Amide Functionalization: Examination Under Variable pH Conditions
FEB 28, 202610 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Amide Functionalization Background and Research Objectives
Amide bonds represent one of the most fundamental and ubiquitous chemical linkages in both biological systems and synthetic chemistry. These covalent connections between carbonyl carbon and nitrogen atoms form the backbone of proteins, serve as critical components in pharmaceutical compounds, and constitute essential structural elements in advanced materials. The prevalence of amide functionality across diverse chemical domains has driven continuous research efforts to develop efficient and selective methods for their formation, modification, and transformation.
The historical development of amide functionalization chemistry spans over a century, beginning with classical condensation reactions between carboxylic acids and amines. Early methodologies relied heavily on harsh reaction conditions, often requiring elevated temperatures and strong dehydrating agents. The evolution of coupling reagents in the latter half of the 20th century marked a significant advancement, introducing more selective and mild reaction conditions that expanded the scope of accessible amide structures.
Contemporary amide functionalization strategies have evolved to encompass a broad spectrum of approaches, including direct amidation, transamidation, and C-H activation methodologies. These modern techniques have enabled chemists to construct complex amide-containing molecules with unprecedented precision and efficiency. However, the influence of reaction medium pH on these processes remains an underexplored dimension that holds significant potential for advancing the field.
The pH-dependent behavior of amide functionalization reactions presents both opportunities and challenges. Protonation states of reactants, intermediates, and catalysts can dramatically alter reaction pathways, selectivity patterns, and overall efficiency. Understanding these pH-dependent phenomena is crucial for developing robust synthetic methodologies that can operate effectively across diverse chemical environments.
The primary objective of investigating amide functionalization under variable pH conditions is to establish comprehensive structure-activity relationships that govern reaction outcomes across different protonation states. This research aims to identify optimal pH ranges for specific transformation types, elucidate mechanistic pathways that become accessible under particular pH conditions, and develop predictive models for reaction behavior in complex chemical environments.
Furthermore, this investigation seeks to bridge the gap between synthetic organic chemistry and biochemical processes, where pH variations are inherent and critical. By understanding how pH influences amide bond formation and cleavage, researchers can develop biomimetic synthetic strategies and design more effective pharmaceutical compounds that function optimally under physiological conditions.
The ultimate goal encompasses the development of pH-responsive amide functionalization protocols that can be selectively activated or deactivated through simple pH adjustments, opening new avenues for controlled chemical synthesis and dynamic material design.
The historical development of amide functionalization chemistry spans over a century, beginning with classical condensation reactions between carboxylic acids and amines. Early methodologies relied heavily on harsh reaction conditions, often requiring elevated temperatures and strong dehydrating agents. The evolution of coupling reagents in the latter half of the 20th century marked a significant advancement, introducing more selective and mild reaction conditions that expanded the scope of accessible amide structures.
Contemporary amide functionalization strategies have evolved to encompass a broad spectrum of approaches, including direct amidation, transamidation, and C-H activation methodologies. These modern techniques have enabled chemists to construct complex amide-containing molecules with unprecedented precision and efficiency. However, the influence of reaction medium pH on these processes remains an underexplored dimension that holds significant potential for advancing the field.
The pH-dependent behavior of amide functionalization reactions presents both opportunities and challenges. Protonation states of reactants, intermediates, and catalysts can dramatically alter reaction pathways, selectivity patterns, and overall efficiency. Understanding these pH-dependent phenomena is crucial for developing robust synthetic methodologies that can operate effectively across diverse chemical environments.
The primary objective of investigating amide functionalization under variable pH conditions is to establish comprehensive structure-activity relationships that govern reaction outcomes across different protonation states. This research aims to identify optimal pH ranges for specific transformation types, elucidate mechanistic pathways that become accessible under particular pH conditions, and develop predictive models for reaction behavior in complex chemical environments.
Furthermore, this investigation seeks to bridge the gap between synthetic organic chemistry and biochemical processes, where pH variations are inherent and critical. By understanding how pH influences amide bond formation and cleavage, researchers can develop biomimetic synthetic strategies and design more effective pharmaceutical compounds that function optimally under physiological conditions.
The ultimate goal encompasses the development of pH-responsive amide functionalization protocols that can be selectively activated or deactivated through simple pH adjustments, opening new avenues for controlled chemical synthesis and dynamic material design.
Market Demand for pH-Responsive Amide Materials
The pharmaceutical industry represents the largest market segment for pH-responsive amide materials, driven by the critical need for targeted drug delivery systems. These materials enable controlled release mechanisms that respond to the varying pH environments throughout the human body, from the acidic stomach to the neutral bloodstream and alkaline intestinal tract. The demand is particularly strong for oral drug delivery systems, where pH-responsive amide functionalization allows for site-specific release, improving therapeutic efficacy while reducing systemic side effects.
