Active Memory in Biomedical Devices: Functional Benefits
MAR 7, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Active Memory in Biomedical Devices Background and Objectives
Active memory technology represents a paradigm shift in biomedical device design, fundamentally altering how medical systems process, store, and respond to physiological data. Unlike traditional passive memory systems that merely store information, active memory incorporates intelligent processing capabilities that enable real-time data analysis, pattern recognition, and adaptive responses within biomedical applications.
The evolution of active memory in biomedical devices traces back to the convergence of several technological domains, including neuromorphic computing, advanced semiconductor materials, and biocompatible electronics. Early developments emerged from research into brain-computer interfaces and implantable medical devices, where the need for intelligent, low-power memory systems became apparent. The technology has progressively evolved from simple data logging capabilities to sophisticated systems capable of learning from patient-specific patterns and adapting treatment protocols accordingly.
Current technological trends indicate a strong movement toward personalized medicine and autonomous medical devices, driving the demand for memory systems that can process complex biological signals in real-time. The integration of artificial intelligence algorithms with memory architectures has created opportunities for devices that not only store patient data but actively interpret and respond to changing physiological conditions without external intervention.
The primary objective of implementing active memory in biomedical devices centers on achieving enhanced diagnostic accuracy through continuous learning and adaptation. These systems aim to reduce false positives and negatives in medical monitoring by developing patient-specific baselines and recognizing subtle pattern variations that might indicate emerging health conditions. The technology seeks to minimize the computational burden on external processing units while maintaining high-fidelity data analysis capabilities.
Another critical objective involves improving patient outcomes through predictive healthcare interventions. Active memory systems are designed to anticipate medical events before they become critical, enabling proactive treatment adjustments and reducing emergency situations. This predictive capability extends to medication management, where memory systems can optimize drug delivery timing and dosages based on individual patient responses and metabolic patterns.
The technology also aims to address the growing need for autonomous medical devices in remote healthcare scenarios. Active memory enables biomedical devices to function independently for extended periods while maintaining sophisticated monitoring and treatment capabilities, particularly valuable in telemedicine applications and underserved geographical regions where immediate medical expertise may not be readily available.
The evolution of active memory in biomedical devices traces back to the convergence of several technological domains, including neuromorphic computing, advanced semiconductor materials, and biocompatible electronics. Early developments emerged from research into brain-computer interfaces and implantable medical devices, where the need for intelligent, low-power memory systems became apparent. The technology has progressively evolved from simple data logging capabilities to sophisticated systems capable of learning from patient-specific patterns and adapting treatment protocols accordingly.
Current technological trends indicate a strong movement toward personalized medicine and autonomous medical devices, driving the demand for memory systems that can process complex biological signals in real-time. The integration of artificial intelligence algorithms with memory architectures has created opportunities for devices that not only store patient data but actively interpret and respond to changing physiological conditions without external intervention.
The primary objective of implementing active memory in biomedical devices centers on achieving enhanced diagnostic accuracy through continuous learning and adaptation. These systems aim to reduce false positives and negatives in medical monitoring by developing patient-specific baselines and recognizing subtle pattern variations that might indicate emerging health conditions. The technology seeks to minimize the computational burden on external processing units while maintaining high-fidelity data analysis capabilities.
Another critical objective involves improving patient outcomes through predictive healthcare interventions. Active memory systems are designed to anticipate medical events before they become critical, enabling proactive treatment adjustments and reducing emergency situations. This predictive capability extends to medication management, where memory systems can optimize drug delivery timing and dosages based on individual patient responses and metabolic patterns.
The technology also aims to address the growing need for autonomous medical devices in remote healthcare scenarios. Active memory enables biomedical devices to function independently for extended periods while maintaining sophisticated monitoring and treatment capabilities, particularly valuable in telemedicine applications and underserved geographical regions where immediate medical expertise may not be readily available.
Market Demand for Smart Biomedical Memory Solutions
The global biomedical device market is experiencing unprecedented growth driven by aging populations, increasing prevalence of chronic diseases, and rising healthcare expenditure worldwide. This demographic shift creates substantial demand for advanced medical technologies that can provide continuous monitoring, real-time data processing, and adaptive therapeutic responses. Smart biomedical memory solutions represent a critical enabling technology for next-generation medical devices that must operate reliably in complex physiological environments while maintaining high performance standards.
