Amide vs Acetals: Comparing Polarity Effects in Performance
FEB 28, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Amide vs Acetals Polarity Background and Research Goals
The fundamental distinction between amides and acetals lies in their molecular polarity characteristics, which significantly influence their performance across various applications. Amides, characterized by the carbonyl group bonded to nitrogen (R-CO-NR2), exhibit strong polar properties due to resonance between the carbonyl oxygen and nitrogen atom. This resonance creates partial charges that enable extensive hydrogen bonding and dipole-dipole interactions. Conversely, acetals feature two alkoxy groups attached to a central carbon atom (R-CH(OR')2), displaying considerably lower polarity due to the absence of highly electronegative atoms in direct conjugation.
The polarity differential between these functional groups has profound implications for material properties, including solubility, thermal stability, mechanical strength, and chemical reactivity. In polymer science, amide-containing materials like nylon demonstrate exceptional tensile strength and thermal resistance attributed to intermolecular hydrogen bonding networks. Meanwhile, acetal-based polymers such as polyoxymethylene exhibit superior dimensional stability and low friction coefficients, largely due to their reduced intermolecular interactions.
Historical development of both chemical classes reveals distinct evolutionary paths driven by their inherent polarity differences. Amide chemistry emerged prominently in the early 20th century with the development of synthetic fibers, where strong intermolecular forces were essential for fiber strength. Acetal chemistry gained prominence later, particularly in precision engineering applications where dimensional stability and chemical inertness were prioritized over mechanical strength.
Contemporary research objectives focus on leveraging these polarity differences to optimize performance in specific applications. Key research goals include developing hybrid materials that combine the mechanical advantages of amides with the stability characteristics of acetals. Additionally, investigations into polarity modification through molecular design aim to fine-tune properties for specialized applications in electronics, biomedical devices, and advanced composites.
Understanding the relationship between molecular polarity and macroscopic performance represents a critical foundation for advancing material science applications. This comparative analysis seeks to establish quantitative correlations between polarity parameters and performance metrics, enabling predictive design of next-generation materials with tailored properties for emerging technological demands.
The polarity differential between these functional groups has profound implications for material properties, including solubility, thermal stability, mechanical strength, and chemical reactivity. In polymer science, amide-containing materials like nylon demonstrate exceptional tensile strength and thermal resistance attributed to intermolecular hydrogen bonding networks. Meanwhile, acetal-based polymers such as polyoxymethylene exhibit superior dimensional stability and low friction coefficients, largely due to their reduced intermolecular interactions.
Historical development of both chemical classes reveals distinct evolutionary paths driven by their inherent polarity differences. Amide chemistry emerged prominently in the early 20th century with the development of synthetic fibers, where strong intermolecular forces were essential for fiber strength. Acetal chemistry gained prominence later, particularly in precision engineering applications where dimensional stability and chemical inertness were prioritized over mechanical strength.
Contemporary research objectives focus on leveraging these polarity differences to optimize performance in specific applications. Key research goals include developing hybrid materials that combine the mechanical advantages of amides with the stability characteristics of acetals. Additionally, investigations into polarity modification through molecular design aim to fine-tune properties for specialized applications in electronics, biomedical devices, and advanced composites.
Understanding the relationship between molecular polarity and macroscopic performance represents a critical foundation for advancing material science applications. This comparative analysis seeks to establish quantitative correlations between polarity parameters and performance metrics, enabling predictive design of next-generation materials with tailored properties for emerging technological demands.
Market Demand for Polarity-Optimized Chemical Solutions
The pharmaceutical industry represents the largest market segment driving demand for polarity-optimized chemical solutions, particularly in the comparison between amide and acetal functionalities. Drug formulation scientists increasingly require precise control over molecular polarity to optimize bioavailability, solubility, and therapeutic efficacy. The growing emphasis on personalized medicine and targeted drug delivery systems has intensified the need for chemical compounds with tailored polarity profiles that can enhance drug-target interactions and improve pharmacokinetic properties.
Advanced materials manufacturing constitutes another significant market driver, where polarity effects directly influence material performance characteristics. Industries producing high-performance polymers, coatings, and adhesives demand chemical solutions that can fine-tune intermolecular interactions through strategic polarity optimization. The electronics sector particularly values compounds that can modulate dielectric properties and surface interactions, making the amide versus acetal polarity comparison crucial for next-generation electronic materials.
