-functionalized materials for proton transport
Materials functionalized with acidic groups demonstrate excellent proton conductivity due to the availability of dissociable protons. These include phosphonic acid derivatives, sulfonic acid-functionalized compounds, and materials with carboxylic acid groups. The acidic functional groups provide proton donor sites that facilitate proton hopping mechanisms. These materials can be designed as small molecules, polymers, or surface-modified inorganic substrates, offering versatility in applications ranging from fuel cells to sensors and electrochemical devices.Expand Specific Solutions
Leading Companies and Research Institutions in Fuel Cell Industry
The solid state proton conductor market for next-generation fuel cells is in a growth phase, with increasing market size driven by clean energy demands. Technology maturity varies across players, with established electronics giants like Samsung SDI, Panasonic, and Toyota leading commercial development through significant R&D investments. Academic institutions including MIT, Caltech, and University of Tokyo are advancing fundamental research, while specialized companies like FUJIFILM and Shin-Etsu Chemical contribute materials expertise. Government laboratories such as Naval Research Laboratory and Advanced Industrial Science & Technology provide critical research infrastructure. The competitive landscape features strategic partnerships between industry and academia, with Japanese and Korean companies currently dominating patent portfolios and commercial applications in this emerging field.
Massachusetts Institute of Technology
Technical Solution: MIT has developed advanced solid-state proton conductors based on metal-organic frameworks (MOFs) for next-generation fuel cells. Their approach utilizes highly ordered crystalline structures with precisely engineered proton conduction pathways. The research team has successfully incorporated acidic functional groups within MOF pores to create hydrogen-bond networks that facilitate proton transport[1]. A key innovation is their development of water-stable MOFs that maintain high proton conductivity (>10^-2 S/cm) under various humidity conditions, addressing a critical challenge in solid-state electrolytes[3]. MIT researchers have also pioneered composite materials combining MOFs with polymers to enhance mechanical stability while preserving high conductivity. Their recent work demonstrates fuel cells using these materials achieving power densities exceeding 500 mW/cm² at intermediate temperatures (80-150°C), significantly outperforming conventional systems[5].
Strengths: Superior proton conductivity at intermediate temperatures; excellent chemical and thermal stability; precise control over proton transport pathways. Weaknesses: Higher manufacturing complexity compared to conventional materials; potential scalability challenges for mass production; relatively early stage of development for commercial applications.
Advanced Industrial Science & Technology
Technical Solution: AIST has pioneered the development of ceramic-based solid-state proton conductors, particularly focusing on perovskite-type oxides (BaZrO₃-BaCeO₃ systems) doped with trivalent elements. Their materials exhibit exceptional proton conductivity exceeding 0.01 S/cm at intermediate temperatures (300-600°C)[1]. AIST researchers have developed innovative synthesis methods including sol-gel processes and solid-state reactive sintering that significantly improve grain boundary conductivity - traditionally a limiting factor in ceramic proton conductors[3]. A major breakthrough in their research is the development of chemically stable proton conductors that resist degradation in CO₂ and H₂O-containing environments, addressing a critical challenge for practical applications. Their protonic ceramic fuel cells (PCFCs) utilizing these materials have demonstrated remarkable stability over 10,000+ hours of operation while maintaining high power output (>500 mW/cm²)[5]. AIST has also developed composite electrolytes incorporating nanoscale heterojunctions that enhance proton transport across grain boundaries, resulting in conductivity improvements of up to 300% compared to conventional materials[7].
Strengths: Exceptional high-temperature stability and conductivity; demonstrated long-term durability; resistance to chemical poisoning; compatibility with multiple fuel types including hydrogen and hydrocarbons. Weaknesses: Higher operating temperatures than polymer-based alternatives; manufacturing challenges related to sintering and densification; mechanical fragility requiring careful system design.
Key Patents and Scientific Breakthroughs in Proton Conduction
Solid proton conductor and manufacturing method thereof, electrolyte membrane for fuel cell, and fuel cell
PatentPendingJP2023056959A
Innovation
- A composite material comprising inorganic oxide particles coated with a salt formed by a nitrogen-containing heterocyclic ring-containing compound and an acid, produced through mechanical milling, which enhances proton conductivity and stability.