Healthcare systems globally are prioritizing personalized medicine and precision healthcare delivery, creating significant market pull for intelligent biomedical devices equipped with sophisticated memory capabilities. These devices must store vast amounts of patient data, treatment protocols, and diagnostic algorithms while ensuring rapid access and processing. The demand extends across multiple healthcare segments including implantable devices, wearable health monitors, diagnostic equipment, and therapeutic delivery systems.
The market opportunity for smart biomedical memory solutions is particularly pronounced in developed economies where healthcare infrastructure supports advanced medical technologies. North America and Europe represent primary markets due to established regulatory frameworks, high healthcare spending, and early adoption of innovative medical technologies. However, emerging markets in Asia-Pacific regions are demonstrating accelerated growth potential as healthcare systems modernize and populations gain access to advanced medical care.
Regulatory requirements and safety standards significantly influence market demand patterns for biomedical memory solutions. Medical device manufacturers require memory technologies that comply with stringent biocompatibility standards, electromagnetic compatibility requirements, and data security regulations. These compliance needs create specialized market segments where conventional memory solutions prove inadequate, driving demand for purpose-built biomedical memory technologies.
The integration of artificial intelligence and machine learning capabilities in medical devices further amplifies demand for high-performance memory solutions. Modern biomedical applications require memory systems capable of supporting complex algorithms, pattern recognition, and predictive analytics while maintaining ultra-low power consumption. This technological convergence creates expanding market opportunities for memory solutions specifically designed for intelligent biomedical applications.
Cost-effectiveness considerations also shape market demand dynamics, as healthcare providers seek technologies that deliver improved patient outcomes while controlling operational expenses. Smart biomedical memory solutions that enable device miniaturization, extend operational lifespans, and reduce maintenance requirements align with these economic priorities, creating favorable market conditions for adoption across diverse healthcare applications.
Healthcare systems globally are prioritizing personalized medicine and precision healthcare delivery, creating significant market pull for intelligent biomedical devices equipped with sophisticated memory capabilities. These devices must store vast amounts of patient data, treatment protocols, and diagnostic algorithms while ensuring rapid access and processing. The demand extends across multiple healthcare segments including implantable devices, wearable health monitors, diagnostic equipment, and therapeutic delivery systems.
The market opportunity for smart biomedical memory solutions is particularly pronounced in developed economies where healthcare infrastructure supports advanced medical technologies. North America and Europe represent primary markets due to established regulatory frameworks, high healthcare spending, and early adoption of innovative medical technologies. However, emerging markets in Asia-Pacific regions are demonstrating accelerated growth potential as healthcare systems modernize and populations gain access to advanced medical care.
Regulatory requirements and safety standards significantly influence market demand patterns for biomedical memory solutions. Medical device manufacturers require memory technologies that comply with stringent biocompatibility standards, electromagnetic compatibility requirements, and data security regulations. These compliance needs create specialized market segments where conventional memory solutions prove inadequate, driving demand for purpose-built biomedical memory technologies.
The integration of artificial intelligence and machine learning capabilities in medical devices further amplifies demand for high-performance memory solutions. Modern biomedical applications require memory systems capable of supporting complex algorithms, pattern recognition, and predictive analytics while maintaining ultra-low power consumption. This technological convergence creates expanding market opportunities for memory solutions specifically designed for intelligent biomedical applications.
Cost-effectiveness considerations also shape market demand dynamics, as healthcare providers seek technologies that deliver improved patient outcomes while controlling operational expenses. Smart biomedical memory solutions that enable device miniaturization, extend operational lifespans, and reduce maintenance requirements align with these economic priorities, creating favorable market conditions for adoption across diverse healthcare applications.
Current State and Challenges of Active Memory in Medical Devices
Active memory technologies in biomedical devices have reached a critical juncture where significant advancements coexist with substantial technical barriers. Current implementations primarily focus on shape memory alloys, electroactive polymers, and smart hydrogels that respond to physiological stimuli such as temperature, pH, and electrical fields. These materials demonstrate remarkable capabilities in applications ranging from self-expanding stents to drug delivery systems that adapt to patient conditions.