The agrochemical market demonstrates substantial growth in demand for polarity-optimized formulations that enhance pesticide and herbicide effectiveness. Agricultural companies seek chemical solutions that can improve soil penetration, plant uptake, and environmental stability through precise polarity control. The increasing focus on sustainable agriculture and reduced environmental impact has created opportunities for innovative chemical designs that leverage polarity differences to minimize off-target effects while maximizing agricultural productivity.
Cosmetics and personal care industries represent emerging markets for polarity-optimized solutions, where consumer preferences drive demand for products with enhanced skin compatibility and performance. Formulators require chemical compounds that can optimize texture, absorption, and stability through careful polarity engineering. The trend toward clean beauty and biocompatible ingredients has created new opportunities for amide and acetal-based solutions that offer superior performance while meeting safety and sustainability requirements.
The specialty chemicals sector shows robust demand for polarity-optimized solutions across diverse applications including catalysis, separation processes, and surface modification. Industrial manufacturers increasingly recognize that polarity optimization can significantly improve process efficiency, product quality, and operational costs. Market growth is particularly strong in applications requiring precise molecular recognition, selective binding, and controlled release mechanisms where polarity effects play determining roles in overall system performance.
Advanced materials manufacturing constitutes another significant market driver, where polarity effects directly influence material performance characteristics. Industries producing high-performance polymers, coatings, and adhesives demand chemical solutions that can fine-tune intermolecular interactions through strategic polarity optimization. The electronics sector particularly values compounds that can modulate dielectric properties and surface interactions, making the amide versus acetal polarity comparison crucial for next-generation electronic materials.
The agrochemical market demonstrates substantial growth in demand for polarity-optimized formulations that enhance pesticide and herbicide effectiveness. Agricultural companies seek chemical solutions that can improve soil penetration, plant uptake, and environmental stability through precise polarity control. The increasing focus on sustainable agriculture and reduced environmental impact has created opportunities for innovative chemical designs that leverage polarity differences to minimize off-target effects while maximizing agricultural productivity.
Cosmetics and personal care industries represent emerging markets for polarity-optimized solutions, where consumer preferences drive demand for products with enhanced skin compatibility and performance. Formulators require chemical compounds that can optimize texture, absorption, and stability through careful polarity engineering. The trend toward clean beauty and biocompatible ingredients has created new opportunities for amide and acetal-based solutions that offer superior performance while meeting safety and sustainability requirements.
The specialty chemicals sector shows robust demand for polarity-optimized solutions across diverse applications including catalysis, separation processes, and surface modification. Industrial manufacturers increasingly recognize that polarity optimization can significantly improve process efficiency, product quality, and operational costs. Market growth is particularly strong in applications requiring precise molecular recognition, selective binding, and controlled release mechanisms where polarity effects play determining roles in overall system performance.
Current Polarity Performance Challenges in Amide-Acetal Systems
The fundamental challenge in amide-acetal systems lies in the inherent polarity mismatch between these two functional groups, which creates significant performance limitations across multiple applications. Amides exhibit strong dipole moments due to resonance between nitrogen lone pairs and carbonyl groups, while acetals demonstrate relatively lower polarity with their ether-like characteristics. This polarity differential leads to phase separation issues, reduced mechanical properties, and compromised chemical stability in composite materials.
Solubility incompatibility represents a critical bottleneck in current amide-acetal formulations. The highly polar nature of amides, particularly those with hydrogen bonding capabilities, creates thermodynamic barriers when combined with less polar acetal components. This incompatibility manifests as precipitation, crystallization, or phase domain formation, ultimately degrading material homogeneity and performance consistency.
Interfacial adhesion problems emerge when amide and acetal phases coexist in polymer blends or composite structures. The polarity mismatch results in weak interfacial bonding, leading to stress concentration points and premature failure under mechanical loading. Current systems often require expensive compatibilizers or surface modification treatments to achieve acceptable interfacial properties.
Thermal stability challenges arise from the different degradation mechanisms of polar amides versus less polar acetals. Amides typically undergo hydrolysis and thermal decomposition at elevated temperatures, while acetals are susceptible to acid-catalyzed cleavage. The polarity differences affect heat transfer rates and thermal expansion coefficients, creating internal stresses during temperature cycling.