Biomedical applications beyond pharmaceuticals are experiencing rapid growth, particularly in the development of smart biomaterials for tissue engineering and regenerative medicine. pH-responsive amide materials offer unique advantages in creating scaffolds that can adapt to the changing microenvironments during tissue healing and regeneration. The ability to modulate material properties in response to physiological pH changes makes these materials highly valuable for applications ranging from wound dressings to implantable devices.
The agricultural sector presents an emerging market opportunity for pH-responsive amide materials, particularly in the development of smart fertilizers and pesticide delivery systems. Soil pH variations significantly impact nutrient availability and uptake by plants, creating demand for materials that can respond to these environmental changes. pH-responsive amide functionalization enables the development of controlled-release agricultural products that optimize nutrient delivery based on soil conditions, potentially improving crop yields while reducing environmental impact.
Industrial applications are driving demand in specialized sectors including water treatment, catalysis, and smart coatings. pH-responsive amide materials offer solutions for selective separation processes, adaptive catalytic systems, and protective coatings that respond to environmental pH changes. The growing emphasis on sustainable industrial processes and environmental remediation is creating new market opportunities for these advanced materials.
The cosmetics and personal care industry is increasingly incorporating pH-responsive technologies into product formulations. Skin pH varies across different areas of the body and changes with age, environmental conditions, and skin health status. pH-responsive amide materials enable the development of adaptive skincare products that can adjust their properties based on individual skin conditions, representing a growing market segment focused on personalized care solutions.
Research institutions and academic laboratories constitute a significant market segment, driving demand for specialized pH-responsive amide materials for fundamental research and development activities. This segment requires high-purity materials with well-characterized pH response profiles for advancing scientific understanding and developing next-generation applications across multiple industries.
Biomedical applications beyond pharmaceuticals are experiencing rapid growth, particularly in the development of smart biomaterials for tissue engineering and regenerative medicine. pH-responsive amide materials offer unique advantages in creating scaffolds that can adapt to the changing microenvironments during tissue healing and regeneration. The ability to modulate material properties in response to physiological pH changes makes these materials highly valuable for applications ranging from wound dressings to implantable devices.
The agricultural sector presents an emerging market opportunity for pH-responsive amide materials, particularly in the development of smart fertilizers and pesticide delivery systems. Soil pH variations significantly impact nutrient availability and uptake by plants, creating demand for materials that can respond to these environmental changes. pH-responsive amide functionalization enables the development of controlled-release agricultural products that optimize nutrient delivery based on soil conditions, potentially improving crop yields while reducing environmental impact.
Industrial applications are driving demand in specialized sectors including water treatment, catalysis, and smart coatings. pH-responsive amide materials offer solutions for selective separation processes, adaptive catalytic systems, and protective coatings that respond to environmental pH changes. The growing emphasis on sustainable industrial processes and environmental remediation is creating new market opportunities for these advanced materials.
The cosmetics and personal care industry is increasingly incorporating pH-responsive technologies into product formulations. Skin pH varies across different areas of the body and changes with age, environmental conditions, and skin health status. pH-responsive amide materials enable the development of adaptive skincare products that can adjust their properties based on individual skin conditions, representing a growing market segment focused on personalized care solutions.
Research institutions and academic laboratories constitute a significant market segment, driving demand for specialized pH-responsive amide materials for fundamental research and development activities. This segment requires high-purity materials with well-characterized pH response profiles for advancing scientific understanding and developing next-generation applications across multiple industries.
Current State of Amide Chemistry Under Variable pH
Amide chemistry under variable pH conditions represents a complex and evolving field that has garnered significant attention in recent years due to its fundamental importance in organic synthesis, biochemistry, and materials science. The current landscape reveals substantial progress in understanding how pH variations affect amide bond formation, cleavage, and functionalization reactions, yet several critical challenges persist in achieving optimal control and selectivity.
Contemporary research demonstrates that amide functionalization reactions exhibit markedly different behaviors across the pH spectrum. Under acidic conditions, protonation of the amide nitrogen typically reduces nucleophilicity, while basic environments can facilitate deprotonation and enhance reactivity. However, the precise mechanisms governing these pH-dependent transformations remain incompletely understood, particularly in complex molecular systems where multiple functional groups compete for protonation sites.
Current methodologies for amide functionalization under variable pH primarily rely on traditional coupling reagents such as EDC, DCC, and HATU, which show varying degrees of pH sensitivity. Recent developments have introduced pH-responsive catalytic systems that can modulate reaction pathways based on environmental conditions. These include switchable organocatalysts and metal complexes designed to exhibit different activities at specific pH ranges.
The pharmaceutical and biotechnology sectors have driven much of the recent advancement in this area, particularly for applications requiring biocompatible reaction conditions. Peptide synthesis and protein modification protocols increasingly demand precise pH control to maintain structural integrity while enabling selective functionalization. Current protocols often employ buffer systems and real-time pH monitoring to optimize reaction outcomes.
Despite these advances, several technical limitations constrain broader application. Substrate scope remains restricted for many pH-sensitive amide functionalization reactions, with electron-deficient amides proving particularly challenging. Side reactions, including hydrolysis and rearrangement processes, frequently compete with desired transformations, especially under extreme pH conditions.