The integration of active memory components in medical devices has achieved notable success in cardiovascular interventions, where nitinol-based stents exhibit superior biocompatibility and reduced restenosis rates compared to traditional materials. Similarly, orthopedic implants incorporating shape memory elements show enhanced osseointegration and reduced mechanical mismatch with natural bone structures. Neural interfaces utilizing electroactive polymers demonstrate improved signal fidelity and reduced inflammatory responses.
Despite these achievements, several fundamental challenges persist across the active memory landscape in medical applications. Material fatigue remains a primary concern, as repeated activation cycles can degrade memory properties and compromise device reliability over extended implantation periods. The unpredictable nature of biological environments introduces variables that can interfere with programmed memory responses, potentially leading to suboptimal therapeutic outcomes.
Biocompatibility issues continue to challenge widespread adoption, particularly regarding long-term tissue interactions and potential immune responses to active memory materials. Manufacturing consistency presents another significant hurdle, as precise control over memory properties requires sophisticated processing techniques that are difficult to scale for mass production while maintaining quality standards.
Power management represents a critical bottleneck for electrically activated memory systems, where energy harvesting from biological sources remains insufficient for reliable operation. The complexity of programming multiple memory states while ensuring fail-safe mechanisms adds layers of technical difficulty that current solutions struggle to address comprehensively.
Regulatory pathways for active memory devices remain unclear, as existing frameworks inadequately address the dynamic nature of these technologies. The lack of standardized testing protocols for memory durability and performance under physiological conditions creates uncertainty for manufacturers and regulatory bodies alike.
Geographic distribution of active memory research reveals concentration in developed nations, with limited accessibility to emerging markets where such technologies could provide significant healthcare improvements. This disparity highlights the need for cost-effective solutions and technology transfer mechanisms to broaden global impact.
The integration of active memory components in medical devices has achieved notable success in cardiovascular interventions, where nitinol-based stents exhibit superior biocompatibility and reduced restenosis rates compared to traditional materials. Similarly, orthopedic implants incorporating shape memory elements show enhanced osseointegration and reduced mechanical mismatch with natural bone structures. Neural interfaces utilizing electroactive polymers demonstrate improved signal fidelity and reduced inflammatory responses.
Despite these achievements, several fundamental challenges persist across the active memory landscape in medical applications. Material fatigue remains a primary concern, as repeated activation cycles can degrade memory properties and compromise device reliability over extended implantation periods. The unpredictable nature of biological environments introduces variables that can interfere with programmed memory responses, potentially leading to suboptimal therapeutic outcomes.
Biocompatibility issues continue to challenge widespread adoption, particularly regarding long-term tissue interactions and potential immune responses to active memory materials. Manufacturing consistency presents another significant hurdle, as precise control over memory properties requires sophisticated processing techniques that are difficult to scale for mass production while maintaining quality standards.
Power management represents a critical bottleneck for electrically activated memory systems, where energy harvesting from biological sources remains insufficient for reliable operation. The complexity of programming multiple memory states while ensuring fail-safe mechanisms adds layers of technical difficulty that current solutions struggle to address comprehensively.
Regulatory pathways for active memory devices remain unclear, as existing frameworks inadequately address the dynamic nature of these technologies. The lack of standardized testing protocols for memory durability and performance under physiological conditions creates uncertainty for manufacturers and regulatory bodies alike.
Geographic distribution of active memory research reveals concentration in developed nations, with limited accessibility to emerging markets where such technologies could provide significant healthcare improvements. This disparity highlights the need for cost-effective solutions and technology transfer mechanisms to broaden global impact.
Existing Active Memory Solutions for Medical Applications
01 Memory enhancement through nutritional supplements and bioactive compounds
Functional benefits for active memory can be achieved through the use of nutritional supplements containing bioactive compounds such as omega-3 fatty acids, phospholipids, and plant extracts. These compounds support cognitive function by promoting neuronal health, enhancing neurotransmitter activity, and protecting against oxidative stress. The formulations can be delivered in various forms including capsules, tablets, and functional foods to improve memory retention, recall, and overall cognitive performance.- Memory enhancement through nutritional supplementation: Functional benefits for active memory can be achieved through nutritional compositions containing specific ingredients that support cognitive function. These formulations may include vitamins, minerals, amino acids, and botanical extracts designed to enhance memory performance, mental clarity, and overall brain health. The compositions are typically delivered in various forms such as beverages, capsules, or food products to provide convenient daily supplementation for memory support.