Processing difficulties in manufacturing represent another significant challenge. The polarity variations between amide and acetal components affect melt viscosity, flow behavior, and processing temperatures. These differences complicate injection molding, extrusion, and other manufacturing processes, often requiring specialized equipment or modified processing parameters that increase production costs and complexity.
Chemical reactivity mismatches further compound performance issues. The electron-rich nature of amides makes them susceptible to oxidation and nucleophilic attack, while acetals are more prone to acid-catalyzed degradation. These different chemical vulnerabilities create challenges in formulating protective additives and stabilization systems that effectively protect both components simultaneously.
Solubility incompatibility represents a critical bottleneck in current amide-acetal formulations. The highly polar nature of amides, particularly those with hydrogen bonding capabilities, creates thermodynamic barriers when combined with less polar acetal components. This incompatibility manifests as precipitation, crystallization, or phase domain formation, ultimately degrading material homogeneity and performance consistency.
Interfacial adhesion problems emerge when amide and acetal phases coexist in polymer blends or composite structures. The polarity mismatch results in weak interfacial bonding, leading to stress concentration points and premature failure under mechanical loading. Current systems often require expensive compatibilizers or surface modification treatments to achieve acceptable interfacial properties.
Thermal stability challenges arise from the different degradation mechanisms of polar amides versus less polar acetals. Amides typically undergo hydrolysis and thermal decomposition at elevated temperatures, while acetals are susceptible to acid-catalyzed cleavage. The polarity differences affect heat transfer rates and thermal expansion coefficients, creating internal stresses during temperature cycling.
Processing difficulties in manufacturing represent another significant challenge. The polarity variations between amide and acetal components affect melt viscosity, flow behavior, and processing temperatures. These differences complicate injection molding, extrusion, and other manufacturing processes, often requiring specialized equipment or modified processing parameters that increase production costs and complexity.
Chemical reactivity mismatches further compound performance issues. The electron-rich nature of amides makes them susceptible to oxidation and nucleophilic attack, while acetals are more prone to acid-catalyzed degradation. These different chemical vulnerabilities create challenges in formulating protective additives and stabilization systems that effectively protect both components simultaneously.
Existing Polarity Comparison Methods and Solutions
01 Amide compounds as polar solvents in chemical synthesis
Amide compounds exhibit significant polarity due to the carbonyl group and nitrogen atom, making them effective polar aprotic solvents in various chemical reactions. Their polarity facilitates dissolution of polar reactants and intermediates, enhancing reaction rates and selectivity. The electron-withdrawing nature of the carbonyl group combined with the electron-donating amino group creates a dipole moment that influences solubility and reactivity patterns in synthetic processes.- Amide compounds as polar solvents in chemical synthesis: Amide compounds exhibit significant polarity due to the carbonyl group and nitrogen atom, making them effective polar aprotic solvents in various chemical reactions. Their polarity facilitates dissolution of polar reactants and intermediates, enhancing reaction rates and selectivity. The electron-withdrawing nature of the carbonyl group combined with the electron-donating amino group creates a dipole moment that influences solubility and reactivity patterns in synthetic processes.
- Acetal formation and stability influenced by polarity: Acetals are formed through the reaction of aldehydes or ketones with alcohols, and their stability is significantly affected by the polarity of the reaction medium. In polar environments, acetal formation equilibrium shifts, affecting both the rate of formation and hydrolysis. The polarity of the solvent system influences the protection and deprotection strategies in organic synthesis, where acetals serve as protecting groups for carbonyl functionalities.
- Polarity effects on amide bond formation and stability: The formation of amide bonds is influenced by the polarity of the reaction environment, affecting both coupling efficiency and product stability. Polar solvents can stabilize charged intermediates during amide bond formation, while the inherent polarity of the amide group itself contributes to hydrogen bonding networks. These polarity effects are crucial in peptide synthesis and polymer chemistry, where amide linkages form the backbone of macromolecular structures.
- Acetal protecting groups in polar reaction conditions: Acetals function as protecting groups for carbonyl compounds, with their effectiveness dependent on the polarity of reaction conditions. The stability of acetal protecting groups varies with solvent polarity, pH, and temperature, allowing selective protection and deprotection in multi-step syntheses. Understanding polarity effects enables optimization of reaction conditions for maintaining acetal integrity during transformations of other functional groups.