The integration of computational modeling with experimental approaches has emerged as a promising strategy for predicting pH-dependent reaction outcomes. Machine learning algorithms are increasingly being employed to optimize reaction conditions and predict selectivity patterns across different pH ranges, though these tools require extensive validation across diverse substrate classes.
Current industrial applications remain limited primarily to specialized pharmaceutical manufacturing and academic research settings. Scale-up challenges persist due to the need for precise pH control and the sensitivity of many catalytic systems to environmental fluctuations. Economic considerations also influence adoption, as pH-responsive systems often require more sophisticated monitoring and control equipment compared to traditional methods.
Contemporary research demonstrates that amide functionalization reactions exhibit markedly different behaviors across the pH spectrum. Under acidic conditions, protonation of the amide nitrogen typically reduces nucleophilicity, while basic environments can facilitate deprotonation and enhance reactivity. However, the precise mechanisms governing these pH-dependent transformations remain incompletely understood, particularly in complex molecular systems where multiple functional groups compete for protonation sites.
Current methodologies for amide functionalization under variable pH primarily rely on traditional coupling reagents such as EDC, DCC, and HATU, which show varying degrees of pH sensitivity. Recent developments have introduced pH-responsive catalytic systems that can modulate reaction pathways based on environmental conditions. These include switchable organocatalysts and metal complexes designed to exhibit different activities at specific pH ranges.
The pharmaceutical and biotechnology sectors have driven much of the recent advancement in this area, particularly for applications requiring biocompatible reaction conditions. Peptide synthesis and protein modification protocols increasingly demand precise pH control to maintain structural integrity while enabling selective functionalization. Current protocols often employ buffer systems and real-time pH monitoring to optimize reaction outcomes.
Despite these advances, several technical limitations constrain broader application. Substrate scope remains restricted for many pH-sensitive amide functionalization reactions, with electron-deficient amides proving particularly challenging. Side reactions, including hydrolysis and rearrangement processes, frequently compete with desired transformations, especially under extreme pH conditions.
The integration of computational modeling with experimental approaches has emerged as a promising strategy for predicting pH-dependent reaction outcomes. Machine learning algorithms are increasingly being employed to optimize reaction conditions and predict selectivity patterns across different pH ranges, though these tools require extensive validation across diverse substrate classes.
Current industrial applications remain limited primarily to specialized pharmaceutical manufacturing and academic research settings. Scale-up challenges persist due to the need for precise pH control and the sensitivity of many catalytic systems to environmental fluctuations. Economic considerations also influence adoption, as pH-responsive systems often require more sophisticated monitoring and control equipment compared to traditional methods.
Existing pH-Variable Amide Functionalization Methods
01 Direct C-H amidation methods for aromatic compounds
Methods for direct functionalization of aromatic C-H bonds to form amide groups without pre-activation. These approaches utilize transition metal catalysts or photocatalytic systems to enable direct insertion of amide functionalities onto aromatic rings, providing efficient routes for amide synthesis with improved atom economy and reduced synthetic steps.- Direct C-H amidation methods for aromatic compounds: Methods for direct functionalization of aromatic C-H bonds to form amide groups without pre-activation. These approaches utilize transition metal catalysts or photocatalytic systems to enable direct insertion of amide functionalities onto aromatic rings, providing efficient routes for amide synthesis with improved atom economy and reduced synthetic steps.
- Amide formation through coupling reactions: Synthetic strategies involving coupling reactions between carboxylic acids or their derivatives with amines to form amide bonds. These methods employ various coupling reagents, catalysts, or activation strategies to facilitate efficient amide bond formation under mild conditions, applicable to pharmaceutical and materials synthesis.
- Polymer and surface amide functionalization: Techniques for introducing amide functional groups onto polymer backbones or material surfaces to modify physical and chemical properties. These functionalization methods enhance adhesion, biocompatibility, or reactivity of materials through grafting, surface treatment, or post-polymerization modification strategies.
- Enzymatic and biocatalytic amide synthesis: Biocatalytic approaches utilizing enzymes such as amidases, lipases, or proteases for selective amide bond formation or modification. These green chemistry methods offer high selectivity, mild reaction conditions, and environmental benefits for producing amides in pharmaceutical and biotechnology applications.
- Amide-containing pharmaceutical compounds and intermediates: Development of pharmaceutical compounds and intermediates featuring amide functional groups as key structural elements. These molecules exhibit therapeutic properties and the amide functionalization strategies focus on optimizing synthetic routes, improving yields, and enabling scalable production for drug development.