- Cognitive function improvement through bioactive compounds: Active memory functional benefits can be delivered through specific bioactive compounds that target neurological pathways. These compounds work by supporting neurotransmitter function, promoting neural connectivity, and protecting brain cells from oxidative stress. The formulations may incorporate phospholipids, omega fatty acids, and antioxidants that have been shown to support memory retention and recall abilities.
- Memory support through herbal and plant-based ingredients: Plant-based formulations provide functional benefits for active memory through the use of traditional and modern herbal ingredients. These natural compounds may include extracts known for their cognitive-supporting properties, which can help maintain mental acuity and support healthy brain aging. The herbal compositions are often combined with other nutrients to create synergistic effects for enhanced memory function.
- Delivery systems for memory-enhancing formulations: Advanced delivery systems are employed to optimize the bioavailability and effectiveness of memory-supporting ingredients. These systems may include specialized encapsulation technologies, time-release mechanisms, and novel formulation matrices that ensure optimal absorption and sustained release of active ingredients. The delivery methods are designed to maximize the functional benefits while improving user compliance and convenience.
- Combination therapies for comprehensive memory support: Comprehensive memory support is achieved through combination formulations that address multiple aspects of cognitive health simultaneously. These multi-ingredient approaches combine various classes of active compounds to provide synergistic benefits for memory function, including support for focus, concentration, and information processing. The formulations are designed to work through complementary mechanisms to provide broad-spectrum cognitive support.
02 Cognitive function improvement through pharmaceutical compositions
Active memory functional benefits can be provided through pharmaceutical compositions that contain specific active ingredients designed to enhance cognitive processes. These compositions may include nootropic compounds, neuroprotective agents, and memory-enhancing substances that work by modulating brain chemistry, improving blood flow to the brain, and supporting synaptic plasticity. The formulations are designed for sustained release and optimal bioavailability to maximize cognitive enhancement effects.Expand Specific Solutions03 Memory support through herbal and natural extracts
Natural and herbal extracts can provide functional benefits for active memory through their neuroprotective and cognitive-enhancing properties. These extracts may include ginkgo biloba, bacopa monnieri, and other botanicals known for their ability to support memory function. The mechanisms include antioxidant activity, anti-inflammatory effects, and enhancement of acetylcholine levels in the brain, which collectively contribute to improved memory formation and retrieval.Expand Specific Solutions04 Technological devices and systems for memory training and enhancement
Active memory functional benefits can be achieved through technological devices and systems designed for cognitive training and memory enhancement. These systems may include electronic devices, software applications, and interactive platforms that utilize neurofeedback, gamification, and adaptive learning algorithms to strengthen memory pathways. The technology provides personalized training programs that target specific aspects of memory function including working memory, episodic memory, and procedural memory.Expand Specific Solutions05 Combination therapies and multi-modal approaches for memory enhancement
Functional benefits for active memory can be optimized through combination therapies that integrate multiple approaches including nutritional supplementation, cognitive training, and lifestyle modifications. These multi-modal strategies work synergistically to address various aspects of memory function, from cellular and molecular mechanisms to behavioral and environmental factors. The combined approach provides comprehensive support for memory enhancement, maintenance of cognitive health, and prevention of age-related memory decline.Expand Specific Solutions
Key Players in Biomedical Active Memory Industry
The active memory technology in biomedical devices represents a rapidly evolving sector within the broader medical technology landscape, currently in its growth phase with significant market expansion potential. The industry demonstrates substantial market size driven by increasing demand for sophisticated implantable devices and smart medical systems. Technology maturity varies considerably across different applications, with established players like Medtronic, Boston Scientific, and St. Jude Medical leading in traditional implantable devices, while companies such as Saluda Medical and Synergia Medical are pioneering next-generation neurostimulation platforms. Memory technology providers including Micron Technology, Macronix International, and Taiwan Semiconductor Manufacturing are advancing the underlying semiconductor foundations. Academic institutions like University of California, North Carolina State University, and Huazhong University of Science & Technology are contributing fundamental research breakthroughs that enhance active memory capabilities in biomedical applications.