- Solvent polarity effects on amide and acetal reactivity: The reactivity of both amides and acetals is modulated by solvent polarity, which affects reaction mechanisms, transition state stabilization, and product distribution. Polar solvents can enhance nucleophilic attack on carbonyl carbons while also influencing the equilibrium between different tautomeric or conformational forms. Selection of appropriate solvent polarity is critical for controlling reaction pathways and achieving desired selectivity in transformations involving these functional groups.
02 Acetal formation and stability influenced by polarity
Acetals are formed through the reaction of aldehydes or ketones with alcohols, and their stability is significantly affected by the polarity of the reaction medium. The polarity of the environment influences the equilibrium between acetal formation and hydrolysis. In less polar environments, acetals tend to be more stable, while polar protic solvents can promote hydrolysis. The protective group strategy utilizing acetals relies on controlling polarity conditions to maintain or cleave these functional groups selectively.Expand Specific Solutions03 Polarity effects on amide bond formation and peptide synthesis
The polarity of reaction media plays a crucial role in amide bond formation, particularly in peptide synthesis. Polar solvents can stabilize charged intermediates and transition states, affecting coupling efficiency and racemization rates. The choice of solvent polarity influences the activation of carboxylic acids and the nucleophilicity of amines, thereby controlling the kinetics and thermodynamics of peptide bond formation. Solvent polarity also affects side reactions and product purity in complex peptide assemblies.Expand Specific Solutions04 Acetal protecting groups in polar and non-polar environments
Acetals serve as important protecting groups for carbonyl compounds, with their effectiveness dependent on environmental polarity. The installation and removal of acetal protecting groups require careful control of reaction polarity. Non-polar conditions favor acetal stability, making them suitable for protecting aldehydes and ketones during reactions involving strong nucleophiles or bases. The selective deprotection can be achieved by adjusting solvent polarity or using acidic conditions in polar media, enabling orthogonal protection strategies in multi-step synthesis.Expand Specific Solutions05 Solvent polarity effects on amide and acetal reactivity in pharmaceutical applications
In pharmaceutical chemistry, the polarity of solvents significantly impacts the reactivity and stability of both amide and acetal functional groups. Polar solvents can enhance the solubility of drug intermediates containing these groups, facilitating purification and formulation processes. The polarity also affects the bioavailability and metabolic stability of pharmaceutical compounds containing amide or acetal moieties. Understanding polarity effects enables optimization of synthetic routes and formulation strategies for improved drug efficacy and stability.Expand Specific Solutions
Key Players in Amide and Acetal Chemical Industry
The competitive landscape for amide versus acetal polarity effects in performance represents a mature technology domain within the advanced materials and specialty chemicals industry. The market demonstrates significant scale, driven by established chemical giants including BASF Corp., DuPont de Nemours, and Dow Global Technologies LLC, alongside specialized players like Covestro Deutschland AG and The Lubrizol Corp. Technology maturity varies across applications, with companies like Asahi Kasei Corp., Sekisui Chemical, and Polyplastics Co. Ltd. leading in polymer engineering, while Axalta Coating Systems and Sika Technology AG focus on coating formulations. The industry shows consolidation trends with major corporations acquiring specialized capabilities, as evidenced by Genentech's integration with Roche and emerging players like LG Energy Solution entering battery applications. Academic institutions including University of York and Shenzhen University contribute fundamental research, indicating ongoing innovation potential despite the technology's established nature.
DuPont de Nemours, Inc.
Technical Solution: DuPont has developed advanced polymer chemistry solutions that leverage both amide and acetal functionalities to optimize polarity effects in high-performance materials. Their approach focuses on engineering molecular structures where amide groups provide strong intermolecular hydrogen bonding for enhanced mechanical properties, while acetal linkages offer controlled polarity and improved processability. The company's proprietary polymer platforms utilize strategic placement of these functional groups to achieve targeted polarity gradients, enabling superior performance in demanding applications such as automotive components, electronics packaging, and industrial membranes. Their research demonstrates that careful balance of amide-acetal ratios can significantly impact material properties including thermal stability, chemical resistance, and mechanical strength.
Strengths: Extensive R&D capabilities and proven track record in advanced polymer chemistry with strong intellectual property portfolio. Weaknesses: High development costs and longer time-to-market for specialized formulations.
BASF Corp.