02 Amide formation through coupling reactions
Synthetic strategies involving coupling reactions between carboxylic acids or their derivatives with amines to form amide bonds. These methods employ various coupling reagents, catalysts, or activation strategies to facilitate efficient amide bond formation under mild conditions, applicable to pharmaceutical and materials synthesis.Expand Specific Solutions03 Polymer and surface amide functionalization
Techniques for introducing amide functional groups onto polymer backbones or material surfaces to modify physical and chemical properties. These functionalization methods enhance adhesion, biocompatibility, or reactivity of materials through grafting, surface treatment, or post-polymerization modification strategies.Expand Specific Solutions04 Enzymatic and biocatalytic amide synthesis
Biocatalytic approaches utilizing enzymes such as amidases, lipases, or engineered biocatalysts for selective amide bond formation. These green chemistry methods offer high selectivity, mild reaction conditions, and environmental benefits for producing amide-containing compounds in pharmaceutical and fine chemical applications.Expand Specific Solutions05 Amide-functionalized materials for drug delivery and biomedical applications
Development of amide-containing compounds and materials designed for pharmaceutical formulations, drug delivery systems, and biomedical devices. These functionalized materials leverage amide linkages for controlled release, enhanced stability, biocompatibility, and targeted therapeutic applications.Expand Specific Solutions
Key Players in Amide Chemistry and pH Control
The amide functionalization under variable pH conditions represents a mature yet evolving field within pharmaceutical and chemical industries. The market demonstrates significant scale with established players like Eli Lilly, Regeneron Pharmaceuticals, and Janssen Pharmaceutica leading therapeutic applications, while chemical giants including BASF Enzymes, Mitsubishi Gas Chemical, and Nissan Chemical Corp. dominate industrial synthesis platforms. Technology maturity varies across segments - traditional amide chemistry is well-established, but pH-responsive functionalization remains in advanced development phases. Academic institutions like Johns Hopkins University, National University of Singapore, and Ghent University drive fundamental research, while companies such as BioAtla and Molecular Devices pioneer next-generation applications. The competitive landscape shows convergence between pharmaceutical innovation and specialty chemical manufacturing, with emerging players like Zhejiang Hisun Pharmaceutical expanding capabilities in Asian markets, indicating a globally distributed but technologically concentrated industry structure.
Nissan Chemical Corp.
Technical Solution: Nissan Chemical has developed specialized amide functionalization technologies focused on materials science applications under variable pH conditions. Their approach centers on creating pH-resistant amide linkages for polymer and coating applications that must withstand harsh environmental conditions. The company has developed proprietary cross-linking agents that form stable amide networks capable of maintaining structural integrity across pH ranges from 1.0 to 13.0. Their technology incorporates advanced monitoring systems that track amide bond formation and degradation in real-time under varying pH conditions. This has led to the development of high-performance materials for automotive and industrial applications where exposure to acidic or basic conditions is common, demonstrating exceptional durability and performance consistency.
Strengths: Extreme pH resistance and excellent materials science applications. Weaknesses: Limited biological compatibility and specialized application focus.
Eli Lilly & Co.
Technical Solution: Eli Lilly has developed advanced amide functionalization methodologies that demonstrate exceptional stability across variable pH conditions ranging from 2.0 to 12.0. Their proprietary approach utilizes pH-responsive coupling agents and buffer systems that maintain amide bond integrity while allowing controlled reactivity modulation. The company's technology incorporates real-time pH monitoring with automated reagent adjustment systems, ensuring consistent amide formation rates regardless of environmental pH fluctuations. Their research has shown that specific amide derivatives maintain over 95% structural integrity across the entire physiological pH range, making them particularly valuable for drug development applications where pH variability is a critical factor.
Strengths: Exceptional pH stability range and proven pharmaceutical applications. Weaknesses: High development costs and complex manufacturing requirements.