Micron Technology, Inc.
Technical Solution: Micron develops advanced memory solutions specifically designed for biomedical applications, including low-power DRAM and NAND flash memory optimized for implantable devices. Their active memory technologies enable real-time data processing and storage in medical devices, supporting continuous patient monitoring and adaptive therapy delivery. The company's biomedical memory solutions feature enhanced reliability, extended operational lifespan, and biocompatibility standards required for implantable applications. These memory systems provide dynamic data management capabilities that allow medical devices to learn from patient responses and adjust treatment parameters automatically.
Strengths: Industry-leading memory technology with proven reliability and low power consumption. Weaknesses: Limited direct biomedical device integration experience compared to specialized medical device manufacturers.
Cochlear (HK) Ltd.
Technical Solution: Cochlear implements active memory technology in their cochlear implant systems to enhance hearing restoration capabilities. Their active memory solutions enable real-time sound processing optimization, storing user preferences and environmental adaptations to improve hearing quality. The technology allows implants to learn from user interactions and automatically adjust sound processing parameters based on different acoustic environments. Their memory systems support advanced features like noise reduction algorithms and speech enhancement protocols that adapt to individual hearing patterns. The active memory also enables remote programming capabilities and stores diagnostic data for clinical follow-up assessments.
Strengths: Specialized expertise in neural interface technology and proven success in hearing restoration applications. Weaknesses: Limited to auditory applications, restricting expansion to other biomedical memory use cases.
Core Innovations in Biomedical Active Memory Patents
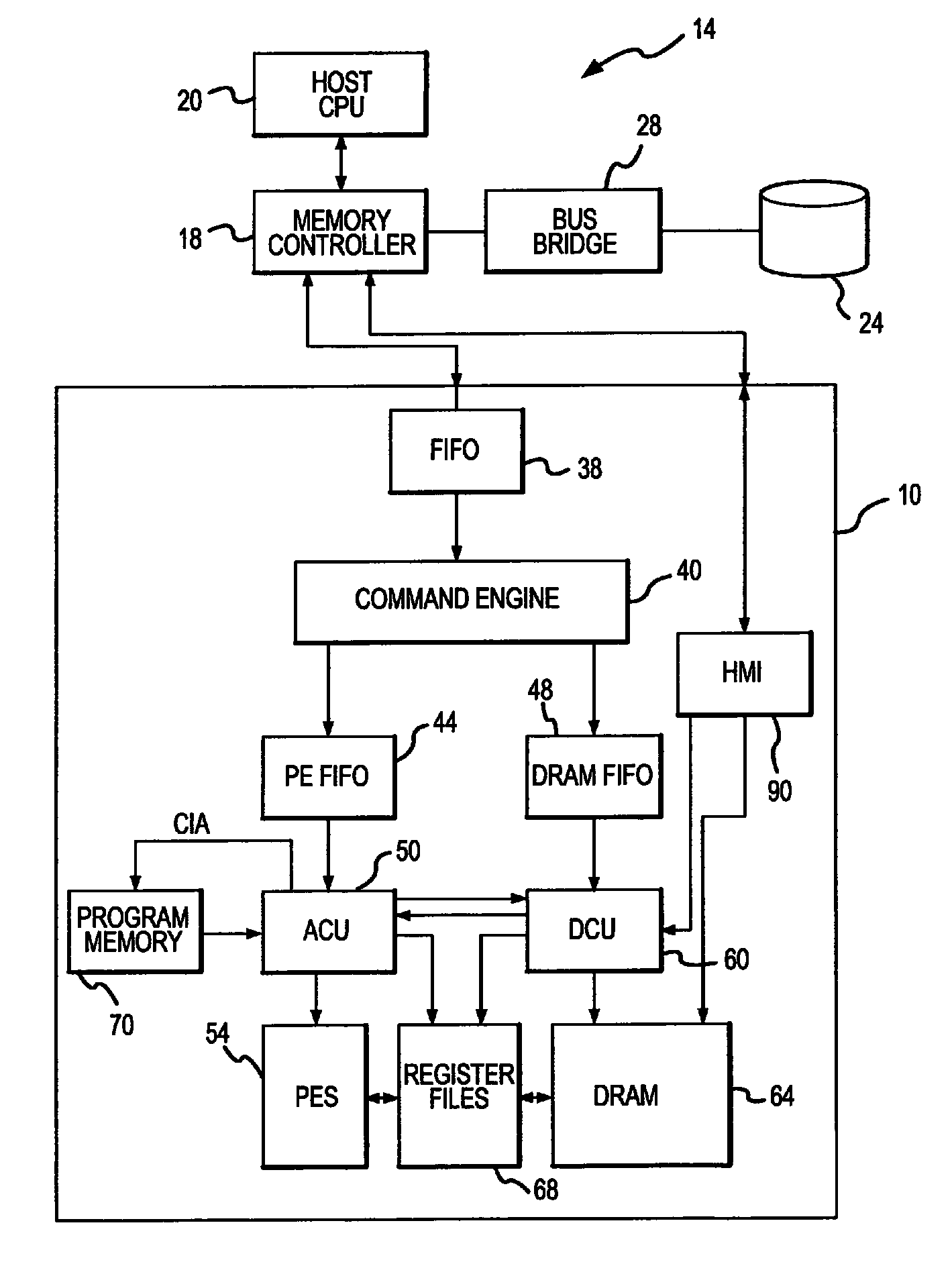
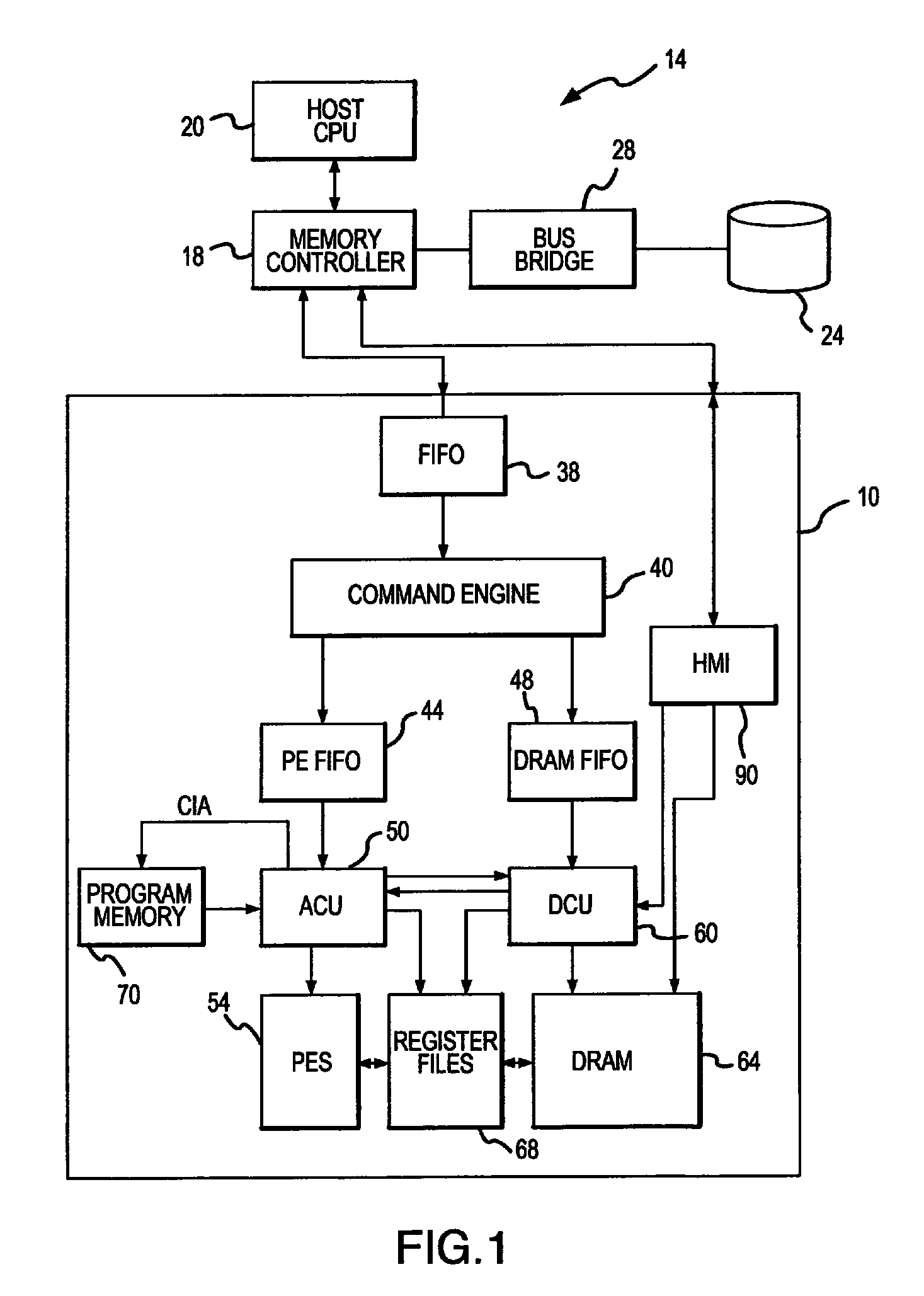
Active memory data compression system and method
PatentActiveUS9015390B2
Innovation