Technical Solution: BASF has developed comprehensive solutions for optimizing polarity effects through their advanced chemical intermediates and polymer additives portfolio. Their technology platform focuses on modifying amide and acetal structures to achieve desired polarity characteristics in various applications. The company's approach includes developing specialized catalysts and reaction conditions that enable precise control over functional group incorporation and distribution. Their research shows that systematic variation of amide versus acetal content can dramatically influence material properties such as adhesion, compatibility, and performance under different environmental conditions. BASF's solutions are particularly effective in coatings, adhesives, and specialty polymer applications where polarity control is critical for performance optimization.
Strengths: Global scale operations with extensive technical support and broad application expertise across multiple industries. Weaknesses: Complex product portfolio may lead to longer customer decision cycles and higher technical service requirements.
Core Innovations in Polarity-Performance Relationship Studies
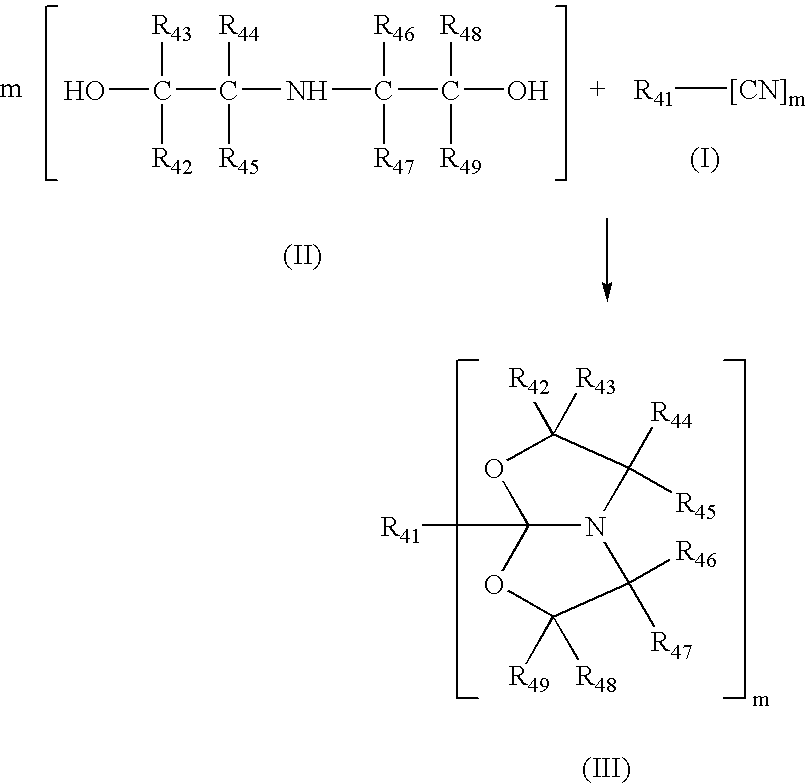
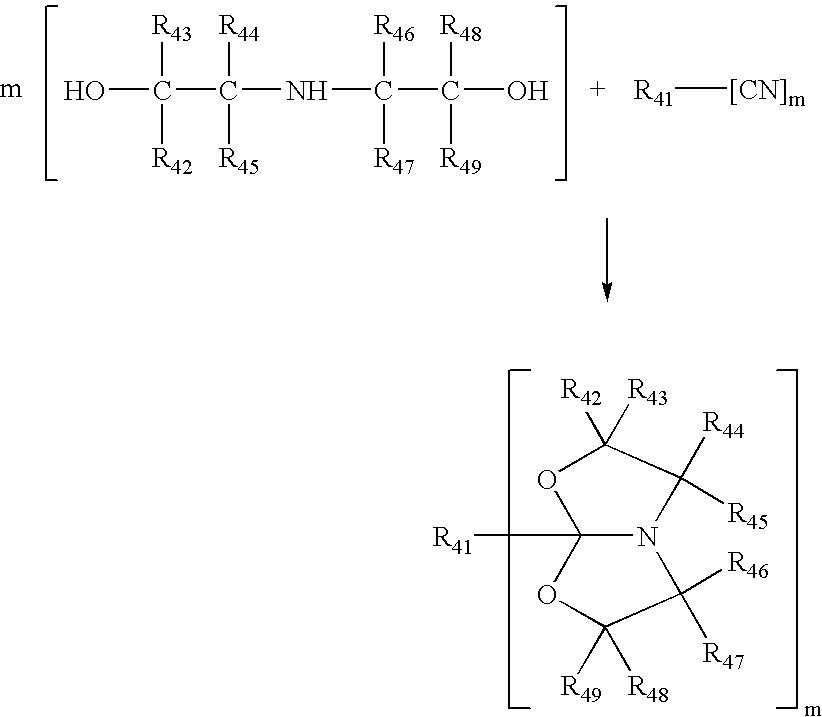
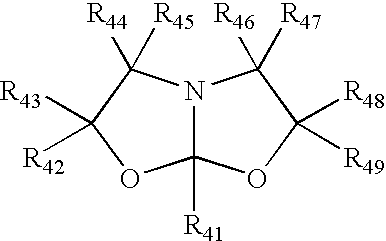
Process for making amide acetals
PatentInactiveUS7230112B2
Innovation
- A catalytic process involving the reaction of organic nitriles with dialkanol amines in the presence of specific metal catalysts, such as scandium, titanium, or zinc, to produce amide acetals that can be rapidly crosslinked via hydrolysis, minimizing volatile organic compound emissions and achieving high conversion rates.