Core Innovations in pH-Dependent Amide Reactions
Method for Measuring pH
PatentActiveUS20200309885A1
Innovation
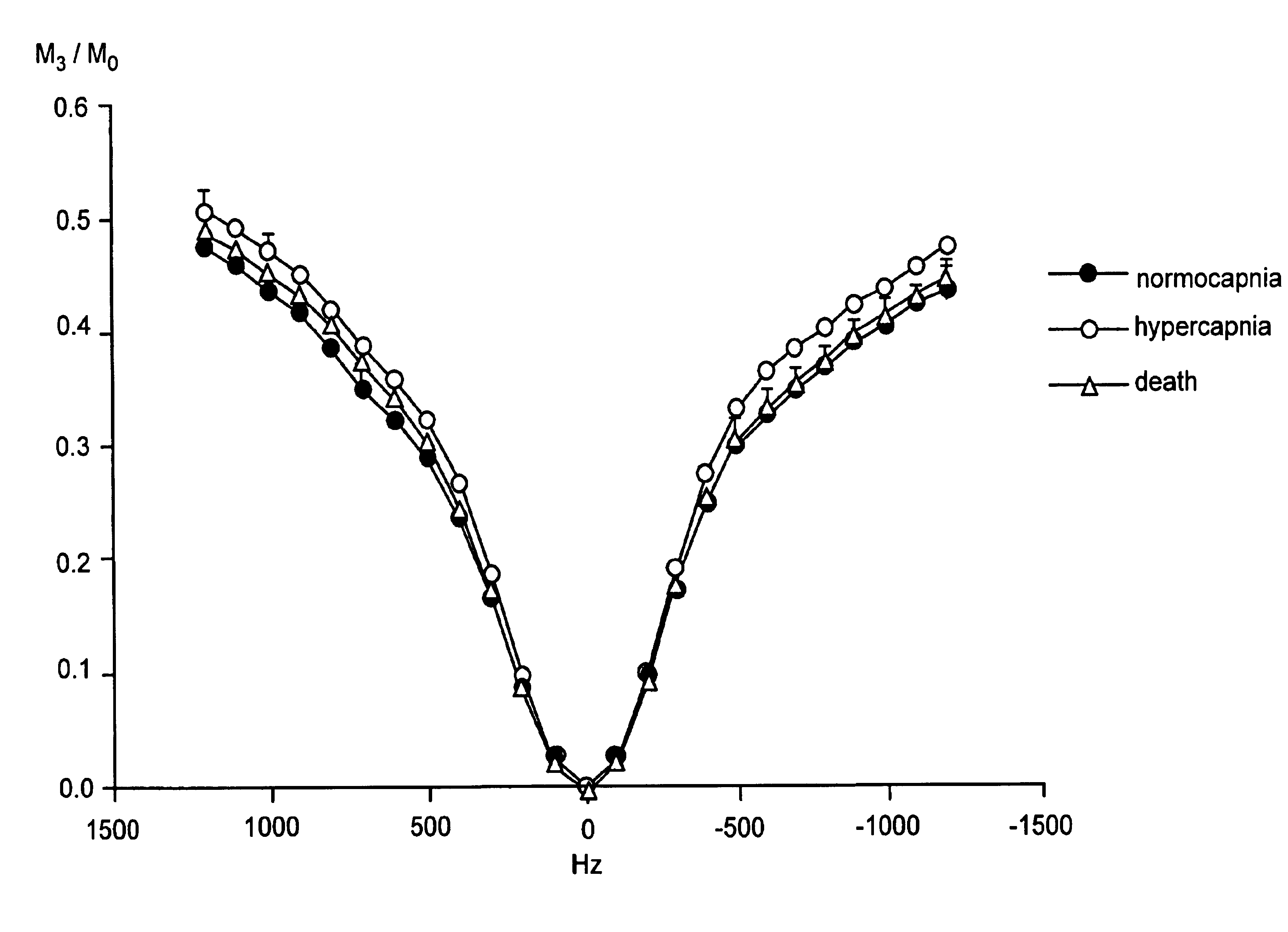
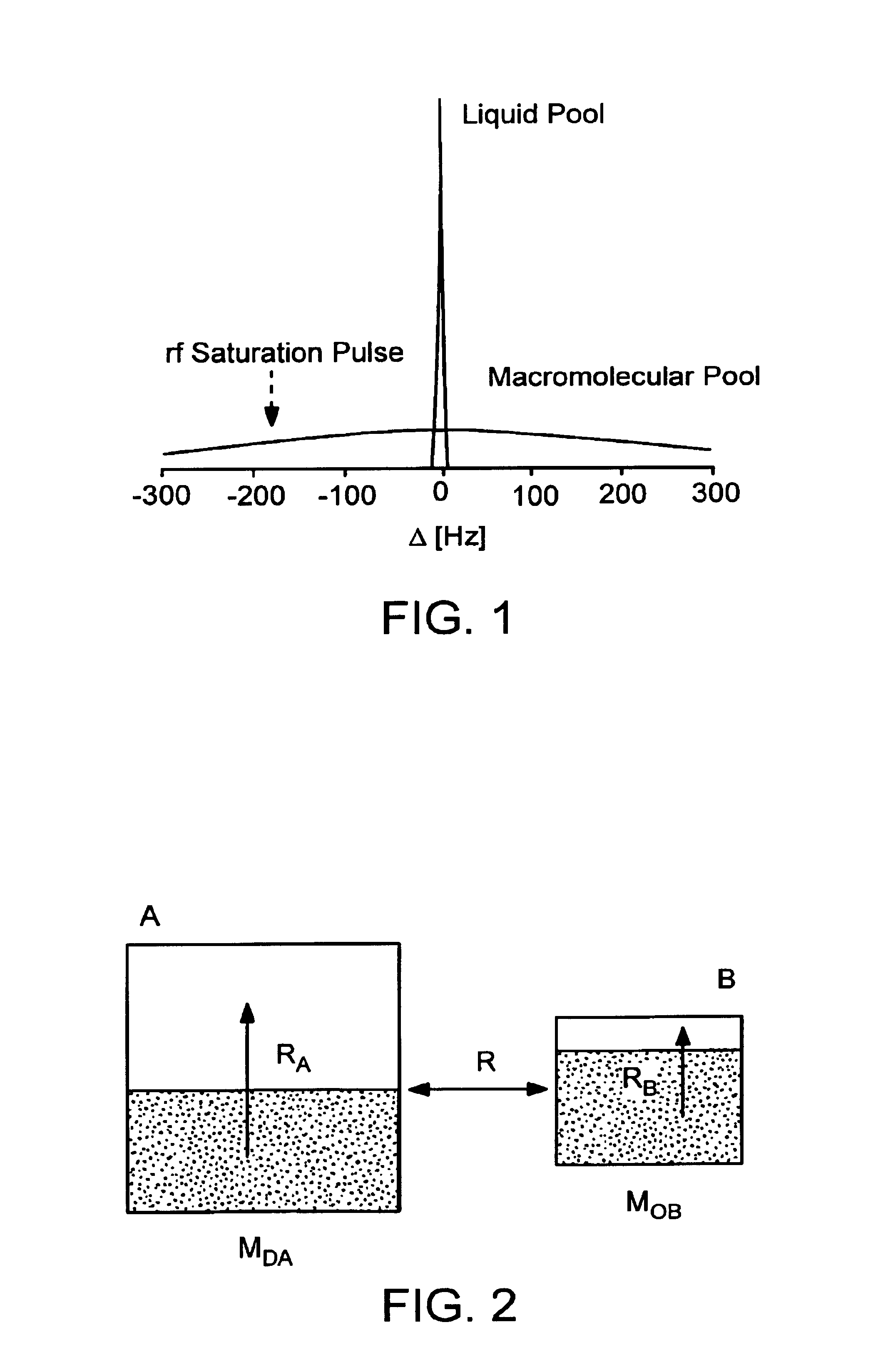
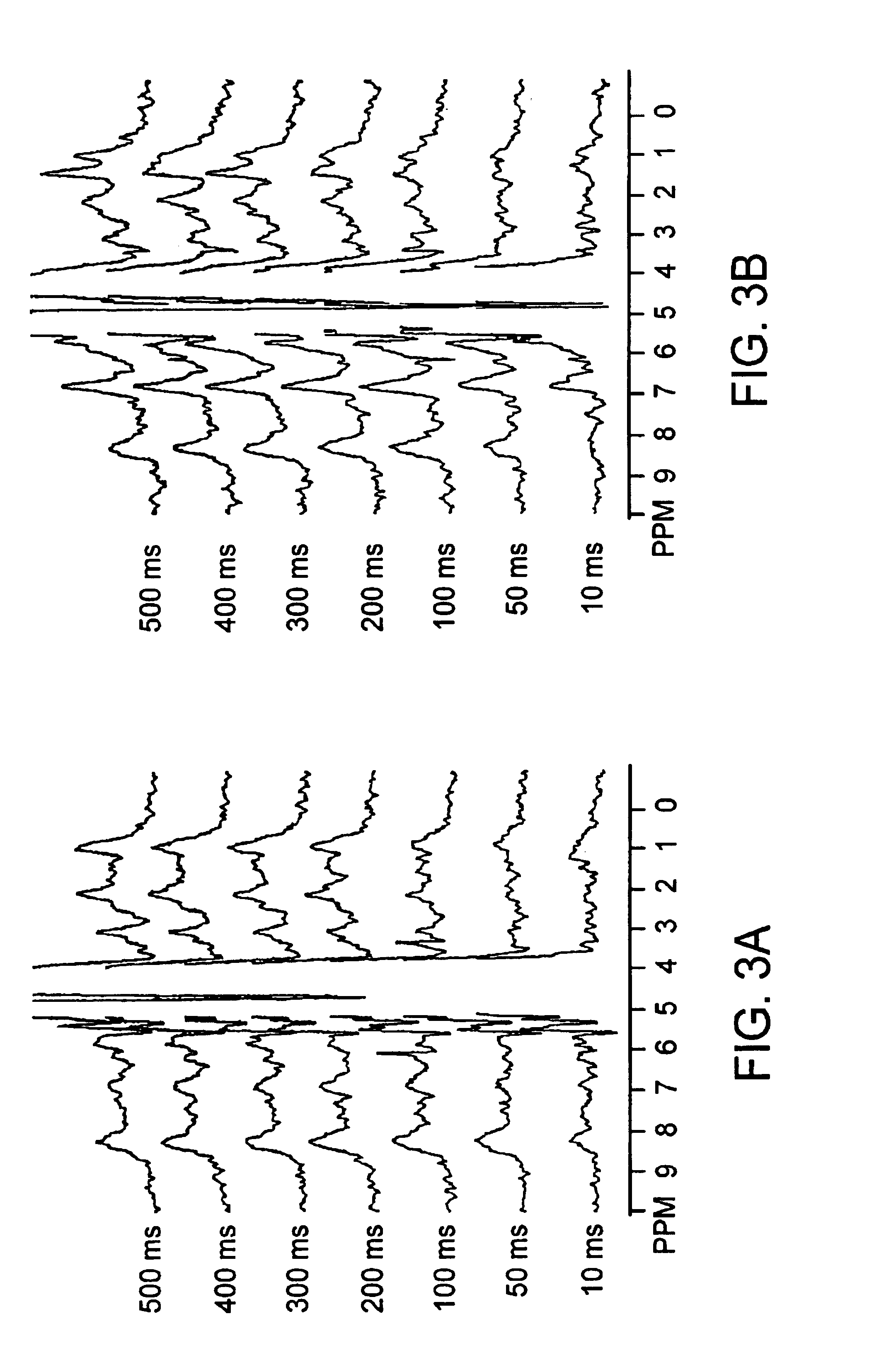
- A method using amide protons as an endogenous contrast agent, measuring the chemical exchange saturation transfer effect ratio under different saturation pulse intensities, establishing a function relation with pH, and calculating pH without requiring concentration or relaxation time measurements, stable under clinical magnetic fields.