- An integrated circuit active memory device with an array of processing elements, such as SIMD or MIMD processors, compresses and decompresses data through a host/memory interface port, enhancing data bandwidth by executing compression and decompression algorithms stored in program memory, thereby increasing data transfer efficiency.
Implantable medical device having a scheme for managing storage of data
PatentWO2017106165A1
Innovation
- A scheme that enables flash memory to operate in a write-once mode, eliminating the need for sector erasure and thus reducing current consumption, while allowing for the preservation of relevant data, by using a configuration where data is stored in a write-once mode and managed through a microprocessor that designates active and inactive data regions within sectors.
Medical Device Regulatory Framework for Active Memory
The regulatory landscape for active memory technologies in biomedical devices presents a complex framework that varies significantly across global jurisdictions. In the United States, the FDA categorizes these devices based on risk levels, with active memory components typically falling under Class II or Class III classifications depending on their clinical application and potential patient impact. The 510(k) premarket notification pathway often applies to devices with established predicate technologies, while novel active memory implementations may require the more rigorous Premarket Approval (PMA) process.
European regulatory frameworks under the Medical Device Regulation (MDR) emphasize conformity assessment procedures that specifically address software and memory components as integral device elements. Active memory systems must demonstrate compliance with essential safety and performance requirements, particularly regarding data integrity, cybersecurity, and electromagnetic compatibility. The notified body assessment process requires comprehensive technical documentation demonstrating the active memory's role in device functionality and patient safety.