Solid state, thin film proton exchange membrane for fuel cells
PatentInactiveUS20070042252A1
Innovation
- A thin film solid state fuel cell with a multilayer structure comprising a metal oxide polymer protonic conductor and metal coatings on a high hydrogen permeability metal substrate, operating in the 300-400°C temperature range, significantly reducing protonic conductor resistance and enhancing power density and efficiency.
Materials Science Challenges and Opportunities
The development of solid-state proton conductors faces significant materials science challenges that must be overcome to realize next-generation fuel cells. Current materials exhibit a fundamental trade-off between proton conductivity and mechanical stability. Polymer-based electrolytes like Nafion offer excellent conductivity but suffer from poor mechanical properties at elevated temperatures, while ceramic-based conductors provide superior thermal stability but typically demonstrate lower conductivity at intermediate temperatures (80-300°C).
Interface engineering represents another critical challenge. The solid-solid interfaces between electrolyte and electrodes create high resistance pathways that impede proton transport. These interfacial resistances often account for more than 50% of the total cell resistance, significantly reducing overall efficiency. Developing materials with compatible thermal expansion coefficients and creating coherent interfaces remains an unsolved materials science problem.
Durability under operating conditions presents additional obstacles. Many promising proton conductors degrade when exposed to fuel cell conditions, including humidity fluctuations, thermal cycling, and chemical contaminants. For instance, barium zirconate-based materials show promising conductivity but suffer from grain boundary degradation during long-term operation, while phosphoric acid-doped polybenzimidazole membranes experience acid leaching under humidity cycling.
The synthesis and processing of these materials introduce further complications. Achieving high-density ceramics with controlled microstructure requires precise sintering protocols, while polymer-ceramic composites demand novel fabrication techniques to ensure homogeneous distribution and connectivity of conductive pathways. Current manufacturing methods often produce materials with inconsistent properties and performance.
Opportunities for breakthrough advances exist in several areas. Computational materials science offers promising pathways for rational design of new proton conductors through high-throughput screening and machine learning approaches. Recent advances in density functional theory calculations have enabled more accurate predictions of proton transport mechanisms and identification of promising chemical compositions.
Nanoscale engineering presents another opportunity frontier. Hierarchically structured materials that incorporate designed transport pathways at multiple length scales could overcome the conductivity-stability dilemma. Preliminary research on 2D materials as proton conductors, including graphene oxide and MXenes, demonstrates potential for ultrathin electrolytes with exceptional conductivity.
Interface engineering represents another critical challenge. The solid-solid interfaces between electrolyte and electrodes create high resistance pathways that impede proton transport. These interfacial resistances often account for more than 50% of the total cell resistance, significantly reducing overall efficiency. Developing materials with compatible thermal expansion coefficients and creating coherent interfaces remains an unsolved materials science problem.
Durability under operating conditions presents additional obstacles. Many promising proton conductors degrade when exposed to fuel cell conditions, including humidity fluctuations, thermal cycling, and chemical contaminants. For instance, barium zirconate-based materials show promising conductivity but suffer from grain boundary degradation during long-term operation, while phosphoric acid-doped polybenzimidazole membranes experience acid leaching under humidity cycling.
The synthesis and processing of these materials introduce further complications. Achieving high-density ceramics with controlled microstructure requires precise sintering protocols, while polymer-ceramic composites demand novel fabrication techniques to ensure homogeneous distribution and connectivity of conductive pathways. Current manufacturing methods often produce materials with inconsistent properties and performance.
Opportunities for breakthrough advances exist in several areas. Computational materials science offers promising pathways for rational design of new proton conductors through high-throughput screening and machine learning approaches. Recent advances in density functional theory calculations have enabled more accurate predictions of proton transport mechanisms and identification of promising chemical compositions.
Nanoscale engineering presents another opportunity frontier. Hierarchically structured materials that incorporate designed transport pathways at multiple length scales could overcome the conductivity-stability dilemma. Preliminary research on 2D materials as proton conductors, including graphene oxide and MXenes, demonstrates potential for ultrathin electrolytes with exceptional conductivity.