(Meth)acrylate amide acetals
PatentActiveUS20060128774A1
Innovation
- A process involving the reaction of hydroxy amide acetals with methacryloyl chloride, ester exchange with methyl (meth)acrylate, or (meth)acrylic anhydride in the presence of a base, such as triethylamine or pyridine, to produce (meth)acrylate amide acetals, which can be used in coatings for rapid drying and cross-linking without VOC emissions.
Environmental Impact of Amide vs Acetal Applications
The environmental implications of amide and acetal applications present distinct profiles that significantly influence their adoption across various industrial sectors. Amide-based compounds, particularly synthetic polymers like nylon and polyacrylamide, demonstrate complex environmental behaviors throughout their lifecycle. These materials typically exhibit slower biodegradation rates due to their stable molecular structures, leading to persistent environmental accumulation. However, their superior mechanical properties often result in longer product lifespans, potentially offsetting disposal concerns through extended utility periods.
Manufacturing processes for amide compounds generally require higher energy inputs and often involve the use of organic solvents and catalysts that may pose environmental risks. The production of polyamides, for instance, generates significant carbon emissions and requires careful management of nitrogen-containing waste streams. Additionally, the disposal of amide-based materials through incineration can release nitrogen oxides, contributing to air quality concerns and requiring advanced emission control systems.
Acetal compounds present a contrasting environmental profile characterized by different degradation pathways and manufacturing impacts. Polyoxymethylene (POM) and other acetal polymers typically demonstrate more predictable thermal decomposition patterns, primarily yielding formaldehyde upon breakdown. While this presents specific handling challenges, it also enables more controlled end-of-life management strategies. The manufacturing of acetal compounds often involves fewer toxic intermediates compared to amide production, though formaldehyde emissions during processing require stringent control measures.
The recyclability characteristics of these material classes differ substantially, with acetals generally showing better reprocessing stability due to their resistance to hydrolysis under controlled conditions. Amide materials, while technically recyclable, often experience property degradation during mechanical recycling processes, limiting their circular economy potential. Chemical recycling approaches for both material types are emerging, though acetals demonstrate more promising depolymerization pathways.
Regulatory frameworks increasingly favor materials with lower environmental persistence and reduced toxicity profiles. This trend particularly impacts long-term strategic decisions regarding material selection, as environmental compliance costs and restrictions continue to evolve. The comparative assessment reveals that while neither material class is inherently superior from an environmental perspective, their specific applications and end-of-life management strategies significantly influence their overall environmental impact profiles.
Manufacturing processes for amide compounds generally require higher energy inputs and often involve the use of organic solvents and catalysts that may pose environmental risks. The production of polyamides, for instance, generates significant carbon emissions and requires careful management of nitrogen-containing waste streams. Additionally, the disposal of amide-based materials through incineration can release nitrogen oxides, contributing to air quality concerns and requiring advanced emission control systems.
Acetal compounds present a contrasting environmental profile characterized by different degradation pathways and manufacturing impacts. Polyoxymethylene (POM) and other acetal polymers typically demonstrate more predictable thermal decomposition patterns, primarily yielding formaldehyde upon breakdown. While this presents specific handling challenges, it also enables more controlled end-of-life management strategies. The manufacturing of acetal compounds often involves fewer toxic intermediates compared to amide production, though formaldehyde emissions during processing require stringent control measures.