International harmonization efforts through ISO 14155 and IEC 62304 provide standardized approaches for clinical evaluation and software lifecycle processes relevant to active memory implementations. These standards establish requirements for risk management, software validation, and post-market surveillance that directly impact regulatory approval timelines and compliance costs.
Regulatory challenges specific to active memory technologies include establishing clear boundaries between hardware and software components, demonstrating long-term memory stability under physiological conditions, and validating data security protocols. Agencies increasingly require evidence of cybersecurity resilience, particularly for networked devices with active memory capabilities that store or transmit patient data.
The evolving regulatory environment reflects growing recognition of active memory as a critical device component rather than merely an accessory function. Recent guidance documents from major regulatory bodies emphasize the need for comprehensive validation protocols that address memory performance degradation, data corruption scenarios, and fail-safe mechanisms. This regulatory evolution directly influences development timelines, requiring manufacturers to integrate compliance considerations early in the design phase rather than treating regulation as a final approval hurdle.
European regulatory frameworks under the Medical Device Regulation (MDR) emphasize conformity assessment procedures that specifically address software and memory components as integral device elements. Active memory systems must demonstrate compliance with essential safety and performance requirements, particularly regarding data integrity, cybersecurity, and electromagnetic compatibility. The notified body assessment process requires comprehensive technical documentation demonstrating the active memory's role in device functionality and patient safety.
International harmonization efforts through ISO 14155 and IEC 62304 provide standardized approaches for clinical evaluation and software lifecycle processes relevant to active memory implementations. These standards establish requirements for risk management, software validation, and post-market surveillance that directly impact regulatory approval timelines and compliance costs.