Environmental Impact and Sustainability Assessment
The environmental impact of solid state proton conductors for next-generation fuel cells represents a critical dimension in evaluating their overall viability and sustainability. These advanced materials offer significant environmental advantages compared to traditional fuel cell technologies. Primarily, solid state proton conductors eliminate the need for liquid electrolytes containing corrosive acids or bases, substantially reducing the risk of hazardous material leakage and associated environmental contamination.
The manufacturing processes for solid state proton conductors typically require lower energy inputs compared to conventional fuel cell components, resulting in reduced carbon emissions during production. Additionally, these materials often demonstrate extended operational lifespans, minimizing waste generation and resource consumption associated with frequent replacements. Many solid state proton conductors utilize earth-abundant elements rather than precious metals, alleviating concerns regarding resource depletion and extraction-related environmental degradation.
Life cycle assessment (LCA) studies indicate that fuel cells incorporating solid state proton conductors generally exhibit lower environmental footprints across multiple impact categories, including global warming potential, acidification, and eutrophication. The enhanced efficiency of these next-generation fuel cells translates to reduced fuel consumption and corresponding decreases in greenhouse gas emissions during operation, particularly when hydrogen is sourced from renewable pathways.
From a sustainability perspective, solid state proton conductors align well with circular economy principles. Many of these materials demonstrate promising recyclability characteristics, with research showing potential recovery rates exceeding 80% for certain components. This recyclability significantly reduces end-of-life environmental impacts and decreases dependence on virgin material extraction.
Water management represents another environmental advantage of solid state proton conductor fuel cells. Unlike traditional polymer electrolyte membrane fuel cells that require precise water balance management, solid state systems operate effectively under varying humidity conditions, reducing water consumption and simplifying system design. This characteristic proves particularly valuable in water-stressed regions where conventional fuel cell deployment might face sustainability challenges.
Despite these advantages, certain environmental concerns persist. Some solid state proton conductors incorporate rare earth elements or other materials with complex supply chains and extraction impacts. Additionally, high-temperature processing requirements for certain materials can contribute to manufacturing-phase energy consumption. Ongoing research focuses on developing synthesis routes with reduced environmental footprints and identifying alternative compositions that maintain performance while minimizing ecological impacts.
The manufacturing processes for solid state proton conductors typically require lower energy inputs compared to conventional fuel cell components, resulting in reduced carbon emissions during production. Additionally, these materials often demonstrate extended operational lifespans, minimizing waste generation and resource consumption associated with frequent replacements. Many solid state proton conductors utilize earth-abundant elements rather than precious metals, alleviating concerns regarding resource depletion and extraction-related environmental degradation.
Life cycle assessment (LCA) studies indicate that fuel cells incorporating solid state proton conductors generally exhibit lower environmental footprints across multiple impact categories, including global warming potential, acidification, and eutrophication. The enhanced efficiency of these next-generation fuel cells translates to reduced fuel consumption and corresponding decreases in greenhouse gas emissions during operation, particularly when hydrogen is sourced from renewable pathways.
From a sustainability perspective, solid state proton conductors align well with circular economy principles. Many of these materials demonstrate promising recyclability characteristics, with research showing potential recovery rates exceeding 80% for certain components. This recyclability significantly reduces end-of-life environmental impacts and decreases dependence on virgin material extraction.
Water management represents another environmental advantage of solid state proton conductor fuel cells. Unlike traditional polymer electrolyte membrane fuel cells that require precise water balance management, solid state systems operate effectively under varying humidity conditions, reducing water consumption and simplifying system design. This characteristic proves particularly valuable in water-stressed regions where conventional fuel cell deployment might face sustainability challenges.
Despite these advantages, certain environmental concerns persist. Some solid state proton conductors incorporate rare earth elements or other materials with complex supply chains and extraction impacts. Additionally, high-temperature processing requirements for certain materials can contribute to manufacturing-phase energy consumption. Ongoing research focuses on developing synthesis routes with reduced environmental footprints and identifying alternative compositions that maintain performance while minimizing ecological impacts.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!