The recyclability characteristics of these material classes differ substantially, with acetals generally showing better reprocessing stability due to their resistance to hydrolysis under controlled conditions. Amide materials, while technically recyclable, often experience property degradation during mechanical recycling processes, limiting their circular economy potential. Chemical recycling approaches for both material types are emerging, though acetals demonstrate more promising depolymerization pathways.
Regulatory frameworks increasingly favor materials with lower environmental persistence and reduced toxicity profiles. This trend particularly impacts long-term strategic decisions regarding material selection, as environmental compliance costs and restrictions continue to evolve. The comparative assessment reveals that while neither material class is inherently superior from an environmental perspective, their specific applications and end-of-life management strategies significantly influence their overall environmental impact profiles.
Safety Regulations for Polar Organic Compound Usage
The regulatory landscape for polar organic compounds, particularly amides and acetals, has evolved significantly in response to growing awareness of their environmental and health impacts. International frameworks such as REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) in Europe and TSCA (Toxic Substances Control Act) in the United States establish comprehensive guidelines for the handling, storage, and disposal of these compounds. These regulations specifically address polarity-related risks, as polar compounds often exhibit enhanced bioavailability and environmental persistence compared to their non-polar counterparts.
Workplace safety standards mandate strict exposure limits for polar organic compounds due to their increased solubility in biological systems. OSHA and equivalent international bodies have established Permissible Exposure Limits (PELs) that are typically lower for polar compounds than non-polar alternatives. For amides, particular attention is given to their potential for dermal absorption and respiratory exposure, while acetals require specific ventilation requirements due to their volatility characteristics. Personal protective equipment specifications must account for the enhanced penetration capabilities of polar solvents through standard materials.
Storage and handling protocols for polar organic compounds emphasize containment systems designed to prevent environmental release. Secondary containment requirements are often more stringent for polar compounds due to their potential for groundwater contamination. Temperature control systems must account for the unique thermal properties of these compounds, with amides requiring careful monitoring to prevent thermal decomposition and acetals needing protection from moisture-induced hydrolysis.
Emergency response procedures for polar organic compound incidents incorporate specialized protocols addressing their enhanced mobility in environmental systems. Spill response teams must utilize polar-compatible absorbents and neutralizing agents, as traditional hydrocarbon spill response materials prove ineffective. Fire suppression systems require polar-solvent compatible foams, particularly for acetal compounds which present unique combustion characteristics.
Waste disposal regulations for polar organic compounds mandate specialized treatment processes that account for their resistance to conventional biological treatment systems. Incineration parameters must be adjusted for complete destruction of polar compounds, while chemical treatment options focus on polarity reduction through controlled reactions. Transportation regulations classify these compounds based on their polarity-enhanced reactivity profiles, requiring specialized packaging and documentation protocols.
Workplace safety standards mandate strict exposure limits for polar organic compounds due to their increased solubility in biological systems. OSHA and equivalent international bodies have established Permissible Exposure Limits (PELs) that are typically lower for polar compounds than non-polar alternatives. For amides, particular attention is given to their potential for dermal absorption and respiratory exposure, while acetals require specific ventilation requirements due to their volatility characteristics. Personal protective equipment specifications must account for the enhanced penetration capabilities of polar solvents through standard materials.
Storage and handling protocols for polar organic compounds emphasize containment systems designed to prevent environmental release. Secondary containment requirements are often more stringent for polar compounds due to their potential for groundwater contamination. Temperature control systems must account for the unique thermal properties of these compounds, with amides requiring careful monitoring to prevent thermal decomposition and acetals needing protection from moisture-induced hydrolysis.
Emergency response procedures for polar organic compound incidents incorporate specialized protocols addressing their enhanced mobility in environmental systems. Spill response teams must utilize polar-compatible absorbents and neutralizing agents, as traditional hydrocarbon spill response materials prove ineffective. Fire suppression systems require polar-solvent compatible foams, particularly for acetal compounds which present unique combustion characteristics.
Waste disposal regulations for polar organic compounds mandate specialized treatment processes that account for their resistance to conventional biological treatment systems. Incineration parameters must be adjusted for complete destruction of polar compounds, while chemical treatment options focus on polarity reduction through controlled reactions. Transportation regulations classify these compounds based on their polarity-enhanced reactivity profiles, requiring specialized packaging and documentation protocols.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!