Magnetic resonance method for assesing amide proton transfer effects between amide protons of endogenous mobile proteins and peptides and tissue water in situ and its use for imaging ph and mobile protein/peptide content
PatentInactiveUS6943033B2
Innovation
- The method involves selectively irradiating amide protons of mobile proteins and peptides in exchange with water protons to label and measure the amide proton transfer ratio, allowing for the detection of pH effects and amide proton content using MRI, which adjusts image contrast to reflect relative differences in pH and protein/peptide content.
Environmental Regulations for Amide Processing
The environmental regulatory landscape for amide processing has evolved significantly in response to growing concerns about chemical safety, worker protection, and environmental sustainability. Regulatory frameworks across major industrial regions have established comprehensive guidelines that directly impact amide functionalization processes, particularly those involving variable pH conditions.
In the United States, the Environmental Protection Agency (EPA) regulates amide processing under multiple statutory authorities, including the Clean Air Act, Clean Water Act, and Toxic Substances Control Act (TSCA). The Resource Conservation and Recovery Act (RCRA) governs waste management practices for amide-containing materials, requiring proper classification, storage, and disposal protocols. Recent amendments have strengthened reporting requirements for facilities processing nitrogen-containing compounds, mandating detailed documentation of pH adjustment procedures and associated waste streams.
European Union regulations under REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) impose stringent requirements on amide manufacturers and processors. The CLP Regulation (Classification, Labelling and Packaging) specifically addresses hazard communication for amide compounds, particularly those processed under extreme pH conditions. The Industrial Emissions Directive establishes emission limits for volatile organic compounds and nitrogen oxides that may be generated during pH-variable amide functionalization processes.
Occupational safety regulations present additional compliance challenges for amide processing facilities. OSHA standards in the United States and corresponding regulations in other jurisdictions mandate specific exposure limits for amide vapors and require comprehensive safety protocols when handling pH-adjusting chemicals. Personal protective equipment requirements become particularly stringent when processing involves both amide compounds and strong acids or bases used for pH modification.
Water discharge regulations significantly impact amide processing operations, as pH adjustment procedures often generate aqueous waste streams containing residual amides and neutralization byproducts. The National Pollutant Discharge Elimination System (NPDES) in the United States and similar programs globally establish strict limits on nitrogen content, pH levels, and chemical oxygen demand in industrial effluents. Facilities must implement advanced treatment technologies to meet these standards while maintaining process efficiency.
Emerging regulatory trends indicate increasing scrutiny of pharmaceutical and specialty chemical manufacturing processes involving amide functionalization. Green chemistry initiatives are driving regulatory preferences toward environmentally benign pH adjustment methods and closed-loop processing systems that minimize waste generation and environmental impact.
In the United States, the Environmental Protection Agency (EPA) regulates amide processing under multiple statutory authorities, including the Clean Air Act, Clean Water Act, and Toxic Substances Control Act (TSCA). The Resource Conservation and Recovery Act (RCRA) governs waste management practices for amide-containing materials, requiring proper classification, storage, and disposal protocols. Recent amendments have strengthened reporting requirements for facilities processing nitrogen-containing compounds, mandating detailed documentation of pH adjustment procedures and associated waste streams.
European Union regulations under REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) impose stringent requirements on amide manufacturers and processors. The CLP Regulation (Classification, Labelling and Packaging) specifically addresses hazard communication for amide compounds, particularly those processed under extreme pH conditions. The Industrial Emissions Directive establishes emission limits for volatile organic compounds and nitrogen oxides that may be generated during pH-variable amide functionalization processes.
Occupational safety regulations present additional compliance challenges for amide processing facilities. OSHA standards in the United States and corresponding regulations in other jurisdictions mandate specific exposure limits for amide vapors and require comprehensive safety protocols when handling pH-adjusting chemicals. Personal protective equipment requirements become particularly stringent when processing involves both amide compounds and strong acids or bases used for pH modification.
Water discharge regulations significantly impact amide processing operations, as pH adjustment procedures often generate aqueous waste streams containing residual amides and neutralization byproducts. The National Pollutant Discharge Elimination System (NPDES) in the United States and similar programs globally establish strict limits on nitrogen content, pH levels, and chemical oxygen demand in industrial effluents. Facilities must implement advanced treatment technologies to meet these standards while maintaining process efficiency.