Regulatory challenges specific to active memory technologies include establishing clear boundaries between hardware and software components, demonstrating long-term memory stability under physiological conditions, and validating data security protocols. Agencies increasingly require evidence of cybersecurity resilience, particularly for networked devices with active memory capabilities that store or transmit patient data.
The evolving regulatory environment reflects growing recognition of active memory as a critical device component rather than merely an accessory function. Recent guidance documents from major regulatory bodies emphasize the need for comprehensive validation protocols that address memory performance degradation, data corruption scenarios, and fail-safe mechanisms. This regulatory evolution directly influences development timelines, requiring manufacturers to integrate compliance considerations early in the design phase rather than treating regulation as a final approval hurdle.
Data Security and Privacy in Active Memory Medical Devices
Active memory technologies in biomedical devices introduce unprecedented data security and privacy challenges that require comprehensive protection frameworks. These devices continuously collect, process, and store sensitive patient information including physiological parameters, treatment responses, and behavioral patterns. The persistent nature of active memory systems creates extended vulnerability windows where protected health information remains accessible to potential security breaches.
The regulatory landscape surrounding active memory medical devices encompasses multiple jurisdictions with varying privacy requirements. HIPAA compliance in the United States mandates strict safeguards for electronic protected health information, while the European Union's GDPR imposes additional consent and data minimization obligations. Medical device manufacturers must navigate these complex regulatory frameworks while ensuring that active memory implementations maintain patient data confidentiality throughout the device lifecycle.
Encryption protocols represent the primary defense mechanism for protecting data stored in active memory systems. Advanced encryption standards must be implemented both for data at rest within the memory modules and data in transit during device communications. The challenge lies in balancing robust encryption with the real-time processing requirements of medical devices, as excessive cryptographic overhead can compromise critical therapeutic functions.
Access control mechanisms in active memory devices require multi-layered authentication systems to prevent unauthorized data retrieval. Biometric authentication, secure key management, and role-based access controls must be integrated without compromising device usability for healthcare providers. The persistent nature of active memory necessitates continuous monitoring for unauthorized access attempts and automatic security protocol updates.
Data anonymization and pseudonymization techniques become particularly complex in active memory environments where longitudinal patient data creates unique identification risks. Traditional de-identification methods may prove insufficient when dealing with continuous physiological monitoring data that can reveal patient identities through behavioral patterns and temporal correlations.
The interconnected nature of modern healthcare systems amplifies privacy risks as active memory devices integrate with electronic health records and telemedicine platforms. Secure communication protocols and network segmentation strategies must be implemented to prevent lateral data movement and maintain patient privacy across the entire healthcare ecosystem while preserving the functional benefits of active memory technologies.
The regulatory landscape surrounding active memory medical devices encompasses multiple jurisdictions with varying privacy requirements. HIPAA compliance in the United States mandates strict safeguards for electronic protected health information, while the European Union's GDPR imposes additional consent and data minimization obligations. Medical device manufacturers must navigate these complex regulatory frameworks while ensuring that active memory implementations maintain patient data confidentiality throughout the device lifecycle.
Encryption protocols represent the primary defense mechanism for protecting data stored in active memory systems. Advanced encryption standards must be implemented both for data at rest within the memory modules and data in transit during device communications. The challenge lies in balancing robust encryption with the real-time processing requirements of medical devices, as excessive cryptographic overhead can compromise critical therapeutic functions.
Access control mechanisms in active memory devices require multi-layered authentication systems to prevent unauthorized data retrieval. Biometric authentication, secure key management, and role-based access controls must be integrated without compromising device usability for healthcare providers. The persistent nature of active memory necessitates continuous monitoring for unauthorized access attempts and automatic security protocol updates.
Data anonymization and pseudonymization techniques become particularly complex in active memory environments where longitudinal patient data creates unique identification risks. Traditional de-identification methods may prove insufficient when dealing with continuous physiological monitoring data that can reveal patient identities through behavioral patterns and temporal correlations.
The interconnected nature of modern healthcare systems amplifies privacy risks as active memory devices integrate with electronic health records and telemedicine platforms. Secure communication protocols and network segmentation strategies must be implemented to prevent lateral data movement and maintain patient privacy across the entire healthcare ecosystem while preserving the functional benefits of active memory technologies.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!