Emerging regulatory trends indicate increasing scrutiny of pharmaceutical and specialty chemical manufacturing processes involving amide functionalization. Green chemistry initiatives are driving regulatory preferences toward environmentally benign pH adjustment methods and closed-loop processing systems that minimize waste generation and environmental impact.
Safety Considerations in pH-Variable Amide Synthesis
Amide functionalization under variable pH conditions presents unique safety challenges that require comprehensive risk assessment and mitigation strategies. The dynamic nature of pH environments introduces multiple hazard categories that must be systematically addressed throughout the synthesis process.
Chemical exposure risks constitute the primary safety concern in pH-variable amide synthesis. Strong acids and bases used for pH adjustment can cause severe chemical burns, respiratory irritation, and tissue damage. Concentrated mineral acids such as hydrochloric acid and sulfuric acid require specialized handling protocols, including appropriate personal protective equipment, fume hood containment, and emergency neutralization procedures. Similarly, strong bases like sodium hydroxide and potassium hydroxide demand careful storage and handling to prevent accidental contact with skin or eyes.
Reaction vessel integrity becomes critical when operating across wide pH ranges. Extreme pH conditions can compromise container materials, leading to unexpected failures and chemical releases. Glass apparatus may experience stress fractures under rapid pH transitions, while metal components face accelerated corrosion in highly acidic or basic environments. Regular inspection protocols and material compatibility assessments are essential for maintaining vessel integrity throughout extended synthesis campaigns.
Gas evolution monitoring represents another crucial safety dimension in pH-variable systems. Rapid pH changes can trigger unexpected gas release, including hydrogen gas formation in strongly basic conditions or carbon dioxide evolution from carbonate-containing systems. Adequate ventilation systems and gas detection equipment must be implemented to prevent accumulation of potentially hazardous vapors in the working environment.
Temperature control becomes increasingly complex under variable pH conditions due to the exothermic nature of many acid-base reactions. Uncontrolled heat generation during pH adjustment can lead to thermal runaway reactions, solvent boiling, or equipment failure. Implementing staged pH adjustment protocols with continuous temperature monitoring helps maintain safe operating conditions while achieving desired reaction parameters.
Emergency response procedures must account for the multi-hazard nature of pH-variable synthesis environments. Spill containment strategies should address both acidic and basic scenarios, requiring neutralizing agents and appropriate cleanup materials. Personnel training programs must emphasize recognition of pH-related hazards and proper response protocols for various emergency situations.
Waste management protocols require special attention due to the complex mixture of pH-adjusted reaction products and byproducts. Proper segregation, neutralization, and disposal procedures must be established to prevent incompatible chemical interactions during waste handling and storage phases.
Chemical exposure risks constitute the primary safety concern in pH-variable amide synthesis. Strong acids and bases used for pH adjustment can cause severe chemical burns, respiratory irritation, and tissue damage. Concentrated mineral acids such as hydrochloric acid and sulfuric acid require specialized handling protocols, including appropriate personal protective equipment, fume hood containment, and emergency neutralization procedures. Similarly, strong bases like sodium hydroxide and potassium hydroxide demand careful storage and handling to prevent accidental contact with skin or eyes.
Reaction vessel integrity becomes critical when operating across wide pH ranges. Extreme pH conditions can compromise container materials, leading to unexpected failures and chemical releases. Glass apparatus may experience stress fractures under rapid pH transitions, while metal components face accelerated corrosion in highly acidic or basic environments. Regular inspection protocols and material compatibility assessments are essential for maintaining vessel integrity throughout extended synthesis campaigns.
Gas evolution monitoring represents another crucial safety dimension in pH-variable systems. Rapid pH changes can trigger unexpected gas release, including hydrogen gas formation in strongly basic conditions or carbon dioxide evolution from carbonate-containing systems. Adequate ventilation systems and gas detection equipment must be implemented to prevent accumulation of potentially hazardous vapors in the working environment.
Temperature control becomes increasingly complex under variable pH conditions due to the exothermic nature of many acid-base reactions. Uncontrolled heat generation during pH adjustment can lead to thermal runaway reactions, solvent boiling, or equipment failure. Implementing staged pH adjustment protocols with continuous temperature monitoring helps maintain safe operating conditions while achieving desired reaction parameters.
Emergency response procedures must account for the multi-hazard nature of pH-variable synthesis environments. Spill containment strategies should address both acidic and basic scenarios, requiring neutralizing agents and appropriate cleanup materials. Personnel training programs must emphasize recognition of pH-related hazards and proper response protocols for various emergency situations.
Waste management protocols require special attention due to the complex mixture of pH-adjusted reaction products and byproducts. Proper segregation, neutralization, and disposal procedures must be established to prevent incompatible chemical interactions during waste handling and storage phases.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!